Abstract
Cold atmospheric plasma is emerging as a non-thermal and non-invasive tool for influencing microbial metabolism by modifying bioreactor conditions. This paper validates the effect of a cold atmospheric helium plasma jet on recombinant thaumatin bioproduction by genetically modified Komagataella phaffii. The yeast produced recombinant thaumatin with methanol serving as the carbon and energy source. The cell-free production medium was treated with a helium plasma jet once for 2, 4, 6, and 8 min before inoculation, while the control bioprocess remained untreated. The same treatments were repeated daily on the cell cultures. The results showed no significant effect of the treatment time (2–8 min) on thaumatin production. However, the application of plasma treatments, regardless of the duration, resulted in significant enhancement of 13.3% in protein production compared to the control. No significant difference was observed between residual methanol concentrations with and without plasma treatments, leading to the conclusion that the yield of thaumatin produced per quantity of methanol consumed increased due to plasma treatments. This study thus confirms the hypothesis that cold atmospheric plasma enhances protein production and highlights the probable role of plasma-generated reactive species in this enhancement.
Similar content being viewed by others
Introduction
Conventional approaches to enhance bioprocesses typically aim to manipulate the microbial environment by addressing process parameters like temperature, pH, aeration rate, gas composition and feeding. Complementary approaches at cell level include genetically engineering microbial hosts via molecular techniques such as codon optimisation, gene copy number amplification, or control of the promoter or terminator genes flanking the gene of interest1,2. Recently, there has been a growing interest in cost-effective and versatile technologies that augment the effects of existing optimisation approaches to further enhance the bioprocess yield or productivity at different stages of the bioprocess. The principle behind these technologies is based on stressing the microorganisms mildly, such that a stimulatory response is triggered that makes the cells more efficient3. However, many of these technologies result in excessive chemical waste or are invasive, with the stimulatory effects resulting from alterations in the physicochemical state of the cell membrane, leading to increased molecular leakage or transport across it4. Moreover, unwanted thermal effects continue to pose a significant challenge to the practise of sub-lethal exposure. A more recent development in this line is the application of a well-established non-thermal technology called cold atmospheric plasma5. Interests in the use of cold atmospheric plasma in biomedical applications began in the mid-1900s6. Fundamentally, plasma comprises a dynamic chemical mixture of high-energy atoms, molecules, positive and negative ions, electrons, free radicals, UV photons, electromagnetic and electric fields and reactive oxygen and nitrogen species (ROS and RNS). The non-thermal nature of cold plasma is attributed to the absence of a thermodynamic equilibrium between high-energy electrons and reactive species. This allowed for the emergence of cold plasma as a non-invasive modality that can influence the microbial metabolism for enhanced bioprocess yield or productivity7,8. The application of cold atmospheric plasma can either be direct or indirect. Direct application involves interaction between the plasma-generated reactive species and the biological material. In contrast, indirect application involves plasma interaction with the culture medium or the solution, where the reactive species generated by plasma are delivered to the target biological specimen through this medium9. The source of generation of plasma-reactive species can either be plasma-air interaction that commonly results in hydroxyl radicals and nitric oxides, or plasma-liquid interaction resulting in nitrates, nitrites and hydrogen peroxide10,11.
Till date, only two other studies have explored the potential of cold atmospheric plasma to enhance recombinant protein bioproduction in K. phaffii. K. phaffii (previously known as Pichia pastoris) is a methylotrophic yeast that possesses a tightly regulated methanol-induced promoter called pAOX1 (alcohol oxidase 1), making the yeast favourable for high level of protein expression under the control of methanol12,13. Farasat et al. (2018) demonstrated that repeated treatments with a cold atmospheric helium plasma jet on K. phaffii X-33, which was genetically modified to produce recombinant phytase under the control of the AOX1 promoter, led to a significant increase in the final concentrations of phytase with increasing treatment duration of up to 4 min14. Another study, by Kabarkouhi et al. (2023), reinforced this finding by plasma-treating the PichiaPink™ expression system that is based on the same yeast and was genetically modified to produce eGFP regulated by the same AOX1 promotor15. Consistent with the findings of Farasat et al. (2018), these authors recorded even up to 36% increase in the final protein production with a single treatment on the cell culture. The mode of action of plasma in microbial enhancement or inactivation has long been proposed to be the plasma-generated reactive species, which could be long-living (O3, H2O2, ∙NO3−) or short-living (∙O2−, ∙OH, HO2−, 1O2, O1, ∙NO, ∙NO2−)4,16,17. Both the previous studies have linked the plasma-induced increase in recombinant protein production with oxidative stress from plasma-generated reactive species, specifically, hydrogen peroxide14,15. Hydrogen peroxide is a reactive oxygen species produced in water through the hydroxyl radicals generated at the plasma-water interface. It is considered to be a key-component in the mechanism of action of plasma because of its ability to diffuse through cell membranes and its role as a signalling molecule involved in redox regulation at the transcription and translation level18,19,20. The plasma treatments in the studies that led to higher recombinant protein concentrations, also resulted in increased expression of stress response genes participating in unfolded protein response and endoplasmic reticulum stress, which are commonly triggered stress responses due to a change in the physiological concentrations of hydrogen peroxide9,21,22. This was found to have possibly contributed to higher protein secretion levels. In both the previous studies, the AOX1 promoter is used as a common promoter regulating the levels of the respective recombinant proteins. Thus, it is important to consider the possibility of a specific effect of cold atmospheric plasma on the AOX1 promoter, besides a general effect of enhanced protein secretion. Although there is no clear evidence suggesting that cold plasma directly stimulates the AOX1 promoter, several studies support that the plasma-derived reactive species act as signalling mediators inducing activation, upregulation and nuclear translocation of transcription factors, e.g., NRF2 (antioxidant stress response) and Mxr1 (activating AOX expression)23,24,25. Additionally, there is also a possibility that these reactive species at certain concentrations lead to epigenetic modifications like DNA methylation or histone protein modification, thereby influencing the binding of transcription factors to the promoter26. This could provide a possible molecular-level explanation for higher levels of recombinant protein observed upon cold atmospheric plasma jet treatments.
Building on the findings of these two previous studies, the goal of the present study was to validate the effects of mild exposure to a cold atmospheric helium plasma jet on a recombinant protein production by K. phaffii. Moreover, the secondary aim was to validate the presence of hydrogen peroxide in the plasma-activated medium as one of the key factors to cause potential effects. This was done to test the previously established correlation of hydrogen peroxide with plasma-induced oxidative stress and associated higher protein levels. To achieve this, a case study on recombinant thaumatin protein production in genetically modified K. phaffii was selected. Thaumatin (E957) is a sweet protein of 22 kDA that is used as flavour enhancer13. It has a sweetness potency of more than 100,000 times that of sucrose on a molar basis and is recognised as a safe ingredient by EFSA since 202327. As thaumatin is originally extracted from the arils of tropically grown Katemfe fruit (Thaumatococcus daniellii), its availability is limited28. This study adopted the two-phase bioprocess for recombinant thaumatin bioproduction in genetically modified K. phaffii as benchmarked by Joseph et al. (2023b), which yielded up to 63 mg/L of the protein in shake flasks. To study the effects of cold atmospheric plasma on the bioproduction of thaumatin, treatments were conducted during the production phase of the bioprocess and the key process parameters were measured daily. To our knowledge, this is the first study that has not only quantified secreted recombinant protein levels but also the substrate (methanol) levels in response to cold atmospheric plasma jet treatments. Unlike previous studies that have focused only on plasma-enhanced protein levels and the potential stress responses, this research also provides crucial insight into the relationship between recombinant thaumatin levels and methanol consumption behaviour of K. phaffii.
Materials and methodology
The effect of cold atmospheric helium plasma jet treatments was studied on the production of recombinant thaumatin by Komagataella phaffii, while also monitoring the main carbon substrate (methanol) and the viable cell population. The experimental design consisted of four different durations of the daily plasma treatments and an untreated control.
K. phaffii cultivation and Recombinant thaumatin production
The mut+ Komagataella phaffii GS115 strain was genetically modified with a pre-thaumatin-II-pro gene derived from Thaumatococcus daniellii, inserted downstream of the AOX1 gene. The expression vector pPICZalphaB was selected to facilitate the secretion of thaumatin II29. The genetic engineering of the strain was performed by VIB Protein Core (Zwijnaarde, Belgium). Modified cells were stored at −80 °C in glycerol stock comprising 5% (w/v) Yeast Peptone Dextrose (YPD, Carl Roth, Karlsruhe, Germany) broth and 25% (w/v) glycerol (99+ %, Chem Lab, Zedelgem, Belgium). To prepare the preculture, 20 mL YPD medium containing 1% w/v yeast extract (Thermo Fisher Scientific, France), 2% w/v peptone (Thermo Fisher Scientific, USA) and 2% w/v anhydrous D(+)-glucose (Thermo Fisher Scientific, Germany) supplemented with 20 µL of 100 mg/L zeocin (Thermo Fisher Scientific, USA) was inoculated with three colonies from a zeocin-containing YPD agar streak plate and incubated for 67 h on a rotary shaker at 220 rpm and 30 °C. For the inoculation of the growth phase, 1 mL of the preculture containing approximately 20 ln (CFU/mL) was added to 40 mL buffered glycerol complex medium (BMGY) in a 100 mL baffled shake flask. BMGY contained 1% w/v glycerol, 1% w/v yeast extract, 2% w/v peptone, 1.34% w/v yeast nitrogen base (Carl Roth GmbH, Germany), 10% w/v 0.1 M potassium phosphate buffer (PBS) at pH 6 and 0.02% w/v biotin (≥ 99%, Sigma-Aldrich Chemie GmbH, Germany). This flask was incubated at 30 °C and 220 rpm on a rotary shaker. After 24 h, the cells were harvested at their maximum cell density (~ 21 ln CFU∙mL∙−1) by centrifugation at 750 g at 20 °C for 15 min and resuspended in buffered methanol complex medium (BMMY) to start the protein production phase. BMMY (pH 6) contained 1% (w/v) methanol (≥ 99.5% VWR, France) as carbon substrate, 1% w/v yeast extract, 2% w/v peptone, 1.34% w/v yeast nitrogen base, 10% w/v 0.1 M potassium phosphate buffer at pH 6 and 0.02% w/v biotin. The entire bioprocess lasted for 102 h, i.e., a 24 h growth phase followed by a 78 h production phase (Fig. 1). During the production phase, cells were induced six times, i.e., at 31, 48, 55, 72, 79 and 96 h of the bioprocess, to express the thaumatin II gene, by feeding 0.5 mL of concentrated methanol (≥ 99.5%). All experiments were conducted at least in quadruplicate in 100 mL baffled shake flasks. Samples for thaumatin and methanol quantification were collected twice a day, starting from the second day of the production phase at 50, 54, 74, 78, 98 and 102 h of the bioprocess.
Scheme of plasma treatments (purple jet), methanol inductions (orange arrow) and sampling (green arrow) during 102 h of the bioproduction process. Transfer of cells from preculture flask to growth phase flask, and from growth phase flask to production phase flask are indicated by black arrows. The time stamps for each of the three activities are marked on the time scale as bars of the respective colours.
Cold atmospheric helium plasma jet treatments on K. phaffii bioprocess
In this study, an in-house plasma generator was employed, comprising a power supply, frequency generator, oscilloscope and plasma jet assembly (Fig. 2). An inverter was used to convert direct current with input power of 17 to19 W and voltage of 28 to 30 V, into high voltage alternating current (5 kV peak to peak). The frequency oscillated between 14 and 15 kHz to ensure the stable emergence of a cold plasma jet. Helium gas was used as the carrier gas at flow rate of 10 standard litres per minute to avoid interference of surrounding air into the helium stream30. The plasma propelled through a Pyrex tube with an inner diameter of 5 mm. Embedded within this tube was a tungsten electrode with diameter of 0.2–0.3 mm, with the tip of the electrode pointing towards the opening of the tube. This tip was surrounded by an aluminium electrode with a height of 10 mm that covered the outside of the Pyrex tube. The distance between the tube end and the sample surface in the flask was 50 mm, equivalent to the length of the plasma jet stream.
The scheme for cold atmospheric plasma treatments is illustrated in Fig. 1. Shortly before the treatments, the cell-free medium or cell culture was transferred into a sterile 50 mL wide-neck conical flask containing a sterile magnetic stirrer. The plasma treatments of 2, 4, 6, or 8 min were applied to the production medium, BMMY, before inoculation as per the methodology of Farasat et al. (2018). After the plasma treatments, cells grown on glycerol medium during the growth phase were transferred to the treated production medium and incubated at 30 °C and 220 rpm. Additional plasma treatments of the same durations were performed every 24 h (48, 72 and 96 h of bioprocess) on the cell cultures. The control bioprocesses remained untreated for comparison.
Metabolite quantification
Viable cells were quantified by plating three 20 µL drops of serially diluted cell culture samples on YPD agar plates and incubating it for 48 h at 30 °C. The colonies were later counted, and cell densities were expressed in ln (CFU/mL).
Quantification of thaumatin in samples started at 50 h of the bioprocess, which is 26 h into the production phase (Fig. 3). In the first hours of the production phase, thaumatin levels were below the detection limit of the HPLC method. For thaumatin quantification, the culture medium was filtered through 0.2 μm filters and 2 mL aliquots were stored at 4 °C. Thaumatin levels were measured using an Agilent LC 1260 Infinity II system equipped with a diode-array detector (G7117C). A Zorbax 300SB-C8 column, (4.6 × 150 mm, 5 μm particle size, Agilent Technologies, USA) was employed for separation and was preceded by a Zorbax SB 300 C8 guard cartridge (4.6 × 12.5 mm, 5 µm, Agilent Technologies, USA), with protein detection via absorbance at 196 nm. The aqueous mobile phase consisted of Milli-Q water (18.2 MΩ cm at 25 °C, Millipore), HPLC grade acetonitrile (≥ 99.9%, Sigma-Aldrich, Germany), and trifluoroacetic acid (TFA) (99.9%, Fisher Scientific, Belgium) in the ratio 950:50:0.5 (vol/vol/vol). The organic mobile phase was made with acetonitrile, Milli-Q water and TFA in the ratio 950:50:0.25 (vol/vol/vol). Each sample underwent analysis for 30 min with a linear gradient of the organic solvent of 25–30% from 0 to 5 min, 30–37% from 5 to 15 min, 37–40% from 15 to 31 min, 40 − 25% from 31 to 32 min followed by 5 min post time. Thaumatin calibration curve was obtained by analysing thaumatin standards of a known range of concentrations (9.4, 18.8, 37.5, 75.0, 112.5, 150.0 and 300.0 mg/L) prepared by diluting commercial thaumatin (Sigma-Aldrich, USA) with sterile Milli-Q water. Each sample was injected at least twice, and data were processed using ChemLab software31.
To understand the substrate consumption behaviour, the methanol concentration was assessed at the same time points as for thaumatin concentrations (Fig. 4). Methanol levels were determined using gas chromatography coupled with a flame ionization detector (8860, Agilent). For sample preparation, 0.772 g of potassium carbonate was added to 700 µL of filtered samples in a sterile 2 mL centrifuge tube. The mixture was agitated for 2 min on a shaker (ZX4 Advanced IR Vortex Mixer, VELP Scientifica Srl) at 800 rpm and room temperature. Subsequently, 700 µL of filtered GC ultra-grade ethyl acetate (≥ 99.9%, Carl Roth GmbH, Germany) was added, followed by mixing for 15 min and centrifugation at 4000 rpm and 20 °C for 2 min. 200 µL of the upper phase was then transferred to a sample vail with insert. Each sample underwent analysis at least three times in the GC instrument. The detector and injector temperatures were set at 300 °C and 225 °C, respectively. Helium served as the carrier gas at a flow rate of 1.5 mL/min, while hydrogen and air flows were set at 40 mL/min and 300 mL/min, respectively. The separation column used was an HP-5 nonpolar GC capillary column (30 m × 320 μm i.d., 0.25 μm film thickness). 1 µL sample was injected with a split ratio of 100:1. Calibration standards were prepared using 100 g/L methanol (≥ 99.5%, VWR, France) in Milli-Q water that was two-fold diluted nine times to obtain a calibration curve of ten concentrations of methanol standards (100.0, 50.0, 25.0 12.5, 6.3, 3.1, 1.6, 0.8, 0.4, and 0.2 g/L). Analysis of each injection was conducted using OpenLab CDS software (Agilent Technologies)32.
Hydrogen peroxide quantification
Hydrogen peroxide was measured using the titanium (IV) oxysulfate (TiOSO4) colorimetric method as reported by Govaert et al. (2020). 100 µL Milli-Q water, BMMY and PBS buffer were treated with plasma for 8 min in a 96-well plate. The distance between plasma jet tube end and the water surface was initially fixed at 50 mm in accordance with treatments during bioproduction. This distance was later reduced to 20 mm as H2O2 concentration were undetectable with a 50 mm distance between plasma source and liquid surface. To quantify H2O2, 10 µL TiOSO4 (27–31% H2SO4basis, Merck, France) was added to the plasma-activated water and the multiwell plate was stored covered in the dark for 10 min. The absorbance of plasma-activated water was recorded at 405 nm in a spectrophotometer (VersaMax tuneable microtiter plate reader, Molecular Devices, Wokingham, UK) and the baseline was subtracted from all the values33. To obtain the calibration curve, hydrogen peroxide solution 35% w/v was two-fold serially diluted to obtain a series of sequentially decreasing concentrations. The corresponding absorbance at 405 nm was plotted against the concentrations to obtain the calibration curve. The detection limit was established at around 39.24 µM.
Statistical analysis
Analysis of variance (ANOVA) was performed using the anovan-function of the Statistics and Machine Learning Toolbox of MATLAB R2023b.
Results
The efficacy of cold atmospheric helium plasma jet to enhance recombinant protein production was tested based on treatments of 2, 4, 6 and 8 min. The cell cultures received daily cold plasma treatments in a wide-neck conical flask during the production phase, while the non-treated cell culture was left untreated except for sample extraction. The effects of ACP treatments on the bioprocess were measured through concentrations of viable cells, thaumatin and methanol.
Cell viability
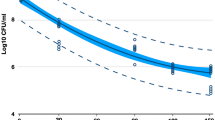
Figure 3 shows the cell density measured throughout the experiments. The initial cell density measurement was taken immediately after inoculation in the growth medium (BMGY). Subsequent measurements were made after the shift to the production phase in BMMY. These measurements were taken 2 h after the first induction of each day. The results showed that the duration of treatment had no substantial impact on cell density, nor the cell density changed over the course of production phase despite repeated plasma treatments. During the production phase, all treated samples maintained a consistent cell density of 20.68 ± 0.38 ln (CFU/mL) while the non-treated cells plateaued at 20.89 ± 0.19 ln (CFU/mL). As such, no significant effect of the plasma treatments on the viability of the cells was observed (p < 0.05).
Cell density as function of time for the untreated control (+) and treatments of 2 ( ), 4 (
), 4 ( ), 6 (
), 6 ( ) and 8 (
) and 8 ( ) min. The cell density at 0 h represents viable cells immediately after inoculating the glycerol medium (BMGY) for biomass growth. Cell densities at 24, 50, 74 and 98 h represent the viables cells in the methanol medium (BMMY) during the production phase. Purple arrows at 24, 48, 72 and 96 h represent plasma treatments.
) min. The cell density at 0 h represents viable cells immediately after inoculating the glycerol medium (BMGY) for biomass growth. Cell densities at 24, 50, 74 and 98 h represent the viables cells in the methanol medium (BMMY) during the production phase. Purple arrows at 24, 48, 72 and 96 h represent plasma treatments.
Recombinant thaumatin
Quantification of recombinant thaumatin began at the 50 h of the bioprocess on the second day of the production phase, which was two hours after the second treatment of the experiment. The measured recombinant thaumatin concentrations are illustrated in Fig. 4. During the second and third day of the production phase, there were no noticeable differences in thaumatin concentration between any of the treatment times. On the contrary, all plasma treated samples of the fourth (last) day of the production phase had a clearly increased thaumatin concentration compared to the control. There was, however, no difference noticed among the thaumatin concentrations for different treatment times.
To validate this observation, an ANOVA was performed to compare for each day all control samples on the one hand and all treatment samples on the other hand. By pooling all data of controls versus treatments, a large number of datapoints was obtained in each group (20 to 38 datapoints respectively) that enabled reliable statistical hypothesis testing. The results of the statistical analysis of thaumatin data are reported in Table 1. Although the mean thaumatin concentration on the second day of the production phase is slightly higher for treated cells compared to untreated cells, the difference is not significant. On the third and fourth day, on the other hand, the mean thaumatin concentration in plasma treated samples was higher than the control bioprocesses by respectively 8.3 and 13.3%, and the difference between the means was statistically significant (p < 0.05). Overall, a significant increase in the thaumatin concentration was attained as a consequence of the cold atmospheric helium plasma jet.
Residual methanol
The concentration of methanol that is present in the medium throughout the production phase is displayed in Fig. 5. Similar to the effect on thaumatin, no relationship is observed between methanol concentrations and plasma exposure time. Neither can a clear distinction be noted between the bioprocesses with and without plasma treatments. To evaluate whether there was a statistically significant effect of plasma treatments on the methanol concentration, daily averages of the methanol concentration in treated samples at all treatment durations were compared with the daily averages of the control samples in Table 2. This analysis demonstrates that, although the methanol concentration in some plasma treated samples is consistently slightly lower than in the control samples, the observed difference was not significant (p > 0.05).
Furthermore, an increasing trend in the residual methanol concentration in the medium was noticed with and without treatments during the production phase, potentially indicating a mismatch between the methanol consumption rate of the cells and the methanol feed rate.
Hydrogen peroxide
Precipitation occurred when adding titanium oxysulfate to the plasma-treated PBS buffer and BMMY medium in a 96-well plate, which inhibited the absorbance measurements for the buffer and the medium. As such, the hydrogen peroxide formation due to plasma treatments could only be assessed in Milli-Q water. This resulted in H2O2 formation equivalent to 630 µM at a treatment distance of 20 mm compared to undetectable levels with a 50 mm distance. The latter distance is consistent with the one maintained during cell culture treatments.
Discussion
The results obtained in this research are largely in line with the findings of the preceding studies where K. phaffii was treated by cold atmospheric helium plasma jet but the specific effect on secreted protein levels was found to be different. With respect to cell growth, treated cells showed similar cell viability as non-treated cells, which is consistent with Kabarkouhi et al. (2023) and Farasat et al. (2018) who also did not report any significant changes even after treatments of 4 min. Although there were some fluctuations, both studies observed no significant changes in the cell density or viability for treatments up to the longest tested duration of 4 min.
With respect to the effect of plasma treatment on protein concentration, in agreement with these studies, the current study also found that the implementation of a cold atmospheric helium plasma jet significantly increased the recombinant protein levels produced by K. phaffii. Specifically, an increase of 13.3% in the secreted protein levels was noted. However, there did not appear to be any noticeable difference between the treatment times of 2, 4, 6 and 8 min. This is in contrast with both the previous studies, in which a consistent increase of the recombinant protein production was seen with increasing plasma treatment time. Moreover, Kabarkouhi et al. (2023) found an increase in the total protein production by 36% for a single 4 min treatment and Farasat et al. (2018) even found a 96% increase in recombinant phytase production due to daily 4 min treatments, confirming the benefit of treatment repetitions. In line with this, the current research found 81% and 63% lower increase in the recombinant protein levels in comparison to Farasat et al. (2018) and Kabarkouhi et al. (2023), respectively.
This could be attributed to the difference in the distance between the plasma source and the liquid surface, which was maintained at 50 mm in the current study as opposed to 10 mm in the previous studies. This is because, with a larger gap, the plasma-generated reactive species in air must travel a longer distance to reach and interact with the medium, reducing their chances of transforming unstable reactive species like hydroxyl radicals into stable reactive species such as hydrogen peroxide. While a longer gap has been shown to be less inhibitory on yeast cells, it also results in lower concentrations of useful reactive species in the liquid medium30. This is supported by the hydrogen peroxide tests performed in 96-well plates on Milli-Q water. These tests yielded H2O2 levels below detection limit (< 39.42 µM) at 50 mm, which was the distance maintained in this study, compared to 630 µM H2O2 at a reduced distance of 20 mm between the plasma source and the liquid surface. This could explain the relatively lower increments observed in the recombinant thaumatin protein levels in this study as both the earlier studies on cold atmospheric plasma application on K. phaffii bioprocesses have linked the increase in protein production to the generation of hydrogen peroxide. In the study of Kabarkouhi et al. (2023), hydrogen peroxide levels were found to be higher than the concentrations obtained in the current study, although consistently lower (~ 80 µM) in yeast-peptone medium than in water (~ 120 µM ) treated with plasma for 8 min.
This could mean that in the current study, the concentration of hydrogen peroxide in plasma-treated production medium, BMMY, might be even lower due to buffering capacity of the medium. Furthermore, the presence of secreted metabolites (e.g., proteins, vitamins and amino acids) and medium components (e.g., glycerol, methanol, buffer salts, yeast extract and peptone) likely affected the stability and generation of hydrogen peroxide through radical chain reaction such as \(\:\cdot\:OH+\:\cdot\:OH+{O}_{2}\to\:{H}_{2}{O}_{2}+{O}_{2}\)34. Given these factors, the overall availability of hydrogen peroxide in the BMMY may be reduced compared to simpler aqueous systems like yeast-peptone medium and water. Therefore, it cannot be confirmed that hydrogen peroxide is the major contributor to the observed effect in the current study.
Figure 5shows methanol concentration in the medium gradually increasing towards about 50 g/L at the end of the experiment regardless of the treatment durations. This is potentially due to a higher methanol feeding rate compared to the methanol consumption rate of the cells. Additionally, methanol metabolism by alcohol oxidase enzyme creates a demand for oxygen. On the other hand, as the oxygen transfer rate during the shake-flask experiments in the study was not controlled, it may have led to partial oxygen limitation, thereby slowing down methanol metabolism and resulting in high methanol levels in the medium35,36,37. Previous research has demonstrated that the optimum methanol concentration for recombinant thaumatin production by K. phaffii lies below 5 g/L38. Recombinant protein production rate decreases with increasing methanol concentrations above this level due to cell death from accumulation of formaldehyde and hydrogen peroxide39. Moreover, it could also be that due to the accumulation of methanol in the culture medium, the cells were forced to redirect metabolism towards eliminating the methanol-induced stress, thereby interfering with the dosage-dependent hormesis effect of the cold atmospheric helium plasma treatments. These considerations would mean that lower methanol concentrations would give rise to an increased production of thaumatin. As per Fig. 4, recombinant thaumatin production increased after exposure to plasma, but no effect of the plasma treatment was observed on the methanol concentrations (Fig. 4). Thus, the increase in thaumatin production cannot be linked to a change in methanol consumption behaviour. Instead, it leads to a conclusion that the increase in the recombinant thaumatin levels is solely an effect of cold atmospheric plasma jet treatments. However, the exact mechanism for amplification of protein production levels could not be unravelled based on the available data. The current study was able to prove that sublethal plasma treatments can indeed increase the levels of secreted recombinant protein but may be highly dependent on additional factors, such as hydrogen peroxide and methanol concentration, during the bioproduction. For future research aiming to enhance recombinant protein expression using cold atmospheric plasma, it should be considered that the overall effect (for e.g., increased protein productivity) may also arise from changes in transcription factor activity, promoter binding, or protein synthesis, secretion and folding. Therefore, analysis should shift from a broader assessment of protein concentrations to exploring promoter regulation, translation efficiency and ribosomal recycling, that can aid in precisely determining the molecular mechanisms responsible for the enhanced effects.
Conclusion
The goal of this research was to validate the effect of cold atmospheric helium plasma jet on enhancing the recombinant protein bioproduction by K. phaffii, as demonstrated in previous studies. In agreement with earlier findings, daily plasma treatments of up to 8 min had no noticeable effect on the cell density. The exposure to plasma was found to significantly increase the thaumatin concentration by 13.3% by the end of the experiment. This increase was found to be relatively consistent independent of the treatment duration.
Although previous research has linked this enhancement to plasma-generated hydrogen peroxide, this link could not be confirmed by the current research as hydrogen peroxide levels remained below detection limit. Nonetheless, recombinant thaumatin production was indeed enhanced by daily cold atmospheric helium plasma jet treatments. On the other hand, methanol concentrations in the culture medium remained unaffected by the plasma treatments. As such, it is hypothesized that the increased thaumatin production is due to plasma-generated reactive oxygen species. However, the precise mechanism that causes it remains undetermined in this study. Therefore, future research should investigate the role of key plasma-generated reactive species, such as, hydrogen peroxide by controlled supplementation in the fermentation medium.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Zhao, M., Ma, J., Zhang, L. & Qi, H. Engineering strategies for enhanced heterologous protein production by Saccharomyces cerevisiae. Microb. Cell. Factories. 23, 32 (2024).
Joseph, J. A., Akkermans, S., Cornillie, E., Deberlanger, J. & Van Impe, J. F. M. Optimal culture medium selection and supplementation for Recombinant thaumatin II production by Komagataella phaffii. Food Bioprod. Process. 139, 190–203 (2023a).
Dong, X. Y., Yuan, X. & Wang, R. J. Interaction of air cold plasma with Saccharomyces cerevisiae in the multi-scale microenvironment for improved ethanol yield. Bioresour Technol. 323, 124621 (2021).
Dong, X. Y. Enhanced Ethanol Production of Saccharomyces cerevisiae Induced by Cold Plasma at Atmospheric Air Pressure. in Fuel Ethanol Production from Sugarcane (eds. Peixoto Basso, T. & Carlos Basso, L.)IntechOpen, (2019). https://doi.org/10.5772/intechopen.78019
Fiebrandt, M., Lackmann, J. & Stapelmann, K. From patent to product? 50 years of low-pressure plasma sterilization. Plasma Process. Polym. 15, 1800139 (2018).
Laroussi, M. Sterilization of contaminated matter with an atmospheric pressure plasma. IEEE Trans. Plasma Sci. 24, 1188–1191 (1996).
Misra, N. N., Yepez, X., Xu, L. & Keener, K. In-package cold plasma technologies. J. Food Eng. 244, 21–31 (2019).
Chaudhary, K., Imam, A. M., Rizvi, S. Z. H. & Ali, J. Plasma Kinetic Theory. in Kinetic Theory (eds. Kyzas, G. Z. & Mitropoulos, A. C.)InTech, (2018). https://doi.org/10.5772/intechopen.70843
Azzariti, A. et al. Plasma-activated medium triggers cell death and the presentation of immune activating danger signals in melanoma and pancreatic cancer cells. Sci. Rep. 9, 4099 (2019).
Von Woedtke, T., Schmidt, A., Bekeschus, S., Wende, K. & Weltmann, K. D. Plasma medicine: A field of applied redox biology. Vivo 33, 1011–1026 (2019).
Takamatsu, T. et al. Investigation of reactive species using various gas plasmas. RSC Adv. 4, 39901–39905 (2014).
Cregg, J. M., Barringer, K. J. & Hessler, A. Y. Pichia pastoris as a host system for transformations. Mol. Cell. Biol. 5, 3376–3385 (1985).
Joseph, J. A., Akkermans, S., Nimmegeers, P. & Van Impe, J. F. M. Bioproduction of the Recombinant sweet protein thaumatin: current state of the Art and perspectives. Front. Microbiol. 10, 695 (2019).
Farasat, M., Arjmand, S., Ranaei Siadat, S. O., Sefidbakht, Y. & Ghomi, H. The effect of non-thermal atmospheric plasma on the production and activity of Recombinant phytase enzyme. Sci. Rep. 8, 16647 (2018).
Kabarkouhi, Z., Arjmand, S., Siadat, R., Shokri, B. & S. O. & Cold atmospheric plasma treatment enhances Recombinant model protein production in yeast Pichia pastoris. Sci. Rep. 13, 6797 (2023).
Kalghatgi, S. et al. Effects of Non-Thermal plasma on mammalian cells. PLoS ONE. 6, e16270 (2011).
López, M. et al. A review on Non-thermal atmospheric plasma for food preservation: mode of action, determinants of effectiveness, and applications. Front. Microbiol. 10, 622 (2019).
Liu, J. et al. Direct synthesis of hydrogen peroxide from plasma-water interactions. Sci. Rep. 6, 38454 (2016).
Gorbanev, Y., O’Connell, D. & Chechik, V. Non-Thermal plasma in contact with water: the origin of species. Chem. – Eur. J. 22, 3496–3505 (2016).
Picazo, C. & Molin, M. Impact of hydrogen peroxide on protein synthesis in yeast. Antioxidants 10, 952 (2021).
Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: oxidative eustress. Redox Biol. 11, 613–619 (2017).
Winterbourn, C. Biological production, detection, and fate of hydrogen peroxide. Antioxid. Redox. Signal. 29, 541–551 (2018).
Yun, J. H., Yang, Y. H., Han, C. H., Kang, S. U. & Kim, C. H. Non-thermal atmospheric pressure plasma induces selective cancer cell apoptosis by modulating redox homeostasis. Cell. Commun. Signal. 22, 452 (2024).
Chang, C. H., Hsiung, H. A., Hong, K. L. & Huang, C. T. Enhancing the efficiency of the Pichia pastoris AOX1 promoter via the synthetic positive feedback circuit of transcription factor Mxr1. BMC Biotechnol. 18, 81 (2018).
Lin-Cereghino, G. P. et al. Mxr1p, a key regulator of the methanol utilization pathway and peroxisomal genes in Pichia pastoris. Mol. Cell. Biol. 26, 883–897 (2006).
Braný, D. et al. Effect of cold atmospheric plasma on epigenetic changes, DNA damage, and possibilities for its use in synergistic Cancer therapy. Int. J. Mol. Sci. 22, 12252 (2021).
EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). et al. Assessment of the feed additive consisting of thaumatin for all animal species for the renewal of its authorisation (ADISSEO France S.A.S). EFSA J. 21, (2023).
Kant, R. Sweet proteins – Potential replacement for artificial low calorie sweeteners. Nutr. J. 4, 5 (2005).
Joseph, J. A., Akkermans, S. & Van Impe, J. F. M. Effects of temperature and pH on Recombinant thaumatin II production by Pichia pastoris. Foods 11, 1438 (2022a).
Ottenheim, C., Nawrath, M. & Wu, J. C. Microbial mutagenesis by atmospheric and room-temperature plasma (ARTP): the latest development. Bioresour Bioprocess. 5, 12 (2018).
Joseph, J. A., Akkermans, S., Tsakali, E. & Van Impe, J. F. M. Determination of Recombinant thaumatin II secreted by Pichia pastoris using reversed-phase high-performance liquid chromatography. Food Bioprod. Process. 137, 93–98 (2023b).
Joseph, J. A., Akkermans, S. & Van Impe, J. F. M. Processing method for the quantification of methanol and ethanol from bioreactor samples using gas Chromatography–Flame ionization detection. ACS Omega. 7, 24121–24133 (2022b).
Govaert, M., Smet, C., Walsh, L., Van Impe, J. F. M. & J. & Influence of plasma characteristics on the inactivation mechanism of cold atmospheric plasma (CAP) for Listeria monocytogenes and Salmonella Typhimurium biofilms. Appl. Sci. 10, 3198 (2020).
Taheri, D. et al. Realtime RONS monitoring of cold plasma-activated aqueous media based on time-resolved phosphorescence spectroscopy. Sci. Rep. 14, 22403 (2024).
Wollborn, D. et al. Predicting high Recombinant protein producer strains of Pichia pastoris MutS using the oxygen transfer rate as an indicator of metabolic burden. Sci. Rep. 12, 11225 (2022).
Lee, C. Y., Lee, S. J., Jung, K. H., Katoh, S. & Lee, E. K. High dissolved oxygen tension enhances heterologous protein expression by Recombinant Pichia pastoris. Process. Biochem. 38, 1147–1154 (2003).
Liang, J. & Yuan, J. Oxygen transfer model in Recombinant Pichia pastoris and its application in biomass Estimation. Biotechnol. Lett. 29, 27–35 (2006).
Parsekar, N., Akkermans, S. & Kozak, D. Effect of the methanol concentration on the production rate of thaumatin and consumption rate of methanol by Pichia pastoris. in Proceedings of FOODSIM 2024 103–106 (Gent, 2024).
Karbalaei, M., Rezaee, S. A. & Farsiani, H. Pichia pastoris: A highly successful expression system for optimal synthesis of heterologous proteins. J. Cell. Physiol. 235, 5867–5881 (2020).
Acknowledgements
This study was supported by the Research Foundation Flanders (FWO) through project G0B4121N. Simen Akkermans was supported by the Research Foundation Flanders (FWO), grant number 1224623 N.
Author information
Authors and Affiliations
Contributions
N.P.: Conceptualisation, Methodology, Software, Validation, Formal analysis, Investigation, Data curation, Writing - Original Draft, Writing - Review & Editing, Visualisation. S.A.: Conceptualisation, Methodology, Software, Validation, Formal analysis, Data curation, Writing - Review & Editing, Visualisation, Supervision, Project Administration, Funding acquisition. D.K.: Conceptualisation, Methodology, Validation, Investigation, Resources, Writing - Review & Editing, Supervision. J.V.I.: Conceptualisation, Resources, Writing - Review & Editing, Supervision, Funding acquisition. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Parsekar, N., Akkermans, S., Kozak, D. et al. Enhancing recombinant thaumatin production by Komagataella phaffii through cold atmospheric helium plasma jet. Sci Rep 15, 39397 (2025). https://doi.org/10.1038/s41598-025-93016-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93016-2





 ), 4 (
), 4 ( ), 6 (
), 6 ( ) and 8 (
) and 8 ( ) min. Purple arrows at 24, 48, 72 and 96 h represent plasma treatments.
) min. Purple arrows at 24, 48, 72 and 96 h represent plasma treatments.
 ), 4 (
), 4 ( ), 6 (
), 6 ( ) and 8 (
) and 8 ( ) min. Purple arrows at 24, 48, 72 and 96 h represent plasma treatments.
) min. Purple arrows at 24, 48, 72 and 96 h represent plasma treatments.