Abstract
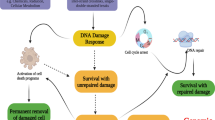
P53-induced protein with a death domain (PIDD) was cloned as a death domain (DD)-containing protein whose expression is induced by p53. It was later described as the core of a molecular platform-activating caspase-2, named the PIDDosome. These first results pointed towards a role for PIDD in apoptosis, in response to DNA damage. Identification of new PIDDosome complexes involved in DNA repair and nuclear factor-κB signaling challenged this early concept. PIDD functions are growing as new complexes and new interaction partners are being discovered, and as additional functions are being revealed. A fascinating feature of PIDD lies within its complex and tight regulation mechanisms, which allow the molecule to fine-tune its different functions: from transcriptional regulation to the expression of different isoforms, and from the interaction with regulatory proteins to an ingenious post-translational cleavage mechanism generating various active fragments with specific functions. Further studies still need to be carried out to provide answers to many unresolved issues and to reconcile conflicting results. This review aims at providing an overview of the current PIDD knowledge status.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- Apaf-1:
-
apoptotic protease-activating factor 1
- CARD:
-
caspase activation and recruitment domain
- Chk1:
-
checkpoint kinase 1
- DD:
-
death domain
- ER:
-
endoplasmic reticulum
- IAP:
-
inhibitor of apoptosis protein
- kDa:
-
kiloDalton
- LRDD:
-
leucine-rich repeats containing protein with a death domain
- LRR:
-
leucine-rich repeats
- ∣NF-κB:
-
nuclear factor-κB
- NLR:
-
Nod-like receptors
- NLRP3:
-
Nod-like receptors protein 3
- PCNA:
-
proliferating cell nuclear antigen
- PIDD:
-
p53-induced protein with a death domain
- RAIDD:
-
RIP-associated ICH-1/CED-3 homologous protein with a death domain
- RIP:
-
receptor-interacting protein
- siRNA:
-
small interfering RNA
- UPA:
-
uncharacterized protein domain in UNC5, PIDD and Ankyrin family of proteins
- VAD-fmk:
-
N-benzyloxycarbonyl-Val-Ala-Asp-fluoromethylketone
- ZUD:
-
Zu5 and DD-containing inhibitor of NF-κB
References
Telliez JB, Bean KM, Lin LL . LRDD, a novel leucine rich repeat and death domain containing protein. Biochim Biophys Acta 2000; 1478: 280–288.
Lin Y, Ma W, Benchimol S . Pidd, a new death-domain-containing protein, is induced by p53 and promotes apoptosis. Nat Genet 2000; 26: 122–127.
Wang R, Wei Z, Jin H, Wu H, Yu C, Wen W et al. Autoinhibition of UNC5b revealed by the cytoplasmic domain structure of the receptor. Mol Cell 2009; 33: 692–703.
Tinel A, Tschopp J . The PIDDosome, a protein complex implicated in activation of caspase-2 in response to genotoxic stress. Science 2004; 304: 843–846.
Pick R, Badura S, Bosser S, Zornig M . Upon intracellular processing, the C-terminal death domain-containing fragment of the p53-inducible PIDD/LRDD protein translocates to the nucleoli and interacts with nucleolin. Biochem Biophys Res Commun 2006; 349: 1329–1338.
Tinel A, Janssens S, Lippens S, Cuenin S, Logette E, Jaccard B et al. Autoproteolysis of PIDD marks the bifurcation between pro-death caspase-2 and pro-survival NF-kappaB pathway. EMBO J 2007; 26: 197–208.
Read SH, Baliga BC, Ekert PG, Vaux DL, Kumar S . A novel Apaf-1-independent putative caspase-2 activation complex. J Cell Biol 2002; 159: 739–745.
Baliga BC, Read SH, Kumar S . The biochemical mechanism of caspase-2 activation. Cell Death Differ 2004; 11: 1234–1241.
Bouchier-Hayes L, Oberst A, McStay GP, Connell S, Tait SW, Dillon CP et al. Characterization of cytoplasmic caspase-2 activation by induced proximity. Mol Cell 2009; 35: 830–840.
Park HH, Logette E, Raunser S, Cuenin S, Walz T, Tschopp J et al. Death domain assembly mechanism revealed by crystal structure of the oligomeric PIDDosome core complex. Cell 2007; 128: 533–546.
Tu S, McStay GP, Boucher LM, Mak T, Beere HM, Green DR . In situ trapping of activated initiator caspases reveals a role for caspase-2 in heat shock-induced apoptosis. Nat Cell Biol 2006; 8: 72–77.
Oliver TG, Meylan E, Chang GP, Xue W, Burke JR, Humpton TJ et al. Caspase-2-mediated cleavage of Mdm2 creates a p53-induced positive feedback loop. Mol Cell 2011; 43: 57–71.
Tinel A, Eckert MJ, Logette E, Lippens S, Janssens S, Jaccard B et al. Regulation of PIDD auto-proteolysis and activity by the molecular chaperone Hsp90. Cell Death Differ 2011; 18: 506–515.
Milleron RS, Bratton SB . Heat shock induces apoptosis independently of any known initiator caspase-activating complex. J Biol Chem 2006; 281: 16991–17000.
Shelton SN, Dillard CD, Robertson JD . Activation of caspase-9, but not caspase-2 or caspase-8, is essential for heat-induced apoptosis in Jurkat cells. J Biol Chem 2010; 285: 40525–40533.
Krumschnabel G, Sohm B, Bock F, Manzl C, Villunger A . The enigma of caspase-2: the laymen's view. Cell Death Differ 2009; 16: 195–207.
Kumar S . Caspase 2 in apoptosis, the DNA damage response and tumour suppression: enigma no more? Nat Rev Cancer 2009; 9: 897–903.
Chen F, Ding X, Ding Y, Xiang Z, Li X, Ghosh D et al. Proinflammatory caspase-2-mediated macrophage cell death induced by a rough attenuated Brucella suis strain. Infect Immun 2011; 79: 2460–2469.
Chen F, He Y . Caspase-2 mediated apoptotic and necrotic murine macrophage cell death induced by rough Brucella abortus. PLoS One 2009; 4: e6830.
Kitevska T, Spencer DM, Hawkins CJ . Caspase-2: controversial killer or checkpoint controller? Apoptosis: An International Journal on Programmed Cell Death 2009; 14: 829–848.
Jesenberger V, Procyk KJ, Yuan J, Reipert S, Baccarini M . Salmonella-induced caspase-2 activation in macrophages: a novel mechanism in pathogen-mediated apoptosis. J Exp Med 2000; 192: 1035–1046.
Vakifahmetoglu-Norberg H, Zhivotovsky B . The unpredictable caspase-2: what can it do? Trends Cell Biol 2010; 20: 150–159.
Berube C, Boucher LM, Ma W, Wakeham A, Salmena L, Hakem R et al. Apoptosis caused by p53-induced protein with death domain (PIDD) depends on the death adapter protein RAIDD. Proc Natl Acad Sci USA 2005; 102: 14314–14320.
Baptiste-Okoh N, Barsotti AM, Prives C . A role for caspase 2 and PIDD in the process of p53-mediated apoptosis. Proc Natl Acad Sci USA 2008; 105: 1937–1942.
O’Reilly LA, Ekert P, Harvey N, Marsden V, Cullen L, Vaux DL et al. Caspase-2 is not required for thymocyte or neuronal apoptosis even though cleavage of caspase-2 is dependent on both Apaf-1 and caspase-9. Cell Death Differ 2002; 9: 832–841.
Enoksson M, Robertson JD, Gogvadze V, Bu P, Kropotov A, Zhivotovsky B et al. Caspase-2 permeabilizes the outer mitochondrial membrane and disrupts the binding of cytochrome c to anionic phospholipids. J Biol Chem 2004; 279: 49575–49578.
Robertson JD, Enoksson M, Suomela M, Zhivotovsky B, Orrenius S . Caspase-2 acts upstream of mitochondria to promote cytochrome c release during etoposide-induced apoptosis. J Biol Chem 2002; 277: 29803–29809.
Robertson JD, Gogvadze V, Kropotov A, Vakifahmetoglu H, Zhivotovsky B, Orrenius S . Processed caspase-2 can induce mitochondria-mediated apoptosis independently of its enzymatic activity. EMBO Rep 2004; 5: 643–648.
Kim IR, Murakami K, Chen NJ, Saibil SD, Matysiak-Zablocki E, Elford AR et al. DNA damage- and stress-induced apoptosis occurs independently of PIDD. Apoptosis: An International Journal on Programmed Cell Death 2009; 14: 1039–1049.
Manzl C, Krumschnabel G, Bock F, Sohm B, Labi V, Baumgartner F et al. Caspase-2 activation in the absence of PIDDosome formation. J Cell Biol 2009; 185: 291–303.
Bergeron L, Perez GI, Macdonald G, Shi L, Sun Y, Jurisicova A et al. Defects in regulation of apoptosis in caspase-2-deficient mice. Genes Dev 1998; 12: 1304–1314.
Troy CM, Rabacchi SA, Hohl JB, Angelastro JM, Greene LA, Shelanski ML . Death in the balance: alternative participation of the caspase-2 and -9 pathways in neuronal death induced by nerve growth factor deprivation. J Neurosci 2001; 21: 5007–5016.
Oliver TG, Mercer KL, Sayles LC, Burke JR, Mendus D, Lovejoy KS et al. Chronic cisplatin treatment promotes enhanced damage repair and tumor progression in a mouse model of lung cancer. Genes Dev 2010; 24: 837–852.
Bouchier-Hayes L, Green DR . Real time with caspase-2. Cell Cycle 2010; 9: 12–13.
McStay GP, Salvesen GS, Green DR . Overlapping cleavage motif selectivity of caspases: implications for analysis of apoptotic pathways. Cell Death Differ 2008; 15: 322–331.
Slee EA, Harte MT, Kluck RM, Wolf BB, Casiano CA, Newmeyer DD et al. Ordering the cytochrome c-initiated caspase cascade: hierarchical activation of caspases-2, -3, -6, -7, -8, and -10 in a caspase-9-dependent manner. J Cell Biol 1999; 144: 281–292.
Janssens S, Tinel A, Lippens S, Tschopp J . PIDD mediates NF-kappaB activation in response to DNA damage. Cell 2005; 123: 1079–1092.
Hur GM, Lewis J, Yang Q, Lin Y, Nakano H, Nedospasov S et al. The death domain kinase RIP has an essential role in DNA damage-induced NF-kappa B activation. Genes Dev 2003; 17: 873–882.
Huang TT, Wuerzberger-Davis SM, Seufzer BJ, Shumway SD, Kurama T, Boothman DA et al. NF-kappaB activation by camptothecin. A linkage between nuclear DNA damage and cytoplasmic signaling events. J Biol Chem 2000; 275: 9501–9509.
Huang TT, Wuerzberger-Davis SM, Wu ZH, Miyamoto S . Sequential modification of NEMO/IKKgamma by SUMO-1 and ubiquitin mediates NF-kappaB activation by genotoxic stress. Cell 2003; 115: 565–576.
Logette E, Schuepbach-Mallepell S, Eckert MJ, Leo XH, Jaccard B, Manzl C et al. PIDD orchestrates translesion DNA synthesis in response to UV irradiation. Cell Death Differ 2011; 18: 1036–1045.
Baliga BC, Colussi PA, Read SH, Dias MM, Jans DA, Kumar S . Role of prodomain in importin-mediated nuclear localization and activation of caspase-2. J Biol Chem 2003; 278: 4899–4905.
Yoo NJ, Lee JW, Kim YJ, Soung YH, Kim SY, Nam SW et al. Loss of caspase-2, -6 and -7 expression in gastric cancers. Apmis 2004; 112: 330–335.
Zohrabian VM, Nandu H, Gulati N, Khitrov G, Zhao C, Mohan A et al. Gene expression profiling of metastatic brain cancer. Oncol Rep 2007; 18: 321–328.
Holleman A, den Boer ML, Kazemier KM, Beverloo HB, von Bergh AR, Janka-Schaub GE et al. Decreased PARP and procaspase-2 protein levels are associated with cellular drug resistance in childhood acute lymphoblastic leukemia. Blood 2005; 106: 1817–1823.
Ho LH, Taylor R, Dorstyn L, Cakouros D, Bouillet P, Kumar S . A tumor suppressor function for caspase-2. Proc Natl Acad Sci USA 2009; 106: 5336–5341.
Bradley G, Tremblay S, Irish J, MacMillan C, Baker G, Gullane P et al. The expression of p53-induced protein with death domain (Pidd) and apoptosis in oral squamous cell carcinoma. Br J Cancer 2007; 96: 1425–1432.
Lin J, Yang Q, Wilder PT, Carrier F, Weber DJ . The calcium-binding protein S100B down-regulates p53 and apoptosis in malignant melanoma. J Biol Chem 2010; 285: 27487–27498.
Heikaus S, Pejin I, Gabbert HE, Ramp U, Mahotka C . PIDDosome expression and the role of caspase-2 activation for chemotherapy-induced apoptosis in RCCs. Cell Oncol 2010; 32: 29–42.
Sidi S, Sanda T, Kennedy RD, Hagen AT, Jette CA, Hoffmans R et al. Chk1 suppresses a caspase-2 apoptotic response to DNA damage that bypasses p53, Bcl-2, and caspase-3. Cell 2008; 133: 864–877.
Andersen JL, Johnson CE, Freel CD, Parrish AB, Day JL, Buchakjian MR et al. Restraint of apoptosis during mitosis through interdomain phosphorylation of caspase-2. EMBO J 2009; 28: 3216–3227.
Castedo M, Perfettini JL, Roumier T, Valent A, Raslova H, Yakushijin K et al. Mitotic catastrophe constitutes a special case of apoptosis whose suppression entails aneuploidy. Oncogene 2004; 23: 4362–4370.
Andersen JL, Kornbluth S . A cut above the other caspases. Mol Cell 2009; 35: 733–734.
Seth R, Yang C, Kaushal V, Shah SV, Kaushal GP . P53-dependent caspase-2 activation in mitochondrial release of apoptosis-inducing factor and its role in renal tubular epithelial cell injury. J Biol Chem 2005; 280: 31230–31239.
Ren J, Shi M, Liu R, Yang QH, Johnson T, Skarnes WC et al. The Birc6 (Bruce) gene regulates p53 and the mitochondrial pathway of apoptosis and is essential for mouse embryonic development. Proc Natl Acad Sci USA 2005; 102: 565–570.
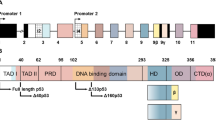
Cuenin S, Tinel A, Janssens S, Tschopp J . p53-induced protein with a death domain (PIDD) isoforms differentially activate nuclear factor-kappaB and caspase-2 in response to genotoxic stress. Oncogene 2008; 27: 387–396.
Huang L, Han D, Yang X, Qin B, Ji G, Yu L . PIDD4, a novel PIDD isoform without the LRR domain, can independently induce cell apoptosis in cytoplasm. Biochem Biophys Res Commun 2011; 407: 86–91.
Rosenblum JS, Blobel G . Autoproteolysis in nucleoporin biogenesis. Proc Natl Acad Sci USA 1999; 96: 11370–11375.
Hodel AE, Hodel MR, Griffis ER, Hennig KA, Ratner GA, Xu S et al. The three-dimensional structure of the autoproteolytic, nuclear pore-targeting domain of the human nucleoporin Nup98. Mol Cell 2002; 10: 347–358.
Paulus H . Protein splicing and related forms of protein autoprocessing. Annu Rev Biochem 2000; 69: 447–496.
Perler FB, Xu MQ, Paulus H . Protein splicing and autoproteolysis mechanisms. Curr Opin Chem Biol 1997; 1: 292–299.
Martinon F, Tschopp J . NLRs join TLRs as innate sensors of pathogens. Trends Immunol 2005; 26: 447–454.
Mayor A, Martinon F, De Smedt T, Petrilli V, Tschopp J . A crucial function of SGT1 and HSP90 in inflammasome activity links mammalian and plant innate immune responses. Nat Immunol 2007; 8: 497–503.
Stuttmann J, Parker JE, Noel LD . Staying in the fold: the SGT1/chaperone machinery in maintenance and evolution of leucine-rich repeat proteins. Plant Signal Behav 2008; 3: 283–285.
Acknowledgements
We thank all members of the group Tschopp who over the years helped in unraveling the functions of this fascinating protein PIDD: S Lippens, S Cuenin, E Logette, M Eckert, and K Ludigs. We would further like to thank N Aebi, S Hertig, and C Mattmann for their excellent technical support and all group Tschopp members for the fantastic discussions over the years.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by G Melino
Rights and permissions
About this article
Cite this article
Janssens, S., Tinel, A. The PIDDosome, DNA-damage-induced apoptosis and beyond. Cell Death Differ 19, 13–20 (2012). https://doi.org/10.1038/cdd.2011.162
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2011.162
Keywords
This article is cited by
-
The caspase-2 substrate p54nrb exhibits a multifaceted role in tumor cell death susceptibility via gene regulatory functions
Cell Death & Disease (2022)
-
Pathogenic variants in PIDD1 lead to an autosomal recessive neurodevelopmental disorder with pachygyria and psychiatric features
European Journal of Human Genetics (2021)
-
Uncovering the PIDDosome and caspase-2 as regulators of organogenesis and cellular differentiation
Cell Death & Differentiation (2020)
-
The expression and role of PIDD in retina after optic nerve crush
Journal of Molecular Histology (2020)
-
Apoptosis regulation by subcellular relocation of caspases
Scientific Reports (2018)