Abstract
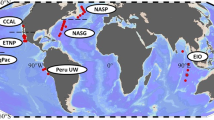
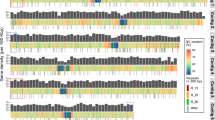
A central goal in ecology is to understand the factors affecting the temporal dynamics and spatial distribution of microorganisms and the underlying processes causing differences in community structure and composition. However, little is known in this respect for photosynthetic picoeukaryotes (PPEs), algae that are now recognised as major players in marine CO2 fixation. Here, we analysed dot blot hybridisation and cloning–sequencing data, using the plastid-encoded 16S rRNA gene, from seven research cruises that encompassed all four ocean biomes. We provide insights into global abundance, α- and β-diversity distribution and the environmental factors shaping PPE community structure and composition. At the class level, the most commonly encountered PPEs were Prymnesiophyceae and Chrysophyceae. These taxa displayed complementary distribution patterns, with peak abundances of Prymnesiophyceae and Chrysophyceae in waters of high (25:1) or low (12:1) nitrogen:phosphorus (N:P) ratio, respectively. Significant differences in phylogenetic composition of PPEs were demonstrated for higher taxonomic levels between ocean basins, using Unifrac analyses of clone library sequence data. Differences in composition were generally greater between basins (interbasins) than within a basin (intrabasin). These differences were primarily linked to taxonomic variation in the composition of Prymnesiophyceae and Prasinophyceae whereas Chrysophyceae were phylogenetically similar in all libraries. These data provide better knowledge of PPE community structure across the world ocean and are crucial in assessing their evolution and contribution to CO2 fixation, especially in the context of global climate change.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
References
Andersen RA . (2004). Biology and systematics of heterokont and haptophyte algae. Am J Bot 10: 1508–1522.
Arrigo KR, Robinson DH, Worthen DL, Dunbar RB, DiTullio GR, VanWoert M et al (1999). Phytoplankton community structure and the drawdown of nutrients and CO2 in the Southern Ocean. Science 283: 365–367.
Arrigo KR . (2005). Marine microorganisms and global nutrient cycles. Nature 437: 349–355.
Bouman HA, Lepère C, Scanlan DJ, Ulloa O . (2012). Phytoplankton community structure in a high-nutrient, low-chlorophyll region of the eastern Pacific Subantarctic region during winter-mixed and summer-stratified conditions. Deep Sea Res I 69: 1–11.
Bouman HA, Ulloa O, Scanlan DJ, Zwirglmaier K, Li WK, Platt T et al (2006). Oceanographic basis of the global surface distribution of Prochlorococcus ecotypes. Science 312: 918–921.
Caron DA . (2009). Past President’s address: protistan biogeography: why all the fuss? J Eukaryot Microbiol 56: 105–112.
Cuvelier ML, Allen A, Monier A, McCrow JP, Messié M, Tringe SG et al (2010). Targeted metagenomics and ecology of globally important uncultured eukaryotic phytoplankton. Proc Natl Acad Sci USA 33: 14679–14684.
Demir-Hilton E, Sudek S, Cuvelier ML, Gentemann CL, Zehr JP, Worden AZ . (2011). Global distribution patterns of distinct clades of the photosynthetic picoeukaryote Ostreococcus. ISME J 5: 1095–1107.
Falkowski PG, Schofield O, Katz ME, Van de Schootbrugge B, Knoll AH . (2004). Why is the land green and the ocean red? In: Thierstein H, Young JR, (eds) Coccolithophores—From Molecular Processes to Global Impact. Elsevier: Amsterdam, pp 429–453.
Finlay BJ, Esteban GF, Brown S, Fenchel T, Hoef-Emden K . (2006). Multiple cosmopolitan ecotypes within a microbial eukaryote morphospecies. Protist 157: 377–390.
Finlay BJ, Fenchel T . (2004). Cosmopolitan metapopulations of free-living microbial eukaryotes. Protist 155: 237–244.
Foissner W . (2006). Biogeography and dispersal of micro-organisms: a review emphasizing protists. Acta Protozool 45: 111–136.
Fuhrman JA, Steele JA, Hewson I, Schwalbach MS, Brown MV, Green JL et al (2008). A latitudinal diversity gradient in planktonic marine bacteria. Proc Natl Acad Sci USA 105: 7774–7778.
Fuller NJ, Campbell C, Allen DJ, Pitt FD, Zwirglmaier K, Le Gall F et al (2006b). Analysis of photosynthetic picoeukaryote diversity at open ocean sites in the Arabian Sea using a PCR biased towards marine algal plastids. Aquat Microb Ecol 43: 79–93.
Fuller NJ, Marie D, Partensky F, Vaulot D, Post AF, Scanlan DJ . (2003). Clade-specific 16S ribosomal DNA oligonucleotides reveal the predominance of a single marine Synechococcus clade throughout a stratified water column in the Red Sea. Appl Environ Microbiol 69: 2430–2443.
Fuller NJ, Tarran GA, Cummings DG, Woodward EMS, Orcutt KM, Yallop M et al (2006a). Molecular analysis of photosynthetic picoeukaryote community structure along an Arabian Sea transect. Limnol Oceanogr 51: 2502–2514.
Green J, Bohannan BJM . (2006). Spatial scaling of microbial biodiversity. Trends Ecol Evol 21: 501–507.
Grob C, Hartmann M, Zubkov MV, Scanlan DJ . (2011). Invariable biomass-specific primary production of taxonomically discrete picoeukaryote groups across the Atlantic Ocean. Environ Microbiol 12: 3266–3274.
Hartmann M, Grob C, Tarran GA, Martina AP, Burkill PH, Scanlan DJ et al (2012). Mixotrophic basis of Atlantic oligotrophic ecosystems. Proc Natl Acad Sci USA 109: 5756–5760.
Hill TCJ, Walsh KA, Harris JA, Mofett BF . (2003). Using ecological diversity measures with bacterial communities. FEMS Micobiol Ecol 43: 1–11.
Härnström K, Ellegaard M, Andersen TJ, Godhe A . (2011). Hundred years of genetic structure in a sediment revived diatom population. Proc Natl Acad Sci USA 108: 4252–4257.
Jardillier L, Zubkov MV, Pearman J, Scanlan DJ . (2010). Significant CO2 fixation by small prymnesiophytes in the subtropical and tropical northeast Atlantic Ocean. ISME J 4: 1180–1192.
Kirkham AR, Jardillier LE, Holland R, Zubkov MV, Scanlan DJ . (2011b). Analysis of phototsynthetic picoeukaryote community structure along an extended Ellett Line transect in the northern North Atlantic reveals a dominance of novel prymnesiophyte and prasinophyte phylotypes. Deep Sea Res I 58: 733–744.
Kirkham AR, Jardillier LE, Tiganescu A, Pearman J, Zubkov MV, Scanlan DJ . (2011a). Basin-scale distribution patterns of photosynthetic picoeukaryotes along an Atlantic Meridional Transect. Environ Microbiol 13: 975–990.
Le Gall F, Rigaut-Jalabert F, Marie D, Garczareck L, Viprey M, Godet A et al (2008). Picoplankton diversity in the South-East Pacific Ocean from cultures. Biogeosciences 5: 203–214.
Legendre P, Legendre L . (1998) Numerical Ecology 2nd edn Elsevier Science BV: Amsterdam.
Lepère C, Vaulot D, Scanlan DJ . (2009). Photosynthetic picoeukaryote community structure in the South East Pacific Ocean encompassing the most oligotrophic waters on Earth. Environ Microbiol 11: 3105–3117.
Li WKW . (1994). Primary production of prochlorophytes, cyanobacteria, and eucaryotic ultraphytoplankton: measurements from flow cytometric sorting. Limnol Oceanogr 39: 169–175.
Litchman E, Klausmeier CA . (2008). Trait-based community ecology of phytoplankton. Annu Rev Ecol Evol Syst 39: 615–639.
Liu H, Probert I, Uits J, Claustre H, Aris-Brosou S, Frada M et al (2009). Extreme diversity in noncalcifying haptophytes explains a major pigment paradox in open oceans. Proc Natl Acad Sci USA 106: 12803–12808.
Longhurst A . (2007) Ecological Geography of the Sea 2nd edn Elsevier, Academic Press.
Lozupone C, Knight R . (2005). UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71: 8228–8235.
Ludwig W, Strunk O, Westram R, Richter L, Meier H, Yadhukumar et al (2004). ARB: a software environment for sequence data. Nucleic Acids Res 32: 1363–1371.
Maguire MJ, Goff LJ, Coleman AW . (1995). In situ plastid and mitochondrial DNA determination: implication of the observed minimal plastid genome number. Am J Bot 82: 1496–1506.
McDonald SM, Sarno D, Scanlan DJ, Zingone A . (2007). Genetic diversity of eukaryotic ultraphytoplankton in the Gulf of Naples during an annual cycle. Aquat Microb Ecol 50: 75–89.
Moon van der Staay SY, De Watchter R, Vaulot D . (2001). Oceanic 18S rDNA sequences from picoplankton reveal unsuspected eukaryotic diversity. Nature 409: 607–610.
Moore LR, Rocap G, Chisholm SW . (1998). Physiology and molecular phylogeny of coexisting Prochlorococcus ecotypes. Nature 393: 464–467.
Nolte V, Pandey RV, Jost S, Medinger R, Ottenwälder B, Boenigk J et al (2010). Constrasting seasonal niche separation between rare and abundant taxa conceals the extent of protist diversity. Mol Ecol 19: 2908–2915.
Not F, Latasa M, Scharek R, Viprey M, Karleskind P, Balague V et al (2008). Protistan assemblages across the Indian Ocean, with a specific emphasis on the picoeukaryotes. Deep Sea Res I 55: 1456–1473.
Not F, Massana R, Latasa M, Marie D, Colson C, Eikrem W et al (2005). Late summer community composition and abundance of photosynthetic eukaryotes in Norwegian and Barents Seas. Limnol Oceanogr 50: 1677–1686.
Papke RT, Ward DM . (2004). The importance of physical isolation to microbial diversification. FEMS Microbiol Ecol 48: 293–303.
Pedros-Alio C . (2006). Marine microbial diversity: can it be determined? Trends Microbiol 14: 257–263.
Pither J . (2007). Comment on ‘Dispersal limitations matter for microbial morphospecies’. Science 316: 1124.
Richardson TL, Jackson GA . (2007). Small phytoplankton and carbon export from the surface ocean. Science 315: 838–840.
Rodríguez F, Derelle E, Guillou L, Le Gall F, Vaulot D, Moreau H . (2005). Ecotype diversity in the marine picoeukaryote Ostreococcus (Chlorophyta, Prasinophyceae). Eniviron Microbiol 7: 853–859.
Romari K, Vaulot D . (2004). Composition and temporal variability of picoeukaryote community at a coastal site of the English Channel from 18S rDNA sequences. Limnol Oceanogr 49: 784–798.
Sanders RW, Gast RJ . (2011). Bacterivory by phototrophic picoplankton and nanoplankton in Arctic waters. FEMS Microbiol Ecol 82: 242–253.
Schnetzer A, Moorthi SD, Countway PD, Gast RJ, Gilg IC, Caron DA . (2011). Depth matters: Microbial eukaryote diversity and community structure in the eastern North Pacific revealed through environmental gene libraries. Deep Sea Res I 58: 16–26.
Shi XL, Lepère C, Scanlan DJ, Vaulot D . (2011). Plastid 16S rRNA gene diversity among eukaryotic picophytoplankton sorted by flow cytometry from the South Pacific Ocean. PLoS One 4: e18979.
Shi XL, Marie D, Jardillier L, Scanlan DJ, Vaulot D . (2009). Groups without cultured representatives dominate eukaryotic picophytoplankton in the Oligotrophic South East Pacific. PLoS One 4: e7657.
Telford RJ, Vandik V, Birks HJB . (2006). Dispersal limitations matter for microbial morphospecies. Science 312: 1015.
Ter Braak CJF . (1986). Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67: 1167–1179.
Tyrell T . (1999). The relative influences of nitrogen and phosphorus on oceanic primary production. Nature 400: 525–531.
Vaulot D, Eikrem W, Viprey M, Moreau H . (2008). The diversity of small eukaryotic phytoplankton (⩽3 μm) in marine ecosystems. FEMS Microbiol Rev 32: 795–820.
Viprey M, Guillou L, Ferréol M, Vaulot D . (2008). Wide genetic diversity of picoplanktonic green algae (Chloroplastida) in the Mediterranean Sea uncovered by a phylum-biased PCR approach. Environ Microbiol 10: 1804–1822.
Weber TS, Deutsch C . (2010). Ocean nutrient ratios governed by plankton biogeography. Nature 467: 550–554.
West NJ, Scanlan DJ . (1999). Niche-partitioning of Prochlorococcus populations in a stratified water column in the eastern North Atlantic Ocean. Appl Environ Microbiol 65: 2585–2591.
West NJ, Schönhuber WA, Fuller NJ, Amann RI, Rippka R, Post AF et al (2001). Closely related Prochlorococcus genotypes show remarkably different depth distributions in two oceanic regions as revealed by in situ hybridisation using 16S rRNA-targeted oligonucleotides. Microbiology 147: 1731–1744.
Worden AZ, Nolan JK, Palenik B . (2004). Assessing the dynamics and ecology of marine picophytoplankton: the importance of the eukaryotic component. Limnol Oceanogr 49: 168–179.
Worden AZ, Not F . (2008). Ecology and diversity of picoeukaryotes. In: Kirchman DL, (eds). Microbial Ecology of the Oceans 2nd edn John Wiley & Sons: Hoboken, NJ, USA, pp 159–205.
Yang J, Smith HG, Sherratt TN, Wilkinson DM . (2010). Is there a size limit for cosmopolitan distribution in free-living microorganisms? A biogeographical analysis of testate amoebae from polar areas. Microb Ecol 59: 635–645.
Zubkov MV, Fuchs BM, Tarran GA, Burkill PH, Amann R . (2003). High rate of uptake of organic nitrogen compounds by Prochlorococcus cyanobacteria as a key to their dominance in oligotrophic oceanic waters. Appl Environ Microbiol 69: 1299–1304.
Zubkov MV, Tarran GA . (2008). High bacterivory by the smallest phytoplankton in the North Atlantic Ocean. Nature 455: 224–226.
Zwirglmaier K, Jardillier L, Ostrowski M, Mazard S, Garczarek L, Vaulot D et al (2008). Global phylogeography of marine Synechococcus and Prochlorococcus reveals a distinct partitioning of lineages among oceanic biomes. Environ Microbiol 10: 147–161.
Acknowledgements
We thank the Captains and crews of all the cruises analysed. This study was supported by the UK Natural Environment Research Council via a PhD studentship award to ARK and NERC Grant NE/C003160/1 to DJS. Sequencing was performed at the NERC Biomolecular Analysis Facility in Edinburgh.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on The ISME Journal website
Supplementary information
Rights and permissions
About this article
Cite this article
Kirkham, A., Lepère, C., Jardillier, L. et al. A global perspective on marine photosynthetic picoeukaryote community structure. ISME J 7, 922–936 (2013). https://doi.org/10.1038/ismej.2012.166
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ismej.2012.166
Keywords
This article is cited by
-
Significant Effects of Associated Microorganisms on the Community of Photosynthetic Picoeukaryotes
Microbial Ecology (2023)
-
Niche partitioning by photosynthetic plankton as a driver of CO2-fixation across the oligotrophic South Pacific Subtropical Ocean
The ISME Journal (2022)
-
Comparison of Deep-Sea Picoeukaryotic Composition Estimated from the V4 and V9 Regions of 18S rRNA Gene with a Focus on the Hadal Zone of the Mariana Trench
Microbial Ecology (2022)
-
Experimental identification and in silico prediction of bacterivory in green algae
The ISME Journal (2021)
-
NanoSIMS single cell analyses reveal the contrasting nitrogen sources for small phytoplankton
The ISME Journal (2019)