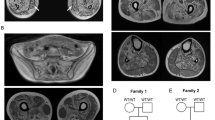
Abstract
Distal hereditary motor neuropathies are pure motor disorders of the peripheral nervous system resulting in severe atrophy and wasting of distal limb muscles1. In two pedigrees with distal hereditary motor neuropathy type II linked to chromosome 12q24.3, we identified the same mutation (K141N) in small heat-shock 22-kDa protein 8 (encoded by HSPB8; also called HSP22). We found a second mutation (K141E) in two smaller families. Both mutations target the same amino acid, which is essential to the structural and functional integrity of the small heat-shock protein αA-crystallin2. This positively charged residue, when mutated in other small heat-shock proteins, results in various human disorders3,4. Coimmunoprecipitation experiments showed greater binding of both HSPB8 mutants to the interacting partner HSPB1. Expression of mutant HSPB8 in cultured cells promoted formation of intracellular aggregates. Our findings provide further evidence that mutations in heat-shock proteins have an important role in neurodegenerative disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Harding, A.E. & Thomas, P.K. Hereditary distal spinal muscular atrophy. A report on 34 cases and a review of the literature. J. Neurol. Sci. 45, 337–348 (1980).
Bera, S., Thampi, P., Cho, W.J. & Abraham, E.C. A positive charge preservation at position 116 of alpha A-crystallin is critical for its structural and functional integrity. Biochemistry 41, 12421–12426 (2002).
Vicart, P. et al. A missense mutation in the alphaB-crystallin chaperone gene causes a desmin-related myopathy. Nat. Genet. 20, 92–95 (1998).
Litt, M. et al. Autosomal dominant congenital cataract associated with a missense mutation in the human alpha crystallin gene CRYAA. Hum. Mol. Genet. 7, 471–474 (1998).
Grohmann, K. et al. Mutations in the gene encoding immunoglobulin μ-binding protein 2 cause spinal muscular atrophy with respiratory distress type 1. Nat. Genet. 29, 75–77 (2001).
Puls, I. et al. Mutant dynactin in motor neuron disease. Nat. Genet. 33 (4), 455–456 (2003).
Antonellis, A. et al. Glycyl tRNA synthetase mutations in Charcot-Marie-Tooth disease type 2D and distal spinal muscular atrophy type V. Am. J. Hum. Genet. 72, 1293–1299 (2003).
Timmerman, V. et al. Distal hereditary motor neuropathy type II (distal HMN II): mapping of a locus to chromosome 12q24. Hum. Mol. Genet. 5, 1065–1069 (1996).
Irobi, J. et al. A clone contig of 12q24.3 encompassing the distal hereditary motor neuropathy type II gene. Genomics 65, 34–43 (2000).
Timmerman, V. et al. Linkage analysis of distal hereditary motor neuropathy type II (distal HMN II) in a single pedigree. J. Neurol. Sci. 109, 41–48 (1992).
Irobi, J. et al. Exclusion of 5 functional candidate genes for distal hereditary motor neuropathy type II (distal HMN II) linked to 12q24.3. Ann. Hum. Genet. 65, 517–529 (2001).
Irobi, J. et al. Mutation analysis of 12 candidate genes for distal hereditary motor neuropathy type II (distal HMN II) linked to 12q24.3. J. Peripher. Nerv. Syst. 7, 87–95 (2002).
Sun, X. et al. Interaction of human HSPB8 (HSPB8) with other small heat shock proteins. J. Biol. Chem. 279, 2394–2402 (2003).
Fontaine, J.M., Rest, J.S., Welsh, M.J. & Benndorf, R. The sperm outer dense fiber protein is the 10th member of the superfamily of mammalian small stress proteins. Cell Stress Chaperones 8, 62–69 (2003).
Kappe, G. et al. The human genome encodes 10 alpha-crystallin-related small heat shock proteins: HspB1-10. Cell Stress Chaperones 8, 53–61 (2003).
Concannon, C.G., Gorman, A.M. & Samali, A. On the role of HSPB1 in regulating apoptosis. Apoptosis 8, 61–70 (2003).
De Jong, W.W., Caspers, G.J. & Leunissen, J.A. Genealogy of the alpha-crystallin-small heat-shock protein superfamily. Int. J. Biol. Macromol. 22, 151–162 (1998).
MacRae, T.H. Structure and function of small heat shock/alpha-crystallin proteins: established concepts and emerging ideas. Cell Mol. Life Sci. 57, 899–913 (2000).
Benndorf, R. et al. HSPB8, a new member of the small heat shock protein superfamily, interacts with mimic of phosphorylated HSPB1 ((3D)HSPB1). J. Biol. Chem. 276, 26753–26761 (2001).
Zobel, A.T.C. et al. Distinct chaperone mechanisms can delay the formation of aggresomes by the myopathy-causing R120G alpha B-crystallin mutant. Hum. Mol. Genet. 12, 1609–1620 (2003).
Bera, S. & Abraham, E.C. The alphaA-crystallin R116C mutant has a higher affinity for forming heteroaggregates with alphaB-crystallin. Biochemistry 41, 297–305 (2002).
Benn, S.C. et al. HSPB1 upregulation and phosphorylation is required for injured sensory and motor neuron survival. Neuron 36, 45–56 (2002).
Andley, U.P., Patel, H.C. & Xi, J.H. The R116C mutation in alpha A-crystallin diminishes its protective ability against stress-induced lens epithelial cell apoptosis. J. Biol. Chem. 277, 10178–10186 (2002).
Kamradt, M.C., Chen, F., Sam, S. & Cryns, V.L. The small heat shock protein alpha B-crystallin negatively regulates apoptosis during myogenic differentiation by inhibiting caspase-3 activation. J. Biol. Chem. 277, 38731–38736 (2002).
Suzuki, A. et al. MKBP, a novel member of the small heat shock protein family, binds and activates the myotonic dystrophy protein kinase. J. Cell Biol. 140, 1113–1124 (1998).
Vleminckx, V. et al. Upregulation of HSPB1 in a transgenic model of ALS. J. Neuropathol. Exp. Neurol. 61, 968–974 (2002).
Carper, S.W., Rocheleau, T.A. & Storm, F.K. cDNA sequence of a human heat-shock protein HSPB1. Nucleic Acids Res. 18, 6457 (1990).
Gettemans, J., De Ville, Y., Waelkens, E. & Vandekerckhove, J. The actin-binding properties of the Physarum actin-fragmin complex. Regulation by calcium, phospholipids, and phosphorylation. J. Biol. Chem. 270, 2644–2651 (1995).
Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Matsudaira, P.T. & Burgess, D.R. SDS microslab linear gradient polyacrylamide gel electrophoresis. Anal. Biochem. 87, 386–396 (1978).
Acknowledgements
We thank the affected individuals and families for their cooperation and participation; the VIB Genetic Service Facility (http://www.vibgeneticservicefacility.be/) for genotyping and sequencing; R. Benndorf for the human HSPB8 antibody; J.P. Timmermans for help with confocal microscopy; J. Theuns for discussions; and B. Ishpekova, M. Rédlová, J. Haberlová and M. Bojar for help with EMG and sampling material from affected individuals. This research project was supported by the Special Research Fund of the Universities of Antwerp, Ghent and Leuven, Fund for Scientific Research-Flanders, Medical Foundation Queen Elisabeth, “Fortis Bank Verzekeringen”, Interuniversity Attraction Poles program P5/19 of the Belgian Federal Science Office and “Association Belge contre les Maladies Neuromusculaires”. A.J. received fellowships from the Belgian Federal Science Office and Fund for Scientific Research-Flanders. N.V. and I.D. received PhD fellowships from the Institute of Science and Technology. W.R. is a clinical investigator of the Fund for Scientific Research-Flanders Flanders, Belgium. P.S. and R.M. were supported by grants from the Internal Grant Agency of Ministry of Health, Czech Republic. P.S. received a fellowship of the European Neurological Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Irobi, J., Impe, K., Seeman, P. et al. Hot-spot residue in small heat-shock protein 22 causes distal motor neuropathy. Nat Genet 36, 597–601 (2004). https://doi.org/10.1038/ng1328
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ng1328
This article is cited by
-
Small heat shock proteins operate as molecular chaperones in the mitochondrial intermembrane space
Nature Cell Biology (2023)
-
Molecular Chaperones as Therapeutic Target: Hallmark of Neurodegenerative Disorders
Molecular Neurobiology (2023)
-
Heat-shock chaperone HSPB1 regulates cytoplasmic TDP-43 phase separation and liquid-to-gel transition
Nature Cell Biology (2022)
-
HSP22 (HSPB8) positively regulates PGF2α-induced synthesis of interleukin-6 and vascular endothelial growth factor in osteoblasts
Journal of Orthopaedic Surgery and Research (2021)
-
A novel deletion in the C-terminal region of HSPB8 in a family with rimmed vacuolar myopathy
Journal of Human Genetics (2021)