Abstract
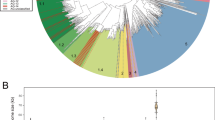
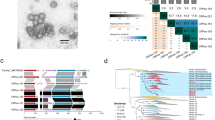
Viral genomes often contain genes recently acquired from microbes. In some cases (for example, psbA) the proteins encoded by these genes have been shown to be important for viral replication. In this study, using a unique search strategy on the Global Ocean Survey (GOS) metagenomes in combination with marine virome and microbiome pyrosequencing-based datasets, we characterize previously undetected microbial metabolic capabilities concealed within the genomes of uncultured marine viral communities. A total of 34 microbial gene families were detected on 452 viral GOS scaffolds. The majority of auxiliary metabolic genes found on these scaffolds have never been reported in phages. Host genes detected in viruses were mainly divided between genes encoding for different energy metabolism pathways, such as electron transport and newly identified photosystem genes, or translation and post-translation mechanism related. Our findings suggest previously undetected ways, in which marine phages adapt to their hosts and improve their fitness, including translation and post-translation level control over the host rather than the already known transcription level control.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Alperovitch A, Sharon I, Rohwer F, Aro E-M, Glaser F, Milo R et al. (2010). Reconstructing a puzzle: existence of cyanophages containing both photosystem-I & photosystem-II gene suites inferred from oceanic matagenomic datasets. Environ Microbiol 13: 24–32.
Angly F, Felts B, Breitbart M, Salamon P, Edwards R, Carlson C et al. (2006). The marine viromes of four oceanic regions. PLoS Biol 4: e368.
Aono J, Kangawa K, Matsuo H, Horiuchi T . (1982). Coliphage 434 tof protein: NH2-terminal amino acid sequence and kinetic and equilibrium measurements of DNA binding. Mol Gen Genet 186: 460–466.
Ayala-Castro C, Saini A, Outten FW . (2008). Fe-S cluster assembly pathways in bacteria. Microbiol Mol Biol Rev 72: 110–125, table of contents.
Battchikova N, Aro E-M . (2007). Cyanobacterial NDH-1 complexes: multiplicity in function and subunit composition. Physiol Plant 131: 22–32.
Beyreuther K, Gronenborn B . (1976). N-terminal sequence of phage lambda repressor. Gen Genet 147: 115–117.
Bingel-Erlenmeyer R, Kohler R, Kramer G, Sandikci A, Antolic S, Maier T et al. (2008). A peptide deformylase-ribosome complex reveals mechanism of nascent chain processing. Nature 452: 108–111.
Comeau AM, Arbiol C, Krisch HM . (2010). Gene network visualization and quantitative synteny analysis of more than 300 marine T4-like phage scaffolds from the GOS metagenome. Mol Biol Evol 27: 1935–1944.
Courey AJ . (2001). Cooperativity in transcriptional control. Curr Biol 11: R250–R252.
Dammeyer T, Bagby SC, Sullivan MB, Chisholm SW, Frankenberg-Dinkel N . (2008). Efficient phage-mediated pigment biosynthesis in oceanic cyanobacteria. Curr Biol 18: 442–448.
Dinsdale EA, Edwards RA, Hall D, Angly F, Breitbart M, Brulc JM et al. (2008a). Functional metagenomic profiling of nine biomes. Nature 452: 629–632.
Dinsdale EA, Pantos O, Smriga S, Edwards RA, Angly F, Wegley L et al. (2008b). Microbial ecology of four coral atolls in the Northern Line Islands. PLoS ONE 3: e1584.
Edgar RC . (2004). MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32: 1792–1797.
Giglione C, Boularot A, Meinnel T . (2004). Protein N-terminal methionine excision. Cell Mol Life Sci 61: 1455–1474.
Giglione C, Fieulaine S, Meinnel T . (2009). Cotranslational processing mechanisms: towards a dynamic 3D model. Trends Biochem Sci 34: 417–426.
Giglione C, Vallon O, Meinnel T . (2003). Control of protein life-span by N-terminal methionine excision. EMBO J 22: 13–23.
Guindon S, Delsuc F, Dufayard JF, Gascuel O . (2009). Estimating maximum likelihood phylogenies with PhyML. Methods Mol Biol 537: 113–137.
Hall A, Karplus PA, Poole LB . (2009). Typical 2-Cys peroxiredoxins: structures, mechanisms and functions. FEBS J 276: 2469–2477.
Hendrix RW, Smith MC, Burns RN, Ford ME, Hatfull GF . (1999). Evolutionary relationships among diverse bacteriophages and prophages: all the world's a phage. Proc Natl Acad Sci USA 96: 2192–2197.
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M et al. (2008). KEGG for linking genomes to life and the environment. Nucleic Acids Res 36: D480–D484.
Kathuria S, Martiny AC . (2011). Prevalence of a calcium-based alkaline phosphatase associated with the marine cyanobacterium Prochlorococcus and other ocean bacteria. Environ Microbiol 13: 74–83.
Klughammer B, Sültemeyer D, Badger MR, Price GD . (1999). The involvement of NAD(P)H dehydrogenase subunits, NdhD3 and NdhF3, in high-affinity CO2 uptake in Synechococcus sp. PCC7002 gives evidence for multiple NDH-1 complexes with specific roles in cyanobacteria. Mol Microbiol 32: 1305–1315.
Kramer G, Boehringer D, Ban N, Bukau B . (2009). The ribosome as a platform for co-translational processing, folding and targeting of newly synthesized proteins. Nat Struct Mol Biol 16: 589–597.
Lindell D, Jaffe JD, Johnson ZI, Church GM, Chisholm SW . (2005). Photosynthesis genes in marine viruses yield proteins during host infection. Nature 438: 86–89.
Lindell D, Sullivan MB, Johnson ZI, Tolonen AC, Rohwer F, Chisholm SW . (2004). Transfer of photosynthesis genes to and from Prochlorococcus viruses. Proc Natl Acad Sci USA 101: 11013–11018.
Maley GF, Guarino DU, Maley F . (1983). Complete amino acid sequence of an allosteric enzyme, T2 bacteriophage deoxycytidylate deaminase. J Biol Chem 258: 8290–8297.
Mann NH, Cook A, Millard A, Bailey S, Clokie M . (2003). Bacterial photosynthesis genes in a virus. Nature 424: 741.
Marsh JJ, Lebherz HG . (1992). Fructose-bisphosphate aldolases: an evolutionary history. Trends Biochem Sci 17: 110–113.
Martiny AC, Huang Y, Li W . (2009). Occurrence of phosphate acquisition genes in Prochlorococcus cells from different ocean regions. Environ Microbiol 11: 1340–1347.
Meinnel T, Blanquet S . (1993). Evidence that peptide deformylase and methionyl-tRNA(fMet) formyltransferase are encoded within the same operon in Escherichia coli. J Bacteriol 175: 7737–7740.
Meinnel T, Lazennec C, Dardel F, Schmitter JM, Blanquet S . (1996). The C-terminal domain of peptide deformylase is disordered and dispensable for activity. FEBS Lett 385: 91–95.
Millard A, Clokie MRJ, Shub DA, Mann NH . (2004). Genetic organization of the psbAD region in phages infecting marine Synechococcus strains. Proc Natl Acad Sci USA 101: 11007–11012.
Ogawa T, Mi H . (2007). Cyanobacterial NADPH dehydrogenase complexes. Photosynth Res 93: 69–77.
Ohkawa H, Pakrasi HB, Ogawa T . (2000). Two types of functionally distinct NAD(P)H dehydrogenases in Synechocystis sp. strain PCC6803. J Biol Chem 275: 31630–31634.
Paul JH . (2008). Prophages in marine bacteria: dangerous molecular time bombs or the key to survival in the seas? ISME J 2: 579–589.
Price MN, Dehal PS, Arkin AP . (2010). FastTree 2—approximately maximum-likelihood trees for large alignments. PLoS ONE 5: e9490.
Pruitt KD, Tatusova T, Klimke W, Maglott DR . (2009). NCBI reference sequences: current status, policy and new initiatives. Nucleic Acids Res 37: D32–D36.
Pruitt KD, Tatusova T, Maglott DR . (2007). NCBI reference sequences (RefSeq): a curated non-redundant sequence database of genomes, transcripts and proteins. Nucleic Acids Res 35: D61–D65.
Rogers MB, Patron NJ, Keeling PJ . (2007). Horizontal transfer of a eukaryotic plastid-targeted protein gene to cyanobacteria. BMC Biol 5: 26.
Rohwer F . (2003). Global phage diversity. Cell 113: 141.
Rohwer F, Thurber RV . (2009). Viruses manipulate the marine environment. Nature 459: 207–212.
Rohwer F, Segall AM, Steward G, Seguritan V, Breitbart M, Wolven F et al. (2000). The complete genomic sequence of the marine phage Roseophage SIO1 shares homology with non-marine phages. Limnol Oceanogr 45: 408–418.
Rusch DB, Halpern AL, Heidelberg KB, Sutton G, Williamson SJ, Yooseph S et al. (2007). The sorcerer II global ocean sampling expedition: I, the northwest Atlantic through the eastern tropical Pacific. PLoS Biol 5: e77.
Sauer RT, Anderegg R . (1978). Primary structure of the lambda repressor. Biochemistry 17: 1092–1100.
Sazanov LA, Hinchliffe P . (2006). Structure of the hydrophilic domain of respiratory complex I from Thermus thermophilus. Science 311: 1430–1436.
Seguritan V, Feng IW, Rohwer F, Swift M, Segall AM . (2003). Genome sequences of two closely related Vibrio parahaemolyticus phages, VP16T and VP16C. J Bacteriol 185: 6434–6447.
Shank PR, Hutchinson III CA, Edgell MH . (1977). Isolation and characterization of the four major proteins in the virion of bacteriophage phiX174. Biochemistry 16: 4545–4549.
Sharon I, Alperovitch A, Rohwer F, Haynes M, Glaser F, Atamna-Ismaeel N et al. (2009). Photosystem-I gene cassettes are present in marine virus genomes. Nature 461: 258–262.
Sharon I, Tzahor S, Williamson S, Shmoish M, Man-Aharonovich D, Rusch DB et al. (2007). Viral photosynthetic reaction center genes and transcripts in the marine environment. ISME J 1: 492–501.
Sullivan MB, Coleman ML, Weigele P, Rohwer F, Chisholm SW . (2005). Three Prochlorococcus cyanophage genomes: signature features and ecological interpretations. PLoS Biol 3: e144.
Sullivan MB, Huang KH, Ignacio-Espinoza JC, Berlin AM, Kelly L, Weigele PR et al. (2010). Genomic analysis of oceanic cyanobacterial myoviruses compared with T4-like myoviruses from diverse hosts and environments. Environ Microbiol 12: 3035–3056.
Tripathi BN, Bhatt I, Dietz KJ . (2009). Peroxiredoxins: a less studied component of hydrogen peroxide detoxification in photosynthetic organisms. Protoplasma 235: 3–15.
Williamson SJ, Rusch DB, Yooseph S, Halpern AL, Heidelberg KB, Glass JI et al. (2008). The sorcerer II global ocean sampling expedition: metagenomic characterization of viruses within aquatic microbial samples. PLoS ONE 3: e1456.
Yooseph S, Sutton G, Rusch DB, Halpern AL, Williamson SJ, Remington K et al. (2007). The sorcerer II global ocean sampling expedition: expanding the universe of protein families. PLoS Biol 5: e16.
Zhang P, Battchikova N, Jansen T, Appel J, Ogawa T, Aro E-M . (2004). Expression and functional roles of the two distinct NDH-1 complexes and the carbon acquisition complex NdhD3/NdhF3/CupA/Sll1735 in Synechocystis sp PCC 6803. Plant Cell 16: 3326–3340.
Acknowledgements
We thank D Lindell for her encouragement and ideas. We are indebted to B Gronenborn (CNRS, Gif, France) for stimulating discussions. This work was supported in part by grant 1203/06 from the Israel Science Foundation (OB), the CoE project 118637 from the Academy of Finland (E-MA), CNRS grant PEPS-2008 and ARC (Association pour la Recherche contre le Cancer, Villejuif) grant 4920 (CG and TM), and grant DBI-0850206 from the National Science Foundation (MB).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on The ISME Journal website
Supplementary information
Rights and permissions
About this article
Cite this article
Sharon, I., Battchikova, N., Aro, EM. et al. Comparative metagenomics of microbial traits within oceanic viral communities. ISME J 5, 1178–1190 (2011). https://doi.org/10.1038/ismej.2011.2
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ismej.2011.2
Keywords
This article is cited by
-
Distribution of soil viruses across China and their potential role in phosphorous metabolism
Environmental Microbiome (2022)
-
Metagenomic and metatranscriptomic insights into sulfate-reducing bacteria in a revegetated acidic mine wasteland
npj Biofilms and Microbiomes (2022)
-
Hybrid assembly of an agricultural slurry virome reveals a diverse and stable community with the potential to alter the metabolism and virulence of veterinary pathogens
Microbiome (2021)
-
Viral elements and their potential influence on microbial processes along the permanently stratified Cariaco Basin redoxcline
The ISME Journal (2020)
-
Diversities and potential biogeochemical impacts of mangrove soil viruses
Microbiome (2019)