Abstract
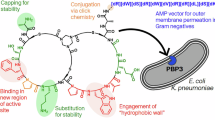
Tunicamycins (TUN) are inhibitors of the UDP-HexNAc: polyprenol-P HexNAc-1-P transferase family of enzymes, which initiate the biosynthesis of bacterial peptidoglycan and catalyze the first step in eukaryotic protein N-glycosylation. The TUN are therefore general and potent toxins to both eukaryotes and prokaryotes. Screening a library of synthetic TUN against Bacillus and yeast identified TUN that are antibacterial, but have significantly reduced eukaryotic toxicity. One of these (Tun-15:0) differs from the native TUN control only by the lack of the conjugated double bond in the tunicaminyl N-acyl group. Tun-15:0 also showed reduced inhibition for protein N-glycosylation in a Pichia-based bioassay. Natural TUN was subsequently modified by chemically reducing the N-acyl double bond (TunR1) or both the N-acyl and uridyl double bonds (TunR2). TunR1 and TunR2 retain their antibacterial activity, but with considerably reduced eukaryotic toxicity. In protein N-glycosylation bioassays, TunR1 is a less potent inhibitor than native TUN and TunR2 is entirely inactive. Importantly, the less toxic TunR1 and TunR2 both enhance the antibacterial activity of β-lactams: oxacillin by 32- to 64-fold, comparable with native TUN, and with similar enhancements for methicillin and penicillin G. Hence, the modified TUNs, TunR1 and TunR2, are potentially important as less-toxic synergistic enhancers of the β-lactams.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Perros, M. Infectious disease. A sustainable model for antibiotics. Science 347, 1062–1064 (2015).
Baker, S. Infectious disease. A return to the pre-antimicrobial era? Science 347, 1064–1066 (2015).
Andersson, D. I. & Hughes, D. Antibiotic resistance and its cost: is it possible to reverse resistance? Nat. Rev. Microbiol. 8, 260–271 (2010).
Wright, G. D. Something old, something new: revisiting natural products in antibiotic drug discovery. Can. J. Microbiol. 60, 147–154 (2014).
Brown, K. Penicillin Man. Alexander Fleming and the Antibiotic Revolution (Sutton Publishing, Stroud, Gloucestershire, 2004).
Drawz, S. M. & Bonomo, R. A. Three decades of β-lactamase inhibitors. Clin. Microbiol. Rev. 23, 160–201 (2010).
Abraham, E. P. & Chain, E. An enzyme from bacteria able to destroy penicillin. Nature 146, 837–837 (1940).
Campbell, J. et al. Synthetic lethal compound combinations reveal a fundamental connection between wall teichoic acid and peptidoglycan biosyntheses in Staphylococcus aureus. ACS Chem. Biol. 6, 106–116 (2011).
Brown, S. et al. Methicillin resistance in Staphylococcus aureus requires glycosylated wall teichoic acids. Proc. Natl Acad. Sci. USA 109, 18909–18914 (2012).
Farha, M. A. et al. Inhibition of WTA synthesis blocks the cooperative action of PBPs and sensitizes MRSA to β-lactams. ACS Chem. Biol. 8, 226–233 (2013).
Reed, P. et al. Staphylococcus aureus survives with a minimal peptidoglycan synthesis machine but sacrifices virulence and antibiotic resistance. PLoS Pathog. 11, e1004891 (2015).
Lee, S. H. et al. TarO-specific inhibitors of wall teichoic acid biosynthesis restore β-lactam efficacy against methicillin-resistant staphylococci. Sci. Transl. Med. 8, 329ra32 (2016).
Takatsuki, A., Arima, K. & Tamura, G. Tunicamycin, a new antibiotic. I. Isolation and characterization of tunicamycin. J. Antibiot. (Tokyo) 24, 215–223 (1971).
Tamura, G. Tunicamycins, Japan Scientific Press, Tokyo, (1982).
Price, N. P. & Tsvetanova, B. Biosynthesis of the tunicamycins: a review. J. Antibiot. (Tokyo) 60, 485–491 (2007).
Heifetz, A., Keenan, R. W. & Elbein, A. D. Mechanism of action of tunicamycin on the UDP-GlcNAc:dolichyl-phosphate GlcNAc-1-phosphate transferase. Biochemistry 18, 2186–2192 (1979).
Mclachlan, K. R. & Krag, S. S. Substrate specificity of N-acetylglucosamine 1-phosphate transferase activity in chinese hamster ovary cells. Glycobiology 2, 313–319 (1992).
Lehrman, M. A. Biosynthesis of N-acetylglucosamine-P-P-dolichol, the committed step of asparagine-linked oligosaccharide assembly. Glycobiology 1, 553–562 (1991).
Price, N. P. J. et al. Quinovosamycins: new tunicamycin-type antibiotics in which the α, β-1″,11′-linked N-acetylglucosamine residue is replaced by N-acetylquinovosamine. J. Antibiot. 69, 637–646 (2016).
Cantagrel, V. et al. SRD5A3 is required for converting polyprenol to dolichol and is mutated in a congenital glycosylation disorder. Cell 142, 203–217 (2010).
Jozwiak, A. et al. Polyprenol reductase2 deficiency is lethal in Arabidopsis due to male sterility. Plant Cell 27, 3336–3353 (2015).
Li, J. & Yu, B. A modular approach to the total synthesis of tunicamycins. Angew. Chem. Int. Ed. Engl. 54, 6618–6621 (2015).
Bannerjee, A. et al. N-Acetylglucosaminyl 1-phosphate transferase: an excellent target for developing new generation breast cancer therapeutic. Adv. Exp. Med. Biol. 842, 355–374 (2015).
Banerjee, A. et al. Unfolded protein response is required in nu/nu mice microvasculature for treating breast tumor with tunicamycin. J. Biol. Chem. 286, 29127–29138 (2011).
Hakulinen, J. K. et al. MraY–antibiotic complex reveals details of tunicamycin mode of action. Nat. Chem. Biol. 13, 265–267 (2017).
Chung, B. C. et al. Crystal structure of MraY, an essential membrane enzyme for bacterial cell wall synthesis. Science 341, 1012–1016 (2013).
Chung, B. C. et al. Structural insights into inhibition of lipid I production in bacterial cell wall synthesis. Nature 533, 557–560 (2016).
Furlong, S. E. & Valvano, M. Characterization of the highly conserved VFMGD motif in a bacterial polyisoprenyl-phosphate N-acetylaminosugar-1-phosphate transferase. Protein Sci. 21, 1366–1375 (2012).
Hartley, M. D. & Imperiali, B. At the membrane frontier: a prospectus on the remarkable evolutionary conservation of polyprenols and polyprenyl-phosphates. Arch. Biochem. Biophys. 517, 83–97 (2012).
Tran, A. T., Huynh, V. A., Friz, E. M., Whitney, S. K. & Cordes, D. B. A general method for the rapid reduction of alkenes and alkynes using sodium borohydride, acetic acid, and palladium. Tetrahedron Lett. 50, 1817–1819 (2009).
Acknowledgements
BY acknowledges the support of the National Natural Science Foundation of China (21372253 and 21432012). We thank WW Metcalf (University of Illinois, Champaign, IL, USA) and JO Rich (ARS-USDA, Peoria, IL, USA) for the initially review of the manuscript. A provisional patent application (patent number 62/450,760) has been filed. Mention of any trade names or commercial products is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Price, N., Hartman, T., Li, J. et al. Modified tunicamycins with reduced eukaryotic toxicity that enhance the antibacterial activity of β-lactams. J Antibiot 70, 1070–1077 (2017). https://doi.org/10.1038/ja.2017.101
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ja.2017.101
This article is cited by
-
The tunicamycin derivative TunR2 exhibits potent antibiotic properties with low toxicity in an in vivo Mycobacterium marinum-zebrafish TB infection model
The Journal of Antibiotics (2024)
-
Synergistic enhancement of beta-lactam antibiotics by modified tunicamycin analogs TunR1 and TunR2
The Journal of Antibiotics (2019)
-
Liposidomycin, the first reported nucleoside antibiotic inhibitor of peptidoglycan biosynthesis translocase I: The discovery of liposidomycin and related compounds with a perspective on their application to new antibiotics
The Journal of Antibiotics (2019)
-
Selective catalytic hydrogenation of the N-acyl and uridyl double bonds in the tunicamycin family of protein N-glycosylation inhibitors
The Journal of Antibiotics (2017)