Abstract
Previous studies have suggested that breast cancer in young women has more aggressive biological features and poorer prognosis. However, the role of biological markers in these patients is not well understood. We aimed to learn more about this disease in a cohort of 125 young women from Singapore, Japan and Hong Kong, aged 35 years or less, with invasive breast cancer by evaluating the expression of vimentin and the basal cytokeratins CK14, CK5/6 and 34βE12. Both standard paraffin sections and tissue microarrays were used in the immunohistochemical evaluation of expression patterns of these four biological markers. CK5/6, CK14, vimentin and 34βE12, in increasing order of proportion, were detected in invasive carcinomas. Basal cytokeratins and vimentin showed significant inverse relationship with estrogen and progesterone receptor status while CK14 expression was found to be directly associated with c-erbB2 status. Basal cytokeratins and vimentin immunoreactivities were directly associated with CD117 and EGFR expression. Vimentin and 34βE12 immunopositivity correlated with tumor size, while vimentin was associated with higher histological grade. Our findings are in concert with reports that expression of basal cytokeratins and vimentin is correlated with adverse pathological parameters.
Similar content being viewed by others
Main
Cytoplasmic intermediate filament proteins which form the cytoskeleton have been categorized into five distinct classes that differ in their biochemical and antigenic properties.1, 2 They are variably expressed in different cell types and their corresponding tumors. Cytokeratins are α-type fibrous polypeptides with diameters of 7–11 nm and are usually expressed in epithelial cells and carcinomas. On the other hand, vimentin is expressed in mesenchymal cells, lymphomas and sarcomas.1, 3
Cytokeratins are generally thought to be the most fundamental markers of epithelial differentiation because their specific compositions reflect both cell type and differentiation.4 In the breast, it has been documented that CK5/6 and CK14 have specificities for basal or high molecular weight keratins, and exhibit immunostaining mainly in the outer myoepithelial cells. 34βE12 is less specific, and can demonstrate immunoreactivity in both myoepithelial as well as luminal epithelial cells.4, 5 Vimentin, however, shows expression in stromal connective tissue, wall of blood vessels, nerves and also in myoepithelial cells.1
Various studies carried out previously have evaluated the expression of these cytokeratins and vimentin in human breast cancer.1, 4, 5, 6, 7 In this paper, we specifically examined the expression patterns of the basal cytokeratins CK5/6, CK14, 34βE12 (CK 1/5/10/14), and of vimentin in breast cancers in young women, defined as those aged 35 years or less, and correlated the findings with conventional pathological parameters.
Materials and methods
Patients
A total of 125 cases were used for this study: 91 from the Department of Pathology, Singapore General Hospital, diagnosed between 1980 and 2005; 11 from the Chinese University of Hong Kong, between 1989 and 2002; and 23 from Tohoku University Hospital, Sendai, Japan, between 1999 and 2005.
Cell Culture
MCF-7 and MDA-MB-231 breast cancer cells purchased from the American Type Culture Collection were cultured in Dulbecco's Modified Eagle's Medium (Sigma) supplemented with 10% fetal bovine serum (FBS) and RPMI-1640 with 10% FBS respectively at 37°C in a 5% carbon dioxide incubator.
Tissue Microarray
The array layout in grid format was planned in advance using Microsoft Excel version 11. Hematoxylin and eosin sections of breast tumors diagnosed at the Singapore General Hospital were reviewed by a pathologist and representative areas marked. Recipient blocks were prepared by melting regular paraffin and pouring it into a 5–10 mm-thick mold. The samples were then arrayed. Punch sizes of 1.0 and 2.0 mm were used for different blocks. Once the array was completed, the block was removed from the instrument and sectioned at 4 μm using a microtome without the use of the adhesive-coated tape system.8 Sectioning of array blocks was carried out in a manner identical to that for routine paraffin blocks.
Immunohistochemistry
The cytokeratin and vimentin antibodies, dilutions and antigen retrieval methods used are detailed in Table 1. Tissue sections were deparaffinized in two five-minute changes of xylene and rehydrated through graded ethanol. Subsequently, sections were microwave-heated for epitope retrieval, washed with distilled water, and antibody staining was done using a DakoCytomation Autostainer with ChemMate™ DAKO Envision™ Detection Kit. Skin sections were used as positive controls for CK14, CK5/6, and 34βE12; sections of uterine fibroid were utilized for vimentin.
For estrogen receptor (ER, NeoMarkers RM9101-S, clone Ra,SP1, 1:70 dilution) and c-erbB2 (NeoMarkers RM9103-S, clone Ra,SP3, 1:200 dilution), antigen retrieval employed pressure cooking in the microwave oven (using Milestone TT Mega, in citrate buffer pH 6 at 115°C for 2 min). Internal positive controls for ER and PR were provided by adjacent benign breast epithelium, whereas a c-erbB2 positive tumor was used as a positive control for c-erbB2. For EGFR and CD117, Dako EGFR pharmDx™ Kit using a monoclonal mouse anti-human EGFR protein, and CD117 antibody (c-kit, Dako A4502, 1:250 dilution) using the Envision ChemMate Kit, were applied with staining demonstrated by the Dako Autostainer. A breast tumor known to be reactive with EGFR antibody was used as positive control, while a gastrointestinal stromal tumor served as the positive control for CD117.
Scoring of Sections
Each section was examined under × 40 to × 400 magnification ( × 10 eyepiece and × 4 to × 40 objectives) of the light microscope after immunostaining. Staining intensity was defined as follows: 0, no staining; 1+, weak; 2+, moderate; 3+ strong. Quantification of positivity (0–100%) was based on an estimate of the percentage of immunoreactive invasive tumor cells in the lesion. The immunostaining pattern for CK14, CK5/6, 34βE12 and vimentin was cytoplasmic staining. ER and PR decorated the nuclei, while c-erbB2 and EGFR highlighted the cytoplasmic membranes. CD117 stained both the cytoplasm and cytoplasmic membranes of tumor cells. A final immunoscore was obtained by multiplying staining intensity with percentage positivity for all the antibodies evaluated. Thus, the range of possible scores was 0–300. Immunoscores and similar semi-quantitative scoring systems had been successfully used previously for tissue microarray evaluation.9, 10, 11
Real-time Reverse Transcription Polymerase Chain Reaction
Extraction of total RNA
The cultured cells were first harvested using 0.25% trypsin/1 mM EDTA, washed and pelleted for RNA extraction using RNeasy Micro Kit (Qiagen) and following the procedures as stated in the manufacturer's protocol. The concentration of RNA extracted was determined by measuring the absorbance at 260 nm using a Bio-Rad® spectrophotometer.
First-strand cDNA synthesis
First-strand cDNA synthesis from total RNA was carried using SuperScript III 1st Strand Synthesis System (Invitrogen) with random hexamer primers following the manufacturer's protocol.
Real-time polymerase chain reaction
The detection of gene expression was done using SYBR green (Qiagen) in Roche Real-time Light Cycler 2.0 System. The cDNA synthesized previously was subjected to real-time polymerase chain reaction using the primer pairs as follows: (forward) 5′-GACAATGCGTCTCTGGCACGTCTT-3′ and (reverse) 5′-TCCTCCGCCTCCTGCAGGTTCTT-3′ for vimentin (Gilles et al, 2003) and (forward) 5′-GAAGGTGAAGGTCGGAGTCAACG-3′ and (reverse) 5′-GAAGATGGTGATGGGATTTC-3′ for GAPDH (Lee et al, 2007). GAPDH gene was included as a control gene. The real-time polymerase chain reaction cycling profile was programmed as follows. Firstly, SYBR Green (QuantiTect) was activated at 95°C for 15 min for one cycle, followed by amplification phase which includes denaturation at 94°C for 15 s, annealing at 60°C for 25 s, extension at 72°C for 20 s for 40 cycles. Melting curve analysis was performed at 65°C for 15 s to verify the specificity of the amplification reaction. The specificity of the polymerase chain reaction was further confirmed by gel electrophoresis using 2% agarose gel containing ethidium bromide and viewed under ultraviolet illuminator. The expression of vimentin was derived using the ΔCT method. ΔCT was calculated by subtracting CT value of GAPDH from that of vimentin. Relative vimentin expression was also calculated by the method.
Cell Cycle Analysis
Cell cycle distribution was determined by flow cytometry after staining the cells with propidium iodide. MCF-7 cells and MDA-MB-231 cells were separately cultured in respective culture media in Costar 6-well plates until they reached 80% confluence before harvesting and fixation with 70% ethanol at 4°C overnight. Before analysis, the cells were centrifuged and washed with phosphate-buffered saline solution to remove ethanol. Cells were then treated with propidium iodide (PI) cocktail, which contains RNaseA (0.2 mg/ml) and PI (20 μg/ml) dissolved in 0.1% triton-X PBS buffer, for 30 min at room temperature. The stained cells were analyzed by CyAn™ ADP Flow Cytometer for 10 000 events per sample. Each experiment was carried in triplicates.
Statistical Analysis
Statistical analysis was performed using the Student's t-test with Welch's correction and Fisher's exact test using the GraphPad Prism v4.03 program. Clinical and pathological details were compared against the results of immunohistochemical scores. A Student's t-test was used to analyze the results of real time-polymerase chain reaction and cell cycle analysis. A P-value of less than 0.05 was considered to reflect a significant correlation.
Database for Analyzing Protein Interactions
The Atlas of Genetics and Cytogenetics in Oncology and Haematology (AGCOH) database at the following URL address: http://www.infobiogen.fr/services/chromcancer/ was used. The ‘IntAct’ function of the database was used for analyzing protein interaction data.
Results
Clinicopathological Features of Patient Samples
The clinicopathological details of the patients in our study are shown in Table 2. The histological subtype in 107 (91.5%) cases was of invasive ductal morphology while five (4.3%) cases presented with lobular form. The mean age of young women with invasive breast cancer in our study was 30.1 years, and 46.4% of the patients presented with histological grade 3 tumors. Invasive tumor size ranged from 2 to 155 mm (mean 30.5 mm, median 22 mm). Nodal metastases were found in 34 (50.7%) cases, with an average involvement of nine lymph nodes. The number of nodes harvested per case ranged from 1 to 56.
Association of Expression of Basal Keratins and Vimentin with Clinicopathological Parameters
The expression of CK14, CK5/6, 34βE12 and vimentin in our study cases is shown in Figure 1 and Table 3.
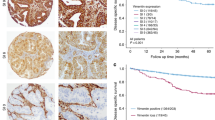
Cytokeratins and vimentin expression in invasive ductal breast carcinoma. (a) Diffuse 3+ staining of invasive carcinoma cells for CK14. Magnification: × 200. (b) 2+ reactivity of tumor cells for CK5/6. Magnification: × 400. (c) 90% of the tumor cells were stained 3+ for 34βE12. Magnification: × 400. (d) Almost all the tumor cells in this section were stained 3+ for vimentin. Magnification: × 200.
There was a lower proportion of cases exhibiting immunopositivity for CK14 and CK5/6 (11.2 and 7.2% respectively) compared to a greater proportion disclosing expression of 34βE12 and vimentin (35.2 and 22.4% respectively). A higher percentage (>50%) of tumor cells were positively stained for 34βE12 and vimentin, whereas a lower percentage of invasive tumor cells was stained for CK14 and CK5/6 (Table 3).
Table 4 shows the results of statistical analysis for associations between clinicopathological parameters and expression of basal cytokeratins and vimentin. All four proteins were expressed at significantly lower levels in breast cancer cells from women aged 31 years and below compared with those from patients between 32 and 35 years old. Vimentin and 34βE12 expression was found to be significantly correlated with tumor size, with proportionately more cases expressing these two biomarkers in tumors larger than 20 mm in size (Table 4). Histological grade of invasive carcinoma was found to correlate with vimentin expression, with higher vimentin immunoscores in grade 3 cancers. However, there was no statistically significant association between nodal status and basal cytokeratin or vimentin expression.
The basal cytokeratins CK14 and CK5/6 as well as vimentin expression in tumor cells revealed significant inverse correlation with ER status (Table 4). PR status in these tumors was found to have reciprocal association with expression of CK5/6, 34βE12 and vimentin. The expression of CK14 was directly associated with c-erbB2 status (Table 4). CK14 was also found to interact with c-erbB2 using the AGCOH database.
There was strong positive correlation between the expression of CD117 (c-kit proto-oncogene) with immunoreactivity of basal cytokeratins and vimentin (Table 4). Expression of CK5/6, 34βE12 and vimentin was also positively associated with epidermal growth factor receptor (EGFR) status. Vimentin was found to interact with tubulin β-2 and tubulin α-3 and both in turn interact with EGFR whereas CK5 and CK14 interact directly with EGFR (ACGOH database).
Association of CK14, CK5/6, 34βE12 and Vimentin Expression
Strong positive associations were noted among the expression of all three basal keratins and vimentin (Table 5). This agrees with the finding that there are interactions among CK1, CK5, CK14 and vimentin (through tubulin β-2 and tubulin α-3) via physical interactions with EGFR (ACGOH database).
Expression of Vimentin and Cell Cycle Analysis in Breast Cancer Cell Lines
As vimentin was most associated with ER status and tumor size in the breast cancer tissue samples, we analyzed its expression and correlation with the cell cycle. At the mRNA level, vimentin expression was found to be significantly higher in ER−/PR− MDA-MB231 breast cancer cells as compared with ER+/PR+ MCF-7 breast cancer cells (Figure 2a). In fact, the relative expression in MDA-MB-231 cells was found to be more than 800 folds higher as compared to MCF-7 cells. Specificity of the primer pairs used were confirmed by melting curve analysis (not shown) as well as by gel electrophoresis of the real-time PCR product (Figure 2b). The MDA-MB231 cells were also more proliferative than MCF-7 cells as shown by the cell cycle analysis (Figure 3). The percentage of cells in the proliferative (S phase+G2/M phase) phase of MDA-MB-231 cells and MCF-7 cells were 41.4±1.3 and 28.9±0.7% respectively.
mRNA expression of vimentin in MCF-7 and MDA-MB231 breast cancer cells (a) ΔCT values of vimentin in MCF-7 and MDA-MB-231 breast cancer cells. The lower the ΔCT value implies the higher the gene expression and vice versa. (b) Gel electrophoresis of real-time PCR products showing specificity of primers. 1=MDA-MB-231 cells; 2=MCF-7 cells.
A representative histogram showing the cell cycle analysis of: (a) MDA-MB-231 and (b) MCF-7 breast cancer cells.
Discussion
The majority of breast cancers in young women are invasive in nature, with T2 disease (20 mm<tumor size ≤50 mm) at presentation and poor histological grade 3 (46.4%).4, 12, 13, 14 The histological subtype in 91.5% of cases in this study was of invasive ductal morphology. This is in line with Maru's report that invasive ductal carcinoma is the most common histological type of breast cancer in young women.12 Our previous clinicopathological study of breast cancer in young women disclosed a significant proportion of high-grade tumors with positive nodal disease.14 The biology of invasive breast carcinoma in this cohort of women is poorly understood and the role of biologic markers not well defined.15, 16 We aimed to learn more about this disease in this young population aged 35 years or less by evaluating the expression of three different basal cytokeratins (CK14, CK5/6 and 34βE12) and vimentin in invasive breast cancers diagnosed in this cohort. Tissue microarray technique was included as a high throughput method for immunohistochemical analysis of the Singapore General Hospital cases.
There were proportionately fewer cases showing expression of CK14 and CK5/6 as compared with 34βE12 and vimentin. This may be due to the 34βE12 antibody recognizing multiple cytokeratins, (CK1, 5, 10, 14).4, 17 Abd El-Rehim et al6 demonstrated in their study that a high proportion of invasive carcinomas expressed only luminal epithelial cell cytokeratins. In contrast, the combined expression of luminal and basal keratins as well as the exclusive expression of basal epithelial markers were found in a lower percentage of this disease. This may explain the small number of cases showing positive immunoreactivity for the basal cytokeratins CK14 and CK5/6 in our study.5 It has been reported that between 2–18% of infiltrative ductal cancers show expression for CK14,18 and our finding of CK14 positivity in 11.2% of breast cancers from our young women lies within this range. Fewer cases were reactive for CK5/6 as compared with CK14, which could be related to differences in sensitivity of the antibodies, as noted in our study on papillary breast lesions.4 Vimentin immunostaining was observed in more than 60% of tumor cells in infiltrating ductal carcinoma,19 similar to what we observed in our positive cases.
In this study, there were inverse associations between expression of basal cytokeratins (CK14 and CK5/6) and vimentin with ER status. CK5/6, 34βE12 and vimentin immunoreactivities were also found to have reciprocal association with tumoral PR status. Likewise, Abd El-Rehim et al6 reported that tumors expressing basal/myoepithelial phenotype were often ER negative.7 Seshadri et al20 convincingly showed that vimentin expression was significantly associated with the absence of hormone receptors. This was comparable to the results reported by Santini et al21 and Korsching et al.22 The in vitro work in the current study also substantiates the finding that expression of vimentin is correlated with the hormonal status as ER−/PR− MDA-MB-231 breast cancer cells had significantly higher expression of vimentin than ER+/PR+ MCF-7 breast cancer cells. Vimentin expression has been documented to be associated with poor prognosis in node negative infiltrative ductal breast cancer,23 and our findings suggest that this may be in part related to hormone receptor negativity. It is proposed that coexpression of vimentin and cytokeratin filaments in human breast cancer cells confers a dedifferentiated or interconverted phenotype, giving them a selective advantage in their interactions with the extracellular matrix.24 The rate of vimentin expression in breast cancers reportedly varies from 7.7 to 21.2%.22, 25 Our figure of 22.4% is on the higher extreme of that reported, and may reflect a bias due to our cohort being that of young women, lending support to cancers in this age group being potentially more aggressive biologically. Recent studies have also provided new information concerning hormone receptor positive breast cancers and the prediction of long-term outcomes in tamoxifen-treated patients,26 linked closely to studies which demonstrated that expression of basal markers and vimentin was associated with poorer prognostic features and behavior.6, 23
Interestingly, we found that CK14 expression was significantly associated with the presence of c-erbB2 (HER2/neu). The presence of a relationship between CK14 and HER2/neu status parallels Abd El-Rehim et al's.6 report that basal markers were associated with poorer prognostic features and behavior.27 There has been some confusion and debate in the literature regarding the interchangeability of basal type breast cancers that express basal keratins, and the ‘triple negative’ tumors that do not express ER, PR or c-erbB2. While many triple negative breast cancers also express basal cytokeratins, this may not always be the case as demonstrated in our study where CK14 positivity is associated with c-erbB2 expression. It is therefore advocated that these terminologies of basal type and triple negative breast cancers are defined separately and not merged into a single entity. Coexpression of CK14 and c-erbB2 in our cases may identify a biologically unique subset that requires further elucidation.
There was a strong positive association between the expression of CD117 (c-kit proto-oncogene) with the immunoreactivities of basal cytokeratins and vimentin. This is in line with the results shown in the reports of Nielsen et al28 and Tsuda et al,29 which stated that c-kit expression was more common in basal-like tumors and that c-kit overexpressing carcinoma tended to coexpress vimentin. Previous studies have reported that expression of c-kit protein appeared to be an indicator of high-grade breast carcinoma, and may be linked to drug resistance and other adverse prognostic factors.29 It may also define a subset of poorly differentiated, HER-2-positive ductal carcinoma in situ (DCIS) with decreased expression of steroid hormone receptors, comedonecrosis, and a solid growth pattern.30
Expression of CK5/6 and 34βE12 was also found to be positively associated with EGFR status. A similar trend has been observed in many recent reports which showed that basal-like tumors expressed EGFR (HER1).28, 31, 32 Coexpression of HER-1, HER-2 and HER-3 was demonstrated to have a negative synergistic effect on patient outcome.33 These studies further showed that expression of basal cytokeratins and vimentin are associated with more aggressive tumor features and poorer disease prognosis.
In our results, immunohistochemical expression of 34βE12 and vimentin was related to the tumor size. This was not found in the study by Seshadri et al, in which there was no association between vimentin expression and tumor size.20 However, our in vitro study showed that highly proliferative MDA-MB231 breast cancer cells had higher vimentin expression than the less proliferative MCF-7 breast cancer cells which also had correspondingly lower vimentin expression. Nevertheless, the strong relationship of histological grade with the presence of vimentin in our study agrees with the results of Seshadri et al.20
Much has been written in the literature regarding molecular subclassification of breast cancers using gene expression profiling, with identification of distinct molecular subtypes that have clinical implications and morphologic correlations. These subgroups include the luminal A, luminal B, basal-like, normal breast-like and c-erbB2 expressing tumors.34, 35 Immunohistochemical surrogates have been proposed as being able to stratify tumors into these molecular categories. In particular, basal-like breast cancers defined using expression profiling are found to be negative for ER and c-erbB2, and positive for vimentin, EGFR, cytokeratin 8/18, and cytokeratin 5/6 on immunohistochemistry.36 Our results reinforce the association between these immunohistochemical markers in our series of breast cancers in young women, and also corroborate our previous finding of conservation of molecular subtypes in women of Asian ethnicity.37
In summary, our study confirms the association of basal cytokeratins and vimentin with adverse pathologic features in our series of young women with breast cancer, with our in vitro work lending support to our conclusions. More work is required to further comprehend if expression of these intermediate filaments independently impacts on biologic behavior, and if therapeutic strategies in this particular group of young women need specific modulation.
References
Heatley M, Whiteside C, Maxwell P, et al. Vimentin expression in benign and malignant breast epithelium. J Clin Pathol 1993;46:441–445.
Fuchs E . Keratins as biochemical markers of epithelial differentiation. Trends Genet 1988;4:277–281.
Domagala W, Lubinski J, Weber K, et al. Intermediate filament typing of tumor cells in fine needle aspirates by means of monoclonal antibodies. Acta Cytol 1986;30:214–224.
Tan PH, Aw MY, Yip GW, et al. Cytokeratins in papillary lesions of the breast: is there a role in distinguishing intraductal papilloma from papillary ductal carcinoma in situ? Am J Surg Pathol 2005;29:625–632.
Dairkee SH, Puett L, Hackett AJ . Expression of basal and luminal epithelium-specific keratins in normal, benign, and malignant breast tissue. J Natl Cancer Inst 1988;80:691–695.
Abd El-Rehim DM, Pinder SE, Paish CE, et al. Expression of luminal and basal cytokeratins in human breast carcinoma. J Pathol 2004;203:661–671.
Malzahn K, Mitze M, Thoenes M, et al. Biological and prognostic significance of stratified epithelial cytokeratins in infiltrating ductal breast carcinomas. Virchows Arch 1998;433:119–129.
Kononen J, Bubendorf L, Kallioniemi A, et al. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 1998;4:844–847.
Rimm DL, Camp RL, Charette LA, et al. Amplification of tissue by construction of tissue microarrays. Exp Mol Pathol 2001;70:255–264.
Callagy G, Cattaneo E, Daigo Y, et al. Molecular classification of breast carcinomas using tissue microarrays. Diagn Mol Pathol 2003;12:27–34.
Selvarajan S, Sii LH, Lee A, et al. Parafibromin expression in breast cancer: a novel marker for prognostication? J Clin Pathol 2008;61:64–67.
Maru D, Middleton LP, Wang S, et al. HER-2/neu and p53 overexpression as biomarkers of breast carcinoma in women age 30 years and younger. Cancer 2005;103:900–905.
Colleoni M, Rotmensz N, Robertson C, et al. Very young women (<35 years) with operable breast cancer: features of disease at presentation. Ann Oncol 2002;13:273–279.
Fernandopulle SM, Cher-Siangang P, Tan PH . Breast carcinoma in women 35 years and younger: a pathological study. Pathology 2006;38:219–222.
Han W, Kim SW, Park IA, et al. Young age: an independent risk factor for disease-free survival in women with operable breast cancer. BMC Cancer 2004;4:82.
Choi DH, Kim S, Rimm DL, et al. Immunohistochemical biomarkers in patients with early-onset breast carcinoma by tissue microarray. Cancer J 2005;11:404–411.
Joshi MG, Lee AK, Pedersen CA, et al. The role of immunocytochemical markers in the differential diagnosis of proliferative and neoplastic lesions of the breast. Mod Pathol 1996;9:57–62.
Jones C, Nonni AV, Fulford L, et al. CGH analysis of ductal carcinoma of the breast with basaloid/myoepithelial cell differentiation. Br J Cancer 2001;85:422–427.
Raymond WA, Leong AS . Co-expression of cytokeratin and vimentin intermediate filament proteins in benign and neoplastic breast epithelium. J Pathol 1989;157:299–306.
Seshadri R, Raymond WA, Leong AS, et al. Vimentin expression is not associated with poor prognosis in breast cancer. Int J Cancer 1996;67:353–356.
Santini D, Ceccarelli C, Taffurelli M, et al. Differentiation pathways in primary invasive breast carcinoma as suggested by intermediate filament and biopathological marker expression. J Pathol 1996;179:386–391.
Korsching E, Packeisen J, Liedtke C, et al. The origin of vimentin expression in invasive breast cancer: epithelial-mesenchymal transition, myoepithelial histogenesis or histogenesis from progenitor cells with bilinear differentiation potential? J Pathol 2005;206:451–457.
Domagala W, Lasota J, Dukowics A, et al. Vimentin expression appears to be associated with poor prognosis in node-negative ductal NOS breast carcinomas. Am J Pathol 1990;137:1299–1304.
Hendrix MJ, Seftor EA, Seftor RE, et al. Experimental co-expression of vimentin and keratin intermediate filaments in human breast cancer cells results in phenotypic interconversion and increased invasive behavior. Am J Pathol 1997;150:483–495.
Fuchs IB, Lichtenegger W, Buehler H, et al. The prognostic significance of epithelial-mesenchymal transition in breast cancer. Anticancer Res 2002;22:3415–3419.
Oh DS, Troester MA, Usary J, et al. Estrogen-regulated genes predict survival in hormone receptor-positive breast cancers. J Clin Oncol 2006;24:1656–1664.
Selvarajan S, Wong KY, Khoo KS, et al. Over-expression of c-erbB-2 correlates with nuclear morphometry and prognosis in breast carcinoma in Asian women. Pathology 2006;38:528–533.
Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 2004;10:5367–5374.
Tsuda H, Morita D, Kimura M, et al. Correlation of KIT and EGFR overexpression with invasive ductal breast carcinoma of the solid-tubular subtype, nuclear grade 3, and mesenchymal or myoepithelial differentiation. Cancer Sci 2005;96:48–53.
Diallo R, Rody A, Jackisch C, et al. C-KIT expression in ductal carcinoma in situ of the breast: co-expression with HER-2/neu. Hum Pathol 2006;37:205–211.
Livasy CA, Karaca G, Nanda R, et al. Phenotypic evaluation of the basal-like subtype of invasive breast carcinoma. Mod Pathol 2006;19:264–271.
Bryan BB, Schnitt SJ, Collins LC . Ductal carcinoma in situ with basal-like phenotype: a possible precursor to invasive basal-like breast cancer. Mod Pathol 2006;19:617–621.
Wiseman SM, Makretsov N, Nielsen TO, et al. Coexpression of the type 1 growth factor receptor family members HER-1, HER-2, and HER-3 has a synergistic negative prognostic effect on breast carcinoma survival. Cancer 2005;103:1770–1777.
Perou CM, Sørlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature 2000;406:747–752.
Sørlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 2001;98:10869–10874.
Livasy CA, Karaca G, Nanda R, et al. Phenotypic evaluation of the basal-like subtype of invasive breast carcinoma. Mod Pathol 2006;19:264–271.
Yu K, Lee CH, Tan PH, et al. Conservation of breast cancer molecular subtypes and transcriptional patterns of tumor progression across distinct ethnic populations. Clin Cancer Res 2004;10:5508–5517.
Acknowledgements
This study was funded by a grant from the Singapore Cancer Syndicate MS0004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, MS., Wai-Cheong Yip, G., Tse, GK. et al. Expression of basal keratins and vimentin in breast cancers of young women correlates with adverse pathologic parameters. Mod Pathol 21, 1183–1191 (2008). https://doi.org/10.1038/modpathol.2008.90
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/modpathol.2008.90
Keywords
This article is cited by
-
Characterization of ductal carcinoma in situ cell lines established from breast tumor of a Singapore Chinese patient
Cancer Cell International (2014)
-
Vimentin, zeb1 and Sip1 are up-regulated in triple-negative and basal-like breast cancers: association with an aggressive tumour phenotype
Breast Cancer Research and Treatment (2013)
-
Targeted and intracellular triggered delivery of therapeutics to cancer cells and the tumor microenvironment: impact on the treatment of breast cancer
Breast Cancer Research and Treatment (2012)
-
Luminal and basal-like breast cancer cells show increased migration induced by hypoxia, mediated by an autocrine mechanism
BMC Cancer (2011)
-
Proteomic profiling of lipid rafts in a human breast cancer model of tumorigenic progression
Clinical & Experimental Metastasis (2011)