Abstract
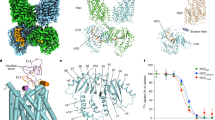
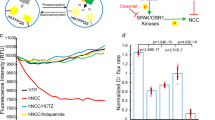
Gitelman syndrome (GS) is an autosomal recessive disorder characterized by hypokalemic metabolic alkalosis in conjunction with significant hypomagnesemia and hypocalciuria. The GS phenotype is caused by mutations in the solute carrier family 12, member 3 (SLC12A3) gene that encodes the thiazide-sensitive NaCl cotransporter (NCC). We analyzed DNA samples of 163 patients with a clinical suspicion of GS by direct sequencing of all 26 exons of the SLC12A3 gene. In total, 114 different mutations were identified, 31 of which have not been reported before. These novel variants include 3 deletions, 18 missense, 6 splice site and 4 nonsense mutations. We selected seven missense mutations to investigate their effect on NCC activity and plasma membrane localization by using the Xenopus laevis oocyte expression system. The Thr392Ile mutant did not display transport activity (probably class 2 mutation), while the Asn442Ser and Gln1030Arg NCC mutants showed decreased plasma membrane localization and consequently function, likely due to impaired trafficking (class 3 mutation). Even though the NaCl uptake was hampered for NCC mutants Glu121Asp, Pro751Leu, Ser475Cys and Tyr489His, the transporters reached the plasma membrane (class 4 mutation), suggesting an effect on NCC regulation or ion affinity. The present study shows the identification of 38 novel mutations in the SLC12A3 gene and provides insight into the mechanisms that regulate NCC.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
References
Gitelman HJ, Graham JB, Welt LG : A new familial disorder characterized by hypokalemia and hypomagnesemia. Trans AssocAm Phys 1966; 79: 221–235.
Melander O, Orho-Melander M, Bengtsson K et al: Genetic variants of thiazide-sensitive NaCl-cotransporter in Gitelman's syndrome and primary hypertension. Hypertension 2000; 36: 389–394.
Cruz DN, Shaer AJ, Bia MJ, Lifton RP, Simon DB : Gitelman's syndrome revisited: an evaluation of symptoms and health-related quality of life. Kidney Int 2001; 59: 710–717.
Spencer RW, Voyce MA : Familial hypokalaemia and hypomagnesaemia. A further family. Acta Paediatr Scand 1976; 65: 505–507.
Calo L, Punzi L, Semplicini A : Hypomagnesemia and chondrocalcinosis in Bartter's and Gitelman's syndrome: review of the pathogenetic mechanisms. Am J Nephrol 2000; 20: 347–350.
Hisakawa N, Yasuoka N, Itoh H et al: A case of Gitelman's syndrome with chondrocalcinosis. Endocr J 1998; 45: 261–267.
Simon DB, Nelson-Williams C, Bia MJ et al: Gitelman's variant of Bartter's syndrome, inherited hypokalaemic alkalosis, is caused by mutations in the thiazide-sensitive Na-Cl cotransporter. Nat Gen 1996; 12: 24–30.
Gamba G, Saltzberg SN, Lombardi M et al: Primary structure and functional expression of a cDNA encoding the thiazide-sensitive, electroneutral sodium-chloride cotransporter. Proc Natl Acad Sci USA 1993; 90: 2749–2753.
Plotkin MD, Kaplan MR, Verlander JW et al: Localization of the thiazide sensitive Na-Cl cotransporter, rTSC1 in the rat kidney. Kidney Int 1996; 50: 174–183.
Chobanian AV, Bakris GL, Black HR et al: The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA 2003; 289: 2560–2572.
Ellison DH, Velazquez H, Wright FS : Thiazide-sensitive sodium chloride cotransport in early distal tubule. Am J Physiol 1987; 253: F546–F554.
De Jong JC, Van Der Vliet WA, Van Den Heuvel LP, Willems PH, Knoers NV, Bindels RJ : Functional expression of mutations in the human NaCl cotransporter: evidence for impaired routing mechanisms in Gitelman's syndrome. J Am Soc Nephrol 2002; 13: 1442–1448.
Kunchaparty S, Palcso M, Berkman J et al: Defective processing and expression of thiazide-sensitive Na-Cl cotransporter as a cause of Gitelman's syndrome. Am J Physiol 1999; 277: F643–F649.
Riveira-Munoz E, Chang Q, Bindels RJ, Devuyst O : Gitelman's syndrome: towards genotype-phenotype correlations? Pediatr Nephrol 2007; 22: 326–332.
Sabath E, Meade P, Berkman J et al: Pathophysiology of functional mutations of the thiazide-sensitive Na-Cl cotransporter in Gitelman disease. Am J Physiol Renal Physiol 2004; 287: F195–F203.
Gamba G : Molecular physiology and pathophysiology of electroneutral cation-chloride cotransporters. Physiol Rev 2005; 85: 423–493.
Koolen DA, Nillesen WM, Versteeg MH et al: Screening for subtelomeric rearrangements in 210 patients with unexplained mental retardation using multiplex ligation dependent probe amplification (MLPA). J Med Genet 2004; 41: 892–899.
Gamba G, Miyanoshita A, Lombardi M et al: Molecular cloning, primary structure, and characterization of two members of the mammalian electroneutral sodium-(potassium)-chloride cotransporter family expressed in kidney. J Biol Chem 1994; 269: 17713–17722.
Cuppen E, van Ham M, Wansink DG, de Leeuw A, Wieringa B, Hendriks W : The zyxin-related protein TRIP6 interacts with PDZ motifs in the adaptor protein RIL and the protein tyrosine phosphatase PTP-BL. Eur J Cell Biol 2000; 79: 283–293.
Moreno E, Cristobal PS, Rivera M, Vazquez N, Bobadilla NA, Gamba G : Affinity-defining domains in the Na-Cl cotransporter: a different location for Cl- and thiazide binding. J Biol Chem 2006; 281: 17266–17275.
Pacheco-Alvarez D, Cristobal PS, Meade P et al: The Na+:Cl- cotransporter is activated and phosphorylated at the amino-terminal domain upon intracellular chloride depletion. J Biol Chem 2006; 281: 28755–28763.
Syren ML, Tedeschi S, Cesareo L et al: Identification of fifteen novel mutations in the SLC12A3 gene encoding the Na-Cl Co-transporter in Italian patients with Gitelman syndrome. Hum Mutat 2002; 20: 78.
Mastroianni N, Bettinelli A, Bianchetti M et al: Novel molecular variants of the Na-Cl cotransporter gene are responsible for Gitelman syndrome. Am J Hum Genet 1996; 59: 1019–1026.
Lemmink HH, Knoers NV, Karolyi L et al: Novel mutations in the thiazide-sensitive NaCl cotransporter gene in patients with Gitelman syndrome with predominant localization to the C-terminal domain. Kidney Int 1998; 54: 720–730.
Shao L, Liu L, Miao Z et al: A novel SLC12A3 splicing mutation skipping of two exons and preliminary screening for alternative splice variants in human kidney. Am J Nephrol 2008; 28: 900–907.
Vargas-Poussou R, Dahan K, Kahila D et al: Spectrum of mutations in Gitelman syndrome. J Am Soc Nephrol 2011; 22: 693–703.
Hoover RS, Poch E, Monroy A et al: N-glycosylation at two sites critically alters thiazide binding and activity of the rat thiazide-sensitive Na(+):Cl(−) cotransporter. J Am Soc Nephrol 2003; 14: 271–282.
Simon DB, Lifton RP : The molecular basis of inherited hypokalemic alkalosis: Bartter's and Gitelman's syndromes. Am J Physiol 1996; 271: F961–F966.
Frischmeyer PA, Dietz HC : Nonsense-mediated mRNA decay in health and disease. Hum Mol Genet 1999; 8: 1893–1900.
Colussi G, Bettinelli A, Tedeschi S et al: A thiazide test for the diagnosis of renal tubular hypokalemic disorders. Clin J Am Soc Nephrol 2007; 2: 454–460.
Brown CR, Hong-Brown LQ, Biwersi J, Verkman AS, Welch WJ : Chemical chaperones correct the mutant phenotype of the delta F508 cystic fibrosis transmembrane conductance regulator protein. Cell Stress Chaperones 1996; 1: 117–125.
Robben JH, Sze M, Knoers NV, Deen PM : Functional rescue of vasopressin V2 receptor mutants in MDCK cells by pharmacochaperones: relevance to therapy of nephrogenic diabetes insipidus. Am J Physiol Renal Physiol 2007; 292: F253–F260.
Acknowledgements
We are grateful to our colleague H Dimke for technical assistance and helpful suggestions. We thank all the patients, their families and the referring doctors for their participation. This study was financially supported by the Netherlands Organization for Scientific Research (ZonMw 9120.6110; ALW 700.55.302), a European Young Investigator award from the European Science Foundation (JGH), the Dutch Kidney foundation (C03.6017) (RJB), (C08.2252) (PS), The Swiss National Centre of Competence in Research (NCCR) Kidney Control of Homeostasis (Kidney.CH 507236) and the European Union Seventh Framework Programme (FP7/2007–2013; no 246539) (BG) and the European Community, FP7 (EUNEFRON 201590) (NVAMK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on European Journal of Human Genetics website
Supplementary information
Rights and permissions
About this article
Cite this article
Glaudemans, B., Yntema, H., San-Cristobal, P. et al. Novel NCC mutants and functional analysis in a new cohort of patients with Gitelman syndrome. Eur J Hum Genet 20, 263–270 (2012). https://doi.org/10.1038/ejhg.2011.189
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ejhg.2011.189
Keywords
This article is cited by
-
Gitelman syndrome with normocalciuria – a case report
BMC Nephrology (2022)
-
R158Q and G212S, novel pathogenic compound heterozygous variants in SLC12A3 of Gitelman syndrome
Frontiers of Medicine (2022)
-
Gitelman syndrome with a novel frameshift variant in SLC12A3 gene accompanied by chronic kidney disease and type 2 diabetes mellitus
CEN Case Reports (2022)
-
Allele-specific RT-PCR for the rapid detection of recurrent SLC12A3 mutations for Gitelman syndrome
npj Genomic Medicine (2021)
-
Early onset children’s Gitelman syndrome with severe hypokalaemia: a case report
BMC Pediatrics (2020)