Abstract
Aim:
Sweroside is an iridoid glycoside with diverse biological activities. In the present study we investigated the effects of sweroside on α-naphthylisothiocyanate (ANIT)-induced cholestatic liver injury in mice.
Methods:
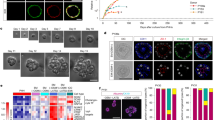
Mice received sweroside (120 mg·kg−1·d−1, ig) or a positive control INT-747 (12 mg·kg−1·d−1, ig) for 5 d, and ANIT (75 mg/kg, ig) was administered on d 3. The mice were euthanized on d 5, and serum biochemical markers, hepatic bile acids and histological changes were analyzed. Hepatic expression of genes related to pro-inflammatory mediators and bile acid metabolism was also assessed. Primary mouse hepatocytes were exposed to a reconstituted mixture of hepatic bile acids, which were markedly elevated in the ANIT-treated mice, and the cell viability and expression of genes related to pro-inflammatory mediators were examined.
Results:
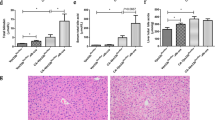
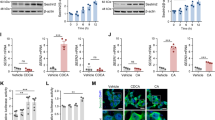
Administration of sweroside or INT-747 effectively ameliorated ANIT-induced cholestatic liver injury in mice, as evidenced by significantly reduced serum biochemical markers and attenuated pathological changes in liver tissues. Furthermore, administration of sweroside or INT-747 significantly decreased ANIT-induced elevation of individual hepatic bile acids, such as β-MCA, CA, and TCA, which were related to its effects on the expression of genes responsible for bile acid synthesis and transport as well as pro-inflammatory responses. Treatment of mouse hepatocytes with the reconstituted bile acid mixture induced significant pro-inflammatory responses without affecting the cell viability.
Conclusion:
Sweroside attenuates ANIT-induced cholestatic liver injury in mice by restoring bile acid synthesis and transport to their normal levels, as well as suppressing pro-inflammatory responses.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Wollbright BL, Li F, Xie Y, Farhood A, Fickert P, Trauner M, et al. Lithocholic acid feeding results in direct hepato-toxicity independent of neutrophil function in mice. Toxicol Lett 2014; 228: 56–66.
Chatterjee S, Bijsmans IT, van Mil SW, Augustijns P, Annaert P . Toxicity and intracellular accumulation of bile acids in sandwich-cultured rat hepatocytes: role of glycine conjugates. Toxicol In Vitro 2014; 28: 218–30.
Malhi H, Guicciardi ME, Gores GJ . Hepatocyte death: a clear and present danger. Physiol Rev 2010; 90: 1165–94.
Faubion WA, Guicciardi ME, Miyoshi H, Bronk SF, Roberts PJ, Svingen PA, et al. Toxic bile salts induce rodent hepatocyte apoptosis via direct activation of Fas. J Clin Invest 1999; 103: 137–45.
Woolbright BL, Dorko K, Antoine DJ, Clarke JI, Gholami P, Li F, et al. Bile acid-induced necrosis in primary human hepatocytes and in patients with obstructive cholestasis. Toxicol Appl Pharmacol 2015; 283: 168–77.
Allen K, Jaeschke H, Copple BL . Bile acids induce inflammatory genes in hepatocytes: a novel mechanism of inflammation during obstructive cholestasis. Am J Pathol 2011; 178: 175–86.
O'Brien KM, Allen KM, Rockwell CE, Towery K, Luyendyk JP, Copple BL . IL-17α synergistically enhances bile acid-induced inflammation during obstructive cholestasis. Am J Pathol 2013; 183: 1498–507.
Paumgartner G . Medical treatment of cholestatic liver diseases: from pathobiology to pharmacological targets. World J Gastroenterol 2006; 12: 4445–51.
Ji F, Deng H, Li Z . Eltrombopag for thrombocytopenic patients with hepatitis C virus infection and cirrhosis. Gastroenterology 2014; 147: 253–4.
Wang L, Wang J, Shi Y, Zhou X, Wang X, Li Z, et al. Identification of a primary biliary cirrhosis associated protein as lysosome-associated membrane protein-2. J Proteomics 2013; 91: 569–79.
Hirschfield GM, Chapman R, Karlsen TH, Lammert F, Lazaridis KN, Mason AL . The genetics of complex cholestatic disorders. Gastroenterology 2013; 144: 1357–74.
Yang F, Xu Y, Xiong A, He Y, Yang L, Wang Z . Evaluation of the protctive effect of Rhei Radix et Rhizoma against α-naphthylisothiocyanate induced liver injury based on metabolic profile of bile acids. J Ethnopharmacol 2012; 18; 144: 599–60.
Ding L, Zhang B, Zhan C, Yang L, Wang Z . Danning tablets attenuates α-naphthylisothiocyanate induced cholestasis by modulating the expression of transporters and metabolic enzymes. BMC Complement Altern Med 2014; 14: 249.
Yan JY, Ai G, Zhang XJ, Xu HJ, Huang ZM . Investigations of the total flavonoids extracted from flowers of Abelmoschus manihot (L) Medic against α-naphthylisothiocyanate-induced cholestatic liver injury in rats. J Ethnopharmacol 2015; 172: 202–13.
Erlinger S . What is cholestasis in 1985? J Hepatol 1985; 1: 687–93.
Kodali P, Wu P, Lahiji PA, Brown EJ, Maher JJ . ANIT toxicity toward mouse hepatocytes in vivo is mediated primarily by neutrophils via CD18. Am J Physiol Gastrointest Liver Physiol 2006; 291: G355–63.
Guo C, He L, Yao D, A J, Cao B, Ren J, et al. Alpha-naphthylisothiocyanate modulates hepatobiliary transporters in sandwich-cultured rat hepatocyte. Toxicol Lett 2014; 224: 93–100.
Choi Y, Yi NJ, Ko JS, Ko JM, Jin US, Kim HS, et al. Living donor liver transplantation for an infant with osteogenesis imperfecta and intrahepatic cholestasis: report of a case. J Korean Med Sci 2014; 29: 441–4.
Hirschfield GM, Heathcote EJ, Gershwin ME . Pathogenesis of cholestatic liver disease and therapeutic approaches. Gastroenterology 2010; 139: 1481–96.
Beuers U, Trauner M, Jansen P, Poupon R . New paradigms in the treatment of hepatic cholestasis: from UDCA to FXR, PXR and beyond. J Hepatol 2015; 62: S25–37.
Fickert P, Pollheimer MJ, Silbert D, Moustafa T, Halilbasic E, Krones E, et al. Differential effects of nor UDCA and UDCA in obstructive cholestasis in mice. J Hepatol 2013; 58: 1201–8.
Fiorucci S, Cipriani S, Mencarelli A, Baldelli F, Bifulco G, Zampella A . Farnesoid X receptor agonist for the treatment of liver and metabolic disorders: focus on 6-ethyl-CDCA. Mini Rev Med Chem 2011; 11: 753–62.
Tan RX, Wolfender JL, Ma WG, Zhang LX, Hostettmann K . Secoiridoids and anti-fungal aromatic acids from Gentiana algida. Phytochemistry 1996; 41: 111–6.
Hase K, Li JX, Basnet P, Xiong Q, Takamura SC, Namba T, et al. Hepatoprotective principles of Swertia japonica Makino on D-galactosamine/lipopolysaccharide-induced liver injury in mice. Chem Pharm Bull 1997; 45: 1823–7.
Mahendran G, Thamotharan G, Sengottuvelu S, Narmtha Bai V . Anti-diabetic activity of Swertia corymbosa (Griseb.) Wight ex C.B. Clarke aerial parts extract in streptozotocin induced diabetic rats. J Ethnopharmacol 2014; 151: 1175–83.
Sun H, Li L, Zhang A, Zhang N, Lv H, Sun W, et al. Protective effects of sweroside on human MG-63 cells and rat osteoblasts. Fitoterapia 2013; 84: 174–9.
Jeong YT, Jeong SC, Hwang JS, Kim JH . Modulation effects of sweroside isolated from the Lonicera japonica on melanin synthesis. Chem Biol Interact 2015; 238: 33–9.
He YM, Zhu S, Ge YW, Kazuma K, Zou K, Cai SQ, et al. The anti-inflammatory secoiridoid glycosides from Gentianae Scabrae Radix: the root and rhizome of Gentiana scabra. J Nat Med 2015; 69: 303–12.
Chen F, Ananthanarayanan M, Emre S, Neimark E, Bull LN, Knisely AS, et al. Progressive familial intrahepatic cholestasis, type 1, is associated with decreased farnesoid X receptor activity. Gastroenterology 2004; 126: 756–64.
Strautnieks SS, Bull LN, Knisely AS, Kocoshis SA, Dahl N, Arnell H, et al. A gene encoding a liver-specific ABC transporter is mutated in progressive familial intrahepatic cholestasis. Nat Genet 1998; 20: 233–8.
van Mil SWC, van der Woerd WL, van der Brugge G, Sturm E, Jansen PLM, Bull LN, et al. Benign recurrent intrahepatic cholestasis type 2 is caused by mutations in ABCB11. Gastroenterology 2004; 127: 379–84.
Cui YJ, Aleksunes LM, Tanaka Y, Goedken MJ, Klaassen CD . Compensatory induction of liver efflux transporters in response to ANIT-induced liver injury is impaired in FXR-null mice. Toxicol Sci 2009; 110: 47–60.
Thatch KA, Schwartz MZ, Yoo EY, Mendelson KG, Duke DS . Modulation of the inflammatory response and apoptosis using epidermal growth factor and hepatocyte growth factor in a liver injury model: a potential approach to the management and treatment of cholestatic liver disease. J Pediatr Surg 2008; 43: 2169–73.
Wang T, Zhou ZX, Sun LX, Li X, Xu ZM, Chen M, et al. Resveratrol effectively attenuates α-naphthyl-isothiocyanate-induced acute cholestasis and liver injury through choleretic and anti-inflammatory mechanisms. Acta Pharmacol Sin 2014; 35: 1527–36.
Liu Y, Binz J, Numerick MJ, Dennis S, Luo G, Desai B, et al. Hepatoprotection by the farnesoid X receptor agonist GW4064 in rat models of intrahepatic and extrahepatic cholestasis. J Clin Invest 2003; 112: 1878–87.
Guo C, He L, Yao D, A J, Cao B, Ren J, et al. Alpha-naphthylisothiocyanate modulates hepatobiliary transporters in sandwich-cultured rat hepatocytes. Toxicol Lett 2014; 224: 93–100.
Keppler D . Multidrug resistance proteins (MRPs, ABCCs): importance for pathophysiology and drug therapy. Handb Exp Pharmacol 2011; 201: 299–323.
Dietrich CG, Ottenhoff R, de Waart DR, Oude Elferink RPJ . Role of Mrp2 and GSH in intrahepatic cycling of toxins. Toxicology 2001; 167: 73–81.
Boyer JL, Trauner M, Mennone A, Soroka CJ, Cai SY, Moustafa T, et al. Up-regulation of a basolateral FXR-dependent bile acid efflux transporter OST alpha-OST beta in cholestasis in humans and rodents. Am J Physiol Gastrointest Liver Physiol 2006; 290: G1124–30.
Zollner G, Fickert P, Zenz R, Fuchsbichler A, Stumptner C, Kenner L, et al. Hepatobiliary transporter expression in percutaneous liver biopsies of patients with cholestatic liver diseases. Hepatology 2001; 33: 633–46.
Zhang Y, Csanaky IL, Lehman-McKeeman LD, Klaassen CD . Loss of organic anion transporting polypeptide 1a1 increases deoxycholic acid absorption in mice by increasing intestinal permeability. Toxicol Sci 2011; 124: 251–60.
Denson LA, Bohan A, Held MA, Boyer JL . Organ-specific alterations in RAR alpha: RXR alpha abundance regulate rat Mrp2 (Abcc2) expression in obstructive cholestasis. Gastroenterology 2002; 123: 599–607.
Jahan A, Chiang JY . Cytokine regulation of human sterol 12alpha-hydroxylase (CYP8B1) gene. Am J Physiol Gastrointest Liver Physiol 2005; 288: G685–95.
Li T, Jahan A, Chiang JY . Bile acids and cytokines inhibit the human cholesterol 7 alpha-hydroxylase gene via the JNK/c-jun pathway in human liver cells. Hepatology 2006; 43: 1202–10.
Geier A, Dietrich CG, Trauner M, Gartung C . Extrahepatic cholestasis down-regulates Oatp1 by TNF-alpha signaling without affecting Oatp2 and Oatp4 expression and sodium-independent bile salt uptake in rat liver. Liver Int 2007; 27: 1056–65.
Acknowledgements
This work was financially supported by the Natural Science Foundations of China (81222053 and 81573581), the Program for New Century Excellent Talents in University (NCET-12-1056) and the National S&T Major Special Projects (2014ZX09301306-007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information is available on the website of Acta Pharmacologica Sinica.
Supplementary information
Supplementary Figure S1 (download DOCX )
Hepatoprotection of sweroside against ANIT-induced hepatotoxicity and cholestasis. (DOCX 119 kb)
Supplementary Figure S2 (download DOCX )
Serum TC, TG, LDL-C and HDL-C levels were affected by sweroside treatment. (DOCX 95 kb)
Supplementary Figure S3 (download DOCX )
Sweroside demonstrated no obvious liver toxicity by histological study and biochemical analysis. (DOCX 1968 kb)
Supplementary Figure S4 (download DOCX )
Cells viability and cells apoptosis were not affected by sweroside exposure. (DOCX 193 kb)
Rights and permissions
About this article
Cite this article
Yang, Ql., Yang, F., Gong, Jt. et al. Sweroside ameliorates α-naphthylisothiocyanate-induced cholestatic liver injury in mice by regulating bile acids and suppressing pro-inflammatory responses. Acta Pharmacol Sin 37, 1218–1228 (2016). https://doi.org/10.1038/aps.2016.86
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2016.86
Keywords
This article is cited by
-
Anti-inflammatory Effects of Sweroside on LPS-Induced ALI in Mice Via Activating SIRT1
Inflammation (2021)
-
Sweroside ameliorated carbon tetrachloride (CCl4)-induced liver fibrosis through FXR-miR-29a signaling pathway
Journal of Natural Medicines (2020)
-
Protective Effects of Sweroside on IL-1β-Induced Inflammation in Rat Articular Chondrocytes Through Suppression of NF-κB and mTORC1 Signaling Pathway
Inflammation (2019)