Abstract
In this study, the magnetic TiO2/Cu/aluminosilicate (MCTA) catalyst was synthesized through a two-step process using the hydrothermal and solvothermal methods. The synthesized materials underwent comprehensive characterization using various analytical techniques. These materials were subsequently applied for the effective elimination of Acid Black 1 dye (AB1). To establish robust correlations among these influential parameters, the research adopted the Box–Behnken design (BBD) within the framework of RSM. This approach ensures a systematic and thorough exploration of the interplay between various variables in the photocatalytic degradation process. In this study, sunlight was utilized as an energy source for the degradation process. The degradation of AB1 reached its peak efficiency under the following optimal conditions: catalyst mass of 0.75 g/L, AB1 concentration of 37.5 mg/L, pH of 3, and a contact time of 45 min. The efficiency, based on the model prediction, was 98.9%, while experimental results showed a 99.1% efficiency. Dark adsorption tests were also conducted, revealing that the dye degradation rate through the photocatalytic process was 18–22 times higher compared to the adsorption process at different concentrations. Toxicity tests revealed that the remaining AB1 in the aquatic environment lost its antibacterial activity after undergoing the studied process. The scavenger experiment provided confirmation that successful photodegradation of AB1 is attributed to the indispensable contribution of both •OH and •O2−. Additionally, MCTA demonstrated outstanding cycle stability in the photocatalytic degradation of AB1. MCTA emerges as a dependable sunlight-responsive heterostructure photocatalyst with significant potential for the photodegradation of pollutants in wastewater treatment.
Similar content being viewed by others
Introduction
Water pollution with toxic and non-degradable chemicals such as heavy metals, artificial dyes, and organic micropollutants threatens the physical and environmental health of billions of people1,2. The estimated usage of dyes is around one million tons per year, widely employed in the production stages of food items, paper, cosmetics, and textiles3. The effluents from these industries contain significant amounts of colorants, leading to long-term environmental pollution, reduced light penetration, decreased dissolved oxygen (DO), increased chemical oxygen demand (COD) in receiving waters, and contamination of groundwater, due to discharge into the environment, even with low concentrations of dyes4.
According to the World Bank research report, textile dyeing and treatment constitute 17–20 percent of industrial water pollution5. Approximately, 2000 different chemical substances are used in the textile industry as dyes, and around 15% of these utilized dyes enter the ecosystem along with wastewater6. Dyes have diverse effects on human health, including allergies, dermatitis, digestive disorders, and eye irritation9. Moreover, dyes are recognized as potential carcinogens that can induce genetic abnormalities. Therefore, the removal of dyes is essential to prevent pollution8.
Among different types of dyes, azo compounds are more commonly used due to their color stability and chemical durability. The azo group is classified into reactive, basic, acidic, direct, dispersed, and sulfur dyes based on one or more different azo (N=N) bonds9. These dyes are categorized as hazardous substances for the environment due to their toxicity and slow degradation10. Acidic dyes are typically used in textile industries (nylon, silk, and wool) within the pH range of 3–7. Acidic dyes often contain metal complexes in their structure. AB1, with the formula of Na2C22H14N6O9S2, is an example of an acidic and anionic dye, which is extensively used in textile industries11.
Although pollution resulting from the use of dyes in the textile industry is substantial, it is challenging to reduce it due to the high demand for colored and printed textiles. However, it is possible to decrease the color concentration in the wastewater of these industries before discharge through various processes13. There are various physical and chemical methods for removing dyes from aquatic environments. Some of them include adsorption14, membrane filtration15, electrocoagulation16, photocatalytic decomposition17, and advanced oxidation processes18. Unfortunately, many of these processes are costly and time-consuming, and they generate a significant amount of sludge, making its disposal challenging. Advanced oxidation processes (AOPs) transform pollutants into harmless substances by generating highly oxidizing free radicals. Consequently, the production of hazardous by-products is avoided19.
AOPs are classified based on the generation of reactive species, including photocatalysis, sonolysis, ozonation, Fenton oxidation, and electrochemical processes20. Among advanced oxidation processes, photocatalytic oxidation has gained significant attention in the past two decades due to its low operational cost, minimal energy consumption, negligible by-product generation, and environmentally friendly structure, making it an effective technique for the safe detoxification of pollutants present in air and wastewater21.
Some materials used as photocatalysts include ZnO22, CuO23, CeO224, WO325, and TiO226. TiO2 is a semiconductor photocatalyst with strong oxidative power, water affinity, stability, non-toxicity, and insolubility in water. It is utilized for degrading different pollutants, including alkenes PCBs and alkenes PAHs27, alcohol phenols28, and dyes29.
Solar energy technology stands as a form of renewable energy that is both sustainable and environmentally friendly and is abundant and cost-effective. Solar radiation alone is also used for decomposing pollutants in water and wastewater. Nowadays, semiconductor photocatalysts are widely employed in utilizing solar energy to modify toxic dyes, leading to cost reduction in these processes30. The use of TiO2 as a photocatalyst is constrained by several significant limitations. The accelerated recombination of electron–hole (e−/h+) pairs curtails the production of various reactive species such as OH, H2O2, and O3 during photocatalytic processes31. As well, due to the relatively large bandgap of TiO2 (3–3.2 eV), its photoresponse is limited to the UV range32. Impurities such as metals, non-metals, or common impurities can enhance the photocatalytic performance of TiO233. In recent times, there has been a notable advancement in surface modification techniques for nanostructured TiO2, involving the incorporation of metals (e.g., Cu, Ag, and Au). This modification has effectively broadened the absorption wavelength of TiO2, extending it into the visible region34,35. Among these, copper has been considered as the most effective impurity for the TiO2 network, significantly reducing the TiO2 bandgap and shifting absorption from UV to the visible region. Additionally, this metal absorbs electrons generated by light excitation, preventing rapid recombination of e−/h+36.
The efficacy of TiO2 as a photocatalyst is contingent upon its surface area, porosity, and crystal structure37. To diminish aggregation and simplify recovery, a diverse spectrum of supports, encompassing zeolites, silica, activated carbon, graphene, and clay minerals, can be effectively employed38. Natural clay minerals possess plentiful adsorption and reaction activation sites, contributing to their outstanding catalytic performance. Their sturdy structure supports effective catalyst recycling, and their unique spatial arrangement allows for the assembly of various dimensional scales. Additionally, their robust cation exchange capacity accelerates catalytic reactions, while surface electronegativity enhances the separation of charge carriers39,40. Aluminosilicate clay minerals (ACM) act as ideal photocatalyst supports, thanks to their expansive specific surface area and abundance of hydroxyl groups on their surface41.
The separation of these particles from treated water, especially from large volumes of water, is a costly and time-consuming process that has limited its use42. Magnetism is a desirable physical property in wastewater treatment because magnetic separation, avoiding centrifugation stages, is an easy method for separating substances from aqueous solutions43. Therefore, the use of TiO2 on magnetic nanoparticles has been proposed as an efficient solution for the formation of magnetic photocatalysts, offering an effective approach for the separation and useful recovery of photocatalysts44.
In view of the minimal studies on the removal of contaminants by TiO2/Cu/aluminosilicate, our study attempts to progress the photocatalytic performance of TiO2/Cu/aluminosilicate by implementing a magnetization process. This study also fully investigates all the related processes (mineralization rate and biodegradability, reaction kinetics, toxicity test, catalyst recovery, effects of anions, EPR spectra and scavenging experiments), which is rarely seen in such a complete study.
Therefore, the current investigation endeavors to fabricate a novel nanocomposite through the utilization of the solvothermal method. The synthesized MCTA nanocomposite was employed as a photocatalyst for eliminating AB1. The optimal conditions for maximizing AB1 degradation were determined using the RSM and BBD. The comparative analysis of the photocatalytic performance involved assessing the synthesized nanocomposite under both sunlight and non-sunlight conditions. Kinetic studies were conducted using the optimized parameters. According to our research findings, we have discovered and reported the degradation of AB1 using the novel MCTA photocatalyst. This significant achievement demonstrates the first documented instance of such degradation using this specific photocatalyst.
Materials and methods
Materials
The chemicals and reagents utilized in this study were sourced from Sigma-Aldrich and adhered to the highest analytical reagent quality standards.
Preparation of Fe3O4 nanoparticles
Magnetite cores were first synthesized using the co-precipitation method with the primary substance and Fe3+ and Fe2+ ions in an alkaline environment. By dissolving 11.66 g of FeCl3·6H2O and 4.31 g of FeCl2·4H2O in 200 ml of deionized water, a solution was initially prepared. The solution was placed on a shaker at 100 °C for one hour, and nitrogen gas bubbling was applied to prevent oxidation. In this step, the precipitation of Fe3+ and Fe2+ occurs in the presence of OH−. While stirring, 70 ml of 30% ammonia solution was added drop by drop to the reactor. At this stage, the shift in color from orange to brown during this stage signaled the development of iron magnetite oxides (Fe3O4) in the solution. At the end of this step, the supernatant was magnetically separated with the assistance of an iron rod.
Preparation of CTA
The CTA nanocomposites were synthesized through the solvothermal method45. Initially, in a beaker, bentonite clay (0.5 g) was combined with ethanol (25 ml) and stirred vigorously at room temperature for two hours until a suspension developed. Afterward, 0.2 g of copper chloride was introduced, blending effortlessly with the bentonite and causing a slight color lightening. After an additional hour of stirring, 1.5 g of TiO2 was added to the solution with ongoing stirring, which turned the mixture greyish-white in color. Following an additional 2 h of stirring, the thick mixture was sealed in an autoclave and heated at 105 °C for 15 min. After centrifugation at 5000 rpm for 15 min, the supernatant was obtained from the resultant mixture. Multiple washes with ethanol were applied to the supernatant, and the product was subsequently dried at 105 °C in an oven for 3 h. The dried product underwent further calcination at 500 °C for 2 h in a furnace to yield the CTA nanocomposite.
Synthesizing MCTA nanocomposites
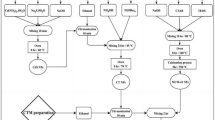
The synthesis procedure for the nanocomposite is comprehensively depicted in Fig. 1. To produce the final composites (MCTA), a hydrothermal method was employed. Initially, 0.5 g of Fe3O4 nanoparticle underwent ultrasonic treatment in 100 mL of deionized water for 2 h. Subsequently, 0.5 g of CTA nanocomposite was added, and the solution underwent further ultrasonication for 1 h. Each solution was then loaded into a 300 ml stainless steel autoclave coated with Teflon and subjected to heating at 200 °C for 12 h, followed by natural cooling to room temperature. The MCTA samples were acquired after washing and drying.
The characterization of the nanocomposite involved various techniques, including UV–VIS Spectrometry (DR5000), FTIR (Thermo Nicolet, USA 370 AVATAR), Scanning Electron Microscopy (TESCAN MIRA2, Czech Republic), XRD (PHILIPS; PW1730), BET (Brunauer–Emmett–Teller), TGA (TA; SDT-Q600), and VSM (MDKB; Iran). Using a potentiostat, EIS was performed with a sinusoidal perturbation voltage set to 2 mV, and the measurements were taken over a frequency range of 0.01 Hz–1 MHz Transient photocurrent.
Photocatalytic and adsorption experiments
The degradation of AB1 solution was employed to gauge the photocatalytic efficiency of MCTA nanoparticles under direct solar radiation, conducted between 10:00 and 14:00, with an average daily temperature of 36 ± 4 °C. Prior to exposure to light, the solutions underwent magnetic stirring in darkness for 60 min to attain a state of adsorption–desorption equilibrium between MCTA and AB1. The preparation of a stock solution involved dissolving AB1 in 1 L of distilled water, resulting in a concentration of 1000 mg/L. Various quantities of the MCTA catalyst were examined for their efficacy in degrading the AB1 solution. Samples were collected at specified time intervals (up to 120 min) and subsequently centrifuged to eliminate the catalyst. The reduction in AB1 concentration was quantified by measuring the absorption at 610 nm using a UV–visible spectrophotometer. The absorbance values were then correlated to concentrations through a standard calibration curve.
Utilizing a TOC analyzer, the levels of Total Organic Carbon (TOC) were assessed. The standard method involving colorimetry and UV–visible spectrophotometry was employed to analyze the initial and final concentrations of Chemical Oxygen Demand (COD). The COD measurement process included adding 2 mL of the wastewater sample to a COD vial containing oxidants, followed by a 2-h digestion at 150°C. The COD concentration was then determined using a DR-5000 at a specific wavelength. Biochemical Oxygen Demand (BOD5) levels were measured with a BOD meter (BD 600).
To assess the pHPZC of the catalyst, an appropriate amount of MCTA catalyst was distributed into 11 flasks, each holding 50 mL NaCl solution (0.01 M). The pH range was adjusted between 2 and 12 using NaOH and HCl (0.1 N). Following agitation at 120 rpm for 24 h, the final pH values were documented. The pHPZC was derived by plotting the initial and final pH values and finding their point of intersection.
Experimental design and statistical analysis
In this investigation, the RSM, employing the BBD in three levels (+ 1, 0, −1), was employed to optimize and analyze the impact of variables, as well as the simultaneous interactions among variables, on the response variable (removal of AB1). Design Expert Version 8 was utilized for this purpose. The variables under examination included pH, time, MCTA mass, and the initial AB1 concentration. Utilizing the response level method with the BBD model, the required number of experiments (N) was determined by the equation N = 2 K(K − 1) + C; here, K represents the number of variables, while C denotes the number of center points. Considering the removal efficiency of AB1 as the dependent variable (response), Table S1 provides details on the four factors studied, including their chosen levels and associated ranges. To ensure experiment reproducibility, all experiments were conducted with three replications, with a criterion of Relative Standard Deviation (RSD) less than 0.05. Central points were chosen for estimating and gauging experimental error, as well as gauging the lack of fit (LOF). LOF was employed, along with an examination of R2 and R2 Adj between laboratory and predicted values, to ascertain the statistical significance of quadratic fit models.
Results and discussion
Characterization of MCTA
The FTIR spectra of the MCTA nanocomposites, as illustrated in Fig. 2a, reveal distinct features. Pre-700 cm−1 absorption peaks signify the bending vibrations of metal oxide bonds, specifically Ti–O at 652.1 cm−1 and Cu–O at 481.4 cm−1, providing confirmation of the successful synthesis of CTA nanocomposites46. A pronounced peak at 1053.88 cm−1 specifies the stretching vibrations associated with Si–O–Si bonds. Additionally, the observed band at 3094 cm−1 corresponds to the stretching mode of hydroxyl groups in water molecules adsorbed on the surface47. The FTIR spectra also exhibit a characteristic band at 591 cm−1, indicative of the stretching vibration of the Fe–O functional group in magnetite.
The magnetic characteristics of Fe3O4 nanoparticles and MCTA nanocomposites were explored, and the findings are depicted in Fig. 2b. The hysteresis loop for Fe3O4 nanoparticles demonstrates superparamagnetic behavior, with magnetic saturation occurring around 40 emu/g. Upon the introduction of CTA nanoparticles and the formation of MCTA, there is a decrease in the magnetic saturation of Fe3O4 nanoparticles. This reduction can be ascribed to the attendance of CTA nanoparticles within the composite, effectively covering the Fe3O4 nanoparticles an observation supported by the hysteresis loop of MCTA as illustrated in Fig. 2b.
The diffuse reflectance UV–visible spectra for TiO2, CTA, and MCTA are depicted in Fig. 2c. TiO2, upon preparation, exhibits heightened absorbance in the UV region, with a peak absorption at around 359 nm. In contrast, a broadened peak ranging from 300 to 800 nm is evident for both CTA and MCTA. This broadening is ascribed to the blue shift of the absorption edges in CTA and MCTA when compared to TiO2. Utilizing the Kubelka–Munk function, band gap energy values for the nanocomposites were computed and are presented in Fig. 2d. The plot indicates an intercept line reaching the x-axis, providing the bandgap energy values for the nanocomposites. TiO2 photocatalyst exhibits a bandgap energy of 3.22 eV, while CTA and MCTA reveal bandgap energies of 1.72 and 1.27 eV, respectively. The reduced bandgap energies in CTA and MCTA, compared to TiO2, are attributed to the integration of magnetic nanoparticles and Cu/aluminosilicate materials into the TiO2 structure. Alterations in size and shape likely contribute to the shift in band gap. The inclusion of magnetic particles facilitates convenient recovery of the nanomaterial from water, and the role of Cu/aluminosilicate is to augment surface area, leading to heightened recombination rates. Ultimately, this enhances pollutant degradation within the specified timeframe.
In Fig. 2e, the Mott-Schottky plots for MCTA are presented. As illustrated, the potentials of the conduction band (CB) in MCTA are recorded at −0.38 eV. Consequently, the valence band (VB) potential is determined to be + 0.89 eV, calculated based on the CB value and the band gap (1.27 eV) obtained from the optical absorption spectrum.
Figure 3a and b present the data obtained from nitrogen sorption isotherms for bentonite clay and MCTA nanocomposites. Observations in Fig. 3a, corresponding to the adsorption isotherm of bentonite clay, point to the existence of micropores within the material structure. Solids with a microporous structure exhibit a type I adsorption isotherm profile, characterized by rapid nitrogen gas uptake at low relative pressures (p/p0). An escalation in relative pressure results in the isotherm adopting a type II profile, featuring a notable H2-type hysteresis loop. Bentonite pore volume and BET surface area are determined to be 0.317 cm3/g and 86.9 m2/g, respectively. Figure 3b demonstrates that the addition of TiO2 and Fe3O4 to bentonite clay alters the sorption isotherm types of the composites from type I/II to type IV, featuring an H3-H4 hysteresis loop. This change implies the attendance of non-rigid aggregates of plate-like particles, indicating the contribution of micropores and mesopores in MCTA. The BET surface area of the MCTA composite is 95.2 m2/g, and the total pore volume is 0.332 cm3/g. Additionally, the pore size distribution (PSD) for bentonite clay and nanocomposites was determined using Density Functional Theory (DFT) with medium regularization, as shown in the insert graphics of Fig. 3a and b. These results reveal predominantly mesoporous structures in all solids.
The thermogram of the MCTA nanocomposites, presented in Fig. 3c, exhibits two distinct stages of thermal weight loss. The initial stage occurs between 60 and 120 °C, resulting in a weight loss of approximately 3–6%. This initial loss is linked to the evaporation of water molecules trapped on the clay’s surface. Weight loss in the 60–120 °C typically signifies the removal of solvent molecules, water, or organic matter contained in the sample. At temperatures of 200–250 °C, the second stage of weight loss constitutes about 30–32% of the total weight loss45. This stage is linked to the removal of water within the layers of bentonite clay, indicating the dehydroxylation of its layers. Moreover, weight loss beyond 200 °C is associated with decomposition processes.
The XRD analysis for the MCTA nanocomposite is depicted in Fig. 3d. Distinct peaks are evident at angles 26.5°, 27.5°, 36.5°, 41.5°, 54.5°, 56.9°, and 69°, corresponding to the specific diffraction angles of the MCTA nanocomposite48. Fe3O4 nanoparticles exhibit seven discernible peaks at 2θ = 31.2°, 35.58°, 43.5°, 53.7°, 57.5°, 63.2°, and 74.2°, aligning with crystallographic planes (220), (311), (311), (400), (422), (511), (440), and (553), respectively. Notable peaks at angles 26.5°, 27.5°, 36.5°, and 41.5° indicate the presence of TiO2 in the sample. Reflection peaks at angles 54.5° and 56.9° suggest the presence of oxygen and silicon atoms (quartz), affirming the existence of bentonite clay. The peak at 69.1° is indicative of copper in the nanocomposite. A sharp peak at 27.5° signifies the anatase phase of TiO2, suggesting a reduction in grain size after the synthesis of nanocomposites, consistent with the general trend of decreasing grain sizes of TiO2 in nanocomposites.
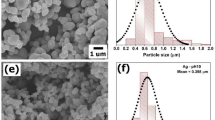
The FESEM analysis images are depicted in Fig. 3e and f. In Fig. 3e, the FESEM image of bentonite reveals particles with a spherical and transparent appearance. Contrastingly, the FESEM image of MCTA in Fig. 3f illustrates the presence of irregularly sized spherical aggregates, appearing to obstruct the bed’s surface. These observations are consistent with findings reported in prior studies.
As shown in Fig. 4, photocurrent (PC) and electrochemical impedance spectroscopy (EIS) analyses were conducted to explore the enhanced charge separation ability and the increased lifetime of photogenerated carriers. During repeated 60-s illumination cycles, the electrodes demonstrated prompt and stable current responses. Figure 4a highlights the photoresponse switching patterns of TiO2, TiO2/Cu, and MCTA nanocomposites. The TiO2 electrode produced a minor photocurrent of 0.22 μA cm−2, whereas the MCTA nanocomposite generated a photocurrent density of 0.84 μA cm−2, nearly 3.8 times higher than that of TiO245. The MCTA nanocomposite exhibited a sharp photocurrent increase to 1.35 μA under illumination, stabilizing near 1 μA before dropping drastically to its original level when the light was turned off. This behavior highlights the substantial improvement in the photocurrent of the quaternary MCTA nanocomposite, with enhanced charge separation efficiency and a prolonged lifespan compared to other materials. To emphasize the benefits of the quaternary MCTA nanocomposite compared to TiO2 and TiO2/Cu samples, Nyquist plots were prepared and analyzed, as shown in Fig. 4b. The MCTA nanocomposite exhibited the smallest impedance arc radius, which reflects a lower charge-transfer resistance. This feature is linked to more efficient separation of photogenerated e−/h+ and an overall improvement in the photocatalytic performance of the material.
Box–Benken statistical analysis
Selecting an appropriate model for the system, capable of predicting results with high and accurate precision, is a crucial initial step in result analysis. In this context, the proposed quadratic model was employed. The model encompasses four terms representing single effects (A, B, C, and D), six terms for interaction effects (AB, AC, AD, BC, BD, and CD), and four terms associated with quadratic effects (A2, B2, C2, and D2). To determine the significance of the model and assess its validity, an ANOVA P-Value analysis was conducted. The ANOVA results concerning the removal of AB1 are reported in Table 1. The P-value for the proposed model, as indicated by the data in this table, is less than 0.0001, suggesting the statistical significance of single effects and their effectiveness (P-Value < 0.05). Considering the remaining parameters in the system, Eq. (1) was derived to forecast the AB1 removal efficiency. It encompasses a combination of single, interaction, and quadratic effects.
In the presented equation:
Y represents the anticipated AB1 removal efficiency in percentage, A signifies the pH, B corresponds to the quantity of nanocomposite (g/L), C denotes the time duration (min), D represents the initial concentration of AB1 (mg/L).
The proposed model performance was assessed using key metrics, including correlation coefficient (R2), adjusted correlation coefficient (R2 Adj), and predicted correlation coefficient (Pred R2). The model demonstrated high accuracy with R2 and R2 Adj values of 0.992 and 0.981, respectively. The close alignment of these values indicates the model’s robust accuracy. This underscores a robust relationship between the experimental findings and the values predicted by the statistical model, confirming the model’s reliability and effectiveness.
In Fig. 5a, the comparison between predicted values and observed values is depicted, while Fig. 5b displays the standardized residues for each run. Analysis of these diagrams suggests that, across all conducted experiments, the standardized residue values fall within an acceptable range. Additionally, Fig. 5c portrays internally studentized residual values, affirming the model acceptance and providing opportunities for variance analysis.
Figure 6 provides a visual representation of the crucial variables influencing AB1 removal, e.g., nanocomposite dose, reaction time, the initial concentration of AB1, and pH. The figure offers insights into the variable ranges, selected intervals, impact magnitudes, and optimal points for each parameter. Al-Musawi et al.48, utilizing titanium dioxide nanoparticles/graphene oxide, investigated the sonocatalytic process for eliminating acid orange 7 dye from aqueous solutions. Through the application of RSM, the quadratic model demonstrated superior fitting when analyzing COD removal data, with the solution pH identified as a highly influential parameter (p < 0.0001).
The adjusted R2 solidifies the high significance of the model (R2 > 0.98). The experiments demonstrate high precision and reliability, as evidenced by the low coefficient of variation (C.V) at 2.4. Furthermore, with a p-value < 0.0001 and an F-value of 85.2, the model demonstrates a meaningful correlation. As observed in Table 1, the Lack of Fit for the ultimate models indicates statistical significance, affirming that the models are in strong agreement with the experimental data.
Moreover, the attained Adequate Precision ratio of 24.2, exceeding 4, indicates a strong signal for the second-order model, suggesting its suitability for application within the design space.
Based on the p-value being less than 0.1, factor effects and interactions for AB1 removal by the process in Table 1 indicate the statistical significance of all main factors. The greatest impact on AB1 removal is observed for the catalyst dosage (F = 854.2), while the least impact is attributed to the initial pH (F = 17.2) among the main factors. Results from Table 1 demonstrate that the overall effects of interactions are less significant than those of the main factors. Of these, Statistical significance is observed for the AB and BC interactions in the model. Regarding second-order interactions, all are statistically significant, with the B2 interaction having the most substantial impact, due to its high F-value, on the AB1 degradation by the MCTA catalyst.
The optimum operating conditions for the AB1 degradation process are as follows: an AB1 concentration of 37.5 mg/L, a catalyst mass of 0.75 g/L, a time of 45 min, and a pH of 3. The efficacy, based on the model prediction, is 98.9%, while experimental results demonstrate an efficiency of 99.1%.
3D response surface plot analysis
Figure 7a shows the surface plots of AB1 photodegradation. The plot represents the relationship between the catalyst dosage and the corresponding irradiation time. The elliptical pattern of the 3D plot indicates that AB1 photodegradation was influenced by the interaction between the catalyst concentration and the duration of irradiation, as illustrated in the figure. Additionally, an increase in MCTA dosage enhanced the removal efficiency. An elevated catalyst concentration improves the access to active surface sites, allowing for the absorption of additional photons. This results in the formation of higher quantities of O2−• and •OH, facilitating the degradation of more AB1 molecules34. Evidence from studies suggests that rising the catalyst dosage develops the degradation of methylene blue dye. This enhancement is explained by the rise in active photocatalytic sites, which absorb more photons and produce greater quantities of O2−• and •OH radicals, leading to more effective pollutant removal29. It was apparent that AB1 degradation was maximized after 60 min of reaction. The results validated the positive influence of extended contact time on the degradation process. Therefore, the enhanced removal of AB1 was attributed to increased contact time, which allowed for a higher probability of interactions between e−/h+ pairs and AB1 molecules. Hitkari et al.23 found that the degradation of congo red dye was significantly enhanced with increasing contact time, rising from 0% to 89.2% between the start and 60 min of the reaction. It was shown by other researchers that naproxen removal efficiency increased with prolonged contact time, as longer contact time generated a greater abundance of active sites on the catalyst surface36.
pH adjustments in the solution are instrumental in governing the dye adsorption process through modifications in the adsorbent’s surface charge, pollutant ionization, and dye structure. By raising the contact time and reduction the pH value, as presented in Fig. 7b, the photodegradation efficiency of AB1 increases.
The effect of pH on AB1 adsorption can be described based on the catalyst pHzpc, which is 7 (Fig S1). At pHzpc, the acidic and basic functional groups do not alter the solution pH. For pH below pHzpc, the MCTA surface becomes positively charged, attracting anionic dyes from the solution. When the pH is above pHzpc, the MCTA surface becomes negatively charged, attracting cationic dyes5. Therefore, at pH < 7, a substantial positive charge density is evident on the MCTA surface, enhancing the adsorption of the AB1 dye, which carries a negative charge. With a high negative charge density on its surface at pH levels above 7, MCTA exhibits decreased adsorption of the negatively charged AB1 dye. The initial pH’s effect on dye adsorption is likely attributable to chemical and electrostatic interactions between the adsorbent and the dye molecules11.
Variations in dye concentration, particularly over a contact period of 10–60 min (see Fig. 7c), are linked to the observed decline in photodegradation performance. While an initial increase in AB1 concentration improves the reaction rate by supplying more reactants to the catalyst’s surface, further increases in dye concentration reduce photocatalytic efficiency. This reduction results from the higher number of pollutant molecules in concentrated solutions, which block light penetration to the photocatalyst surface. As a result, fewer hydroxyl radicals are generated, limiting their ability to degrade contaminants effectively20. Nevertheless, over time, the adsorption of dye molecules on the catalyst surface hinders the adsorption of oxygen and water molecules, thereby limiting the formation of •OH19. Moreover, an increase in AB1 concentration leads to light scattering, while the dye’s electrons can become excited upon absorbing energy from the light. Hence, the absorption of emitted light by excited dye molecules increases with rising dye concentration, which consequently lowers the generation of hydroxyl radicals by the photocatalyst21.
Reaction kinetics
Pseudo First Order (PFO) or the Hinshelwood-Langmuir model can be employed to aptly characterize the photocatalytic reaction kinetics involving organic compounds. The mentioned model intricately relates the degradation rate to the pollutant concentration, as succinctly expressed in Eq. (2) 49:
In the given equation, K represents the main constant rate (1/min), and kd signifies the absorption equilibrium constant (l/mg).
In scenarios where adsorption is relatively low or the concentration of the organic compound is low, Eq. (2) can be reduced to PFO. This form includes a constant rate (Kapp = kd K, 1/min), as defined in Eq. (3):
In the mentioned equation, C0 and Ce refer to the concentrations (mg/L) of AB1 at the beginning and after a designated time (t, min) of the photocatalytic reaction, respectively.
The half-life constant is a pivotal parameter in the pollutant removal process, signifying the time required for the pollutant to degrade by 50%. This constant is determined using the equation represented below50:
The results obtained for both processes conform to the first-order kinetics model, exhibiting a high regression coefficient. Table 2 presents the reaction rate coefficients for various concentrations in both the adsorption and photocatalytic processes. Notably, a rise in concentration causes a reduction in the reaction rate coefficient, which matches the observed target percentage. Moreover, the photocatalytic process demonstrates a markedly higher reaction rate coefficient compared to the adsorption process, ranging from 18 to 22 times higher across different concentrations. Based on these findings, the half-life constant is calculated for concentrations from 10 to 100 mg/L for both processes. In the adsorption process, the half-life constant exhibits an increasing trend with concentration, ranging from 96.2 to 288.7 min. In contrast, the corresponding half-life constant for the photocatalytic process is considerably shorter, varying from 5.13 to 13.32 min for the same concentrations. The adsorption rates for concentrations of 10, 25, 50, 75, and 100 mg/L are 4.6, 9.25, 13.2, 20.4, and 24.6 mg/g, respectively. Comparable findings were seen in research by Al-Hawary et al.51, where the photocatalytic process exhibited a reaction rate coefficient tenfold higher compared to the adsorption process during the degradation of acid red 14, with respective half-life constants of 11.2 and 121.4 min. Additionally, Yilmaz et al.20 observed comparable trends in their study on the degradation of acid blue 113 by Fe3O4/ZnO/GO, reporting a greater reaction rate coefficient for the photocatalytic process compared to adsorption.
Effects of anions
Anionic species hold considerable significance in the chemical composition of aqueous systems (63). The photodegradation of AB1 using the MCTA photocatalyst is analyzed in this section, focusing on the impact of anions. Specifically, 0.1 M solutions of Na2CO3, NaCl, NaNO3, Na3PO4, and Na2SO4 were individually employed to assess how carbonate, chloride, nitrate, phosphate, and sulfate ions impact the reaction rate. The experiments utilized an initial concentration of 37.5 mg/L of AB1 with 0.75 g/L of MCTA. As depicted in Fig. 8a, the presence of these anions marginally inhibited the degradation of AB1, resulting in prolonged reaction times. This inhibition effect aligns with previous observations involving anionic species, e.g., NaCl, NaNO3, and Na2SO452. Competing for active sites on the photocatalyst surface or inducing deactivation is a potential consequence of the presence of these anions. Additionally, some studies propose that these anions may function as scavengers for active species (h+ and •OH), generating less reactive radicals such as Cl• or ClOH−• (Eqs. 5 and 6). The formation of these fewer active radicals subsequently decelerates the catalytic degradation rate53,54.
Mineralization rate and biodegradability
The effectiveness of an oxidation process in eliminating organic matter is significantly influenced by its mineralization and biodegradability. In this study, the mineralization rate of AB1 in the MCTA catalyst was assessed using COD and TOC indices. As depicted in Fig. 8b, the TOC removal efficiency escalates from 0 (41.2 mg/L) to 91.1% (3.67 mg/L) with an increasing reaction time of 0 to 120 min, while the AB1 removal rate remains steady at 99.1%. These findings corroborate the COD removal results, indicating a reduction in COD from 117.4 to 7.8 mg/L at 120 min. It may be attributed to the good mineralization of AB1 molecules into CO2 and H2O, while others are converted into more resistant organic compounds that resist degradation by active radicals55. Adekunle et al.56 detected similar results for the mineralization rate of Eriochrome black T using the Enhanced Nickel Oxide system.
To evaluate the biodegradability of photocatalyzed AB1, the ratios of BOD5 to COD (BOD5/COD) and BOD5 to TOC (BOD5/TOC) were examined for the AB1 solution, as presented in Fig. 8b. The figure illustrates an improvement in the BOD5/COD ratio from 0.26 to 0.72 and for BOD5/TOC from 0.745 to 1.38 with the extension of irradiation time (0 to 120 min). As the threshold values for the BOD5/COD and BOD5/TOC ratios are 0.4 and 0.8, respectively, the treated solution meets such criteria57. Furthermore, the results highlight that the MCTA process effectively converts AB1 into biodegradable compounds, particularly with reaction times exceeding 60 min. In line with earlier studies, the confirmation of effluent biodegradability can be achieved through the utilization of the carbon oxidation state (COS) and average oxidation state (AOS) techniques, as specified in Eqs. (7) and (8) 58. In the context of these equations, COD signifies the chemical oxygen demand concentration at reaction time t (mg/L), TOC refers to the initial total organic carbon concentration (mg/L), and TOCt indicates the concentration of soluble TOC at reaction time t (mg/L). Figure 7c displays graphs showing the COS and AOS values of the effluent solution over various reaction times. Over a reaction time of 120 min, the AOS and COS values gradually rise from -0.27 and + 1.64 to 1.75 and 3.93, respectively. This trend indicates the transformation of AB1 molecules into degradable compounds through the photocatalytic process, making the effluent suitable for introduction into the biological treatment process.
Halomoan et al.57, using CuO-Gd2Ti2O7 for the degradation of malachite green dye, reported removal efficiencies of 92% for TOC and 88% for COD were reported. Moreover, the BOD5/COD ratio rose from 0.27 to 0.7, signifying the transformation of non-biodegradable wastewater into biodegradable wastewater.
Catalyst recovery
One of the most important parameters for the economic viability of a catalyst is its recovery and reuse. In this study, recovery and reuse experiments were conducted in five stages. The experiments were performed at the optimum amount obtained from the RSM optimization method. After each use, the catalyst was separated using iron wool, underwent multiple wash cycles with distilled water, subjected to ultrasonic treatment for 10 min, and finally dried at 105 °C for further testing. After five recovery cycles, the catalyst still maintains its efficiency, although the removal percentage has decreased (Fig. 8d). It can be justified for two reasons. Firstly, the reduction in the catalyst during collection, where approximately a 2% reduction in the collection amount was observed at each stage. Secondly, the saturation of active surface sites of the catalyst during preparation can be mentioned59. This saturation is similar to the study conducted by Hitkari et al.23, which aimed at Congo red dye removal using copper-zinc oxide nanocomposite, where a reduction was also observed during the recovery stage, with approximately 20% reduction in the weight of the adsorbent during the separation stage, attributed to its magnetic properties in this study.
In the study of Yilmaz et al.20 for removing the AB113 using the photocatalytic process with the Fe3O4/ZnO/GO catalyst, the recovery rate of the catalyst is 95%, and during the five recovery stages, about 25% of the catalyst has been lost, while in this study, 10 percent of the catalyst was reduced during the 5 steps. The decrease in the amount of metal in the period of evaluation can be considered as one of the reasons for the decrease in pollutant degradation.
Toxicity test
To assess the remaining antibacterial effectiveness of the AB1 solution treated with MCTA, a microbiological antibacterial activity assay was conducted using E. coli as a reference. In Fig. 8e, it is evident that, with an extended photocatalytic duration, the bacteriostatic ring exhibited a diminishing diameter, reducing from 9.5 mm to 0 mm after 120 min, indicating the loss of antibacterial activity in the residual AB1 within the water environment post-photocatalytic reaction. Figure 8f presents a comparative analysis of the antibacterial test results for AB1 dye treated exclusively with the catalyst through adsorption. Notably, the persistence of the bacteriostatic ring post-adsorption suggests the retention of antibacterial activity in the antibiotics. These findings imply a substantial reduction in the antibacterial properties of AB1 after the photocatalytic reaction, thereby offering notable advantages for the efficient recycling of dye wastewater.
Identification of active species and elucidation of the mechanism
The photocatalytic degradation of AB1 can be ascribed to the joint action of active species, including h+, e−, O2−•, and OH•. To assess how each active species contributes, scavenging experiments were conducted using isopropanol (IPA) to trap OH•, ammonium oxalate (AO) to scavenge H+ ions, K2S2O8 to capture e−, and 1,4-benzoquinone (BQ) to quench O2−• 60. Figure 9a summarizes the results of AB1 degradation experiments with MCTA catalyst in the absence and presence of these scavengers. The introduction of IPA (1 mM) significantly reduces the initial efficiency of 99.1% to 61.2% after 1 h of irradiation. This observation highlights the crucial involvement of OH• in the degradation mechanism. Similarly, upon introducing 1 mM BQ, the efficiency dropped to 70.3% within 60 min, suggesting that O2−• is a key factor in the degradation process of AB1 while the inhibition effects of AO and K2S2O8 were relatively minor, suggesting that the involvement of H+ and e− in the photocatalytic degradation of AB1 is minimal.
The existence of •OH and O2−• was reaffirmed through the Electron Spin Resonance (ESR) spin-trap technique employing 5,5-dimethylpyridine-N-oxide (DMPO). The achieved ESR spectra using the MCTA/sunlight system (Fig. 9b) exhibited characteristic signals for DMPO-•OH and DMPO-•O2− spin adducts, further substantiating the involvement of these radicals in AB1 degradation61. Therefore, the proposed photocatalytic system primarily relies on O2−• and •OH for AB1 degradation, with supplemental contributions from photogenerated holes.
Based on DRS, trapping, and EPR spectra experiments, Fig. 8c displays a proposed schematic diagram elucidating the potential mechanism for the photocatalytic degradation of AB1. Before interpreting the possible mechanism, the conduction band (CB) and valence band (VB) energies of TiO2 and Fe3O4 particles were determined through Eqs. (9) and (10) 61.
The CB potential of the semiconductor is denoted by ECB, while EVB represents the VB potential of the semiconductor. The electronegativity of the Mulliken catalyst is represented by χ, with values of 5.78 and 5.81 eV for Fe3O4 and TiO2, respectively62,63. Ec symbolizes the energy of free electrons at 4.5 eV on the hydrogen scale, and Eg represents the energy associated with the band gap of particles.
According to Eg TiO2 (3.22 eV) and χ stated in the earlier study63 (5.81 eV), ECB and EVB were calculated to be − 0.3 eV and 2.92 eV, respectively. For Fe3O4, ECB and EVB values reported by Channei et al.61 were used (+ 0.48 eV and + 2.08 eV, respectively). When the MCTA nanocomposite is exposed to sunlight, every component of the MCTA nanocomposite becomes excited, giving rise to the production of e− and h+. The photogenerated e− is transferred from the TiO2 conduction band to the Cu surface because the Cu surface is more positive than the TiO2 nanoparticle CB. Next, some of e− is directed from Cu to the Fe₃O₄ valence band, owing to the lower positioning of the Fermi level of Cu metal relative to the energy of the valence band62. This event is beneficial for the photocatalysis process through declining e−/h+ recombination. In this way, the photogenerated electron in Fe3O4 is transferred to the aluminosilicate surface. The electron on the aluminosilicate surface converts the O2 molecule into the O2•− radical63. The holes produced in VB TiO2 and VB Fe3O4 can oxidize H2O or OH− molecules to generate •HO because the positive value of VB particles is greater than the potential of H2O/HO• (E0 = + 2.34 eV vs. NHE) and OH−/HO• (E0 = + 1.99 eV vs. NHE)64. Finally, all the active species produced can decompose AB1 in two phases, soluble and solid, into CO2, H2O compounds and low biological risk products.
Comparison with studies
To evaluate the effectiveness of the synthesized catalyst, its performance was compared with results from prior studies (Table 3). The analysis revealed that all the examined processes could achieve dye removal efficiencies exceeding 80%. However, this high efficiency was obtained under a range of laboratory conditions, complicating direct comparisons among the findings. Notably, textile industry wastewater generally exhibits a low pH, and conventional wastewater treatment plants have shown limited success in dye removal. Table 3 indicates that acidic environments and increased catalyst dosages enhance the efficiency of most advanced oxidation processes. The MCTA photocatalytic system demonstrated complete AB1 removal under acidic conditions (pH 3). Achieving 100% AB1 degradation within a mere 60 min, our catalyst surpassed the efficiency of other semiconductor-based photocatalytic systems. These results highlight the superior ability of our photocatalyst to produce reactive species necessary for the efficient breakdown of AB1.
Conclusion
The analysis of the prepared photocatalytic nanocomposite involved a comprehensive examination using various microscopic and spectroscopic methods. By assessing the optimal nanocomposite sample, it was tested as a nanostructured catalyst for the photodecomposition of AB1. Trapping experiments demonstrated that hydroxyl radicals exhibited superior efficacy in decomposing AB1 molecules when facilitated by the MCTA nanophotocatalyst. The Box–Behnken design disclosed that all the main factors were statistically significant, with the catalytic dosage having the most significant impact (F = 854.2), and the initial pH exhibiting the least impact (F = 17.2) among the main factors. Furthermore, in this study, catalyst recovery experiments were conducted, and the findings demonstrated a consistently high efficiency of the catalyst over five consecutive cycles. Also, the results of the biodegradability tests showed that the BOD5/COD ratio improved from 0.26 to 0.72, and for BOD5/TOC, it was 0.745–1.38 as the duration of irradiation extended from 0 to 120 min. This indicates the process of transforming wastewater from a non-biodegradable to a biodegradable composition. Furthermore, as the photocatalytic duration extended, the bacteriostatic ring diameter progressively diminished from 9.5 to 0 mm within 120 min. Analysis of the impact of anions indicated a subtle inhibition in AB1 degradation, resulting in extended reaction times. The findings from this experimental study underscore that incorporating Cu/aluminosilicate into TiO2, coupled with the hydrothermal and solvothermal synthesis of the MCTA nanocomposite, yielded an innovative high-performance nanocatalyst for applications in water remediation and environmental purification.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Balarak, D., Mostafapour, F. K. & Joghatayi, A. Adsorption of Acid Blue 225 dye by multi walled carbon nanotubes: Determination of equilibrium and kinetics parameters. Pharm. Chem. 8, 138–145 (2016).
Al-Musawi, T. J., Mengelizadeh, N. & Al-Rawi, O. Capacity and modeling of acid blue 113 dye adsorption onto chitosan magnetized by Fe2O3 nanoparticles. J. Polym. Environ. 30, 344–359 (2022).
Sillanpää, M. & Mahvi, A. H. Adsorption of Acid orange 7 dyes from aqueous solution using Polypyrrole/nanosilica composite: Experimental and modelling. Int. J. Environ. Anal. Chem. 103, 212–229 (2021).
Gou, Z. et al. Removal of dye pollution by an oxidase derived from mutagenesis of the Deuteromycete Myrothecium with high potential in industrial applications. Environ. Pollut. 310, 119726 (2022).
Rafiq, A. et al. Photocatalytic degradation of dyes using semiconductor photocatalysts to clean industrial water pollution. J. Ind. Eng. Chem. 97, 111–128 (2021).
Kumar, N., Pandey, A. & Sharma, Y. C. A review on sustainable mesoporous activated carbon as adsorbent for efficient removal of hazardous dyes from industrial wastewater. J. Water Process Eng. 54, 104054 (2023).
Vinayagam, V. et al. Recent developments on advanced oxidation processes for degradation of pollutants from wastewater with focus on antibiotics and organic dyes. Environ. Res. 240, 117500 (2023).
Sadia, M. et al. Synthesis and characterization of MIPs for selective removal of textile dye acid Black-234 from wastewater sample. Molecules 28, 1555 (2023).
Rafaqat, S., Perveen, B., Imran, W., Hussain, A. & Ali, N. Development of manganese peroxidase based voltammetric biosensor for detection of textile Azo dyes RR 195 & RB 221. Mater. Chem. Phys. 312, 128647 (2023).
Ali, F. et al. Synthesis, column packing and liquid chromatography of molecularly imprinted polymers for the acid black 1, acid black 210, and acid Brown 703 dyes. RSC Adv. 12(196), 1–23 (2022).
Khatoon, J. et al. Convenient pH-responsive removal of Acid Black 1 by green l-histidine/iron oxide magnetic nanoadsorbent from water: performance and mechanistic studies. RSC Adv. 9(6), 2978–2996 (2019).
Elbadawy, H. A., Sadik, W. A., Elhusseiny, A. F. & Hussein, S. M. Design of economic photocatalytic system with low energy consumption, and high quantum yield, for the degradation of acid red 37 textile dye. Process. Saf. Environ. Prot. 148, 1191–1206 (2021).
Fernandes, J. V., Rodrigues, A. M. & Menezes, R. R. Adsorption of anionic dye on the acid-functionalized bentonite. Materials 13(16), 3600 (2020).
Chen, H., Huang, M., Liu, Y., Meng, L. & Ma, M. Functionalized electrospun nanofiber membranes for water treatment: A review. Sci. Total. Environ. 739, 139944 (2020).
Taheri, M. Techno-economical aspects of electrocoagulation optimization in three acid azo dyes’ removal comparison. Cleaner. Chem. Eng. 2, 100007 (2022).
Ahmed, J. et al. Synthesis, characterization, and significant photochemical performances of delafossite AgFeO2 nanoparticles. J. Sol-Gel Sci. Technol. 94, 493–503 (2020).
Ma, D. et al. Critical review of advanced oxidation processes in organic wastewater treatment. Chemosphere 275, 130104 (2021).
Raninga, M., Mudgal, A., Patel, V. K., Patel, J. & Sinha, M. K. Modification of activated carbon-based adsorbent for removal of industrial dyes and heavy metals: A review. Mater. Today: Proc. 77, 286–294 (2023).
Navidpour, A. H., Abbasi, S., Li, D., Mojiri, A. & Zhou, J. L. Investigation of advanced oxidation process in the presence of TiO2 semiconductor as photocatalyst: property, principle, kinetic analysis, and photocatalytic activity. Catalysts. 13(2), 232 (2023).
Yilmaz, M., Mengelizadeh, N., Khodadadi Saloot, M. & Balarak, D. Facile synthesis of Fe3O4/ZnO/GO photocatalysts for decolorization of acid blue 113 under solar, visible and UV lights. Mater. Sci. Semicond. Process. 144, 106593 (2022).
Tichapondwa, S. M., Newman, J. & Kubheka, O. Effect of TiO2 phase on the photocatalytic degradation of methylene blue dye. Phys. Chem. Earth. 118, 102900 (2020).
Dalal, C. et al. Sunlight-assisted photocatalytic degradation of azo-dye using zinc-sulfide embedded reduced graphene oxide. Solar Energy 251, 315–324 (2023).
Hitkari, G., Chowdhary, P., Kumar, V., Singh, S. & Motghare, A. Potential of Copper-Zinc Oxide nanocomposite for photocatalytic degradation of congo red dye. Cleaner Chem. Eng. 1, 100003 (2022).
Rauf, H. et al. New insight in photocatalytic degradation of textile dyes over CeO2/Ce2S3 composite. Phys. B. 632, 413760 (2022).
Warsi, A.-Z. et al. Synthesis and characterisation of WO3 and Ag2O nanoparticles and their nanocomposite for photocatalytic degradation by taguchi technique and detection by solid phase nano extraction. Iranian J. Chem. Chem. Eng. 40(5), 1541–1553 (2021).
Ashkezari, H. D., Kalal, H. S., Hoveidi, H., Almasian, M. & Ashoor, M. Fabrication of UV/TiO2 nanotubes/Pd system by electrochemical anodization for furfural photocatalytic degradation. Casp. J. Environ. Sci. 15, 1–11 (2017).
McQueen, A. D. et al. Photocatalytic degradation of polycyclic aromatic hydrocarbons in water by 3D printed TiO2 composites. ACS ES&T Water. 2(1), 137–147 (2021).
Choquette-Labbé, M., Shewa, W. A., Lalman, J. A. & Shanmugam, S. R. Photocatalytic degradation of phenol and phenol derivatives using a Nano-TiO2 catalyst: Integrating quantitative and qualitative factors using response surface methodology. Water 6(6), 1785–1806 (2014).
Kurniawan, T. A. et al. Functionalizing TiO2 with graphene oxide for enhancing photocatalytic degradation of methylene blue (MB) in contaminated wastewater. J. Environ. Manage. 270, 110871 (2020).
Nawaz, A. et al. Solar light driven degradation of textile dye contaminants for wastewater treatment–studies of novel polycationic selenide photocatalyst and process optimization by response surface methodology desirability factor. Chemosphere 328, 138476 (2023).
Pandiyaraj, K. N. et al. Dye wastewater degradation by the synergetic effect of an atmospheric pressure plasma treatment and the photocatalytic activity of plasma-functionalized Cu-TiO2 nanoparticles. J. Hazard Mater. 405, 124264 (2021).
Sultana, M. et al. Strategic development of metal doped TiO2 photocatalysts for enhanced dye degradation activity under UV–Vis irradiation: A review. Curr. Res. Green Sustain. Chem. 7, 100383 (2023).
Qureshi, A. A. et al. Strategic design of Cu/TiO2-based photoanode and rGO-Fe3O4-based counter electrode for optimized plasmonic dye-sensitized solar cells. Opt. Mater. 109, 110267 (2020).
Chakhtouna, H., Benzeid, H. & Zari, N. Recent progress on Ag/TiO2 photocatalysts: Photocatalytic and bactericidal behaviors. Environ Sci Pollut Res. 28, 44638–44666 (2021).
Lee, B. H., Park, S. & Kim, M. Reversible and cooperative photoactivation of single-atom Cu/TiO2 photocatalysts. Nat. Mater. 18, 620–626 (2019).
Al-Musawi, T. J., Yilmaz, M. & Ramirez-Coronel, A. A. Degradation of amoxicillin under a UV or visible light photocatalytic treatment process using Fe2O3/bentonite/TiO2: Performance, kinetic, degradation pathway, energy consumption, and toxicology studies. Optik. 272, 170230 (2023).
Mansourian, R. & Mousavi, S. CeO2/TiO2/SiO2 nanocatalyst for the photocatalytic and sonophotocatalytic degradation of chlorpyrifos. Can. J. Chem. Eng. 100, 451–464 (2022).
Osgouei, M. S. & Khatamian, M. Preparation of TiO2/Metalosilicate composites via acrylamide gel method and investigation of their photocatalytic performance for degradation of Brown NG. Mater. Sci. Semicond. Process. 144, 106605 (2022).
Freitas, W. et al. The role of clay mineral-derived photocatalysts in insights of remediation. Ceramics 5(4), 862–882 (2022).
Li, C. et al. A review of clay based photocatalysts: Role of phyllosilicate mineral in interfacial assembly, microstructure control and performance regulation. Chemosphere 273, 129723 (2021).
Zou, Y. et al. Application of aluminosilicate clay mineral-based composites in photocatalysis. J. Environ. Sci. 115, 190–214 (2022).
Li, Z. et al. Magnetic carbon nanotube modified S-scheme TiO2-x/g-C3N4/CNFe heterojunction coupled with peroxymonosulfate for effective visible-light-driven photodegradation via enhanced interfacial charge separation. Sep. Purif. Technol. 308, 122897 (2023).
Mapossa, A. B., Mhike, W., Adalima, J. L. & Tichapondwa, S. Removal of organic dyes from water and wastewater using magnetic ferrite-based titanium oxide and zinc oxide nanocomposites: A review. Catalysts 11(12), 1543 (2021).
Madima, N., Kefeni, K. K., Mishra, S. B., Mishra, A. K. & Kuvarega, A. T. Fabrication of magnetic recoverable Fe3O4/TiO2 heterostructure for photocatalytic degradation of rhodamine B dye. Inorg. Chem. Commun. 145, 109966 (2022).
Farooq, M. U. et al. Cu-TiO2-aluminosilicate nanocomposite as highly efficient photocatalyst for sustainable removal of cefixime antibiotic: Synthesis, optimization, and toxicity evaluation. Environ. Process. 11, 33 (2024).
Leong, C. Y., Teh, H., Ching, C. & Lee, S. Effect of synthesis methods on properties of copper oxide doped titanium dioxide photocatalyst in dye photodegradation of rhodamine B. Sci. Technol. Indones. 7, 91–97 (2022).
Ahmad, S. & Saeed, A. Synthesis of metal/silica/titania composites for the photocatalytic removal of methylene blue dye. J. Chem. 2019, 1–6. https://doi.org/10.1155/2019/9010289 (2019).
Al-Musawi, T. J., Rajiv, P., Mengelizad, N. & Mohammed, I. A. Development of sono-photocatalytic process for degradation of acid orange 7 dye by using titanium dioxide nanoparticles/graphene oxide nanocomposite as a catalyst. J. Environ. Manag. 292, 112777 (2021).
Yao, Y. R., Huang, W. Z. & Zhou, H. A novel Fe3O4@SiO2@BiOBr photocatalyst with highly active visible light photocatalytic properties. Mater. Chem. Phys. 148(3), 896–902 (2014).
Rezaei, M., Mengelizadeh, N. & Berizi, Z. Synthesis of MMT−CuFe2O4 composite as a peroxymonosulfate activator for the degradation of reactive black 5. ChemistrySelect. 8(2), 10–19 (2023).
Al-Hawary, S. I. S., Rahimpoor, R., Rahmani, A., Romero-Parra, R. M. & Ramírez-Coronel, A. A. Enhanced sonophotocatalytic degradation of acid red 14 using Fe3O4@SiO2/PAEDTC@MIL-101 (Fe) based on metal-organic framework. Catalysts 13(2), 411. https://doi.org/10.3390/catal13020411 (2023).
Rajiv, P., Mengelizadeh, N. & McKay, G. Photocatalytic degradation of ciprofloxacin with Fe2O3 nanoparticles loaded on graphitic carbon nitride: Mineralisation, degradation mechanism and toxicity assessment. Int. J. Environ. Anal. Chem. 101, 1–11 (2021).
Balarak, D., Mengelizadeh, N., Rajiv, P. & Chandrika, K. Photocatalytic degradation of amoxicillin from aqueous solutions by titanium dioxide nanoparticles loaded on graphene oxide. Environ. Sci. Pollut. Res. 28(36), 49743–49754 (2021).
Al-Musawi, T. J., Mengelizadeh, N. & Alwared, A. I. Photocatalytic degradation of ciprofloxacin by MMT/CuFe2O4 nanocomposite: Characteristics, response surface methodology, and toxicity analyses. Environ. Sci. Pollut. Res. 30, 70076–70093 (2023).
Al-Musawi, T. J., Alghamdi, M. I. & Alhachami, F. R. The application of a new recyclable photocatalyst γ-Fe2O3@SiO2@ZIF8-Ag in the photocatalytic degradation of amoxicillin in aqueous solutions. Environ. Monit. Assess. 195, 372 (2023).
Adekunle, A. S., Oyekunle, J. A. O. & Durodola, S. S. Photocatalytic degradation of dyes in wastewater using solar enhanced nickel oxide (NiO) nanocatalysts prepared by chemical methods. J Fluoresc. https://doi.org/10.1007/s10895-023-03507-6 (2023).
Halomoan, I., Yulizar, Y., Surya, R. M. & Apriandanu, D. O. B. Facile preparation of CuO-Gd2Ti2O7 using Acmella uliginosa leaf extract for photocatalytic degradation of malachite green. Mater. Res. Bull. 150, 111726 (2022).
Vinesh, V., Ashokkumar, M. & Neppolian, B. rGO supported self-assembly of 2D nano sheet of (g-C3N4) into rod-like nano structure and its application in sonophotocatalytic degradation of an antibiotic. Ultrason. Sonochem. 68, 105218 (2020).
Talukdar, K., Saravanakumar, K., Kim, Y., Fayyaz, A. & Kim, G. Rational construction of CeO2–ZrO2@MoS2 hybrid nanoflowers for enhanced sonophotocatalytic degradation of naproxen: Mechanisms and degradation pathways. Compos. Part B Eng. 215, 108780 (2021).
Yang, Y. et al. In-situ fabrication of a spherical-shaped Zn-Al hydrotalcite with BiOCl and study on its enhanced photocatalytic mechanism for perfluorooctanoic acid removal performed with a response surface methodology. J. Hazard. Mater. 399, 123070 (2020).
Ai, C., Bian, Z., Xue, Z., Zhang, W. & Zhang, Y. Assembling Bi3TaO7 nanodots on tubular carbon nitride to boost photocatalytic degradation of tetracycline under visible light irradiation. J. Alloys Compd. 1007, 176416 (2024).
Alameri, A. A., Alfilh, R. H. C., Awad, S. A. et al. Ciprofloxacin adsorption using magnetic and ZnO nanoparticles supported activated carbon derived from Azolla filiculoides biomass. Biomass Conv. Bioref. 14, 27001–27014. https://doi.org/10.1007/s13399-022-03372-6 (2024).
Liyanaarachchi, H., Thambiliyagodage, C., Liyanaarachchi, C. & Samarakoon, U. Efficient photocatalysis of Cu doped TiO2/g-C3N4 for the photodegradation of methylene blue. Arab. J. Chem. 16(6), 104749 (2023).
Landge, V. K. et al. Solar-energy-driven Cu-ZnO/TiO2 nanocomposite photocatalyst for the rapid degradation of Congo Red azo dye. Catalysts 12(6), 605 (2022).
Dlamini, M. C., Dlamini, M. L., Mente, P. & Tlhaole, B. E. Photocatalytic abatement of phenol on amorphous TiO2-BiOBr-bentonite heterostructures under visible light irradiation. J. Ind. Eng. Chem. 111, 419–436 (2022).
Acknowledgements
The authors extend their gratitude to Zahedan University of Medical Sciences for funding this research (IR.ZAUMS.REC.1402.314).
Author information
Authors and Affiliations
Contributions
FM: analysis of colored wastewater, NA and NM: Writing–original draft, Visualization. DB: Writing—review & editing, Visualization. MA: analysis of catalyst characteristics.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
No artifacts or human participants are involved in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mollaie, F., Afroomand, M., Ahmadi, N. et al. Optimization of photodegradation of acid blue 1 dye on aluminosilicate supported Cu doped TiO2 magnetic nanocatalyst using response surface methodology. Sci Rep 15, 5550 (2025). https://doi.org/10.1038/s41598-025-89968-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89968-0
Keywords
This article is cited by
-
Green synthesis and comparative analysis of zinc and cobalt-based oxide and ferrite nanocatalysts for efficient Actacryl Red GRL dye removal
Chemical Papers (2026)
-
Sonophotocatalytic efficiency of nickel and copper supported ferrite nanoparticles doped MWCNTs nanocatalyst as a novel catalyst for amoxicillin degradation
Scientific Reports (2025)
-
Chemical degradation of remazol dye by using Tb-doped TiO2 catalyst and Photo-Fenton’s process under UV light
Scientific Reports (2025)
-
Green synthesis and characterization of ZnO-NPs and Ag/ZnO-NPs using Mariposa Christia vespertilionis for the photodegradation of pesticides: endosulfan and Aldrin
Emergent Materials (2025)
-
Synergistic sonophotocatalytic degradation of Diazinon with cobalt ferrite on carbon nanotubes: investigation of toxicity, biodegradation, recovery, kinetics, and energy consumption
Chemical Papers (2025)