Abstract
Paraquat (PQ) toxicity is a common problem in the world, associated with oxidative stress, inflammation, and apoptosis. Therefore, the use of agents that reduce these disorders can be effective in the treatment of PQ toxicity. The protective effects of dimethyl fumarate (DMF) on liver disorders have been suggested in many reports. In this study, mice were divided into 6 groups; control, PQ (30 mg/kg, i.p., at day 4), DMF (100 mg/kg, p.o.), and PQ groups pretreated by DMF in three doses 10, 30, and 100 mg/kg, respectively. DMF was administered for 7 days to counteract PQ-induced liver toxicity. On the 8th day, mice were euthanized with ketamine/xylazine, and serum factors, oxidative stress markers, apoptosis index, and inflammatory markers were measured. PQ significantly increased the activity level of serum enzymes, thiobarbituric acid reactive substances, apoptotic factor (Bax/Bcl-2 ratio), inflammatory factors (NF-κB protein expression, tumor necrosis factor-α, interleukin-1β), nitric oxide, and Nrf-2 protein expression. Furthermore, PQ decreased hepatic total thiol and activity levels of catalase, superoxide dismutase, and glutathione peroxidase. However, DMF reduced the harmful effects caused by the imbalance in the oxidant and antioxidant system and histopathological damage in PQ-poisoned mice and improved the damage caused by inflammation and apoptosis.
Similar content being viewed by others
Introduction
Paraquat (PQ) is a powerful, non-selective herbicide that was first introduced in 1955. It’s widely used to help control weeds by acting as a desiccant and defoliant for crops. However, one of the concerning aspects of PQ is that non-target organisms can easily absorb it, making them vulnerable through ingestion, inhalation, skin contact, or eye exposure. In plants, toxicity of PQ arises from its ability to produce free radicals when it interacts with oxygen, which disrupts the photosynthesis process. In animals, PQ is rapidly distributed throughout various tissues and organs. Unlike some chemicals, it isn’t metabolized in the body; instead, it transforms into an unstable free radical. This radical is then re-oxidized, resulting in the formation of a superoxide anion and a cation, both of which are highly toxic to humans and animals alike1,2,3,4,5.
PQ is known as a potent free radical generator6 and one of the widely used agriculture herbicides worldwide7. Among the most serious poisonings related to this herbicide, we can mention accidental poisoning and intentional suicide, so PQ is a serious public health problem8. According to epidemiological data, PQ poisoning is responsible for more than 50% of pesticide-related fatality cases that affect organ systems including the respiratory, reproductive, neurobehavioral, and gastrointestinal systems9,10,11,12. Poisoning with a moderate concentration of PQ can cause serious heart, kidney, and liver failure within a few days to a few weeks13. Inflammation and oxidative stress play an essential role in the pathological processes of these changes14. The liver is the body’s primary organ for maintaining homeostasis between intracellular free radicals and the antioxidant defense system. Following PQ exposure, an excessive rise in free radicals and a reduction in antioxidant capacity are brought on by creating cellular superoxide anion, hydroxyl free radical, and nitric oxide (NO), which can result in oxidative stress, tissue damage, and liver malfunction15,16,17. It has been reported that PQ can cause many histopathological and biochemical changes in liver tissue18 Free radicals produced by PQ bind to important body molecules such as proteins, lipids, and DNA and cause liver dysfunction, so many patients poisoned with PQ suffer from hepatotoxicity19. Oxidative stress imbalance activates redox-sensitive signaling pathways, including nuclear factor erythroid-2 (Nrf-2) and nuclear factor-kappa B (NF-κB)20. With the increase of oxidative stress, Nrf-2 located in the cytoplasm is separated from Keap1 and migrates to the cell nucleus, which causes the activity of antioxidant enzymes21,22. Meanwhile, the NF-κB which is a transcription regulator also migrates into the nucleus and increases the expression of pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1 beta (IL-β1)23. PQ-induced apoptosis has been shown to activate the intrinsic mitochondrial pathway by increasing pro-apoptotic and anti-apoptotic proteins (Bax/Bcl-2 ratio) and activating caspases by releasing cytochrome C from mitochondria. Bax induces cell death and Bcl-2 prevents Bax-induced apoptosis, so overexpression of Bcl-2 prevents PQ- induced apoptosis24,25.
Dimethyl fumarate (DMF), known as an anti-inflammatory and antioxidant drug, has been used in patients with multiple sclerosis26,27 and psoriasis28. DMF blocks the activity of the cellular immune system by inhibiting glycolysis29. The use of DMF treatment may be helpful for individuals with psoriasis because it activates the (Nuclear factor erythroid-derived 2-like 2) Nrf-2 pathway, which provides protection against oxidative stress30. DMF has been suggested to have a potential protective effect on the nervous system, which is believed to be due to its ability to activate the Nrf-2 pathway in astrocytes, oligodendrocytes, and neurons. This pathway is triggered by an initial increase in oxidative stress caused by DMF binding and depleting Glutathione (GSH), which leads to an increase in reactive oxygen species (ROS). However, this increase in ROS ultimately results in the activation of antioxidant gene transcription and an increase in intracellular GSH concentration, which can protect against further oxidative damage31. According to the many studies that have been conducted on the safety of DMF, digestive problems are one of the most unwanted side effects of this drug, however, an increase in serum liver factors, including aspartate aminotransferase (AST) and alanine aminotransferase (ALT), has rarely been seen31,32,33,34. It has been reported that DMF improves liver function and antioxidant and inflammatory status by up-expression of antioxidant enzymes catalase (CAT) and glutamate cysteine ligase, as well as regulating a key inflammatory mediator known as NF-κB35. Also, DMF can regulate inflammatory pathways via inhibition of the Nrf-2 factor35,36. In this way, DMF has a common pathway with PQ herbicide, and it seems that the effects of increasing Nicotinamide adenine dinucleotide phosphate (NADPH) by DMF can be prevented in the treatment of adverse effects caused by the reduction of PQ. Considering that PQ is a multi-organ toxicant, the mechanisms of toxicity in different organs are important to investigate separately. In a study by our research team, the protective effects of DMF against pulmonary toxicity of PQ were investigated37. Therefore, knowing that the liver is one of the vital organs in the detoxification and metabolism of toxicants, it may also be exposed to the toxic effects of PQ11. Accordingly, the aim of the present study was to investigate the effects of DMF on the incidence of oxidative stress, apoptosis, and inflammation caused by PQ in the mouse liver.
Results
DMF effects on liver function biomarkers
In our study, we found that the activities of ALT, AST, and ALP were significantly elevated in the PQ treatment group when compared to the control group (P < 0.001). This elevation indicates potential hepatotoxicity associated with PQ exposure, reflecting liver damage or dysfunction. Notably, when PQ was co-treated with DMF at doses of 30 and 100 mg/kg, a significant decrease in serum enzyme activity levels was observed compared to the PQ group (P < 0.001). This reduction suggests that DMF may exert a protective effect against the liver damage induced by PQ, potentially restoring liver function or mitigating the toxic effects of PQ. These findings underscore the therapeutic potential of DMF in managing PQ-induced hepatotoxicity and warrant further investigation into its mechanisms of action and clinical applicability. A visual representation of these results is shown in Fig. 1.
The effects of DMF (dimethyl fumarate) on the activity level of (A): alanine aminotransferase: (ALT), (B): aspartate aminotransferase (AST), (C): alkaline phosphatase (ALP) in PQ (paraquat)-induced hepatotoxicity in mice (n = 7–8). *Significant difference compared to the control group (***P < 0.001). #Significant difference compared to the PQ group (###P < 0.001).
DMF effects on oxidative stress factors
We observed a significant increase in the formation of TBARS and NO in the PQ treatment group compared to the control group (P < 0.001). As illustrated in (Fig. 2), PQ administration led to a marked decrease in TT and the activities of key antioxidant enzymes, including GPx, SOD, and CAT, relative to the control group (P < 0.001). Conversely, co-treatment with DMF at doses of 30 and 100 mg/kg significantly mitigated the increases in NO and TBARS levels and enhanced the activities of GPx, SOD, CAT, and restored TT levels compared to the PQ group. These results suggest that DMF may effectively counteract the oxidative stress induced by PQ, promoting antioxidant defenses and improving overall cellular health.
The effects of DMF (dimethyl fumarate) on the level of (A): TBARS (thiobarbituric acid reactive substances), (B): NO (nitric oxide), (C): TT (total thiol), (D): GPx (glutathione peroxidase), (E): SOD (superoxide dismutase), and (F): CAT (catalase) in PQ (paraquat)-induced liver injury in mice (n = 7–8). *Significant difference compared to the control group (***P < 0.001). #Significant difference compared to the PQ group (#P < 0.05, ##P < 0.01 and ###P < 0.001).
DMF effects on inflammatory factors
We found that the levels of TNF-α and IL-1β were significantly elevated in the PQ treatment group when compared to the control group (P < 0.001). This increase indicates an inflammatory response triggered by PQ exposure. However, in the groups pretreated with DMF, we observed a significant reduction in the levels of both TNF-α and IL-1β compared to the PQ group, as shown in (Fig. 3). These findings suggest that DMF may possess anti-inflammatory properties that help to mitigate the inflammatory cytokine response associated with PQ toxicity and highlighting its potential role in protecting against inflammation-induced damage.
The effects of DMF (dimethyl fumarate) on the level of (A): tumor necrosis factor-α (TNF-α) and (B): interleukin-1β (IL-1β) in PQ (paraquat)-induced hepatotoxicity in mice (n = 7–8). *Significant difference compared to the control group (***P < 0.001). #Significant difference compared to PQ group (###P < 0.001).
Effects of DMF on the expression of NF-κB, Nrf-2, Bax, and Bcl-2 proteins
As illustrated in (Fig. 4) the Nrf-2 to NF-κB proteins as regulators of oxidative stress and inflammation were significantly elevated in mice exposed to PQ. Treatment with DMF at doses of 30 and 100 mg/kg resulted in overexpression of Nrf-2, but reduced NF-κB levels compared to the PQ group. These changes suggest that DMF may enhance the cellular antioxidant response while suppressing pro-inflammatory signaling pathways. Furthermore, we observed that the expression level of the pro-apoptotic protein Bax was significantly increased in mice intoxicated with PQ (P < 0.001), whereas the anti-apoptotic protein Bcl-2 exhibited a significant decrease (P < 0.001). Notably, treatment with DMF resulted in a reduction in Bax expression (P < 0.001) and a significant elevation in Bcl-2 levels (P < 0.001) in mice administered 30 and 100 mg/kg DMF compared to the PQ group. In addition, the ratio of Bax to Bcl-2 in the PQ-treated group was markedly elevated compared to the control group (P < 0.001). However, DMF-treated groups (10, 30, and 100 mg/kg) remarkably prevented the increase in the ratio of Bax to Bcl-2 compared to the PQ group (P < 0.001). These findings indicate that DMF not only modulates the expression of key proteins involved in oxidative stress and inflammation but also plays a crucial role in regulating apoptosis, thereby potentially enhancing cell survival in PQ toxicity.
The effects of DMF (dimethyl fumarate) on protein expression of (A): Nrf-2, (B): NF-κB, (C): Bax, (D): Bcl-2 proteins, (E): Bax/Bcl-2 ratio and (F): Western blot bands in PQ (paraquat)-induced hepatotoxicity in mice (n = 3). *Significant difference compared to the control group (***P < 0.001). #Significant difference compared to the PQ group (##P < 0.01 and ###P < 0.001).
Histopathologic findings
Histological examination of liver sections from control animals revealed a normal tissue structure, characterized by well-defined cellular structure and absence of pathological changes. In contrast, liver tissues from the PQ exposed group exhibited significant disruption of normal tissue patterns, including inflammatory cell infiltration and congestion. Notably, treatment with DMF at a dose of 100 mg/kg demonstrated a significant protective effect against PQ-induced histological damage. This treatment effectively reduced inflammatory cell infiltration and congestion within the liver tissue, as shown in (Fig. 5; Table 1). These findings suggest that DMF has the potential to preserve liver histology and mitigate the adverse effects of PQ exposure, highlighting its role as a protective agent in maintaining liver integrity under toxic conditions.
Discussion
Suicide and accidental poisoning with bipyridinium herbicides such as PQ quickly cause liver, kidney, and lung tissue damage38 By damaging the lipid cell membrane and lipid peroxidation, PQ causes widespread and multisystem toxicity in the body39. It has been shown in various clinical studies that PQ causes severe toxicity to various organs, including the liver40,41. Treatment of PQ poisoning is difficult due to its rapid distribution with a distribution volume of 1.2–1.6 L/kg in the liver, muscle, kidney, and lung42. Due to the mentioned reason, if PQ poisoning is not treated in a timely manner, the accumulation of PQ in the body can lead to rapid liver failure43. There is still no specific antidote for treatment, therefore the various proposed treatments have always been considered according to the mechanism of PQ toxicity40,42. In animal modeling studies, PQ administration with an acute dose in mice causes necrosis and inflammation of the liver due to oxidative damage to cells and the production of free radicals such as superoxide onions, hydrogen peroxide, hydroxyl free radicals, and peroxynitrite. Oxidative stress is one of the most important damage factors that leads to lipid peroxidation, mitochondrial membrane damage, cell death, and massive toxicity44,45,46. The redox cycle of PQ disrupts important biochemical processes by preventing the reduction of NADP+ to NADPH in the region of the electron transport chain, and induces GSH reduction and lipid peroxidation47, which can cause oxidative stress. Excessive production of ROS in the presence of PQ results in rapid and prolonged oxidation of NADPH, which is primarily caused by the cyclic reduction and reoxidation of PQ48. This oxidative reaction leads to significant consumption of NADPH, as it serves as a cofactor for glutathione reductase in the regeneration of oxidized glutathione (GSSG) back to its reduced form (GSH)49. As a consequence, the levels of intracellular SH groups decrease due to the oxidation of GSH, resulting in the formation of mixed disulfides between glutathione and protein thiols50. It has been reported that PQ increases the activity level of serum enzymes due to oxidative damage to liver cells and many pathological conditions such as liver and kidney damage, cancer, and diabetes51,52. In the study conducted by Semeniuk et al., ALT and AST increased in PQ group and decreased in genistein treatment groups53. Also, in a study that was induced by ischemia’s liver injury; Liver enzymes, including AST and ALT, increased, and DMF has had good recovery effects on damage54. The results of the present study show that the administration of PQ increased the serum enzymes AST, ALT, and ALP. However, a decrease in the activity of liver enzymes was observed in the group treated with DMF.
Inflammation and oxidative stress play important roles in the pathological processes of these changes. In addition to inducing oxidative stress, free oxygen radicals are responsible for direct cell damage with the regulated synthesis of nitrite oxide, which ultimately causes inflammation and tissue destruction. In the referenced studies, the group receiving PQ increased NO, and it decreased in the treatment groups in a dose-dependent manner55,56. DMF has been shown in a study to have a beneficial effect on acetaminophen hepatotoxicity by raising SOD and TT levels while lowering NO and TBARS57. As the results of the present study, DMF is a good antioxidant and reducing agent that could preserve the cellular and extracellular content of glutathione and NADPH-dependent enzyme mechanisms and glutathione enzyme activity. The results of the present study demonstrate that PQ reduces the TT, activity of antioxidant enzymes like SOD and CAT and subsequently increases TBARS, which play a role in detoxifying free radicals, and in this way, antioxidant enzymes do have not the capacity to remove oxygen free radicals then the amount of lipid peroxidation increases. Under oxidative stress, Nrf-2 is dissociated from Keap1 and subsequently moves into the nucleus, where it binds to the antioxidant response element (ARE) sequence, thereby activating the Nrf-2-ARE pathway. NF-κB serves as a key regulator of inflammatory responses, driving the expression of numerous pro-inflammatory genes. In the presence of oxidative stress, IκBα proteins undergo degradation through polyubiquitination following phosphorylation, allowing NF-κB to be released. NF-κB translocated to the nucleus, where it binds to its specific DNA response elements, resulting in the transcription of pro-inflammatory mediators. The crosstalk between Nrf-2 and NF-κB in the context of oxidative stress and inflammation is significant. When Nrf-2 translocates to the nucleus and binds to the ARE sequence, it enhances the expression of various antioxidant enzymes (such as SOD, CAT, GPx). Additionally, Nrf-2 can reduce the degradation of IκB-α, thereby supporting antioxidant defenses and potentially functioning independently of ROS. Subsequently, NF-κB nuclear translocation is suppressed. NF-κB has the ability to elevate levels of pro-inflammatory cytokines (such as TNF-α, IL-1β, IL-6)58. The Nrf-2/ARE pathway is considered an important signaling pathway related to antioxidant activity, but NF-κB is known as a pro-inflammatory factor59 that can activate cytokines like TNF-α and IL-1β. In several studies, Nrf-2 upregulation against oxidative stress conditions has been pointed out as a key defense factor against free radicals60,61. In Qian et al.‘s study, the expression of Nrf-2 was significantly increased in PQ-induced lung toxicity62. In this study, PQ increased the expression of Nrf-2 protein, which indicates the activity of the defense system to create compensatory mechanisms against oxidative stress of acute PQ toxicity. The results of the protective effect of DMF in the present study are consistent with the evidence of previous studies and caused a significant increase in Nrf-2 in the treatment groups compared to the PQ group. NF-κB increased in the group receiving PQ, which caused an increase in the levels of TNF-α and IL-1β. DMF, as an anti-inflammatory agent, was able to inhibit PQ-induced inflammation in a dose-dependent manner63,64. In the Abdelrahman and Abdel-Rahman study, the TNF- α factor increased significantly in the group treated with acetaminophen, while it decreased in the group treated with dimethyl fumarate. In our study, this is also similar to the mentioned study57. DMF has been reported in past studies to modulate the Nrf-2 pathway as well as the nuclear factor NF-κB65. Bax is a pro-apoptotic member of the Bcl-2 family that promotes apoptosis by forming pores in the mitochondrial membrane, leading to the release of cytochrome c and activation of downstream caspases. In contrast, Bcl-2 is an anti-apoptotic protein that inhibits Bax’s activity, preventing mitochondrial permeability transition and promoting cell survival. During hepatotoxic events, the ratio of Bax to Bcl-2 can rise by increased Bax expression and decreased Bcl-2 levels. This apoptotic signaling pathway, contributing to liver cell death and exacerbating liver injury66. In our findings, exposure to PQ was shown to increase the Bax/Bcl-2 ratio and shift the balance towards apoptosis. This alteration enhances oxidative stress within the cells, contributing to mitochondrial dysfunction and cell death. Our study corroborates previous research that highlights the significant interplay between oxidative stress and the apoptotic pathway, emphasizing the importance of these molecular changes in understanding the mechanisms underlying cellular responses to environmental stressors67,68,69. In many studies, PQ has caused inflammation in the liver tissue, and antioxidants have been able to reduce this inflammation70,71,72. The histopathological findings in our study show that PQ has led to the damage to mouse liver tissue, these damages are observed due to the infiltration of inflammatory cells and vascular congestion in the group receiving PQ.
Materials and methods
Chemicals
Most of the materials needed for research, including paraquat, dimethyl fumarate, thiobarbituric acid (TBA), Ellman’s Reagent, Bradford solution reagent, phosphate buffered saline, trichloroacetic acid, and phosphoric acid were obtained from Sigma Aldrich Company. All kits used for glutathione peroxidase (GPx) and superoxide dismutase (SOD) (Catalog Number: ZB-SOD-96 A /48A, NO (Catalog Number: ZB-NO-96 A/48A (and TNF-α (Cat. No: ZB-10082 C-H9648) tests were purchased from ZellBio Company. BT lab kit was used for IL-1β (Catalog Number: E0192Mo) test. Kits for measuring serum factors were purchased from Pars Azmoon Company. Bax (catalog number: sc-7480), anti-rabbit lgG (catalog number: sc-2357) and Bcl-2 (catalog number: sc-7382) antibodies were purchased from Santa Cruz Biotechnology company. GAPDH (catalog number: #5174), Nrf-2 (catalog number: #12721), NF-κB (catalog number: #8242), antibodies were purchased from Cell Signaling Technology Company.
Animals
Adult male NMRI mice (6–8 weeks of age) and (weight 23–27 g) were obtained from the Center of Animal House of Ahvaz Jundishapur University of Medical Sciences (AJUMS), Ahvaz, Iran. Before starting the experiment, to accustom the animals to the laboratory environment conditions such as lighting cycle (12:12 h), temperature regulation (22–25 °C), and free access to food and water for 7 days were provided. The study received approval from the Ethical Committee of Ahvaz Jundishapur University of Medical Science, which is in accordance with the ARRIVE guidelines as well as the instructions of the National Institute of Health (NIH) for the care and use of laboratory animals. The working methods of animal experiments and euthanasia were approved by the AJUMS ethics committee (Ethics approval ID: IR.AJUMS.ABHC.REC.1401.031).
Experimental method
Animals were randomly placed in different groups: a control group, a DMF group, a PQ group, and three DMF pretreatment groups. Each group consisted of 7–8 mice. Mice in the DMF-treated groups received oral DMF in 10% DMSO as a vehicle at doses of 10, 30, and 100 mg/kg/day35, for seven consecutive days (four days before and three days after PQ administration) (Fig. 6). Mice in PQ and DMF pretreated groups were intraperitoneally injected just with a single dose of 30 mg/kg PQ73. Based on the strict ethical protocols of working with laboratory animals and with the presence of selected observers from the AJUMS, mice were anesthetized with ketamine (90 mg/kg) and xylazine (10 mg/kg)74,75, intraperitoneally. Samples of blood were obtained after heart puncture. Performing of deep anesthesia, exsanguination, and disruption of the diaphragm induce animal euthanasia. Collected sera were used to measure liver enzyme activity. After collecting the liver tissue, one part was stored in phosphate-buffered formalin solution (10%) for sliding and checking histological criteria by the histologist, and the other parts were frozen at − 70 °C until the subsequent evaluation of hepatic markers. The protein concentration of the samples was measured using the standard Bradford method76.
Scheme of experimental protocol to study ameliorative effects of DMF (dimethyl fumarate) on PQ (paraquat)-induced of the liver injury in mice. Mice were pretreated with DMF (10, 30 and 100 mg/kg) or vehicle (10% DMSO in saline) orally 4 days before and continued 3 days after paraquat administration.
Liver tissue preparation
Samples were homogenized (1000 rpm) in phosphate buffer saline at a ratio (1:10 w/v) for 3 min. Then, the homogenized samples were centrifuged (8000 × g (for 10 min. In the next step, the tissues were sedimented, and the supernatants were collected and transferred to microtubes. To avoid damaging the samples, all steps were performed on ice and in the presence of a protease inhibitor.
Biochemical assays
Hepatic function biomarkers
After collecting the sera, the activities of AST, alkaline phosphatase (ALP), and ALT were assessed based on the kit protocols of the manufacturer.
Hepatic oxidative stress
The measurement of total thiol (TT) level77, thiobarbituric acid reactive substances (TBARS) level78, and CAT activity79 were performed according to mentioned studies. The analysis of colorimetric tests like NO, SOD and GPX were performed according to the method provided by the ZellBio manufacturer and read with plate reader.
Hepatic cytokine
To measure IL-1β and TNF-α in the liver supernatant samples, the methods of BT lab and ZellBio kits were used respectively.
Hepatic Western blot assay
Liver tissue was homogenized in RIPA lysis buffer with 1mM phenylmethylsulphonyl fluoride. Then the homogenates were centrifuged for 10 min at, 14,000 × g and the supernatants were transferred to new microtubes. The protein concentration of the supernatant was measured by the Bradford method, and the samples were kept at − 70 °C until further analysis. Fifty µg proteins were loaded with 10% SDS-PAGE and transferred to the nitrocellulose membrane. The membranes were blocked with 5% Skim Milk for 2 h and then incubated overnight with primary antibodies against GAPDH, Nrf-2, NF-κB, Bcl-2, and Bax at 4 ℃. After two washes, the membrane was incubated with anti-rabbit lgG for 2 h. Finally, the immunoreactive bands were detected using the Excellent Chemiluminescent Substrate (ECL) kit and quantified using JS 2000 (BonninTec, China) scanner. The protein expressions of Nrf-2, NF-κB, Bcl-2, and Bax were performed by analyzing the density of target proteins to GAPDH.
Histological procedure
For histopathological evaluation, liver tissue was fixed in a formalin solution and embedded in paraffin. The paraffin samples were sectioned at a thickness of 4 μm, placed on glass slides, and stained with H&E. Three slides were prepared from each animal tissue, and 5 microscopic fields from each slide were randomly scored numerically including: no lesion (grade 0), mild lesion (grade 1), moderate lesion (grade 2), and severe lesion (grade 3)80.
Statistical method
Data are shown as mean ± standard deviation. The data set of this research was compared using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons as a post hoc test. P values < 0.05 were considered statistically significant. All these analyses were performed with Prism 9 software.
Limitations
This study provides valuable insights into the potential protective effects of DMF against PQ-induced liver toxicity, but has limitations that should be considered. One limitation of this study is that we measured total protein expression of NF-κB and Nrf-2 without distinguishing between their phosphorylated or nuclear forms, which are indicators of their functional activity. However, alterations in the downstream pathway components of these factors may suggest the involvement of their active forms. Additionally, while Bax and Bcl-2 protein expressions were assessed as apoptosis markers, the study did not evaluate caspase-3 and caspase-9, key mediators in the apoptotic cascade. Future research should include these proteins and complementary methods like the TUNEL assay to provide direct evidence of apoptosis. Despite these limitations, the Bax/Bcl-2 ratio remains a valuable indicator supporting the study’s conclusions.
Conclusion
PQ inhibits antioxidant enzymes, induces lipid peroxidation, inflammation, and apoptosis in the liver, which ultimately leads to liver dysfunction (Fig. 7). Due to the positive effects of DMF as an anti-inflammatory, antioxidant, and antiapoptotic agent, it can be used against hepatotoxicity caused by PQ. Therefore, antioxidants and immunosuppressive drugs such as DMF, as an FDA-approved drug, may be suggested as effective and promising agent against PQ toxicity.
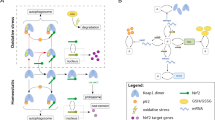
Graphical abstract shows the effects of dimethyl fumarate on paraquat-induced hepatotoxicity in mice. Parts of the graphical abstract were drawn by using pictures from Servier Medical Art, provided by Servier, licensed under a Creative Commons Attribution 3.0 unported license (https:/ /creativecommons.org/licenses/by/3.0/).
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Onur, B., Çavuşoğlu, K., Yalçin, E. & Acar, A. Paraquat toxicity in different cell types of Swiss albino mice. Sci. Rep. 12, 4818 (2022).
Chohan, M. S., Tahir, M., Lone, K. P., Sami, W. & Munir, B. Paraquat induced hepatotoxicity in albino mice. Pakistan J. Zool. 42 (2010).
Haripriya, B., Lakshman, M. & Sudha, V. Influence of paraquat induced acute toxicity on body weights and haemoto-biochemical parameters in experimental. Int. J. Sci. Res. 6, 396–398 (2017).
Reddy, K., Jeevanalatha, M., Lakshman, M. & Rani, M. U. The toxic effects of paraquat (PQ) on body weights and haematological parameters in male albino wistar rats and its amelioration with vitamin C. Int. J. Curr. Microbiol. App Sci. 8, 314–320 (2019).
Shariati, S. et al. The ameliorative effects of allopurinol on paraquat-induced pulmonary fibrosis in rats. Pharm. Sci. 25, 11–16 (2019).
Juchau, M. R., Fantel, A. G., Harris, C. & Beyer, B. K. The potential role of redox cycling as a mechanism for chemical teratogenesis. Environ. Health Perspect. 70, 131–136 (1986).
Strasberg, S. et al. The Brisbane 2000 terminology of liver anatomy and resections. Hpb 2, 333–339 (2000).
Rappaport, A., Borowy, Z., Lougheed, W. & Lotto, W. Subdivision of hexagonal liver lobules into a structural and functional unit. Bole in hepatic physiology and pathology. Anat. Rec. 119, 11–33 (1954).
Delirrad, M., Majidi, M. & Boushehri, B. Clinical features and prognosis of paraquat poisoning: a review of 41 cases. Int. J. Clin. Exp. Med. 8, 8122 (2015).
Ko, D. R. et al. Effects of paraquat ban on herbicide poisoning-related mortality. Yonsei Med. J. 58, 859 (2017).
Peiró, A. M. et al. Hepatotoxicity related to paraquat and diquat absorption through intact skin. Dig. Dis. Sci. 52, 3282–3284 (2007).
Shariati, S., Kalantar, H., Pashmforoosh, M., Mansouri, E. & Khodayar, M. J. Epicatechin protective effects on bleomycin-induced pulmonary oxidative stress and fibrosis in mice. Biomed. Pharmacother. 114, 108776 (2019).
Jungermann, K. in Seminars in liver disease. 329–341 (© 1988 by Thieme Medical Publishers, Inc.).
Arias, I. M. et al. The Liver: Biology and Pathobiology. (Wiley, 2020).
Liu, Z., Wang, X., Li, L., Wei, G. & Zhao, M. Hydrogen sulfide protects against paraquat-induced acute liver injury in rats by regulating oxidative stress, mitochondrial function, and inflammation. Oxidative Medicine and Cellular Longevity 2020 (2020).
Qi, M. et al. Ellagic acid ameliorates paraquat-induced liver injury associated with improved gut microbial profile. Environ. Pollut. 293, 118572 (2022).
Shirani, K., Yousefsani, B. S., Shirani, M. & Karimi, G. Protective effects of naringin against drugs and chemical toxins induced hepatotoxicity: a review. Phytother. Res. 34, 1734–1744 (2020).
Luo, L. et al. Assessment of serum bile acid profiles as biomarkers of liver injury and liver disease in humans. PloS One 13, e0193824 (2018).
Yang, C. J. et al. Spectrum of toxic hepatitis following intentional paraquat ingestion: analysis of 187 cases. Liver Int. 32, 1400–1406 (2012).
Zhang, L., Guo, Z., Wang, Y., Geng, J. & Han, S. The protective effect of kaempferol on heart via the regulation of Nrf2, NF-κβ, and PI3K/Akt/GSK‐3β signaling pathways in isoproterenol‐induced heart failure in diabetic rats. Drug Dev. Res. 80, 294–309 (2019).
David, J. A., Rifkin, W. J., Rabbani, P. S. & Ceradini, D. J. The Nrf2/Keap1/ARE pathway and oxidative stress as a therapeutic target in type II diabetes mellitus. J. Diabetes Res. 2017 (2017).
Badibostan, H., Eizadi-Mood, N., Hayes, A. W. & Karimi, G. Protective effects of natural compounds against paraquat-induced pulmonary toxicity: the role of the Nrf2/ARE signaling pathway. Int. J. Environ. Health Res. 34, 611–624. https://doi.org/10.1080/09603123.2022.2163985 (2024).
Hoffmann, A. & Baltimore, D. Circuitry of nuclear factor κB signaling. Immunol. Rev. 210, 171–186 (2006).
Zhang, Z. D. et al. Aspirin eugenol ester attenuates paraquat-induced hepatotoxicity by inhibiting oxidative stress. Front. Physiol. 11, 582801 (2020).
Jang, Y. J. et al. Paraquat induces apoptosis through a mitochondria-dependent pathway in RAW264. 7 cells. Biomolecules Ther. 23, 407 (2015).
Jordan, A. L., Yang, J., Fisher, C. J. & Racke, M. K. Mao-Draayer, Y. Progressive multifocal leukoencephalopathy in dimethyl fumarate-treated multiple sclerosis patients. Multiple Scler. J. 28, 7–15 (2022).
Bresciani, G. et al. Novel potential pharmacological applications of dimethyl fumarate—an overview and update. Front. Pharmacol. 14, 1264842 (2023).
Sulaimani, J. et al. Dimethyl fumarate modulates the Treg–Th17 cell axis in patients with psoriasis. Br. J. Dermatol. 184, 495–503 (2021).
Diebold, M. et al. High-dimensional immune profiling identifies a biomarker to monitor dimethyl fumarate response in multiple sclerosis. Proceedings of the National Academy of Sciences 119, e2205042119 (2022).
Lee, Y. J. et al. Pro-oxidant status and Nrf2 levels in psoriasis vulgaris skin tissues and dimethyl fumarate-treated HaCaT cells. Arch. Pharm. Res. 40, 1105–1116 (2017).
Dubey, D. et al. Dimethyl fumarate in relapsing–remitting multiple sclerosis: rationale, mechanisms of action, pharmacokinetics, efficacy and safety. Expert Rev. Neurother. 15, 339–346 (2015).
Wynn, D., Lategan, T. W., Sprague, T. N., Rousseau, F. S. & Fox, E. J. Monomethyl fumarate has better gastrointestinal tolerability profile compared with dimethyl fumarate. Multiple Scler. Relat. Disorders 45, 102335. https://doi.org/10.1016/j.msard.2020.102335 (2020).
Linker, R. A. & Gold, R. Dimethyl fumarate for treatment of multiple sclerosis: mechanism of action, effectiveness, and side effects. Curr. Neurol. Neurosci. Rep. 13, 1–7 (2013).
Reszke, R. & Szepietowski, J. C. A safety evaluation of dimethyl fumarate in moderate-to-severe psoriasis. Exp. Opin. Drug Saf. 19, 373–380 (2020).
Vanani, A. R. et al. Dimethyl fumarate reduces oxidative stress, inflammation and fat deposition by modulation of Nrf2, SREBP-1c and NF-κB signaling in HFD fed mice. Life Sci. 283, 119852 (2021).
Li, Y., Chu, L., Liu, C., Zha, Z. & Shu, Y. Protective effect of GSK-3β/Nrf2 mediated by dimethyl fumarate in middle cerebral artery embolization reperfusion rat model. Curr. Neurovasc. Res. 18, 456–464 (2021).
Mahmoudi, Z., Kalantar, H., Mansouri, E., Mohammadi, E. & Khodayar, M. J. Dimethyl fumarate attenuates paraquat-induced pulmonary oxidative stress, inflammation and fibrosis in mice. Pestic. Biochem. Physiol. 190, 105336 (2023).
Smith, P., Heath, D., Fishman, A. P. & Paraquat CRC Crit. Reviews Toxicol. 4, 411–445 (1976).
Bus, J. S., Aust, S. D. & Gibson, J. E. Paraquat toxicity: proposed mechanism of action involving lipid peroxidation. Environ. Health Perspect. 16, 139–146 (1976).
Shadnia, S. et al. Delayed death following paraquat poisoning: three case reports and a literature review. Toxicol. Res. 7, 745–753 (2018).
Yu, G. et al. A case report of acute severe paraquat poisoning and long–term follow–up. Experimental Therapeutic Med. 8, 233–236 (2014).
Houze, P. et al. Toxicokinetics of paraquat in humans. Hum. Exp. Toxicol. 9, 5–12 (1990).
Gawarammana, I. B. & Buckley, N. A. Medical management of paraquat ingestion. Br. J. Clin. Pharmacol. 72, 745–757 (2011).
Asaduzzaman, M. et al. Paraquat-induced acute kidney and liver injury: Case report of a survivor from Bangladesh. Clin. Case Rep. 9, e05020 (2021).
Autor, A. Biochemical mechanisms of paraquat toxicity. (1977).
Morán, J. M., Ortiz-Ortiz, M. A., Ruiz‐Mesa, L. M. & Fuentes, J. M. Nitric oxide in paraquat‐mediated toxicity: a review. J. Biochem. Mol. Toxicol. 24, 402–409 (2010).
Pontes, M. S. et al. Chitosan/tripolyphosphate nanoformulation carrying paraquat: insights on its enhanced herbicidal activity. Environ. Science: Nano 8, 1336–1351 (2021).
Castello, P. R., Drechsel, D. A. & Patel, M. Mitochondria are a major source of paraquat-induced reactive oxygen species production in the Brain*. J. Biol. Chem. 282, 14186–14193. https://doi.org/10.1074/jbc.M700827200 (2007).
Dinis-Oliveira, R. et al. Paraquat poisonings: mechanisms of lung toxicity, clinical features, and treatment. Crit. Rev. Toxicol. 38, 13–71 (2008).
Dinis-Oliveira, R. J. et al. P-glycoprotein induction: an antidotal pathway for paraquat-induced lung toxicity. Free Radic. Biol. Med. 41, 1213–1224. https://doi.org/10.1016/j.freeradbiomed.2006.06.012 (2006).
Ajayi, A. M., Adedapo, A. D., Badaki, V. B., Oyagbemi, A. A. & Adedapo, A. A. Chrysophyllum albidum fruit ethanol extract ameliorates hyperglycaemia and elevated blood pressure in streptozotocin-induced diabetic rats through modulation of oxidative stress, NF-κB and PPAR-γ. Biomed. Pharmacother. 141, 111879 (2021).
Lamia, S. S. et al. Coenzyme Q10 and silymarin reduce CCl4-induced oxidative stress and liver and kidney injury in ovariectomized rats—implications for protective therapy in chronic liver and kidney diseases. Pathophysiology 28, 50–63 (2021).
Semeniuk, M. et al. Protective effect of genistein pre-treatment on paraquat hepatotoxicity in rats. Toxicol. Appl. Pharmcol. 426, 115636 (2021).
Takasu, C. et al. Treatment with dimethyl fumarate ameliorates liver ischemia/reperfusion injury. World J. Gastroenterol. 23, 4508 (2017).
Shen, H. et al. Toll-like receptor 9 mediates paraquat-induced acute lung injury: an in vitro and in vivo study. Life Sci. 178, 109–118 (2017).
Shen, H. et al. Chloroquine attenuates paraquat-induced lung injury in mice by altering inflammation, oxidative stress and fibrosis. Int. Immunopharmacol. 46, 16–22 (2017).
Abdelrahman, R. S. & Abdel-Rahman, N. Dimethyl fumarate ameliorates acetaminophen-induced hepatic injury in mice dependent of Nrf-2/HO-1 pathway. Life Sci. 217, 251–260 (2019).
Wu, S. et al. Antioxidant and anti-inflammation effects of dietary phytochemicals: the Nrf2/NF-κB signalling pathway and upstream factors of Nrf2. Phytochemistry 204, 113429 (2022).
Henderson, W. R., Chi, E. Y., Teo, J. L., Nguyen, C. & Kahn, M. A small molecule inhibitor of redox-regulated NF-κB and activator protein-1 transcription blocks allergic airway inflammation in a mouse asthma model. J. Immunol. 169, 5294–5299 (2002).
Khodayar, M., Kalantari, H., Khorsandi, L., Rashno, M. & Zeidooni, L. Upregulation of Nrf2-related cytoprotective genes expression by acetaminophen-induced acute hepatotoxicity in mice and the protective role of betaine. Hum. Exp. Toxicol. 39, 948–959 (2020).
Shi, Z. et al. The LipoxinA4 receptor agonist BML-111 ameliorates intestinal disruption following acute pancreatitis through the Nrf2-regulated antioxidant pathway. Free Radic. Biol. Med. 163, 379–391 (2021).
Qian, J. et al. Anthrahydroquinone-2-6-disulfonate is a novel, powerful antidote for paraquat poisoning. Sci. Rep. 11, 20159 (2021).
Dwivedi, D. K., Jena, G. & Kumar, V. Dimethyl fumarate protects thioacetamide-induced liver damage in rats: studies on Nrf2, NLRP3, and NF‐κB. J. Biochem. Mol. Toxicol. 34, e22476 (2020).
Xu, Y. et al. Rapamycin protects against paraquat-induced pulmonary fibrosis: activation of Nrf2 signaling pathway. Biochem. Biophys. Res. Commun. 490, 535–540 (2017).
Scuderi, S. A., Ardizzone, A., Paterniti, I., Esposito, E. & Campolo, M. Antioxidant and anti-inflammatory effect of Nrf2 inducer dimethyl fumarate in neurodegenerative diseases. Antioxidants 9, 630 (2020).
Sharma, S., Singh, R. & Kakkar, P. Modulation of Bax/Bcl-2 and caspases by probiotics during acetaminophen induced apoptosis in primary hepatocytes. Food Chem. Toxicol. 49, 770–779 (2011).
Rai, N. K., Tripathi, K., Sharma, D. & Shukla, V. K. Apoptosis: a basic physiologic process in wound healing. Int. J. Low. Extrem. Wounds 4, 138–144 (2005).
Willis, S., Day, C. L., Hinds, M. G. & Huang, D. C. The bcl-2-regulated apoptotic pathway. J. Cell Sci. 116, 4053–4056 (2003).
Wong, W. W. L. & Puthalakath, H. Bcl-2 family proteins: the sentinels of the mitochondrial apoptosis pathway. IUBMB life 60, 390–397 (2008).
Zeinvand-Lorestani, H., Nili-Ahmadabadi, A., Balak, F., Hasanzadeh, G. & Sabzevari, O. Protective role of thymoquinone against paraquat-induced hepatotoxicity in mice. Pestic. Biochem. Physiol. 148, 16–21 (2018).
Awadalla, E. A. Efficacy of vitamin C against liver and kidney damage induced by paraquat toxicity. Exp. Toxicol. Pathol. 64, 431–434 (2012).
Shalaby, M., Emam, S. & Soliman, A. Protective effect of vitamin e against herbicide paraquat-induced enzymatic leakage and oxidative damage in the liver of rats. Adv. Anim. Vet. Sci. 8, 639–646 (2020).
Shen, H., Wu, N., Liu, Z., Zhao, H. & Zhao, M. Epigallocatechin-3-gallate alleviates paraquat-induced acute lung injury and inhibits upregulation of toll-like receptors. Life Sci. 170, 25–32 (2017).
Shariati, S., Shirani, M., Azadnasab, R., Khorsandi, L. & Khodayar, M. J. Betaine protects mice from Cardiotoxicity triggered by Sodium Arsenite through Antioxidative and anti-inflammatory pathways. Cardiovasc. Toxicol. 24, 539–549 (2024).
Oh, S. S. & Narver, H. L. Mouse and rat anesthesia and Analgesia. Curr. Protocols 4, e995 (2024).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Ellman., G. L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82, 70–77 (1959).
Uchiyama, M. & Mihara, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem. 86, 271–278 (1978).
Shangari, N. & O’Brien, P. J. Catalase activity assays. Curr. Protocols Toxicol. 27(7), 16 (2006). 71-7.7.
Mirzaei, M., Moosavi, M., Mansouri, E., Mohtadi, S. & Khodayar, M. J. Diosmin exerts hepatoprotective and antihyperglycemic effects against sodium arsenite-induced toxicity through the modulation of oxidative stress and inflammation in mice. J. Trace Elem. Med Biol. 78, 127154 (2023).
Funding
This manuscript is taken from the pharmacy thesis of Maryam Kavianinia and was financially supported by the Toxicology Research Center of Ahvaz Jundishapur University of Medical Sciences (Grant No: TRC-0114).
Author information
Authors and Affiliations
Contributions
M.K.: material preparation, methodology, writing original draft and investigation. H.K.: study conception and design, editing and revising manuscript, supervision. M.S.: writing original draft, study conception and design. L.K.: pathological analysis. S.S.: writing original draft, investigation, material preparation, S.M.: editing and revising manuscript. M.J.K.: material preparation, methodology, investigation, writing original draft, editing and revising manuscript, supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Animal experiments working methods and euthanasia were performed based on animal care and use methods approved by the Ethics Committee of AJUMS (Ethics Approval ID: IR.AJUMS.ABHC.REC.1401.031).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kavianinia, M., Kalantar, H., Salehcheh, M. et al. Dimethyl fumarate effects on paraquat-induced hepatotoxicity in mice via anti-oxidative, anti-inflammatory, and anti-apoptotic activities. Sci Rep 15, 3897 (2025). https://doi.org/10.1038/s41598-025-88461-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-88461-y
Keywords
This article is cited by
-
Comparative study of adipose tissue derived mesenchymal stem cells with rapamycin on paraquat-induced acute lung injury and pulmonary fibrosis in a mouse model: histological and biochemical study
Stem Cell Research & Therapy (2025)
-
Boric acid and quercetin supplementations alleviated paraquat-induced neurotoxic and irritation effects in human SH-SY5Y cells and in ovo models
BMC Complementary Medicine and Therapies (2025)
-
Antioxidative Effects of Crocin-loaded Noisome in Paraquat-induced Oxidative Stress in Lung Rat
Cell Biochemistry and Biophysics (2025)