Abstract
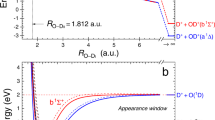
At the transition from the gas to the liquid phase of water, a wealth of new phenomena emerge, which are absent for isolated H2O molecules. Many of those are important for the existence of life, for astrophysics and atmospheric science. In particular, the response to electronic excitation changes completely as more degrees of freedom become available. Here we report the direct observation of an ultrafast transfer of energy across the hydrogen bridge in (H2O)2 (a so-called water dimer). This intermolecular coulombic decay leads to an ejection of a low-energy electron from the molecular neighbour of the initially excited molecule. We observe that this decay is faster than the proton transfer that is usually a prominent pathway in the case of electronic excitation of small water clusters and leads to dissociation of the water dimer into two H2O+ ions. As electrons of low energy (∼0.7–20 eV) have recently been found to efficiently break-up DNA constituents1,2, the observed decay channel might contribute as a source of electrons that can cause radiation damage in biological matter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boudaiffa, B., Cloutier, P., Hunting, D., Huels, M. A. & Sanche, L. Resonant formation of DNA strand breaks by low-energy (3–20 eV) electrons. Science 287, 1658–1660 (2000).
Hanel, G. et al. Electron attachment to uracil: Effective destruction at subexcitation energies. Phys. Rev. Lett. 90, 188104 (2003).
Angle, L. & Stace, A. J. Dissociation patterns of (H2O)n+ cluster ions, for n=26. Chem. Phys. Lett. 345, 277–281 (2001).
Belau, L., Wilson, K. R., Leone, S. R. & Ahmed, M. Vacuum ultraviolet (VUV) photoionization of small water clusters. J. Phys. Chem. A 111, 10075–10083 (2007).
Dong, F., Heinbuch, S., Rocca, J. J. & Bernstein, E. R. Dynamics and fragmentation of van der Waals clusters: (H2O)(n), (CH3OH)(n), and (NH3)(n) upon ionization by a 26.5 eV soft X-ray laser. J. Chem. Phys. 124, 224319 (2006).
Shiromaru, H., Shinohara, H., Washida, N., Yoo, H. S. & Kimura, K. Synchrotron radiation measurements of appearance potentials for (H2O)+2, (H2O)+3, (H2O)2H+ and (H2O)3H+ in supersonic jets. Chem. Phys. Lett. 141, 7–11 (1987).
Radi, P. P. et al. Femtosecond photoionization of (H2O)n and (D2O)n clusters. J. Chem. Phys. 111, 512–518 (1999).
Tachikawa, H. Ionization dynamics of the small-sized water clusters: A direct ab initio trajectory study. J. Phys. Chem. A 108, 7853–7862 (2004).
Furuhama, A., Dupuis, M. & Hirao, K. Reactions associated with ionization in water: A direct ab initio dynamics study of ionization in (H2O)17 . J. Chem. Phys. 124, 164310 (2006).
Cederbaum, L. S., Zobeley, J. & Tarantelli, F. Giant intermolecular decay and fragmentation of clusters. Phys. Rev. Lett. 79, 4778–4781 (1997).
Marburger, S., Kugeler, O., Hergenhahn, U. & Möller, T. Experimental evidence for interatomic coulombic decay in Ne clusters. Phys. Rev. Lett. 93, 203401 (2003).
Jahnke, T. et al. Experimental observation of interatomic coulombic decay in neon dimers. Phys. Rev. Lett. 93, 163401 (2004).
Morishita, Y. et al. Experimental evidence of interatomic coulombic decay from the Auger final states in argon dimers. Phys. Rev. Lett. 96, 243402 (2006).
Barth, S. et al. Observation of resonant interatomic coulombic decay in Ne clusters. J. Chem. Phys. 122, 241102 (2005).
Aoto, T. et al. Properties of resonant interatomic coulombic decay in Ne dimers. Phys. Rev. Lett. 97, 243401 (2006).
Jahnke, T. et al. Experimental separation of virtual photon exchange and electron transfer in interatomic coulombic decay of neon dimers. Phys. Rev. Lett. 99, 153401 (2007).
Kreidi, K. et al. Localization of inner shell photo electron emission and ICD in neon dimers. J. Phys. B 41, 101002 (2008).
Kreidi, K. et al. Relaxation processes following 1s photoionization and Auger decay in Ne2 . Phys. Rev. A 78, 043422 (2008).
Ueda, K. et al. Interatomic coulombic decay following the Auger decay: Experimental evidence in rare-gas dimers. J. Electron Spectrosc. Relat. Phenom. 3–10, 166–167 (2008).
Aziz, E. F., Ottosson, N., Faubel, M., Hertel, I. V. & Winter, B. Interaction between liquid water and hydroxide revealed by core–hole de-excitation. Nature 455, 89–91 (2008).
Müller, I. B. & Cederbaum, L. S. Ionization and double ionization of small water clusters. J. Chem. Phys. 125, 204305 (2006).
Dörner, R. et al. Cold target recoil ion momentum spectroscopy: A ‘momentum microscope’ to view atomic collision dynamics. Phys. Rep. 330, 96–192 (2000).
Ullrich, J. et al. Recoil-ion and electron momentum spectroscopy: Reaction-microscopes. Rep. Prog. Phys. 66, 1463–1545 (2003).
Jahnke, T. et al. Multicoincidence studies of photo and Auger electrons from fixed-in-space molecules using the COLTRIMS technique. J. Electron Spectrosc. Relat. Phenom. 73, 229–238 (2004).
Jagutzki, O. et al. A broad-application microchannel-plate detector system for advanced particle or photon detection tasks: Large area imaging, precise multi-hit timing information and high detection rate. NIM A 477, 244–249 (2002).
Gislason, E. A. Series expansion for Franck–Condon factors 1. Linear potential and reflection approximation. J. Chem. Phys. 58, 3702–3707 (1973).
Wiley, W. C. & McLaren, I. H. Time-of-flight mass spectrometer with improved resolution. Rev. Sci. Instrum. 26, 1150–1157 (1955).
Mucke, M. et al. A hitherto unrecognized source of low-energy electrons in water. Nature Phys. 10.1038/nphys1500 (2010).
Acknowledgements
We would like to thank the staff at BESSY, especially H. Pfau and G. Reichardt, for extraordinary support during the beamtime. Many discussions on ICD with L. Cederbaum and his group are gratefully acknowledged. This work was supported by the DFG and BESSY GmbH.
Author information
Authors and Affiliations
Contributions
Experiment design and set-up (T.J., T.H., K.K., H.S., O.J.), beamtime (T.J., H.S., T.H., K.K., C.S., M.M., M.S., N.N., R.W., S.V., F.A.), data analysis (T.J.), interpretation of data (T.J., R.D., T.W., A.M., H.S.B.), manuscript preparation (T.J., R.D., T.W., M.S., A.C.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Jahnke, T., Sann, H., Havermeier, T. et al. Ultrafast energy transfer between water molecules. Nature Phys 6, 139–142 (2010). https://doi.org/10.1038/nphys1498
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/nphys1498
This article is cited by
-
X-ray radiation damage cycle of solvated inorganic ions
Nature Communications (2024)
-
Prebiotic chemical origin of biomolecular complementarity
Communications Chemistry (2023)
-
Directly imaging excited state-resolved transient structures of water induced by valence and inner-shell ionisation
Nature Communications (2023)
-
Radiation damage by extensive local water ionization from two-step electron-transfer-mediated decay of solvated ions
Nature Chemistry (2023)
-
Ortho-para interconversion of nuclear states of H2O through replica transition state: prospect of quantum entanglement at homodromic Bjerrum defect site
Journal of Molecular Modeling (2023)