Abstract
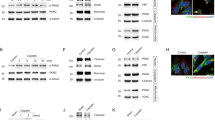
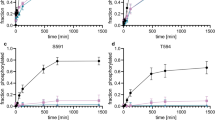
Treatment of Jurkat cells with specific inhibitors of protein kinase CK2 induces apoptosis. Here we provide evidence that the antiapoptotic effect of CK2 can be at least partially mediated by upregulation of the Akt/PKB pathway. Such a conclusion is based on the following observations: (1) inhibition of CK2 by cell treatment with two structurally unrelated CK2 inhibitors induces downregulation of Akt/PKB, as judged from decreased phosphorylation of its physiological targets, and immunoprecipitate kinase assay; (2) similar results are observed upon reduction of CK2 catalytic subunit by the RNA-interference technique; (3) Akt/PKB Ser129 is phosphorylated by CK2 in vitro and in vivo; (4) such a phosphorylation of activated Akt/PKB correlates with a further increase in catalytic activity. These data disclose an unanticipated mechanism by which constitutive phosphorylation by CK2 may be required for maximal activation of Akt/PKB.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- CK2:
-
casein kinase 2
- GSK3:
-
glycogen synthase kinase 3
- IQA:
-
5-oxo-5,6-dihydroindolo-(1,2-a)quinazolin-7-yl]acetic acid
- MALDI:
-
matrix-assisted laser desorbtion and ionization
- MS/MS:
-
tandem mass spectrometry
- PDK1:
-
3-phosphoinositide-dependent protein kinase-1
- PH:
-
pleckstrin homology
- PI3-kinase:
-
phosphatidylinositol 3-kinase
- PKB:
-
protein kinase B (Akt)
- PTEN:
-
phosphatase and tensin homolog deleted on chromosome ten
- RNAi:
-
RNA interference
- SGK:
-
serum- and glucocorticoid-induced kinase
- siRNA:
-
short interfering RNA
- TBB:
-
4,5,6,7-tetrabromobenzotriazole
- WB:
-
Western blot
- w.t.:
-
wild type
References
Meggio F and Pinna LA (2003) One-thousand-and-one substrates of protein kinase CK2? FASEB J. 17: 349–368
Guerra B and Issinger O-G (1999) Protein kinase CK2 and its role in cellular proliferation, development and pathology. Electrophoresis 20: 20391–20408
Tawfic S, Yu S, Wang H, Faust R, Davis A and Ahmed K (2001) Protein kinase CK2 signal in neoplasia. Histol. Histopathol. 16: 573–582
Seldin DC and Leder P (1995) Casein kinase II α transgene-induced murine lymphoma: relation to theileriosis in cattle. Science 267: 894–897
Kelliher MA, Seldin DC and Leder P (1996) Tal-1 induces T cell acute lymphoblastic leukemia accelerated by casein kinase IIα. EMBO J. 15: 5160–5166
Landesman-Bollag E, Channavajhala PL, Cardiff RD and Seldin DC (1998) p53 deficiency and misexpression of protein kinase CK2α collaborate in the development of thymic lymphomas in mice. Oncogene 16: 2965–2974
Faust RA, Tawfic S, Davis AT, Bubash LA and Ahmed K (2000) Antisense oligonucleotides against protein kinase CK2-α inhibit growth of squamous cell carcinoma of the head and neck in vitro. Head Neck 22: 341–346
Romieu-Mourez R, Landesman-Bollag E, Seldin DC and Sonenshein GE (2002) Protein kinase CK2 promotes aberrant activation of nuclear factor-κB, transformed phenotype, and survival of breast cancer cells. Cancer Res. 62: 6770–6778
Litchfield DW (2003) Protein kinase CK2: structure, regulation and role in cellular decisions of life and death. Biochem. J. 369: 1–15
Guo C, Yu S, Wang H, Davis AT, Green JE and Ahmed K (2001) A potential role of nuclear matrix-associated protein kinase CK2 in protection against drug-induced apoptosis in cancer cells. J. Biol. Chem. 276: 5992–5999
Ruzzene M, Penzo D and Pinna LA (2002) Protein kinase CK2 inhibitor 4,5,6,7-tetrabromobenzotriazole (TBB) induces apoptosis and caspase-dependent degradation of haematopoietic lineage cell-specific protein 1 (HS1) in Jurkat cells. Biochem. J. 364: 41–47
Vlietstra RJ, van Alewijk DC, Hermans KG, van Steenbrugge GJ and Trapman J (1998) Frequent inactivation of PTEN in prostate cancer cell lines and xenografts. Cancer Res. 58: 2720–2723
Shan X, Czar MJ, Bunnell SC, Liu P, Liu Y, Schwartzberg PL and Wange RL (2000) Deficiency of PTEN in Jurkat T cells causes constitutive localization of Itk to the plasma membrane and hyperresponsiveness to CD3 stimulation. Mol. Cell. Biol. 20: 6945–6957
Miller SJ, Lou DY, Seldin DC, Lane WS and Neel BG (2002) Direct identification of PTEN phosphorylation sites. FEBS Lett. 528: 145–153
Hanada M, Feng J and Hemmings BA (2004) Structure, regulation and function of PKB/AKT – a major therapeutic target. Biochim. Biophys. Acta. 1697: 3–16
Scheid MP and Woodgett JR (2003) Unravelling the activation mechanisms of protein kinase B/Akt. FEBS Lett. 546: 108–112
Nicholson KM and Anderson NG (2002) The protein kinase B/Akt signalling pathway in human malignancy. Cell. Signal. 14: 381–395
Besson A, Robbins SM and Yong VW (1999) PTEN/MMAC1/TEP1 in signal transduction and tumorigenesis. Eur. J. Biochem. 263: 605–611
Sarno S, Reddy H, Meggio F, Ruzzene M, Davies SP, Donella-Deana A, Shugar D and Pinna LA (2001) Selectivity of 4,5,6,7-tetrabromobenzotriazole, an ATP site-directed inhibitor of protein kinase CK2 (‘casein kinase-2’). FEBS Lett. 496: 44–48
Sarno S, de Moliner E, Ruzzene M, Pagano MA, Battistutta R, Bain J, Fabbro D, Schoepfer J, Elliott M, Furet P, Meggio F, Zanotti G and Pinna LA (2003) Biochemical and three-dimensional-structural study of the specific inhibition of protein kinase CK2 by [5-oxo-5,6-dihydroindolo-(1,2-a)quinazolin-7-yl]acetic acid (IQA). Biochem. J. 374: 639–646
Vanhaesebroeck B and Alessi DR (2000) The PI3K-PDK1 connection: more than just a road to PKB. Biochem. J. 346: 561–576
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P and Hemmings BA. (1996) Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J. 15: 6541–6551
Sato S, Fujita N and Tsuruo T (2000) Modulation of Akt kinase activity by binding to Hsp90. Proc. Natl. Acad. Sci. USA 97: 10832–10837
Miyata Y and Nishida E (2004) CK2 controls multiple protein kinases by phosphorylating a kinase-targeting molecular chaperone, Cdc37. Mol. Cell. Biol. 24: 4065–4074
Lin R, Beauparlant P, Makris C, Meloche S and Hiscott J (1996) Phosphorylation of IκBα in the C-terminal PEST domain by casein kinase II affects intrinsic protein stability. Mol. Cell. Biol. 16: 1401–1409
Shen J, Channavajhala P, Seldin DC and Sonenshein GE (2001) Phosphorylation by the protein kinase CK2 promotes calpain-mediated degradation of IκBα. J. Immunology 167: 4919–4925
Kato Jr T, Delhase M, Hoffmann A and Karin M (2003) CK2 Is a C-Terminal IκB Kinase responsible for NF-κB activation during the UV response. Mol. Cell. 12: 829–839
Torres J, Rodriguez J, Myers MP, Valiente M, Graves JD, Tonks NK and Pulido R (2003) Phosphorylation-regulated cleavage of the tumor suppressor PTEN by caspase-3: implications for the control of protein stability and PTEN–protein interactions. J. Biol. Chem. 278: 30652–30660
Pinna LA (2002) Protein kinase CK2: a challenge to canons. J. Cell Sci. 115: 3873–3878
Li PF, Li J, Muller EC, Otto A, Dietz R and von Harsdorf R (2002) Phosphorylation by protein kinase CK2: a signaling switch for the caspase-inhibiting protein ARC. Mol. Cell. 10: 247–258
Ubeda M and Habener JF (2003) CHOP transcription factor phosphorylation by casein kinase 2 inhibits transcriptional activation. J. Biol. Chem. 278: 40514–40520
Marin O, Meggio F and Pinna LA (1994) Design and synthesis of two new peptide substrates for the specific and sensitive monitoring of casein-kinase-1 and -2. Biochem. Biophys. Res. Commun. 198: 898–905
Facchin S, Lopreiato R, Stocchetto S, Arrigoni G, Cesaro L, Marin O, Carignani G and Pinna LA (2002) Structure–function analysis of yeast piD261/Bud32, an atypical protein kinase essential for normal cell life. Biochem. J. 364: 457–463
Szyska R, Grankowski N, Felczak K and Shugar D (1995) Halogenated benzimidazoles and benzotriazoles as selective inhibitors of protein kinases CK I and CK II from Saccharomyces cerevisiae and other sources. Biochem. Biophys. Res. Commun. 208: 418–424
Elbashir SM, Harborth J, Weber K and Tuschl T (2002) Analysis of gene function in somatic mammalian cells using small interfering RNAs. Methods 26: 199–213
Perich JW, Meggio F, Reynolds EC, Marin O and Pinna LA (1992) Role of phosphorylated aminoacyl residues in generating atypical consensus sequences which are recognized by casein kinase-2 but not by casein kinase-1. Biochemistry 31: 5893–5897
Cross DA, Alessi DR, Cohen P, Andjelkovich M and Hemmings BA (1995) Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378: 785–789
Gharahdaghi F, Weinberg CR, Meagher DA, Imai BS and Mische SM (1999) Mass spectrometric identification of proteins from silver-stained polyacrylamide gel: a method for the removal of silver ions to enhance sensitivity. Electrophoresis 20: 601–605
Meyer HE, Swiderek K, Hoffmann-Posorske E, Korte H and Heilmeyer Jr LM (1987) Quantitative determination of phosphoserine by high-performance liquid chromatography as the phenylthiocarbamyl-S-ethylcysteine. Application to picomolar amounts of peptides and proteins. J. Chromatogr. 397: 113–121
Resing KA, Johnson RS and Walsh KA (1995) Mass spectrometric analysis of 21 phosphorylation sites in the internal repeat of rat profilaggrin, precursor of an intermediate filament associated protein. Biochemistry 34: 9477–9487
Hunt DF, Yates III JR, Shabanowitz J, Winston S and Hauer CR (1986) Protein sequencing by tandem mass spectrometry. Proc. Natl. Acad. Sci. USA 83: 6233–6237
Acknowledgements
We thank Sir Philip Cohen (University of Dundee, Scotland) for critical reading of the manuscript, Joseph Schoepfer (Novartis, Basel, Switzerland) for providing IQA, the protein production team of the Division of Signal Transduction Therapy at the University of Dundee, Scotland (coordinated by Hilary McLauchlan and James Hastie) for providing the active and inactive full-length Akt, Peter James for the availability of the Proteomic Facility at Wallemberg Laboratory II, Lund University (Sweden), Sonia Facchin (University of Padova, Italy) for providing piD261, Sandra Marmiroli (University of Modena e Reggio Emilia, Italy) for providing the cDNA for HA-tagged myrAkt1. This work was supported by grants from the Italian MIUR (PRIN 2003), AIRC, and European Commission (PRO-KINASERESEARCH 503467).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by JA Trapani
Rights and permissions
About this article
Cite this article
Di Maira, G., Salvi, M., Arrigoni, G. et al. Protein kinase CK2 phosphorylates and upregulates Akt/PKB. Cell Death Differ 12, 668–677 (2005). https://doi.org/10.1038/sj.cdd.4401604
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.cdd.4401604
Keywords
This article is cited by
-
SLC39A10 promotes malignant phenotypes of gastric cancer cells by activating the CK2-mediated MAPK/ERK and PI3K/AKT pathways
Experimental & Molecular Medicine (2023)
-
Oncoprotein SET dynamically regulates cellular stress response through nucleocytoplasmic transport in breast cancer
Cell Biology and Toxicology (2023)
-
Molecular targets and therapeutic strategies for triple-negative breast cancer
Molecular Biology Reports (2023)
-
CSNK2 in cancer: pathophysiology and translational applications
British Journal of Cancer (2022)
-
Simultaneous CK2/TNIK/DYRK1 inhibition by 108600 suppresses triple negative breast cancer stem cells and chemotherapy-resistant disease
Nature Communications (2021)