Abstract
Aim:
Paeoniflorin has shown to attenuate bleomycin-induced pulmonary fibrosis (PF) in mice. Because the epithelial-mesenchymal transition (EMT) in type 2 lung endothelial cells contributes to excessive fibroblasts and myofibroblasts during multiple fibrosis of tissues, we investigated the effects of paeoniflorin on TGF-β mediated pulmonary EMT in bleomycin-induced PF mice.
Methods:
PF was induced in mice by intratracheal instillation of bleomycin (5 mg/kg). The mice were orally treated with paeoniflorin or prednisone for 21 d. After the mice were sacrificed, lung tissues were collected for analysis. An in vitro EMT model was established in alveolar epithelial cells (A549 cells) incubated with TGF-β1 (2 ng/mL). EMT identification and the expression of related proteins were performed using immunohistochemistry, transwell assay, ELISA, Western blot and RT-qPCR.
Results:
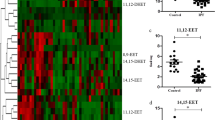
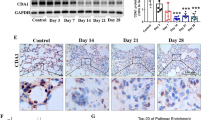
In PF mice, paeoniflorin (50, 100 mg·kg−1·d−1) or prednisone (6 mg·kg−1·d−1) significantly decreased the expression of FSP-1 and α-SMA, and increased the expression of E-cadherin in lung tissues. In A549 cells, TGF-β1 stimulation induced EMT, as shown by the changes in cell morphology, the increased cell migration, and the increased vimentin and α-SMA expression as well as type I and type III collagen levels, and by the decreased E-cadherin expression. In contrast, effects of paeoniflorin on EMT disappeared when the A549 cells were pretreated with TGF-β1 for 24 h. TGF-β1 stimulation markedly increased the expression of Snail and activated Smad2/3, Akt, ERK, JNK and p38 MAPK in A549 cells. Co-incubation with paeoniflorin (1–30 μmol/L) dose-dependently attenuated TGF-β1-induced expression of Snail and activation of Smad2/3, but slightly affected TGF-β1-induced activation of Akt, ERK, JNK and p38 MAPK. Moreover, paeoniflorin markedly increased Smad7 level, and decreased ALK5 level in A549 cells.
Conclusion:
Paeoniflorin suppresses the early stages of TGF-β mediated EMT in alveolar epithelial cells, likely by decreasing the expression of the transcription factors Snail via a Smad-dependent pathway involving the up-regulation of Smad7.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Bouros D . Pirfenidone for idiopathic pulmonary fibrosis. Lancet 2011; 377: 1727–9.
Raghu G, Weycker D, Edelsberg J, Bradford WZ, Oster G . Incidence and prevalence of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 2006; 174: 810–6.
Gross TJ, Hunninghake GW . Idiopathic pulmonary fibrosis. N Engl J Med 2001; 345: 517–25.
Polyakova V, Loeffler I, Hein S, Miyagawa S, Piotrowska I, Dammer S, et al. Fibrosis in endstage human heart failure: Severe changes in collagen metabolism and MMP/TIMP profiles. Int J Cardiol 2011; 151: 18–33.
Crouch E . Pathobiology of pulmonary fibrosis. Am J Physiol 1990; 259: L159–84.
Raghu G, Striker LJ, Hudson LD, Striker GE . Extracellular matrix in normal and fibrotic human lungs. Am Rev Respir Dis 1985; 131: 281–9.
Gabbiani G . The biology of the myofibroblast. Kidney Int 1992; 41: 530–2.
Willis BC, duBois RM, Borok Z . Epithelial origin of myofibroblasts during fibrosis in the lung. Proc Am Thorac Soc 2006; 3: 377–82.
Tanjore H, Xu XC, Polosukhin VV, Degryse AL, Li B, Han W, et al. Contribution of epithelial-derived fibroblasts to bleomycin-induced lung fibrosis. Am J Respir Crit Care Med 2009; 180: 657–65.
Li JZ, Wu JH, Yu SY, Shao QR, Dong XM . Inhibitory effects of paeoniflorin on lysophosphatidylcholine-induced inflammatory factor production in human umbilical vein endothelial cells. Int J Mol Med 2013; 31: 493–7.
Zhou H, Bian D, Jiao X, Wei Z, Zhang H, Xia Y, et al. Paeoniflorin protects against lipopolysaccharide-induced acute lung injury in mice by alleviating inflammatory cell infiltration and microvascular permeability. Inflamm Res 2011; 60: 981–90.
Zheng YQ, Wei W, Zhu L, Liu JX . Effects and mechanisms of Paeoniflorin, a bioactive glucoside from paeony root, on adjuvant arthritis in rats. Inflamm Res 2007; 56: 182–8.
Ji Y, Wang T, Wei ZF, Lu GX, Jiang SD, Xia YF, et al. Paeoniflorin, the main active constituent of Paeonia lactiflora roots, attenuates bleomycin-induced pulmonary fibrosis in mice by suppressing the synthesis of type I collagen. J Ethnopharmacol 2013; 149: 825–32.
Pirozzi G, Tirino V, Camerlingo R, Franco R, La Rocca A, Liguori E, et al. 2011. Epithelial to mesenchymal transition by TGFβ-1 induction increases stemness characteristics in primary non small cell lung cancer cell line. PLoS One 2011; 6: e 21548.
Smith BN, Odero-Marah VA . The role of Snail in prostate cancer. Cell Adh Migr 2012; 6: 433–41.
Kalluri R, Weinberg RA . The basics of epithelial-mesenchymal transition. J Clin Invest 2009; 199: 1420–8.
Habiel DM, Hogaboam C . Heterogeneity in fibroblast proliferation and survival in idiopathic pulmonary fibrosis. Front Pharmacol 2014; 5: 2.
Günther A, Korfei M, Mahavadi P, von der Beck D, Ruppert C, Markart P . Unravelling the progressive pathophysiology of idiopathic pulmonary fibrosis. Eur Respir Rev 2012; 21: 152–60.
Willis BC, Borok Z . TGF-beta-induced EMT: mechanisms and implications for fibrotic lung disease. Am J Physiol Lung Cell Mol Physiol 2007; 293: L525–34.
Yang H, Zhan L, Yang T, Wang L, Li C, Zhao J, et al. Ski prevents TGF-β-induced EMT and cell invasion by repressing SMAD-dependent signaling in non-small cell lung cancer. Oncol Rep 2015. doi: 10.3892/or.2015.3961.
Zhang F, Zhang X, Meng J, Zhao Y, Liu X, Liu Y, et al. ING5 inhibits cancer aggressiveness via preventing EMT and is a potential prognostic biomarker for lung cancer. Oncotarget 2015; 6: 16239–52.
Song P, Zheng JX, Xu J, Liu JZ, Wu LY, Liu C . β-Catenin induces A549 alveolar epithelial cell mesenchymal transition during pulmonary fibrosis. Mol Med Rep 2015; 11: 2703–10.
Zeisberg M, Neilson EG . Biomarkers for epithelial-mesenchymal transitions. J Clin Invest 2009; 119: 1429–37.
Yang J, Weinberg RA . Epithelial-mesenchymal transition: at the crossroads of development and tumor metastasis. Dev Cell 2008; 14: 818–29.
Felton VM, Borok Z, Willis BC . N-acetylcysteine inhibits alveolar epithelial-mesenchymal transition. Am J Physiol Lung Cell Mol Physiol 2009; 297: L805–12.
Ramos C, Becerril C, Montaño M, García-De-Alba C, Ramírez R, Checa M, et al. FGF-1 reverts epithelial-mesenchymal transition induced by TGF-{beta}1 through MAPK/ERK kinase pathway. Am J Physiol Lung Cell Mol Physiol 2010; 299: L222–31.
Maeda M, Johnson KR, Wheelock MJ . Cadherin switching: essential for behavioral but not morphological changes during an epithelium-to-mesenchyme transition. J Cell Sci 2005; 118: 873–87.
Xu J, Lamouille S, Derynck R . TGF-β-induced epithelial to mesenchymal transition. Cell Res 2009; 19: 156–72.
Nieto MA . The snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol 2002; 3: 155–66.
Olmeda D, Jordá M, Peinado H, Fabra A, Cano A . Snail silencing effectively suppresses tumour growth and invasiveness. Oncogene 2007; 26: 1862–74.
Peinado H, Olmeda D, Cano A . Snail, Zeb and bHLH factors in tumour progression: an alliance against the epithelial phenotype? Nat Rev Cancer 2007; 7: 415–28.
Postigo AA, Depp JL, Taylor JJ, Kroll KL . Regulation of Smad signaling through a differential recruitment of co-activators and co-repressors by ZEB proteins. EMBO J 2003; 22: 2453–62.
Massari ME, Murre C . Helix-loop-helix proteins: regulators of transcription in eucaryotic organisms. Mol Cell Biol 2000; 20: 429–40.
Vesuna F, van Diest P, Chen JH, Raman V . Twist is a transcriptional repressor of E-cadherin gene expression in breast cancer. Biochem Biophys Res Commun 2008; 367: 235–41.
Derynck R, Zhang YE . Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature 2003; 425: 577–84.
Flanders KC . Smad3 as a mediator of the fibrotic response. Int J Exp Pathol 2004; 85: 47–64.
Derynck R, Akhurst RJ, Balmain A . TGF-beta signaling in tumor suppression and cancer progression. Nat Genet 2001; 29: 117–29.
Sato M, Muragaki Y, Saika S, Roberts AB, Ooshima A . Targeted disruption of TGF-beta1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J Clin Invest 2003; 112: 1486–94.
Moustakas A, Heldin CH . Non-Smad TGF-β signals. J Cell Sci 2005; 118: 3572–84.
Yan W, Fu Y, Tian D, Liao J, Liu M, Wang B, et al. PI3 kinase/Akt signaling mediates epithelial-mesenchymal transition in hypoxic hepatocellular carcinoma cells. Biochem Biophys Res Commun 2009; 382: 631–6.
Xie L, Law BK, Chytil AM, Brown KA, Aakre ME, Moses HL . Activation of the Erk pathway is required for TGF-beta1-induced EMT in vitro. Neoplasia 2004; 6: 603–10.
Kolosova I, Nethery D, Kern JA . Role of Smad2/3 and p38 MAP kinase in TGF-β1-induced epithelial-mesenchymal transition of pulmonary epithelial cells. J Cell Physiol 2011; 226: 1248–54.
Santibañez JF . JNK mediates TGF-beta1-induced epithelial mesenchymal transdifferentiation of mouse transformed keratinocytes. FEBS Lett 2006; 580: 5385–91.
Kavsak P, Rasmussen RK, Causing CG, Bonni S, Zhu H, Thomsen GH, et al. Smad7 binds to Smurf2 to form an E3 ubiquitin ligase that targets the TGF beta receptor for degradation. Mol Cell 2000; 6: 1365–75.
Jung SM, Lee JH, Park J, Oh YS, Lee SK, Park JS, et al. Smad6 inhibits non-canonical TGF-β1 signalling by recruiting the deubiquitinase A20 to TRAF6. Nat Commun 2013; 4: 2562.
Hneino M, François A, Buard V, Tarlet G, Abderrahmani R, Blirando K, et al. The TGF-β/Smad repressor TG-interacting factor 1 (TGIF1) plays a role in radiation-induced intestinal injury independently of a Smad signaling pathway. PLoS One 2012; 7: e35672.
Acknowledgements
This work was funded by the Innovative Training Plan for Graduate Students of Jiangsu Province (No CXZZ11_0829) and the Priority Academic Program Development of the Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary information is available at the Acta Pharmacologica Sinica's website.
Supplementary information
Supplementary Information Figure S1 (download TIF )
The effects of paeoniflorin (Pae) and prednisone (Pred) on the expression levels of FSP-1, α-SMA and E-cadherin on bleomycin-induced fibrosis in mouse lung tissues. (TIF 5337 kb)
Supplementary Information Figure S2 (download TIF )
The effects of different concentrations of paeoniflorin (Pae) on TGF-β1-induced epithelial-mesenchymal transition in A549 cells.A549 cells were seeded into plates andincubated with serum-free RPMI 1640 for 2 h. (TIF 2305 kb)
PowerPoint slides
Rights and permissions
About this article
Cite this article
Ji, Y., Dou, Yn., Zhao, Qw. et al. Paeoniflorin suppresses TGF-β mediated epithelial-mesenchymal transition in pulmonary fibrosis through a Smad-dependent pathway. Acta Pharmacol Sin 37, 794–804 (2016). https://doi.org/10.1038/aps.2016.36
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2016.36
Keywords
This article is cited by
-
Biomaterial-based mechanical regulation facilitates scarless wound healing with functional skin appendage regeneration
Military Medical Research (2024)
-
Scutellarin Alleviates Ovalbumin-Induced Airway Remodeling in Mice and TGF-β-Induced Pro-fibrotic Phenotype in Human Bronchial Epithelial Cells via MAPK and Smad2/3 Signaling Pathways
Inflammation (2024)
-
Protective effects of Qing-Re-Huo-Xue formula on bleomycin-induced pulmonary fibrosis through the p53/IGFBP3 pathway
Chinese Medicine (2023)
-
PRDX1 negatively regulates bleomycin-induced pulmonary fibrosis via inhibiting the epithelial-mesenchymal transition and lung fibroblast proliferation in vitro and in vivo
Cellular & Molecular Biology Letters (2023)
-
Asarinin attenuates bleomycin-induced pulmonary fibrosis by activating PPARγ
Scientific Reports (2023)