Abstract
Background/Objectives:
The orally administered para-amino benzoic acid (PABA) is known to have near 100% excretion in urine and is used as a measure of 24-h urine collection completeness (referred to as PABAcheck). The purpose was to examine the effect of including urine collections deemed incomplete based on PABAcheck in a dietary measurement error study.
Subjects/Methods:
The Observing Protein and Energy Nutrition (OPEN) study was conducted in 1999–2000 and included 484 men and women aged 40–69 years. A food frequency questionnaire and 24-h dietary recalls were evaluated using recovery biomarkers that included urinary nitrogen and potassium from two 24-h urine collections. Statistical modeling determined the measurement error properties of dietary assessment instruments. In the original analyses, PABAcheck was used as a measure of complete urine collection; incomplete collections were either excluded or adjusted to acceptable levels. The OPEN data were reanalyzed including all urine collections and by using criteria based on self-reported missing voids to assess the differences.
Results:
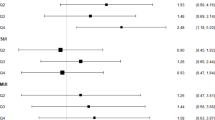
Means and coefficients of variation for biomarker-based protein and potassium intakes, and measurement error model-based correlations and attenuation factors were similar regardless of whether PABAcheck or missed voids were considered.
Conclusion:
PABAcheck may not be required in large population-based biomarker studies. However, until there are more analyses evaluating the necessity of a PABAcheck, it is recommended that PABA be given to all participants, but not necessarily analyzed. Then, PABAcheck could be used selectively as a marker of completeness among the collections in which low levels of biomarker are detected or for which noncompliance is suspected.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kaaks RJ . Biochemical markers as additional measurements in studies of the accuracy of dietary questionnaire measurements: conceptual issues. Am J Clin Nutr 1997; 65: 1232S–1239S.
Bingham S . Biomarkers in nutritional epidemiology. Public Health Nutr 2002; 5: 821–827.
Huse DM, Nelson RA, Briones ER, Hodgson PA . Urinary nitrogen excretion as objective measure of dietary intake. Am J Clin Nutr 1974; 27: 771–773.
Isaksson B . Urinary nitrogen output as validity test in dietary surveys. Am J Clin Nutr 1980; 33: 4–5.
Bingham SA, Cummings JH . Urine nitrogen as an independent validatory measure of dietary intake: a study of nitrogen balance in individuals consuming their normal diet. Am J Clin Nutr 1985; 42: 1276–1289.
Subar AF, Kipnis V, Troiano RP, Midthune D, Schoeller DA, Bingham S et al. Using intake biomarkers to evaluate the extent of dietary misreporting in a large sample of adults: The OPEN Study. Am J Epidemiol 2003; 158: 1–13.
Kipnis V, Subar AF, Midthune D, Freedman LS, Ballard-Barbash R, Troiano RP et al. The structure of dietary measurement error: results of the OPEN biomarker study. Am J Epidemiol 2003; 158: 14–21.
Neuhouser ML, Tinker L, Shaw PA, Schoeller D, Bingham SA, Van Horn L et al. Use of recovery biomarkers to calibrate nutrient consumption self-reports in the Women’s Health Initiative. Am J Epidemiol 2008; 67: 1247–1259.
Preis SR, Spiegelman D, Zhao BB, Moshfegh A, Baer DJ, Willett WC . Application of repeat-measure biomarker measurement error model to 2 validation studies: examination of the effect of within-person variation in biomarker measurements. Am J Epidemiol 2011; 173: 683–694.
Crispin SP, de Vries JHM, Geelen A, Souverein OW, Hulshof PJM, Lafay L et al. Two non-consecutive 24-h recalls using EPIC-Soft software are sufficiently valid for comparing protein and potassium intake between five European centers—results from the European Food Consumption Validation (EFCOVAL) study. Br J Nutr 2011; 105: 447–458.
Tasevska N, Runswick SA, Bingham SA . Urinary potassium is as reliable as urinary nitrogen for use as a recovery biomarker in dietary studies of free living individuals. J Nutr 2006; 136: 1334–1340.
Bingham SA, Cummings JH . 4 amino benzoic acid as a marker for the assessment of completeness of 24 h urine collections. Clin Sci 1983; 64: 629–635.
Roberts SB, Morrow FD, Evans WJ, Shepard DC, Dallal GE, Meredith CN et al. Use of p-aminobenzoic acid to monitor compliance with prescribed dietary regimens during metabolic balance studies in man. Am J Clin Nutr 1990; 51: 385–388.
Leclerq C, Maiani G, Polito A, Ferro-Luzzi A . Use of PABA test to check completeness of 24-h urine collections in elderly subjects. Nutrition 1991; 7: 350–354.
Jakobsen J, Pedersen AN, Ovesen L . Para-aminobenzoic acid (PABA) used as a marker for completeness of 24 h urine: effects of age and dosage scheduling. Eur J Clin Nutr 2003; 57: 138–142.
Arab L, Tseng C-H, Ang A, Jardack P . Validity of a multipass, web-based 24-hour self-administered recall for assessment of total energy intake in blacks and whites. Am J Epidemiol 2011; 174: 1256–1265.
Subar AF, Thompson FE, Kipnis V, Midthune D, Hurwitz P, McNutt S et al. Comparative validation of the Block, Willett, and National Cancer Institute food frequency questionnaires: the Eating at America's Table Study. Am J Epidemiol 2001; 154: 1089–1099.
Raper N, Perloff B, Ingwersen L, Steinfeldt L, Anand J . An overview of USDA’s Dietary Intake Data System. J Food Comp Anal 2004; 17: 545–555.
Bingham SA, Williams DRR, Cole TJ, Price CP, Cummings JH . Reference values for analytes of 24 h urine samples known to be complete. Ann Clin Biochem 1988; 25: 610–619.
Johansson G, Bingham S, Vahter M . A method to compensate for incomplete 24 h urine collections in nutritional epidemiology studies. Public Health Nutr 1999; 2: 587–591.
Jakobsen J, Ovesen L, Fagt S, Pederson AN . Para-aminobenzoic acid used as a marker for completeness of 24 h urine: assessment of control limits for a specific HPLC method. Eur J Clin Nutr 1997; 51: 514–519.
Berg JD, Chesner I, Lawson N . Practical Assessment of the NBT-PABA pancreatic function test using HPLC determination of p-aminobenzoic acid in urine. Ann Clin Biochem 1985; 22: 586–590.
Freedman LS, Midthune D, Carroll RJ, Krebs-Smith S, Subar AF, Troiano RP et al. Adjustments to improve the estimation of usual dietary intake distributions in the population. J Nutr 2004; 134: 1836–1843.
Schatzkin A, Kipnis V, Carroll RJ, Midthune D, Subar AF, Bingham S et al. A comparison of a food frequency questionnaire with a 24-hour recall for use in an epidemiological cohort study: results from the biomarker-based Observing Protein and Energy Nutrition (OPEN) study. Int J Epidemiol 2003; 32: 1054–1062.
Carroll RJ, Midthune D, Freedman LS, Kipnis V . Seemingly unrelated measurement error models, with application to nutritional epidemiology. Biometrics 2006; 62: 75–84.
Freedman LS, Schatzkin A, Midthune D, Kipnis V . Dietary measurement error in nutritional cohort studies. J Natl Cancer Inst 2011; 103: 1086–1092.
Acknowledgements
This work was funded by the US National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Subar, A., Midthune, D., Tasevska, N. et al. Checking for completeness of 24-h urine collection using para-amino benzoic acid not necessary in the Observing Protein and Energy Nutrition study. Eur J Clin Nutr 67, 863–867 (2013). https://doi.org/10.1038/ejcn.2013.62
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ejcn.2013.62
Keywords
This article is cited by
-
Methods for the dietary assessment of adult kidney stone formers: a scoping review
Journal of Nephrology (2022)
-
Validity of an online 24-h recall tool (myfood24) for dietary assessment in population studies: comparison with biomarkers and standard interviews
BMC Medicine (2018)
-
Validation of the use of p-aminobenzoic acid to determine completeness of 24 h urine collections in surveys of diet and nutrition
European Journal of Clinical Nutrition (2018)
-
Applying recovery biomarkers to calibrate self-report measures of sodium and potassium in the Hispanic Community Health Study/Study of Latinos
Journal of Human Hypertension (2017)