Abstract
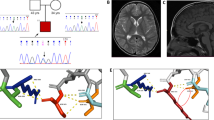
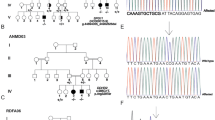
Variants in family 1 kinesin (KIF1A), which encodes a kinesin axonal motor protein, have been described to cause variable neurological manifestations. Recessive missense variants have led to spastic paraplegia, and recessive truncations to sensory and autonomic neuropathy. De novo missense variants cause developmental delay or intellectual disability, cerebellar atrophy and variable spasticity. We describe a family with father-to-son transmission of de novo variant in the KIF1A motor domain, in a phenotype of pure spastic paraplegia. Structural modeling of the predicted p.(Ser69Leu) amino acid change suggested that it impairs the stable binding of ATP to the KIF1A protein. Our study reports the first dominantly inherited KIF1A variant and expands the spectrum of phenotypes caused by heterozygous KIF1A motor domain variants to include pure spastic paraplegia. We conclude that KIF1A should be considered a candidate gene for hereditary paraplegias regardless of inheritance pattern.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Hirokawa N, Nitta R, Okada Y : The mechanisms of kinesin motor motility: lessons from the monomeric motor KIF1A. Nat Rev Mol Cell Biol 2009; 10: 877–884.
Okada Y, Yamazaki H, Sekine-Aizawa Y, Hirokawa N : The neuron-specific kinesin superfamily protein KIF1A is a unique monomeric motor for anterograde axonal transport of synaptic vesicle precursors. Cell 1995; 81: 769–780.
Shin H, Wyszynski M, Huh KH et al: Association of the kinesin motor KIF1A with the multimodular protein liprin-alpha. J Biol Chem 2003; 278: 11393–11401.
Yonekawa Y, Harada A, Okada Y et al: Defect in synaptic vesicle precursor transport and neuronal cell death in KIF1A motor protein-deficient mice. J Cell Biol 1998; 141: 431–441.
Riviere JB, Ramalingam S, Lavastre V et al: KIF1A, an axonal transporter of synaptic vesicles, is mutated in hereditary sensory and autonomic neuropathy type 2. Am J Hum Genet 2011; 89: 219–230.
Erlich Y, Edvardson S, Hodges E et al: Exome sequencing and disease-network analysis of a single family implicate a mutation in KIF1A in hereditary spastic paraparesis. Genome Res 2011; 21: 658–664.
Klebe S, Lossos A, Azzedine H et al: KIF1A missense mutations in SPG30, an autosomal recessive spastic paraplegia: distinct phenotypes according to the nature of the mutations. Eur J Hum Genet 2012; 20: 645–649.
Klebe S, Azzedine H, Durr A et al: Autosomal recessive spastic paraplegia (SPG30) with mild ataxia and sensory neuropathy maps to chromosome 2q37.3. Brain 2006; 129: 1456–1462.
Fink JK : Hereditary spastic paraplegia: clinico-pathologic features and emerging molecular mechanisms. Acta Neuropathol 2013; 126: 307–328.
Lee JR, Srour M, Kim D et al: De novo Mutations in the motor domain of KIF1A cause cognitive impairment, spastic paraparesis, axonal neuropathy and cerebellar atrophy. Hum Mutat 2014; doi:10.1089/hum.2012.003.
Okamoto N, Miya F, Tsunoda T et al: KIF1A mutation in a patient with progressive neurodegeneration. J Hum Genet 2014; 59: 639–64.
Hamdan FF, Gauthier J, Araki Y et al: Excess of de novo deleterious mutations in genes associated with glutamatergic systems in nonsyndromic intellectual disability. Am J Hum Genet 2011; 88: 306–316.
Ylikallio E, Johari M, Konovalova S et al: Targeted next-generation sequencing reveals further genetic heterogeneity in axonal Charcot-Marie-Tooth neuropathy and a mutation in HSPB1. Eur J Hum Genet 2014; 22: 522–527.
Sulonen AM, Ellonen P, Almusa H et al: Comparison of solution-based exome capture methods for next generation sequencing. Genome Biol 2011; 12: R94.
Robinson JT, Thorvaldsdottir H, Winckler W et al: Integrative genomics viewer. Nat Biotechnol 2011; 29: 24–26.
Lim ET, Wurtz P, Havulinna AS et al: Distribution and Medical Impact of Loss-of-Function Variants in the Finnish Founder Population. PLoS Genet 2014; 10: e1004494.
Kumar KR, Blair NF, Vandebona H et al: Targeted next generation sequencing in SPAST-negative hereditary spastic paraplegia. J Neurol 2013; 260: 2516–2522.
Adzhubei IA, Schmidt S, Peshkin L et al: A method and server for predicting damaging missense mutations. Nat Methods 2010; 7: 248–249.
Schwarz JM, Cooper DN, Schuelke M, Seelow D : MutationTaster2: mutation prediction for the deep-sequencing age. Nat Methods 2014; 11: 361–362.
DeLano W : PyMOL molecular viewer: updates and refinements. Abstr Pap Am Chem Soc 2009; 238: available at www.pymol.org.
Fiser A, Do RK, Sali A : Modeling of loops in protein structures. Protein Sci 2000; 9: 1753–1773.
Kikkawa M, Sablin EP, Okada Y, Yajima H, Fletterick RJ, Hirokawa N : Switch-based mechanism of kinesin motors. Nature 2001; 411: 439–445.
Nitta R, Kikkawa M, Okada Y, Hirokawa N : KIF1A alternately uses two loops to bind microtubules. Science 2004; 305: 678–683.
Esteves T, Durr A, Mundwiller E et al: Loss of association of REEP2 with membranes leads to hereditary spastic paraplegia. Am J Hum Genet 2014; 94: 268–277.
Lo Giudice T, Lombardi F, Santorelli FM, Kawarai T, Orlacchio A : Hereditary spastic paraplegia: clinical-genetic characteristics and evolving molecular mechanisms. Exp Neurol 2014; 261C: 518–539.
Yue Y, Sheng Y, Zhang HN et al: The CC1-FHA dimer is essential for KIF1A-mediated axonal transport of synaptic vesicles in C. elegans. Biochem Biophys Res Commun 2013; 435: 441–446.
Huo L, Yue Y, Ren J et al: The CC1-FHA tandem as a central hub for controlling the dimerization and activation of kinesin-3 KIF1A. Structure 2012; 20: 1550–1561.
Acknowledgements
We would like to thank the family for participation in the study. Riitta Lehtinen is thanked for her technical help. We also acknowledge the target enrichment, sequencing and variant calling pipeline analyses performed by the Institute for Molecular Medicine Finland. We wish to thank the following funding sources for support: Sigrid Jusélius Foundation (for HT), the Finnish Neuromuscular Disorders Association (for MA), University of Helsinki (for HT), the Academy of Finland (for HT and EY), Svenska kulturfonden (for EY), Arvid and Greta Olin’s Foundation (for EY), Finska Läkaresällskapet (for EY) and the Institute for Basic Science (for EK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Human Genetics website
Supplementary information
Rights and permissions
About this article
Cite this article
Ylikallio, E., Kim, D., Isohanni, P. et al. Dominant transmission of de novo KIF1A motor domain variant underlying pure spastic paraplegia. Eur J Hum Genet 23, 1427–1430 (2015). https://doi.org/10.1038/ejhg.2014.297
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ejhg.2014.297
This article is cited by
-
Clinical genetics of Charcot–Marie–Tooth disease
Journal of Human Genetics (2023)
-
Monoallelic KIF1A-related disorders: a multicenter cross sectional study and systematic literature review
Journal of Neurology (2022)
-
Challenges and Controversies in the Genetic Diagnosis of Hereditary Spastic Paraplegia
Current Neurology and Neuroscience Reports (2021)
-
KIF1A-related autosomal dominant spastic paraplegias (SPG30) in Russian families
BMC Neurology (2020)
-
KIF1A variants are a frequent cause of autosomal dominant hereditary spastic paraplegia
European Journal of Human Genetics (2020)