Abstract
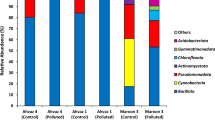
To compare microbial functional diversity in different oil-contaminated fields and to know the effects of oil contaminant and environmental factors, soil samples were taken from typical oil-contaminated fields located in five geographic regions of China. GeoChip, a high-throughput functional gene array, was used to evaluate the microbial functional genes involved in contaminant degradation and in other major biogeochemical/metabolic processes. Our results indicated that the overall microbial community structures were distinct in each oil-contaminated field, and samples were clustered by geographic locations. The organic contaminant degradation genes were most abundant in all samples and presented a similar pattern under oil contaminant stress among the five fields. In addition, alkane and aromatic hydrocarbon degradation genes such as monooxygenase and dioxygenase were detected in high abundance in the oil-contaminated fields. Canonical correspondence analysis indicated that the microbial functional patterns were highly correlated to the local environmental variables, such as oil contaminant concentration, nitrogen and phosphorus contents, salt and pH. Finally, a total of 59% of microbial community variation from GeoChip data can be explained by oil contamination, geographic location and soil geochemical parameters. This study provided insights into the in situ microbial functional structures in oil-contaminated fields and discerned the linkages between microbial communities and environmental variables, which is important to the application of bioremediation in oil-contaminated sites.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Aislabie J, Balks M, Foght J, Waterhouse E . (2004). Hydrocarbon spills on Antarctic soils: Effects and management. Environ Sci Technol 38: 1265–1274.
Al-Hashem M, Brain P, Omar S . (2007). Effects of oil pollution at Kuwait′s greater Al-Burgan oil field on polycyclic aromatic hydrocarbon concentrations in the tissues of the desert lizard Acanthodactylus scutellatus and their ant prey. Ecotoxicology 16: 551–555.
Al-Sarawi M, Massoud M . (1998). Preliminary assessment of oil contamination levels in soils contaminated with oil lakes in the greater Burgan oil fields, Kuwait. Water Air Soil Pollut 106: 493–504.
Allen J, Atekwana E, Atekwana E, Duris J, Werkema D, Rossbach S . (2007). The microbial community structure in petroleum-contaminated sediments corresponds to geophysical signatures. Appl Environ Microbiol 73: 2860–2870.
Atlas R . (1981). Microbial-degradation of petroleum-hydrocarbons: an environmental perspective. Microbiol Rev 45: 180–209.
Bhattacharya D, Sarma P, Krishnan S, Mishra S, Lal B . (2003). Evaluation of genetic diversity among Pseudomonas citronellolis strains isolated from oily sludge-contaminated sites. Appl Environ Microbiol 69: 1435–1441.
Braddock J, Ruth M, Catterall P, Walworth J, Mccarthy K . (1997). Enhancement and inhibition of microbial activity in hydrocarbon-contaminated arctic soils: Implications for nutrient-amended bioremediation. Environ Sci Technol 31: 2078–2084.
Din S, Dousari A, Literathy P . (2008). Evidence of hydrocarbon contamination from the Burgan oil field, Kuwait—Interpretations from thermal remote sensing data. J Environ Manage 86: 605–615.
Eisen M, Spellman P, Brown P, Botstein D . (1998). Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95: 14863–14868.
Gogoi B, Dutta N, Goswami P, Mohan T . (2003). A case study of bioremediation of petroleum-hydrocarbon contaminated soil at a crude oil spill site. Adv Environ Res 7: 767–782.
Gomes N, Kosheleva I, Abraham W, Smalla K . (2005). Effects of the inoculant strain Pseudomonas putida KT2442 (pNF142) and of naphthalene contamination on the soil bacterial community. FEMS Microbiol Ecol 54: 21–33.
Hamamura N, Olson S, Ward D, Inskeep W . (2006). Microbial population dynamics associated with crude-oil biodegradation in diverse soils. Appl Environ Microbiol 72: 6316–6324.
Hamid H, Kassim W, El Hishir A, El-Jawashi S . (2008). Risk assessment and remediation suggestion of impacted soil by produced water associated with oil production. Environ Monit Assess 145: 95–102.
He J, Xu Z, Hughes J . (2006). Molecular bacterial diversity of a forest soil under residue management regimes in subtropical Australia. FEMS Microbiol Ecol 55: 38–47.
He Z, Gentry T, Schadt C, Wu L, Liebich J, Chong S et al. (2007). GeoChip: a comprehensive microarray for investigating biogeochemical, ecological and environmental processes. ISME J 1: 67–77.
He Z, Xu M, Deng Y, Kang S, Kellogg L, Wu L et al. (2010). Metagenomic analysis reveals a marked divergence in the structure of belowground microbial communities at elevated CO2 . Ecol Lett 13: 564–575.
Head I, Jones D, Roling W . (2006). Marine microorganisms make a meal of oil. Nat Rev Microbiol 4: 173–182.
Heitkamp M, Franklin W, Cerniglia C . (1988). Microbial-metabolism of polycyclic aromatic-hydrocarbons-isolation and characterization of a pyrene-degrading bacterium. Appl Environ Microbiol 54: 2549–2555.
Huesemann M . (1995). Predictive model for estimating the extent of petroleum hydrocarbon biodegradation in contaminated soils. Environ Sci Technol 29: 7–18.
Joner E, Johansen A, Loibner A, Dela Cruz M, Szolar O, Portal J et al. (2001). Rhizosphere effects on microbial community structure and dissipation and toxicity of polycyclic aromatic hydrocarbons (PAHs) in spiked soil. Environ Sci Technol 35: 2773–2777.
Kanaly R, Harayama S . (2000). Biodegradation of high-molecular-weight polycyclic aromatic hydrocarbons by bacteria. J Bacteriol 182: 2059–2067.
Langworthy D, Stapleton R, Sayler G, Findlay R . (1998). Genotypic and phenotypic responses of a riverine microbial community to polycyclic aromatic hydrocarbon contamination. Appl Environ Microbiol 64: 3422–3428.
Legendre L, Legendre P . (1998). Numerical ecology. Elsevier Press: Amsterdam, The Netherlands.
Leigh M, Pellizari V, Uhlik O, Sutka R, Rodrigues J, Ostrom N et al. (2007). Biphenyl-utilizing bacteria and their functional genes in a pine root zone contaminated with polychlorinated biphenyls (PCBs). ISME J 1: 134–148.
Liang Y, Li G, Van Nostrand J, He Z, Wu L, Den Y et al. (2009a). Microarray-based analysis of microbial functional diversity along an oil contamination gradient in oil field. FEMS Microbiol 70: 324–333.
Liang Y, Van Nostrand J, Wang J, Zhang X, Zhou J, Li G . (2009b). Microarray-based functional gene analysis of soil microbial communities during ozonation and biodegradation of crude oil. Chemosphere 75: 193–199.
Lindstrom J, Barry R, Braddock J . (1999). Long-term effects on microbial communities after a subarctic oil spill. Soil Biol Biochem 31: 1677–1689.
Lu RK . (1999). Soil agricultural chemical analysis. China Agricultural Science and Technology Press: Nanjing, China.
Lundegard P, Johnson P . (2006). Source zone natural attenuation at petroleum hydrocarbon spill sites-II: Application to a former oil field. Ground Water Monit Remed 26: 93–106.
Maila M, Randima P, Dronen K, Cloete T . (2006). Soil microbial communities: Influence of geographic location and hydrocarbon pollutants. Soil Biol Biochem 38: 303–310.
Margesin R, Labbe D, Schinner F, Greer C, Whyte L . (2003). Characterization of hydrocarbon-degrading microbial populations in contaminated and pristine alpine soils. Appl Environ Microbiol 69: 3085–3092.
Megharaj M, Singleton I, McClure N, Naidu R . (2000). Influence of petroleum hydrocarbon contamination on microalgae and microbial activities in a long-term contaminated soil. Arch Environ Contam Toxicol 38: 439–445.
Oh Y, Sim D, Kim S . (2001). Effects of nutrients on crude oil biodegradation in the upper intertidal zone. Mar Pollut Bull 42: 1367–1372.
Paisse S, Coulon F, Goni-Urriza M, Peperzak L, McGenity T, Duran R . (2008). Structure of bacterial communities along a hydrocarbon contamination gradient in a coastal sediment. FEMS Microbiol Ecol 66: 295–305.
Ramette A, Tiedje J . (2007a). Biogeography: an emerging cornerstone for understanding prokaryotic diversity, ecology, and evolution. Microb Ecol 53: 197–207.
Ramette A, Tiedje J . (2007b). Multiscale responses of microbial life to spatial distance and environmental heterogeneity in a patchy ecosystem. Proc Natl Acad Sci USA 104: 2761–2766.
Rhee S, Liu X, Wu L, Chong S, Wan X, Zhou J . (2004). Detection of genes involved in biodegradation and biotransformation in microbial communities by using 50-mer oligonucleotide microarrays. Appl Environ Microbiol 70: 4303–4317.
Rooney-Varga J, Giewat M, Duddleston K, Chanton J, Hines M . (2007). Links between archaeal community structure, vegetation type and methanogenic pathway in Alaskan peatlands. FEMS Microbiol Ecol 60: 240–251.
Saul D, Aislabie J, Brown C, Harris L, Foght J . (2005). Hydrocarbon contamination changes the bacterial diversity of soil from around Scott Base, Antarctica. FEMS Microbiol Ecol 53: 141–155.
Shiaris M . (1989). Seasonal biotransformation of naphthalene, phenanthrene, and benzo[a]pyrene in surficial estuarine sediments. Appl Environ Microbiol 55: 1391–1399.
ter Braak CJF, Smilauer P . (1998). CANOCO Reference Manual and User's Guide to CANOCO for Windows: Software for Canonical Community Ordination, Version 4 edn. Microcomputer Power: New York, USA.
Tiquia S, Wu L, Chong S, Passovets S, Xu D, Xu Y et al. (2004). Evaluation of 50-mer oligonucleotide arrays for detecting microbial populations in environmental samples. Biotechniques 36: 664–675.
Torsvik V, Ovreas L . (2002). Microbial diversity and function in soil: from genes to ecosystems. Curr Opin Microbiol 5: 240–245.
Treves D, Xia B, Zhou J, Tiedje J . (2003). A two-species test of the hypothesis that spatial isolation influences microbial diversity in soil. Microb Ecol 45: 20–28.
van Beilen J, Smits T, Whyte L, Schorcht S, Rothlisberger M, Plaggemeier T et al. (2002). Alkane hydroxylase homologues in Gram-positive strains. Environ Microbiol 4: 676–682.
Van Hamme J, Singh A, Ward O . (2003). Recent advances in petroleum microbiology. Microbiol Mol Biol Rev 67: 503–549.
Van Nostrand J, Wu W, Wu L, Deng Y, Carley J, Carroll S et al. (2009). GeoChip-based analysis of functional microbial communities during the reoxidation of a bioreduced uranium-contaminated aquifer. Environ Microbiol 11: 2611–2626.
Wang F, Zhou H, Meng J, Peng X, Jiang L, Sun P et al. (2009). GeoChip-based analysis of metabolic diversity of microbial communities at the Juan de Fuca Ridge hydrothermal vent. Proc Natl Acad Sci USA 106: 4840–4845.
Ward D, Brock T . (1978). Hydrocarbon biodegradation in hypersaline environments. Appl Environ Microbiol 35: 353–359.
Whyte L, Hawari J, Zhou E, Bourbonniere L, Inniss W, Greer C . (1998). Biodegradation of variable-chain-length alkanes at low temperatures by a psychrotrophic Rhodococcus sp. Appl Environ Microbiol 64: 2578–2584.
Wu L, Kellogg L, Devol A, Tiedje J, Zhou J . (2008). Microarray-based characterization of microbial community functional structure and heterogeneity in marine sediments from the gulf of Mexico. Appl Environ Microbiol 74: 4516–4529.
Wu L, Liu X, Schadt C, Zhou J . (2006). Microarray-based analysis of subnanogram quantities of microbial community DNAs by using whole-community genome amplification. Appl Environ Microbiol 72: 4931–4941.
Wu L, Thompson D, Li G, Hurt R, Tiedje J, Zhou J . (2001). Development and evaluation of functional gene arrays for detection of selected genes in the environment. Appl Environ Microbiol 67: 5780–5790.
Xiong Z, Hu H, Wang Y, Fu G, Tan Z, Yan G . (1997). Comparative analyses of soil contaminant levels and plant species diversity at developing and disused oil well sites in Qianjiang Oilfield, China. Bull Environ Contam Toxicol 58: 667–672.
Xu M, Wu W, Wu L, He Z, Nostrand DVJ, Deng Y et al. (2010). Responses of microbial community functional structures to pilot-scale uranium in situ bioremediation. ISME J 1: 1–11.
Yergeau E, Arbour M, Brousseau R, Juck D, Lawrence J, Masson L et al. (2009). Microarray and real-time PCR analyses of the responses of high Arctic soil bacteria to hydrocarbon pollution and bioremediation treatments. Appl Environ Microbiol 75: 6258–6267.
Yergeau E, Kang S, He Z, Zhou J, Kowalchuk G . (2007). Functional microarray analysis of nitrogen and carbon cycling genes across an Antarctic latitudinal transect. ISME J 1: 163–179.
Yuste L, Corbella M, Turiegano M, Karlson U, Puyet A, Rojo F . (2000). Characterization of bacterial strains able to grow on high molecular mass residues from crude oil processing. FEMS Microbiol Ecol 32: 69–75.
Zhou J, Bruns M, Tiedje J . (1996). DNA recovery from soils of diverse composition. Appl Environ Microbiol 62: 316–322.
Zhou J, Kang S, Schadt C, Garten C . (2008). Spatial scaling of functional gene diversity across various microbial taxa. Proc Natl Acad Sci USA 105: 7768–7773.
Zhou J, Xia B, Treves D, Wu L, Marsh T, O’Neill R et al. (2002). Spatial and resource factors influencing high microbial diversity in soil. Appl Environ Microbiol 68: 326–334.
Acknowledgements
This work was supported by the National Natural Scientific Foundation of China (No. 40730738), the United States Department of Energy under the Environmental Remediation Science Program, and was part of the Virtual Institute for Microbial Stress and Survival (http://VIMSS.lbl.gov), which was supported by the U. S. Department of Energy, Office of Science, Office of Biological and Environmental Research, Genomics Program: GTL through contract DE-AC02-05CH11231 between Lawrence Berkeley National Laboratory and the U. S. Department of Energy, and was also supported by the Oklahoma Center for the Advancement of Science and Technology under the Oklahoma Applied Research Support Program.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on The ISME Journal website
Rights and permissions
About this article
Cite this article
Liang, Y., Van Nostrand, J., Deng, Y. et al. Functional gene diversity of soil microbial communities from five oil-contaminated fields in China. ISME J 5, 403–413 (2011). https://doi.org/10.1038/ismej.2010.142
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ismej.2010.142
Keywords
This article is cited by
-
Oil contamination drives the transformation of microbial communities and metabolic pathways involved in Phosphorous-cycling in coastal soil
Journal of Soils and Sediments (2024)
-
Spatiotemporal Dynamics of Bacterial Taxonomic and Functional Profiles in Estuarine Intertidal Soils of China Coastal Zone
Microbial Ecology (2023)
-
Diagnosing bioremediation of crude oil-contaminated soil and related geochemical processes at the field scale through microbial community and functional genes
Annals of Microbiology (2020)
-
Effects of soil properties on the remediation of diesel-contaminated soil by Triton X-100-aided washing
Environmental Science and Pollution Research (2020)
-
Computational-based insights into the phylogeny, structure, and function of Rhodococcus alkane-1-monooxygenase
3 Biotech (2020)