Abstract
Photopolymers are promising materials for in-space manufacturing, whether for continuous extrusion or structural bonding. However, they must be processed as a liquid under high vacuum, which can alter viscosity, curing behavior, or mechanical properties. Furthermore, high outgassing can contaminate spacecraft surfaces. To study the influence of high vacuum on the resins in their liquid phase, Delo Katiobond GE680, Delo Katiobond OB678, Polymer-G EPV9511, and Loctite AA3301 LC were exposed to high vacuum. Outgassing was quantified for both liquid and cured samples. Viscosity increased in all resins, and UV-curing efficiency generally decreased. EPV9511 maintained stable mechanical properties, whereas the Delo and Loctite resins exhibited reduced stiffness at 45 °C. All resins exceeded 1% total mass loss in their liquid state, but cured GE680 and OB678 met low-outgassing criteria. GE680 and EPV9511 showed the highest vacuum stability and appear suitable for in-space processing when volatile components are minimized through pre-degassing.
Similar content being viewed by others
Introduction
Photopolymers are recommended for use as structural or adhesive materials in in-space manufacturing, assembly, and repair missions because they require low curing energy at a fast-curing rate1, are mechanically strong, and demonstrate high storage efficiency as they are present in their liquid form2. They can be utilized for structural bonding, additive manufacturing, and continuous extrusion of support elements for large space systems, by extruding the resin through a nozzle and curing it with UV-light3,4. Examples include joint fabrication for lightweight trusses5, linearly extruded spacecraft booms3, and mechanically supported membrane structures to support flexible solar arrays, or drag sails4.
However, processing photopolymers in their liquid state under high vacuum (10−6 to 10−9 mbar) presents significant challenges. Outgassing of volatile resin components can contaminate sensitive spacecraft surfaces6, and the evaporation of resin constituents may alter their physicochemical properties. Spacecraft materials must therefore meet low-outgassing standards such as those from American Society for Testing and Materials (ASTM E595) or the European Cooperation for Space Standardization (ECSS-Q-ST-70-02C).
These issues arise because photopolymers can contain monomers, oligomers, solvents, and photoinitiators or thermal inhibitors, which have low-molecular weights, making them prone to evaporation under vacuum7. A loss of these components can alter the resin’s viscosity, curing behavior, and mechanical performance. Since the viscosity of a resin is mainly defined by the length of the molecules, side chains, and polar groups8, evaporation of low-molecular-weight components is expected to increase it. An increase in viscosity can reduce extrudability as a higher extrusion pressure is required4, or impair its surface wetting capabilities, which are critical in microgravity where surface tension dominates liquid behavior9. As photopolymerization is determined by the concentration of reactive molecules and photoinitiator10, evaporation of components may reduce curing depth, curing speed, or the achievable degree of conversion.
Previous studies on photopolymers processed under high vacuum have reported bubble formation in the extruded material, leading to mechanical degradation and dimensional inaccuracies as a result of high outgassing during processing4,11,12. However, bubble formation is not solely attributable to the evaporation of volatile resin constituents; it may also result from air entrapped during resin handling13, moisture-related degassing13, or from gas release originating from other system components. Trapped air and moisture therefore contribute to the measured total mass loss (\(\delta {m}_{{TML}}\)), but not to contamination, as these gases are not expected to condense in high vacuum and can to a large extent, be removed from the resin by pre-degassing14.
A custom photopolymer formulation based on urethane acrylates was demonstrated in a mild vacuum of 6 mbar, showing only microbubbles in the extruded element which were attributed to the process setup7. Composites with a photopolymer matrix were cured under vacuum using UV-light and plasma10,15. However, the studies primarily addressed the curability in vacuum, rather than the changes in resin properties caused by vacuum exposure. To date, no systematic study has evaluated the behavior of commercially available photopolymers after high-vacuum exposure in their liquid state, in terms of their viscosity, curing behavior, mechanical performance, and outgassing properties.
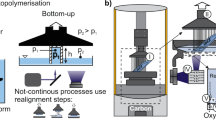
To address this gap, this study systematically evaluates the behavior of four commercially available photopolymers exposed to high vacuum for 24 h, following the experimental workflow summarized in Fig. 1. The investigated properties include viscosity, UV- and thermal-curing behavior, viscoelastic properties, and outgassing characteristics for both liquid and cured states.
Initially, unexposed resins as received from the manufacturer were characterized in both liquid and cured states. (1) Low-outgassing measurements were performed for liquid and cured resins by mass determination before and after vacuum exposure under defined temperature and pressure conditions. (2) Additionally, liquid resins were exposed to high vacuum for 24 h at 25 °C and pressures below 1 × 10⁻³ mbar. Following vacuum treatment, changes in material properties were assessed, including (3) mechanical properties determined by dynamic mechanical thermal analysis (DMTA), (4) viscosity measured using a rheometer, (5) UV-curing behavior evaluated by correlating curing energy with cured layer thickness, and (6) thermal-curing behavior analyzed by differential scanning calorimetry (DSC). Numbers in circles indicate the sequence of experimental steps.
Results
The following section presents the results comparing non-exposed and vacuum-exposed resin samples, as defined in the methodology.
Outgassing of the resins under high vacuum
The measured outgassing of the liquid resins exposed to high vacuum for 24 hours and heated to 60 °C showed significant values for \(\delta {m}_{{TML}}\), as shown in Table 1. None of the tested resins met the threshold of \(\delta {m}_{{TML}}=\, < 1 \%\). However, the \(\delta {m}_{{CVCM}}\) values are close to being acceptable for the Delo Katiobond GE680 (0.11%) while Polymer-G EPV9511 and Loctite AA3301 LC meet the <0.1% criterion for the \(\delta {m}_{{CVCM}}\). A low \(\delta {m}_{{CVCM}}\) value indicates reduced risk of long-term contamination, particularly important for sensitive optical or solar array surfaces in space applications. The standard deviation of the \(\delta {m}_{{CVCM}}\) values is relatively high for Katiobond GE680 and Katiobond OB678. This observation is examined in more detail in the discussion section. The Polymer-G resin showed the lowest \(\delta {m}_{{TML}}\), with only 4.37%, and the Delo Katiobond OB678 showed the highest value, at 57.12%.
In the case of the cured resin samples, the outgassing behavior differs significantly from that of the liquid resin samples. As shown in Table 2, Delo Katiobond GE680 and Delo Katiobond OB678 exhibit a \(\delta {m}_{{TML}}\) below the acceptable threshold of 1%. The values were also within acceptable limits (<0.1%) for all samples, except for the Loctite AA3301 LC, which reached exactly the limit. Among the tested resins, Delo Katiobond GE680 showed the lowest \(\delta {m}_{{TML}}\) at 0.08%, and Loctite AA3301 LC showed the highest at 4.36%. Both Loctite AA3301 LC and Polymer-G EPV9511 exceeded the 1% \(\delta {m}_{{TML}}\) threshold in their polymerized state and are therefore not compliant with the standard outgassing criteria.
Relative viscosity changes before and after vacuum exposure
The average relative change in viscosity across the entire measurement range between vacuum-exposed and non-vacuum-exposed samples was 34% for Katiobond OB678, 15% for Polymer-G EPV 9511, and 4% for Katiobond GE680, as shown in the graphs of Fig. 2a–c. All resins exhibited pseudoplastic behavior, with viscosity decreasing as shear rate increased. For Polymer-G EPV9511, the shear rate increase was so pronounced that, beyond 10 rpm, the rheometer exceeded its measurable range for both unexposed and exposed resin. Loctite AA3301 LC resin changed from a low-viscosity, watery state to a thixotropic, gum-like consistency, making it impossible to measure its viscosity with the rheometer setup. For a quantitative comparison, see Fig. 2d, e, where the difference in viscosity is clearly visible.
a–c Comparison of normalized viscosity for photopolymer samples before and after exposure to high vacuum. The viscosity values are normalized to the initial viscosity measured prior to vacuum exposure. a Shows Delo Katiobond OB678, b Delo Katiobond GE680, and c Polymer-G EPV9511. Qualitative comparison of the viscosity of the Loctite AA3301 LC resin before vacuum exposure (d) and after 24 h of vacuum treatment (e).
Effects of high-vacuum exposure on residual curing enthalpy and UV curing behavior
The non-treated and vacuum-treated resins were measured to assess any impact on the resin’s thermal reactivity. As the measurement results showed some fluctuations in the curing enthalpy, which may be attributable to factors such as segregation, sedimentation, light-induced curing during handling, mass deviations, or contamination in the DSC hardware. To ensure data quality, five measurements were performed for each resin, and a 95% confidence interval was calculated to evaluate the normality of the data. Outliers identified through this analysis were excluded, leaving at least three valid measurements per resin for evaluation. Further details, including probability plots, are provided in the Supplementary Information.
The compilation of the averaged graphs in Fig. 3 illustrates the exothermic curing reaction as a distinct peak deviating from the reference measurement. The area of this peak corresponds to the curing enthalpy relative to the reference measurement of the fully cured resin (overlayed for comparison). The results indicate that vacuum-exposed samples of Delo Katiobond GE680, Katiobond OB678, and Polymer-G EPV9511 show a strong correlation with the non-exposed samples, suggesting similar curing behavior. In contrast, the Loctite AA3301 LC resin behaves differently. The non-exposed resin does not exhibit any thermal-curing reaction and remains liquid after heating to 280 °C during the DSC test. An endothermic peak appears between 120 °C and 220 °C. However, the vacuum-exposed resin does show an exothermal peak from 150 °C to 210 °C.
a–d Differential scanning calorimetry (DSC) heat-flow curves of photopolymer resins measured as a function of temperature. a Shows Delo Katiobond GE680, b Delo Katiobond OB678, c Polymer-G EPV9511, and d Loctite AA3301 LC. Exothermic heat flow is plotted in the downward direction. Dashed black lines represent reference measurements of fully cured resins after the second heating cycle. Solid blue lines indicate the average response of non-vacuum-exposed samples, and solid orange lines indicate the average response of vacuum-exposed samples. In panel (d), thin colored lines show individual DSC measurements of non-vacuum-exposed (i2–i5) and vacuum-exposed (vi2–vi5) samples, while thicker lines represent the corresponding averaged curves.
Table 3 shows the average exotherm peak temperatures and reaction enthalpies, before and after vacuum exposure, for each resin. For the Delo resins, a reduction in the reaction enthalpies was measured of −2% or −2.6%, respectively, after vacuum exposure. The Polymer-G resin showed an increase of +4.6%. The average peak temperature in the curing reaction was very similar across all measurements. It should be noted that Delo Katiobond OB678 exhibited a high standard deviation (SD) for its enthalpy measurements compared to the other resins.
The UV-curing behavior before and after vacuum exposure was assessed by creating resin working curves, as shown in Fig. 4. Notable differences shown by the graphs are insignificant for Polymer-G EPV9511 and Delo Katiobond GE680. In contrast, Delo Katiobond OB678 exhibits significantly shallower curing at low UV exposure, but its curing depth gradually approaches the non-exposed curve at higher UV exposures. For Loctite AA3301 LC, the vacuum-exposed sample reached a significantly lower curing depth compared to the non-exposed sample.
a–d Working curves showing the curing depth as a function of the applied curing energy for photopolymer resins measured under non-vacuum and vacuum-exposed conditions. a Shows Delo Katiobond GE680, b Delo Katiobond OB678, c Polymer-G EPV9511, and d Loctite AA3301 LC. Discrete data points represent experimentally measured curing depths. Blue symbols indicate non-vacuum-exposed samples, and orange symbols indicate vacuum-exposed samples. Solid blue lines show fitted working curves for non-vacuum-exposed samples, while solid orange lines show fitted working curves for vacuum-exposed samples. The curing energy is expressed as the maximum applied energy density per unit area.
The critical curing energy \({E_c}\) and penetration depth \({D_p}\) are detailed in the graph of Fig. 5. For Delo Katiobond OB678, a significant increase in the critical curing energy \({E_c}\) was observed as it changed from 27 to 232 mJ/cm², and the penetration depth \({D_p}\) from 0.97 to 1.5 mm. Delo Katiobond GE680 showed an increase in \({E_c}\) from 61 to 97 mJ/cm² compared to the vacuum-exposed resin, but only a slight deviation in \({D_p}\) from 1.03 to 1.13 mm. No significant changes in the UV-curing behavior were observed for the Polymer-G resin due to the vacuum influence. The Loctite AA3301 LC showed a low penetration depth of 0.31 mm for the non-exposed resin, with no significant change after vacuum exposure. However, the critical energy needed to cure the resin increased from 31.6 to 49.4 mJ/cm².
Critical curing energy \({{\boldsymbol{E}}}_{{\boldsymbol{c}}}\) and penetration depth \({{\boldsymbol{D}}}_{{\boldsymbol{p}}}\) determined from the resin working curves for photopolymer resins under non-vacuum and vacuum-exposed conditions. Bar plots show the critical curing energy \({{\boldsymbol{E}}}_{{\boldsymbol{c}}}\) for non-vacuum-exposed samples (blue bars) and samples exposed to vacuum for 24 h (orange bars). Dashed lines with open symbols represent the corresponding penetration depth \({{\boldsymbol{D}}}_{{\boldsymbol{p}}}\) values for non-vacuum-exposed and vacuum-exposed samples. Data are shown for Delo Katiobond OB678, Delo Katiobond GE680, Polymer-G EPV9511, and Loctite AA3301 LC.
Change of the mechanical properties
The DMTA tests aimed to determine whether mechanically relevant substances or curing agents are outgassed during vacuum treatment of the liquid state. Each sample was measured twice to capture the effects of thermal post-curing caused by the initial heating cycle. For Katiobond GE680, Fig. 6 shows thermal post-curing as indicated by the dent in the \(\tan \,\delta\) curve, and the shift in the storage modulus during the first heating cycle. This indicates that the resin was not fully cured when cured only by UV light. The second heating curve shows no further curing reaction, confirming that post-curing was completed in the first cycle. However, the thermally post-cured sample exhibits a slightly higher storage modulus than the UV-only cured sample. The untreated resin shows an average storage modulus of 12 GPa at 50 °C, whereas the vacuum-treated resin measures ~11 GPa, indicating a reduction in stiffness. The \(\tan \,\delta\) peak of the vacuum-treated resin is higher (0.18) compared to the untreated resin, suggesting increased damping, while the peak temperatures remain unchanged. Overall, vacuum exposure affects the mechanical properties of this resin, but the effect is limited to a reduction of less than 10% in the storage modulus. Between 120 °C and 180 °C, the vacuum-treated samples show a slightly softer behavior, as seen in the \(\tan \,\delta\) curve.
Panels a and b show the storage modulus for the first and second measurement, respectively, while panels c and d show the corresponding loss factor (tan δ). Solid colored lines represent individual measurements of non-vacuum-exposed and vacuum-exposed samples. Dashed lines indicate the averaged response of non-vacuum-exposed samples, and dash-dotted lines indicate the averaged response of vacuum-exposed samples.
For Delo Katiobond OB678 (Fig. 7), the thermal post-curing effect during the first heating is evident in the \(\tan \,\delta\) curve, which shows an almost linear increase up to the peak, rather than a parabola-like shape as for the second measurement of Katiobond GE680, accompanied by an offset between both curves. After the second heating cycle, the storage modulus falls below that of the untreated sample, suggesting a significant reduction in mechanical properties. The difference in modulus between 80 °C and 140 °C is nearly 300 MPa. Overall, the resin is relatively soft, with a storage modulus of 2300 MPa to 2500 MPa at 45 °C. The effect of vacuum exposure on mechanical properties is visible, with a reduction of ~20% in storage modulus at 100 °C. A shift of 10 °C is visible in the corresponding peak of the \(\tan \,\delta\) curve after vacuum exposure in the second measurement, indicating a higher crosslinking density.
Panels a and b show the storage modulus for the first and second measurement, respectively, while panels c and d show the corresponding loss factor (tan δ). Solid colored lines represent individual measurements of non-vacuum-exposed and vacuum-exposed samples. Dashed lines indicate the averaged response of non-vacuum-exposed samples, and dash-dotted lines indicate the averaged response of vacuum-exposed samples.
The Polymer-G EPV9511 (Fig. 8) exhibits a strong thermal post-cure response. At 45 °C, the storage modulus for both the vacuum-treated and untreated samples is approximately 2500 MPa. However, the \(\tan \,\delta\) curve reveals the post-curing effect through two distinct peaks, where the first peak is 0.05 lower for the vacuum-treated sample compared to the untreated one. In the second measurement, the storage modulus nearly doubles, reaching 4000 MPa at 45 °C for both groups, confirming a significant thermal post-cure effect. Additionally, the \(\tan \,\delta\) curves of the vacuum-treated and untreated samples become nearly identical after the second heating cycle. It is important to note that above 120 °C, the material becomes soft and its storage modulus drops to 100 MPa.
Panels a and b show the storage modulus for the first and second measurement, respectively, while panels c and d show the corresponding loss factor (tan δ). Solid colored lines represent individual measurements of non-vacuum-exposed and vacuum-exposed samples. Dashed lines indicate the averaged response of non-vacuum-exposed samples, and dash-dotted lines indicate the averaged response of vacuum-exposed samples.
The Loctite AA3301 LC (Fig. 9) does not show a significant thermal-induced post-cure reaction between the first and second measurements of the same sample, which aligns with the DSC curing enthalpy experiment results. The storage modulus of the average vacuum-exposed samples is 248 MPa lower than that of the non-vacuum-exposed samples. However, the non-treated samples at 90 °C lose their complete stiffness, as evidenced by a noisy \(\tan \,\delta\) plot. This suggests that vacuum treatment has a significant impact on the mechanical properties of the resin. While vacuum slightly reduces overall stiffness, it causes a minor increase at temperatures above 80 °C.
Panels a and b show the storage modulus for the first and second measurement, respectively, while panels c and d show the corresponding loss factor (tan δ). Solid colored lines represent individual measurements of non-vacuum-exposed and vacuum-exposed samples. Dashed lines indicate the averaged response of non-vacuum-exposed samples, and dash-dotted lines indicate the averaged response of vacuum-exposed samples.
The glass transition temperatures were obtained from the maxima of the \(\tan \delta\) curves during the second heating cycle and are summarized in Table 4. For Delo Katiobond GE680, Delo Katiobond OB678, and Polymer-G EPV9511, no significant difference between the non-exposed and vacuum-exposed specimens was observed, with all changes falling within the respective standard deviations. In contrast, Loctite AA3301 LC exhibited a pronounced increase of 25.6 °C after vacuum exposure.
Table 5 summarizes the averaged results from the vacuum tests, with the findings discussed in the subsequent section. The \(\delta {m}_{{TML}}\) and \(\delta {m}_{{CVCM}}\) are given as absolute values for both liquid and solid states. For the viscosity, curing behavior, and differences in the mechanical properties, the average percentages relative to the untreated and vacuum-exposed resin were calculated. For the Loctite AA3301 LC resin, the measurement range of the rheometer was exceeded after vacuum exposure; therefore, the upper limit of the device was used to obtain an approximate quantitative estimate of the increased viscosity. Differences in the storage modulus (\({E}^{{\prime} }\)) at two specific temperatures of the second heating cycle (45 °C and 120 °C) are included. This approach was chosen because the first heating cycle exhibited thermal post-curing effects for all four resins, as UV curing alone did not fully cure the material.
Discussion
The analysis of the relative resin viscosity change due to high vacuum showed an increase in viscosity for all resins. However, the extent of the increase varies among different formulations between 4 and 34%. One resin experienced a significant increase in viscosity of over 420%, surpassing the maximum measurement limit of the rheometer. A local polymerization based on the absence of oxygen in high vacuum could be responsible for this behavior7. It is expected that the increase in viscosity can be attributed to the evaporation of low-molecular-weight substances in the resin mixtures, resulting in a more viscous mixture. For ISM, an increase in viscosity can cause process implications which may impair wetting and increase extrusion pressures.
Change in the curing mechanism due to high vacuum exposure showed that the thermal-curing mechanism was only slightly affected, indicating that any thermal inhibitors present in the formulations remained effective and did not lead to an increase in reaction enthalpy. One resin showed a substantial increase ( + 769%) in the critical curing energy which is attributed to outgassing of photoinitiators that initiate polymerization. However, another possible explanation is that the change in viscosity have altered the resin’s optical behavior, reducing the effective UV penetration and thus the curing efficiency.
Changes in mechanical properties due to high vacuum revealed that some of the vacuum-exposed resins exhibited a decrease in storage modulus after the second heating cycle of the DMTA, ranging from 12 to 35%, while others remained stable or showed slight increases depending on the temperature. However, when comparing the \({T}_{g}\) it was apparent that it didn’t change significantly for GE680, OB678, and EPV9511 which indicates that these photopolymers didn’t change chemically. Therefore, macroscopic effects like an increased void content related to outgassing are attributed to their lowered storage modulus. For Loctite AA3301 LC an increase in its \({T}_{g}\) (+26 °C) at lowered \({E}^{{\prime} }\) (−35%) was observed, which is counterintuitive as a higher crosslinking density should come with a higher storage modulus. However, its still a possible explanation that increased void content led to a reduced stiffness even at higher crosslinking density.
For space structures, this level of stiffness reduction is expected to remain within a range that can be accommodated through appropriate safety margins in the structural design.
The results showed that UV curing alone did not produce a fully cross-linked polymer network under the selected curing parameters as thermal-induced post curing was observed. Depending on the orbit and orientation of a photopolymer structure in space, thermally induced post-curing will likely occur naturally due to solar heating which can be up to 120 °C in LEO16, allowing the material to reach a higher degree of conversion over time. However, the structure must either be designed to withstand loads in the initial under-cured state, which requires empirical evaluation for the respective system configuration, or an additional curing step should be implemented.
The photopolymer also offers the possibility to embed carbon fibers, for example in endless extruded tubes3,17. The mechanical properties of these reinforcement fibers would not be affected by vacuum, as these are solids, but they would contribute significantly to the effective Young’s modulus of the resin-fiber composite; therefore, the contribution of the resin’s stiffness reduction under high vacuum is expected to be far less. However, fibers can hinder UV curing because of different transmission or shadowing17 and require good wetting, which may be compromised when vacuum exposure increases the viscosity.
The total mass loss measurements showed for all resins in their liquid state a \(\delta {m}_{{TML}}\) above the acceptable limit when exposed to vacuum at 60 °C for 24 h. The selected temperature deviates from the ECSS-Q-ST-70-02C standard but is high enough to initiate thermal curing and is therefore unsuitable for ISM processing, but it was chosen to simulate a worst-case outgassing scenario and to enable \(\delta {m}_{{CVCM}}\) measurements. The resins were not degassed before testing to avoid artificially altering the outgassing results. However, pre-degassing would be a standard step in processing resin used in high-vacuum environments to remove trapped air, moisture, and other volatiles beforehand, which would likely significantly reduce the \(\delta {m}_{{TML}}\). Using vacuum degassing a significant reduction in bubble volume fraction from 29 to 5% for a Delo Katiobond GE680 extruded and cured structure under high-vacuum was already demonstrated14. The comparatively low \(\delta {m}_{{CVCM}}\) values, compared to the \(\delta {m}_{{TML}}\), indicate that trapped air and moisture are probably most responsible for the high \(\delta {m}_{{TML}}\), as otherwise, elements would likely deposit on the collector plates. Therefore, measuring \(\delta {m}_{{TML}}\) again after proper degassing is advisable to confirm this hypothesis. In addition, 24-hour exposure to high vacuum is significantly longer than expected for an ISM application, where the resin is extruded and cured within seconds. Therefore, the measured \(\delta {m}_{{TML}}\) likely overestimates the mass loss contribution form the resin in a liquid state for an ISM application.
The outgassing values of the cured resins were significantly lower when exposed to vacuum at 125 °C for 24 h, in accordance with ECSS-Q-ST-70-02C. This was expected as volatile content is greatly reduced after crosslinking. Both Delo Katiobond GE680 and OB678 met the specification and therefore comply with the ECSS in their cured state.
Delo Katiobond GE680 showed high TML in its liquid state, likely due to trapped air and volatile constituents, consistent with the foam-like bubble formation observed in earlier high-vacuum extrusion tests4. After curing, however, the resin met the ECSS-Q-ST-70-02C requirements and showed slightly reduced mechanical properties but stable curing behavior, making it the most promising candidate among the tested formulations. Further analyses of the liquid-phase outgassed species and improved degassing protocols are recommended.
Delo Katiobond OB678 exhibited a high mass loss in its liquid state indicating evaporation of a substantial fraction of the resin under high vacuum. This was accompanied by increased viscosity and a pronounced rise in the critical curing energy, suggesting that key photoinitiators or reactive species were depleted during vacuum exposure. Despite these changes, the resin could still cure in 5 mm thick sections. Due to its extreme volatility and altered curing behavior, OB678 is not suitable for processing in its liquid state under high-vacuum conditions, even though the cured resin itself meets ASTM E595 and ECSS-Q-ST-70-02C requirements as stated by the manufacturer, which aligns with the results of this study18.
Polymer-G EPV9511 showed the lowest mass loss in its liquid state among the resins tested, accompanied by low contamination potential. In its cured state, however, it shows a high mass loss above the acceptable limit. Besides the comparable high outgassing properties, the viscosity, curing performance, and mechanical properties showed the least variation among all resins tested, suggesting good overall stability and suitability as a candidate for in-space manufacturing (ISM). As the tested batch was nearly two years past its shelf life, it could have shown lowered outgassing values because any potential chemical degradation may have already taken place. This should be considered for all resin related results presented in this study.
Loctite AA3301 LC is the only tested resin based on acylated urethane. It showed strong outgassing in both liquid and cured states, and its viscosity increased drastically under vacuum, causing the resin to transition from a liquid to a gum-like state. The resin shows a low stiffness after vacuum exposure at lower temperatures (e.g. −35% at 45 °C), while at higher temperatures above 85 °C it gained stiffness compared to the unexposed resin. However, above 100 °C the total stiffness of the resin was very low and it seems not suitable for most applications. Based on these findings, Loctite AA3301 LC is not recommended for being processed in high vacuum for ISM, based on its volatility, uncertainty in its viscosity, and stiffness under elevated temperatures.
The findings of this study indicate that the photopolymers Delo Katiobond GE680 and Polymer-G EPV9511 are, with certain limitations, promising candidates for high-vacuum processing in ISM. Although the total mass loss values of the resins in their liquid state exceeded the ECSS-Q-ST-70-02C low-outgassing limits, the significantly shorter processing times characteristic of ISM and the option to pre-degas the resins are expected to substantially reduce the risk of contamination. Importantly, all investigated photopolymers remained curable after high-vacuum exposure. The results further showed that the polymer networks remained largely intact, while reductions in mechanical properties were primarily attributed to macroscopic effects such as void formation induced by vacuum exposure. Despite these reductions, the retained stiffness levels appear sufficient for structural applications in space and for ISM processes in particular.
Future work should address long-term environmental durability, bubble-mitigation through improved degassing, and the development of formulations specifically optimized for high-vacuum curing.
Methods
Selection of photopolymer resins
Different resins were compared based on the datasheet values for this screening, and four were selected. The selection criteria included a wide operation temperature as there are large temperature fluctuations to expect on orbit, a high Young’s modulus as a high stiffness is required, and a high curable layer thickness as structures are intended to be cured in a single pass. The individual substances of the resin mixture are disclosed because the formulations are commercial products. Nevertheless, the majority of included molecules are given in the safety data sheets (SDS). A summary of the selected resins is shown in Table 6, and a short intro is subsequently provided.
Delo Katiobond GE680 was successfully tested in microgravity and medium vacuum on a sounding rocket and parabolic flights in microgravity11,19. It consists of two substances according to its SDS and is free of solvents20,21. It contains a mineral-based filler and has a honey-like viscosity (70,000 mPa∙s), a wide operating temperature of −40 to 150 °C in a cured state, and the highest stiffness of 16,800 MPa20,21.
Delo Katiobond OB678 contains no fillers, consists of five substances according to the SDS, and is free of solvents18,22. Its viscosity is significantly lower with 8400 mPa∙s, compared to Delo Katiobond GE680. Compared to the other resins of this study, it offers the widest operational temperature range of −40 to 180 °C for its cured state. While its stiffness is relatively low at 3400 MPa, the low viscosity allows for easier extrusion with less power, and its improved surface-wetting properties are beneficial for bonding or impregnating fibers18,22. This resin is low outgassing in accordance with ASTM E595 and ECSS-Q-ST-70-02C18.
Polymer-G EPV 9511 is a micro glass fiber-reinforced epoxy resin23. It consists of three substances, according to the SDS24. A lower operating temperature is not given for the resin; however, the upper temperature of 95 °C is on the low end for the temperatures expected on-orbit. The viscosity is the highest at 130,000 mPa∙s23. In its cured state, it shows about a third of the Young’s Modulus of the Delo Katiobond GE680 with 5500 MPa23. At the time of testing, the resin was already two years past the shelf-life date specified by the manufacturer, but no notable difference in the resin handling and curing behavior was observed; however, it may have influenced the absolute data.
Loctite AA3301 LC is the most exotic of the selected resins. According to the SDS, unlike the other resins, it is not epoxy-based but an acrylated urethane made up of 9 substances25,26. It was selected as Yerazunis et al. developed a formulation using such oligomers for in-space manufacturing7. It contains no fillers and shows a very low viscosity between 90 and 210 mPa∙s25,26.
Vacuum treatment of the resin
To investigate the effects of vacuum exposure, every resin was dispensed into cups containing 10 g of material and placed inside a high-vacuum chamber capable of reaching pressures down to 10−6 mbar as shown in Fig. 1 (2). Due to differences in the outgassing behavior, the chamber reached a pressure of <9 × 10⁻⁵ mbar for Delo Katiobond GE680, Delo Katiobond OB678, and Polymer-G EPV9511, and <9 × 10⁻³ mbar for Loctite AA3301 LC. Because the pumps operated at a constant power level in both cases, the final pressure reached is determined by the amount of outgassing from the investigated materials. All samples were kept under high-vacuum for 24 h at a temperature of 25 °C. After the vacuum treatment, the liquid resin samples were analyzed for their viscosity, curing behavior, and curing enthalpy as well as for their mechanical (viscoelastic) properties in their cured state.
Low-outgassing test procedure
In addition to the vacuum exposure experiments, an outgassing analysis was performed in accordance with the ECSS-Q-ST-70-02C space product assurance standard. For each photopolymer, the total mass loss (\(\delta {m}_{{TML}}\)), the collected volatile condensable material (\(\delta {m}_{{CVCM}}\)), as well as the recovered mass loss (\(\delta {m}_{{RML}}\)) were measured in both the liquid and the solidified state, as shown in Fig. 1 (1).
For this purpose, three crucibles per resin were filled with 100–300 mg of either liquid or UV-cured material. Three additional empty crucibles were included to monitor potential contamination and to account for any outgassing originating from the crucibles themselves.
All crucibles were tested in the micro-Volatile Component Material facility of the European Space Agency. Both the liquid and the cured resin samples were exposed to a vacuum below 1 × 10−4 mbar for 24 h. The UV-cured and thus solidified resins were additionally heated to 125 °C as specified by the ECSS standard. In contrast, the liquid resin samples were tested at 60 °C, which deviates from the ECSS. This reduced temperature was necessary because the photopolymers can start to thermally cure at temperatures above 60 °C which was not intended to observe here as it would alter its outgassing behavior. Nevertheless, a certain degree of heating was necessary as the collector plate was cooled to 25 °C by ECSS guidelines for the collection of \(\delta {m}_{{CVCM}}\). In the context of ISM this procedure is seen as appropriate, as during the manufacturing phase the resin is extruded and exposed to high vacuum as a liquid before it’s cured. The chosen conditions therefore reflect the highest temperature at which the resin remains in a stable liquid state and provides a realistic upper-bound measure of its outgassing prior to curing.
To determine the \(\delta {m}_{{TML}}\) for both the liquid and the cured resin, mass measurements of each sample were taken at the beginning (\({m}_{0}\)) and directly after the vacuum exposure (\({m}_{f}\)) using a balance. The difference between \({m}_{0}\) and \({m}_{f}\) corresponds to the mass lost due to outgassing in vacuum and was calculated using Eq. (1) according to ECSS-Q-ST-70-02C27.
The \(\delta {m}_{{CVCM}}\) was measured using separate collector plates placed above each sample and cooled to 25 °C to condense volatile species. The \(\delta {m}_{{CVCM}}\) value was calculated by subtracting the final plate mass, \({m}_{g},\) from the initial mass, \({m}_{p}\), and the resulting mass difference was normalized to the initial sample mass \({m}_{m}\) as shown by Eq. (2).
A third value, \(\delta {m}_{{RML}}\), indicates how much moisture the material reabsorbs after the test. For this, the samples are stored for 24 h at 22 ± 3 °C and 55 ± 10% relative humidity after the test, before the sample weight \({m}_{r}\) is measured and calculated according to Eq. (3).
For in-space manufacturing, where the material is processed in orbit, the \(\delta {m}_{{RML}}\) is not necessarily relevant, as there is no humidity present in orbit that the material could absorb. However, this value is still presented here for completeness. All outgassing related mass measurements were carried out using a microbalance (Cubis II MCA6.6S, Sartorius) with a resolution of 0.001 mg. The acceptable limits defined by the ECSS require an \(\delta {m}_{{RML}}\) of <1% and a \(\delta {m}_{{CVCM}}\) of <0.1%. In this study, however, the \(\delta {m}_{{TML}}\) and the \(\delta {m}_{{CVCM}}\) are used as the primary acceptance criteria, with a \(\delta {m}_{{TML}}\) limit of less than 1%, since no water reabsorption is expected under space conditions.
Relative viscosity measurement
The relative resin viscosity before and after vacuum exposure was measured using the rheometer RC01-L of the company Rheotec, as illustrated in Fig. 1 (4) to quantify its relative change. Due to limited sample volumes, the viscosity measurement spindles were immersed to a reduced but reproducible depth between 4 and 22 mm, depending on the viscosity of the respective resin, rather than to the full immersion depth as specified for each spindle. As a result, the measurements provide internally consistent relative viscosity values that enable reliable comparison between samples but are not suitable for deriving absolute viscosities.
The resin was measured at ambient temperature, ensuring that the temperature difference between measurements did not exceed \(\pm\)2 °C, as temperature can significantly affect the resin’s viscosity. The shear rates applied were iterated from 1 to 100 rpm within the operational limits of the instrument. When subjected to shear (e.g., during extrusion), fluids exhibit varying absolute viscosities. For Newtonian fluids, this behavior is linear.
Curing characteristics evaluation procedure
To evaluate the thermal-curing characteristics, Differential Scanning Calorimetry (DSC) measurements were conducted to assess the curing enthalpy of the resin exposed to vacuum versus resin that was not exposed, as shown in Fig. 1 (6). For the DSC measurements, the device 214 Polyma, Erich NETZSCH B.V. & Co. Holding KG, was used. This method compares the heat absorption or release of a sample-filled crucible against an empty reference crucible during linear heating in accordance with DIN EN ISO 11357-1:2023.
To evaluate the effect of vacuum exposure on thermal curing, 15 mg (±1 mg) of resin was placed in each crucible. The temperature limits as well as the heating rate were selected based on preliminary DSC measurements conducted prior to the main experiments. These preliminary measurements were used to identify the temperature range required to fully capture the exothermic curing reaction without reaching thermal degradation, and to select an appropriate heating rate. The final parameters applied in this study are listed in Table 7. The curing enthalpy and peak temperature were determined graphically, and the vacuum-exposed and non-exposed resins were compared. During resin curing, the exothermic heat release is added to the constant heat flow (\(Q\)), resulting in a DSC peak. This peak corresponds to the peak temperature of the exothermal curing reaction. The curing enthalpy (\(\Delta Q\)) is the area between the measured DSC curve and the constant heat flow. This enthalpy is quantified using the integral in Eq. (4), where \(\beta\) denotes the continuous heating and cooling rate28.
For each resin, five samples were tested to improve statistical reliability. In addition, one sample was measured twice to verify full thermal curing; this sample is referred to as the “reference sample” in the plots.
The UV-curing characteristics were identified by creating resin working curves according to Jacobs29. To create these curves, vacuum-exposed resin and non-vacuum-exposed resin were poured into five circular 8 mm molds with a depth of 5 mm, as shown in Fig. 1 (5). After that, the samples were cured with an energy of 305 mW/cm² at a wavelength of 365 nm (respectively 395 nm for Polymer-G EPV9511, following the manufacturer’s datasheet), where the exposure time per sample was varied between 1 and 30 s. The samples were removed from the mold after 24 h, and excess resin was removed to measure the thickness according to the exposure dose. This thickness is the curing depth \({C}_{d}\) for a known energy \({E}_{\max }\). Jacobs adopted the Beer-Lambert law to characterize the curing behavior of different photopolymer resins for stereolithography 3D-printers as shown in Eq. (5). According to this equation, the cure depth \({C}_{d}\) is proportional to the natural logarithm of the maximum exposure energy \({E}_{\max }\)29.
The relationship between cure depth and exposure energy is called the resin working curve. The slope of this curve corresponds to \({D}_{p}\) (penetration depth), which indicates the depth at which the penetrating light intensity falls to 1/e30,31. The x-intercept of the curve represents the critical curing energy \({E}_{c}\)30. The value of \({E_c}\) is associated with the gel point of the photopolymer30. Therefore, \({E}_{c}\) represents the minimum energy needed to cure a resin. Resin working curves provide key parameters for evaluating the curing performance of a resin.
\({D}_{p}\) and \({E_c}\) were determined using Eq. (5) and MATLAB’s curve-fitting functionality, including a 95% confidence check.
Evaluation of the mechanical properties
For the mechanical tests, the resin was cured by UV light into solid samples and stored in a desiccator for at least 24 h to keep the exposure to moisture equal between all specimens and to give the resin time to fully crosslink.
To assess the influence of vacuum treatment on the viscoelastic properties over a temperature range, a Dynamic Mechanical Thermal Analysis (DMTA) was performed in accordance with ISO 6721-1. The samples were prepared in a rectangular shape with 60 × 8 × 3 – 4 mm, depending on the Young’s modulus of the resin, as shown in Fig. 1 (3). A silicon mold was filled with non-exposed and vacuum-exposed resin and cured by UV light with an intensity of 305 mW/cm². GE680, OB678, and EPV9511 were cured for 60 s, while the slower-curing Loctite resin required an exposure time of 120 s followed by at least 24 h post curing phase for all samples. The curing exposure times were established empirically through preliminary exposure tests, selecting a conservative duration that ensured complete solidification and a tack-free surface. Although no independent conversion analysis was performed, all specimens were cured using identical exposure conditions to ensure consistency. Uniform curing was further ensured by positioning all samples in the same region of the UV field and maintaining identical distance and orientation during irradiation.
Any changes due to vacuum exposure on a molecular level influence the molecular movements and, therefore, the reaction on time-dependent deformation32. The tests were performed on each specimen twice to account for potential thermal-induced post-cure reactions from the heating of the DMTA. The setup for the measurement was performed using a DMA 242, Erich NETZSCH B.V. & Co. Holding KG, in a 3-point bending configuration. In DMTA, a sinusoidal oscillation is applied to a polymer sample to measure its viscoelastic material properties8. The oscillation had a constant frequency of 1 Hz at a constant amplitude. The primary outputs from a DMTA are the storage modulus (\({E}^{{\prime} }\)), loss modulus (\({E}^{{\prime}{\prime} }\)), and damping factor (tan δ) plotted over a temperature range. The storage modulus \({E}^{{\prime} }\) represents the elastic or stored energy component8. A high value represents a stiff material, and a low value represents a highly elastic material. The loss modulus \({E}^{{\prime}{\prime} }\) shows the amount of input energy that gets irreversibly dissipated in the material8. The \(\tan \,\delta\) is defined as the ratio between loss modulus and storage modulus \({E}^{{\prime}{\prime} }\)/\({E}^{{\prime} }\) and represents the damping characteristics of a material8. A peak in the \(\tan \,\delta\) curve typically corresponds to the glass transition temperature \({T_g}\). This paper shows the storage modulus and \(\tan \,\delta\) to compare any shifts in the mechanical properties.
The specimens were tested at a temperature range from 30 to 190 °C, depending on the photopolymer, as shown in Table 8. The upper temperature limit was chosen based on the \({T_g}\) value given in the data sheet, with an additional 20 to 30 °C added based on prior tests. A temperature dwell time of ten minutes after reaching the starting temperature was selected to ensure uniform temperature distribution within the specimen. The heating rate was set to 2 K/min as the ISO 6721-1 standard recommended.
Data availability
All data can be found either in the manuscript or in the supplementary data.
References
Crivello, J. V. & Reichmanis, E. Photopolymer materials and processes for advanced technologies. Chem. Mater. 26, 533–548 (2014).
Kringer, M., Lafont, U., Pietras, M., Böhrer, C. & Sinn, T. In-space manufacturing of external satellite structures: A review of existing concepts and discussion of their feasibility. In IAC 2022 Congress Proceedings vol. 73 (IAF Astro, 2022).
Kringer, M., Böhrer, C., Sinn, T. & Pietras, M. Endless photopolymer tube extrusion for on-orbit manufacturing of satellite booms. In IAC 2022 Congress Proceedings vol. 73 (IAF Astro, 2022).
Kringer, M. et al. In-space manufacturing for flexible membranes: process, applications, and vacuum test insights. In (IEEE Xplore, 2025). https://doi.org/10.1109/AERO63441.2025.11068679.
Glaser, T. et al. AMOCSIS, On-orbit manufacturing of composite truss structures. In Proceedings 16th European Conference on Spacecraft Structures, Materials and Environmental Testing ECSSMET 2021 (ECSSMET, 2023).
Tribble, A. C. The Space Environment Implications for Spacecraft Design (The Princeton University Press, 2003).
Yerazunis, W., Weiss, A., Radyjowski, P. & Cottrell, R. Resin development for on-orbit fabrication of spacecraft structures by direct solar photopolymerization. In Proceedings of the 33rd Annual International Solid Freeform Fabrication Symposium (Solid Freeform Fabrication Symposium, 2022).
Habenicht, G. Kleben: Grundlagen, Technologien, Anwendungen (Springer, 2009). https://doi.org/10.1007/978-3-540-85266-7.
Meseguer, J. et al. Surface tension and microgravity. Eur. J. Phys. 35, 055010 (2014).
Kondyurin, A. Design and Fabrication of Large Polymer Constructions in Space. (Elsevier, 2023). https://doi.org/10.1016/C2018-0-01458-9.
Kringer, M. et al. Effects of microgravity and reduced atmospheric pressure on manufacturing photopolymer specimens. Acta Astronaut 218, 314–325 (2024).
Yerazunis, W., Weiss, A., Radyjowski, P. & Cottrell, R. On-orbit additive manufacturing of parabolic reflectors via solar photopolymerization. In 70th International Astronautical Congress Proceedings (IAF Astro, 2019).
Afendi, M., Banks, W. M. & Kirkwood, D. Bubble free resin for infusion process. Compos. Part Appl. Sci. Manuf. 36, 739–746 (2005).
Pimpi, J., Kringer, M., Sinn, T. & Pietras, M. Fluid Photopolymer Extrusion under High Vacuum Environment: Enabling In-Space Manufacturing of Solar Arrays (IAF Astro, Sydney, 2025).
Reiss, P., Breunig, E., Zimmerhakl, P., Newie, N. & Zeiner, A. Investigating New Space Structures with the Focus Experiment (Hyère, France, 2011).
Finckenor, M. M. & de Groh, K. K. A. Researcher´s Guide to: International Space Station Space Environmental Effects. https://www.nasa.gov/sites/default/files/files/NP-2015-03-015-JSC_Space_Environment-ISS-Mini-Book-2015-508.pdf (2015).
Pimpi, J., Kringer, M., Sinn, T. & Pietras, M. Generative in-space manufacturing of large space structures using fiber-reinforced photopolymers. In IAC 2024 Congress Proceedings (IAF Astro, 2024).
Delo GmbH. DELO KATIOBOND OB678 (Delo GmbH, 2023).
Kringer, M., Böhrer, C., Frey, M., Pimpi, J. & Pietras, M. Direct Robotic Extrusion of Photopolymers (DREPP): influence of microgravity on an in-space manufacturing method. Front. Space Technol. 3, 899242 (2022).
Delo GmbH. DELO KATIOBOND GE680. https://www.delo.de/service-center/downloadbereich/downloads/datasheet/DELOKATIOBOND_GE680_TIDB-de.pdf?show=1&type=5001&cHash=39489453a92d624c4dafb22f7ea499a5 (2023).
Delo GmbH. DELO KATIOBOND GE680 (Delo GmbH, 2022).
Delo GmbH. DELO KATIOBOND OB678 (Delo GmbH, 2022).
Polymer Gvulot Ltd. EPV-9511. https://www.polymer-g.com/wp-content/uploads/EPV-9511.pdf (2020).
Polymer Gvulot Ltd. Polymer-G EPV9511 (Polymer Gvulot Ltd., 2020).
Henkel AG & Co. KGaA. HenkelTM-Loctite®-AA-3301-LC (Henkel AG & Co., 2014).
Henkel AG & Co. KGaA. HenkelTM-Loctite®-AA-3301-LC (Henkel AG & Co, 2023).
ESA Requirements and Standards Division. Space Product Assurance: Thermal Vacuum Outgassing Test for the Screening of Space Materials (ECSS-Q-ST-70-02C) (ESA Requirements and Standards Division, 2008).
DIN e.V. DIN E. N. ISO 11357-1, Kunststoffe Dynamische Differenzkalorimetrie (DSC) Teil 1: Allgemeine Grundlagen. (2023).
Jacobs, Paul F. Rapid Prototyping & Manufacturing: Fundamentals of Stereolithography (Society Of Manufacturing Engineers, 1992).
Jacobs, P. F. Stereolithography and Other RP&M Technologies from Rapid Prototyping to Rapid Tooling (Society Of Manufacturing Engineers, 1995).
Bennett, J. Measuring UV curing parameters of commercial photopolymers used in additive manufacturing. Addit. Manuf. 18, 203–212 (2017).
DIN e.V. Kunststoffe Bestimmung dynamisch-mechanischer Eigenschaften Teil 1: Allgemeine Grundlagen (ISO 6721-1:2019); Deutsche Fassung EN ISO 6721-1:2019 (2019).
Acknowledgements
This study is funded by Dcubed GmbH and by the European Space Agency under a co-funded research program (ESA contract number 400013618721/NL/GLC/my). The author gratefully acknowledges financial support through a qualification position at Munich University of Applied Sciences.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
M.K. contributed to conceptualization, data curation, methodology, investigation, and writing of the original draft. J.P. contributed to writing the original draft. U.L. contributed to supervision, methodology and resources. T.S. contributed to project administration and supervision. P.R. contributed to supervision, methodology, review and editing, and the definition of overarching research goals. M.P. contributed to project administration, supervision, review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kringer, M., Pimpi, J., Sinn, T. et al. Screening of liquid photopolymer resins exposed to high-vacuum. npj Adv. Manuf. 3, 5 (2026). https://doi.org/10.1038/s44334-025-00066-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44334-025-00066-5