Abstract
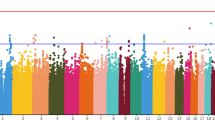
Neurocognitive deficits are a core feature of schizophrenia and, therefore, represent potentially critical outcome variables for assessing antipsychotic treatment response. We performed genome-wide association studies (GWAS) with 492K single nucleotide polymorphisms (SNPs) in a sample of 738 patients with schizophrenia from the Clinical Antipsychotic Trials of Intervention Effectiveness study. Outcome variables consisted of a neurocognitive battery administered at multiple time points over an 18-month period, measuring processing speed, verbal memory, vigilance, reasoning, and working memory domains. Genetic mediation of improvements in each of these five domains plus a composite neurocognitive measure was assessed for each of five antipsychotics (olanzapine, perphenazine, quetiapine, risperidone, and ziprasidone). Six SNPs achieved genome-wide significance using a pre-specified threshold that ensures, on average, only 1 in 10 findings is a false discovery. These six SNPs were located within, or in close proximity to, genes EHF, SLC26A9, DRD2, GPR137B, CHST8, and IL1A. The more robust findings, that is those significant across multiple neurocognitive domains and having adjacent SNPs showing evidence for association, were rs286913 at the EHF gene (p-value 6.99 × 10−8, q-value 0.034, mediating the effects of ziprasidone on vigilance), rs11240594 at SLC26A9 (p-value 1.4 × 10−7, q-value 0.068, mediating the effects of olanzapine on processing speed), and rs11677416 at IL1A (p-value 6.67 × 10−7, q-value 0.081, mediating the effects of olanzapine on working memory). This study has generated several novel candidate genes for antipsychotic response. However, our findings will require replication and functional validation. To facilitate replication efforts, we provide all GWAS p-values for download.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Altshuler D, Daly MJ, Lander ES (2008). Genetic mapping in human disease. Science 322: 881–888.
Anttila S, Kampman O, Illi A, Rontu R, Lehtimaki T, Leinonen E (2007). Association between 5-HT2A, TPH1 and GNB3 genotypes and response to typical neuroleptics: a serotonergic approach. BMC Psychiatry 7: 22.
Arranz MJ, de Leon J (2007). Pharmacogenetics and pharmacogenomics of schizophrenia: a review of last decade of research. Mol Psychiatry 12: 707–747.
Banks WA, Farr SA, La Scola ME, Morley JE (2001). Intravenous human interleukin-1α impairs memory processing in mice: dependence on blood-brain barrier transport into posterior division of the septum. J Pharmacol Exp Ther 299: 536–541.
Basile VS, Masellis M, Potkin SG, Kennedy JL (2002). Pharmacogenomics in schizophrenia: the quest for individualized therapy. Hum Mol Genet 11: 2517–2530.
Beecham GW, Martin ER, Li YJ, Slifer MA, Gilbert JR, Haines JL et al (2009). Genome-wide association study implicates a chromosome 12 risk locus for late-onset Alzheimer disease. Am J Hum Genet 84: 35–43.
Benjamini Y, Hochberg Y (1995). Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57: 289–300.
Benton AL H, K (1978). Multilingual Aphasia Examination Manual (Revised). University of Iowa: Iowa City, IA.
Black MA (2004). A note on the adaptive control of false discovery rates. J R Stat Soc B 66: 297–304.
Brandt J (1991). The hopkins verbal learning test: development of a new memory test with six equivalent forms. Clin Neuropsychol 5: 125–142.
Brown BW, Russell K (1997). Methods of correcting for multiple testing: operating characteristics. Stat Med 16: 2511–2528.
Bukszar J, McClay JL, van den Oord EJ (2009). Estimating the posterior probability that genome-wide association findings are true or false. Bioinformatics 25: 1807–1813.
Burmester JK, Sedova M, Shapero MH, Mansfield E (2010). DMET microarray technology for pharmacogenomics-based personalized medicine. Methods Mol Biol 632: 99–124.
Campbell DB, Ebert PJ, Skelly T, Stroup TS, Lieberman J, Levitt P et al (2008). Ethnic stratification of the association of RGS4 variants with antipsychotic treatment response in schizophrenia. Biol Psychiatry 63: 32–41.
Cornblatt BA, Risch NJ, Faris G, Friedman D, Erlenmeyer-Kimling L (1988). The continuous performance test, identical pairs version (CPT-IP): I new findings about sustained attention in normal families. Psychiatry Res 26: 223–238.
Dorow DS, Devereux L, Dietzsch E, De Kretser T (1993). Identification of a new family of human epithelial protein kinases containing two leucine/isoleucine-zipper domains. Eur J Biochem 213: 701–710.
Dorwart MR, Shcheynikov N, Wang Y, Stippec S, Muallem S (2007). SLC26A9 is a Cl(−) channel regulated by the WNK kinases. J Physiol 584 (Pt 1): 333–345.
Efron BT, Storey R, Tusher JD, V (2001). Empirical Bayes analysis of a microarray experiment. J Am Stat Assoc 96: 1151–1160.
Fernandez E, Collins MO, Uren RT, Kopanitsa MV, Komiyama NH, Croning MD et al (2009). Targeted tandem affinity purification of PSD-95 recovers core postsynaptic complexes and schizophrenia susceptibility proteins. Mol Syst Biol 5: 269.
First MS, Gibbon R, Williams M, J (1994). Structured Clinical Interview for DSM-IV Axis I Disorders—Administration Booklet. American Psychiatric Press: Washington DC.
Garriock HA, Kraft JB, Shyn SI, Peters EJ, Yokoyama JS, Jenkins GD et al (2010). A genomewide association study of citalopram response in major depressive disorder. Biol Psychiatry 67: 133–138.
Gold JM, Carpenter C, Randolph C, Goldberg TE, Weinberger DR (1997). Auditory working memory and wisconsin card sorting test performance in schizophrenia. Arch Gen Psychiatry 54: 159–165.
Goldberg TE, Goldman RS, Burdick KE, Malhotra AK, Lencz T, Patel RC et al (2007). Cognitive improvement after treatment with second-generation antipsychotic medications in first-episode schizophrenia: is it a practice effect? Arch Gen Psychiatry 64: 1115–1122.
Goldstein H (1995). Multilevel Statistical Models. Arnold: London.
Goring HH, Terwilliger JD, Blangero J (2001). Large upward bias in estimation of locus-specific effects from genomewide scans. Am J Hum Genet 69: 1357–1369.
Green MF (1996). What are the functional consequences of neurocognitive deficits in schizophrenia? Am J Psychiatry 153: 321–330.
Greenwood TA, Braff DL, Light GA, Cadenhead KS, Calkins ME, Dobie DJ et al (2007). Initial heritability analyses of endophenotypic measures for schizophrenia: the consortium on the genetics of schizophrenia. Arch Gen Psychiatry 64: 1242–1250.
Hadi AS, Simonoff JS (1993). Procedures for the identification of multiple outliers in linear-models. J Am Stat Assoc 88: 1264–1272.
Hamdani N, Tabeze JP, Ramoz N, Ades J, Hamon M, Sarfati Y et al (2008). The CNR1 gene as a pharmacogenetic factor for antipsychotics rather than a susceptibility gene for schizophrenia. Eur Neuropsychopharmacol 18: 34–40.
He G, Wang HR, Huang SK, Huang CL (2007). Intersectin links WNK kinases to endocytosis of ROMK1. J Clin Invest 117: 1078–1087.
Heaton RK, Chelune GJ, Taley JL, Kay GG, Curtiss G (1993). Wisconsin Card Sorting Test Manual: Revised and Expanded. Psychological Assessment Resources: Odessa, FL.
Hill SK, Harris MS, Herbener ES, Pavuluri M, Sweeney JA (2008). Neurocognitive allied phenotypes for schizophrenia and bipolar disorder. Schizophr Bull 34: 743–759.
Hwang R, Shinkai T, De Luca V, Ni X, Potkin SG, Lieberman JA et al (2007). Association study of four dopamine D1 receptor gene polymorphisms and clozapine treatment response. J Psychopharmacol 21: 718–727.
Hyman SE, Fenton WS (2003). Medicine. What are the right targets for psychopharmacology? Science 299: 350–351.
Ioannidis JP, Ntzani EE, Trikalinos TA, Contopoulos-Ioannidis DG (2001). Replication validity of genetic association studies. Nat Genet 29: 306–309.
Kahle KT, Macgregor GG, Wilson FH, Van Hoek AN, Brown D, Ardito T et al (2004). Paracellular Cl- permeability is regulated by WNK4 kinase: insight into normal physiology and hypertension. Proc Natl Acad Sci USA 101: 14877–14882.
Kane JM (1999). Pharmacologic treatment of schizophrenia. Biol Psychiatry 46: 1396–1408.
Kapur S, Zipursky RB, Remington G (1999). Clinical and theoretical implications of 5-HT2 and D2 receptor occupancy of clozapine, risperidone, and olanzapine in schizophrenia. Am J Psychiatry 156: 286–293.
Kas K, Finger E, Grall F, Gu X, Akbarali Y, Boltax J et al (2000). ESE-3, a novel member of an epithelium-specific ets transcription factor subfamily, demonstrates different target gene specificity from ESE-1. J Biol Chem 275: 2986–2998.
Keefe RS, Bilder RM, Davis SM, Harvey PD, Palmer BW, Gold JM et al (2007a). Neurocognitive effects of antipsychotic medications in patients with chronic schizophrenia in the CATIE Trial. Arch Gen Psychiatry 64: 633–647.
Keefe RS, Bilder RM, Harvey PD, Davis SM, Palmer BW, Gold JM et al (2006). Baseline neurocognitive deficits in the CATIE schizophrenia trial. Neuropsychopharmacology 31: 2033–2046.
Keefe RS, Malhotra AK, Meltzer HY, Kane JM, Buchanan RW, Murthy A et al (2008). Efficacy and safety of donepezil in patients with schizophrenia or schizoaffective disorder: significant placebo/practice effects in a 12-week, randomized, double-blind, placebo-controlled trial. Neuropsychopharmacology 33: 1217–1228.
Keefe RS, Mohs RC, Bilder RM, Harvey PD, Green MF, Meltzer HY et al (2003). Neurocognitive assessment in the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) project schizophrenia trial: development, methodology, and rationale. Schizophr Bull 29: 45–55.
Keefe RS, Sweeney JA, Gu H, Hamer RM, Perkins DO, McEvoy JP et al (2007b). Effects of olanzapine, quetiapine, and risperidone on neurocognitive function in early psychosis: a randomized, double-blind 52-week comparison. Am J Psychiatry 164: 1061–1071.
Kirchheiner J, Nickchen K, Bauer M, Wong ML, Licinio J, Roots I et al (2004). Pharmacogenetics of antidepressants and antipsychotics: the contribution of allelic variations to the phenotype of drug response. Mol Psychiatry 9: 442–473.
Kremen WS, Seidman LJ, Faraone SV, Toomey R, Tsuang MT (2004). Heterogeneity of schizophrenia: a study of individual neuropsychological profiles. Schizophr Res 71: 307–321.
Lafayette Instrument Company (1989). Grooved Pegboard Instruction Manual (Model 32025). Lafayette Instrument Company: Lafayette, IN.
Lei SF, Yang TL, Tan LJ, Chen XD, Guo Y, Guo YF et al (2009). Genome-wide association scan for stature in Chinese: evidence for ethnic specific loci. Hum Genet 125: 1–9.
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO et al (2005). Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 353: 1209–1223.
Liu YZ, Guo YF, Wang L, Tan LJ, Liu XG, Pei YF et al (2009). Genome-wide association analyses identify SPOCK as a key novel gene underlying age at menarche. PLoS Genet 5: e1000420.
Loriol C, Dulong S, Avella M, Gabillat N, Boulukos K, Borgese F et al (2008). Characterization of SLC26A9, facilitation of Cl(−) transport by bicarbonate. Cell Physiol Biochem 22: 15–30.
Lyons-Warren A, Lillie R, Hershey T (2004). Short- and long-term spatial delayed response performance across the lifespan. Dev Neuropsychol 26: 661–678.
Malhotra AK, Murphy Jr GM., Kennedy JL (2004). Pharmacogenetics of psychotropic drug response. Am J Psychiatry 161: 780–796.
Mancama D, Mata I, Kerwin RW, Arranz MJ (2007). Choline acetyltransferase variants and their influence in schizophrenia and olanzapine response. Am J Med Genet B Neuropsychiatr Genet 144B: 849–853.
McClay JL, Adkins DE, Aberg K, Stroup S, Perkins DO, Vladimirov VI et al (2009). Genome-wide pharmacogenomic analysis of response to treatment with antipsychotics. Mol Psychiatry; e-pub ahead of print 1 September 2009, PMID 19721433.
Meary A, Brousse G, Jamain S, Schmitt A, Szoke A, Schurhoff F et al (2008). Pharmacogenetic study of atypical antipsychotic drug response: involvement of the norepinephrine transporter gene. Am J Med Genet B Neuropsychiatr Genet 147B: 491–494.
Meisenzahl EM, Rujescu D, Kirner A, Giegling I, Kathmann N, Leinsinger G et al (2001). Association of an interleukin-1beta genetic polymorphism with altered brain structure in patients with schizophrenia. Am J Psychiatry 158: 1316–1319.
Meltzer HY, Okayli G (1995). Reduction of suicidality during clozapine treatment of neuroleptic-resistant schizophrenia: impact on risk-benefit assessment. Am J Psychiatry 152: 183–190.
Mesholam-Gately RI, Giuliano AJ, Goff KP, Faraone SV, Seidman LJ (2009). Neurocognition in first-episode schizophrenia: a meta-analytic review. Neuropsychology 23: 315–336.
Need AC, Keefe RS, Ge D, Grossman I, Dickson S, McEvoy JP et al (2009). Pharmacogenetics of antipsychotic response in the CATIE trial: a candidate gene analysis. Eur J Hum Genet 17: 946–957.
O'Donovan MC, Craddock N, Norton N, Williams H, Peirce T, Moskvina V et al (2008). Identification of loci associated with schizophrenia by genome-wide association and follow-up. Nat Genet 40: 1053–1055.
Pinheiro JC, Bates DM (2000). Mixed-Effects Models in S and S-Plus. Springer: New York, NY.
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al (2007). PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81: 559–575.
Roses AD (2000). Pharmacogenetics and the practice of medicine. Nature 405: 857–865.
Sanger DJ (2004). The search for novel antipsychotics: pharmacological and molecular targets. Expert Opin Ther Targets 8: 631–641.
Searle SR, Casella G, McCulloch CE (1992). Variance Components. Wiley: New York, NY.
Shi J, Levinson DF, Duan J, Sanders AR, Zheng Y, Pe'er I et al (2009). Common variants on chromosome 6p22.1 are associated with schizophrenia. Nature 460: 753–757.
Storey J (2003). The positive false discovery rate: a Bayesian interpretation and the q-value. Ann Stat 31: 2013–2035.
Storey JD, Tibshirani R (2003). Statistical significance for genomewide studies. Proc Natl Acad Sci USA 100: 9440–9445.
Stroup TS, McEvoy JP, Swartz MS, Byerly MJ, Glick ID, Canive JM et al (2003). The National Institute of Mental Health Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) project: schizophrenia trial design and protocol development. Schizophr Bull 29: 15–31.
Sullivan PF, Lin D, Tzeng JY, van den Oord E, Perkins D, Stroup TS et al (2008). Genomewide association for schizophrenia in the CATIE study: results of stage 1. Mol Psychiatry 13: 570–584.
Thierry-Mieg D, Thierry-Mieg J (2006). AceView: a comprehensive cDNA-supported gene and transcripts annotation. Genome Biol 7 (Suppl 1): 11–S12 14.
Tsukita S, Furuse M (2000). Pores in the wall: claudins constitute tight junction strands containing aqueous pores. J Cell Biol 149: 13–16.
Tugores A, Le J, Sorokina I, Snijders AJ, Duyao M, Reddy PS et al (2001). The epithelium-specific ETS protein EHF/ESE-3 is a context-dependent transcriptional repressor downstream of MAPK signaling cascades. J Biol Chem 276: 20397–20406.
van den Oord EJ, Adkins DE, McClay J, Lieberman J, Sullivan PF (2009). A systematic method for estimating individual responses to treatment with antipsychotics in CATIE. Schizophr Res 107: 13–21.
van den Oord EJ, Sullivan PF (2003). False discoveries and models for gene discovery. Trends Genet 19: 537–542.
Wechsler D (1981). WAIS-R Manual: Wechsler Adult Intelligence Scale-Revised. Psychological Corporation: New York, NY.
Wechsler D (1991). Wechsler Intelligence Scale for Children. Psychological Corporation: San Antonio, TX.
Wheeler DL, Church DM, Federhen S, Lash AE, Madden TL, Pontius JU et al (2003). Database resources of the National Center for Biotechnology. Nucleic Acids Res 31: 28–33.
Williams M (2003). Genome-based drug discovery: prioritizing disease-susceptibility/disease-associated genes as novel drug targets for schizophrenia. Curr Opin Investig Drugs 4: 31–36.
Zandi PP, Judy JT (2010). The promise and reality of pharmacogenetics in psychiatry. Psychiatr Clin North Am 33: 181–224.
Zaykin DV, Young SS, Westfall PH (2000). Using the false discovery rate approach in the genetic dissection of complex traits: a response to Weller et al. Genetics 154: 1917–1918.
Acknowledgements
The CATIE project was supported by NIMH contract N01 MH90001. Dr Sullivan was supported by R01s MH074027 and MH077139.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Eli Lilly funded the GWAS genotyping performed at Perlegen Sciences. Dr Sullivan reports receiving research funding from Eli Lilly in connection with this project. Dr Stroup reports that in the past 3 years he has received consulting fees from Lilly and Janssen. Dr Lieberman reports having received research funding from AstraZeneca Pharmaceuticals, Bristol-Myers Squibb, GlaxoSmithKline, Janssen Pharmaceutica and Pfizer, and consulting and educational fees from AstraZeneca Pharmaceuticals, Bristol-Myers Squibb, Eli Lilly, Forest Pharmaceuticals, GlaxoSmithKline, Janssen Pharmaceutica, Novartis, Pfizer, and Solvay. Dr Keefe reports that he currently or in the past 12 months has received investigator-initiated research funding support from the National Institute of Mental Health, Allon, Novartis and the Singapore National Medical Research Council, and an unrestricted educational grant from Astra-Zeneca. He currently or in the past 12 months has received honoraria or served as a consultant or advisory board member for Abbott, Astra-Zeneca, BiolineRx, Bristol Myers Squibb, Cephalon, Dainippon Sumitomo Pharma, Eli Lilly, Johnson & Johnson, Lundbeck, Memory Pharmaceuticals, Merck, Neurosearch, Orion, Orexigen, Otsuka, Pfizer, Roche, Targacept, Sanofi/Aventis, Shire, Wyeth, and Xenoport. In the past, he has received honoraria or served as a consultant or advisory board member for Acadia, Cortex, Cyberonics, Forest, Gabriel, GlaxoSmithKline, Repligen, Saegis, Shering-Plough, and has received research funding from Astra-Zeneca, Eli Lilly, Janssen, and Pfizer. Dr Keefe receives royalties from the Brief Assessment of Cognition in Schizophrenia (BACS) testing battery and the MATRICS Battery (BACS Symbol Coding). Dr McEvoy declares receiving honoraria from Lilly, research grants and consulting fees from Sunovion, research grants from GlaxoSmithKline and Novartis, and serving on an advisory board with Merck. Drs McClay, Adkins, Åberg, Bukszár, Khachane, Perkins, Vann, Beardsley, and van den Oord declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Neuropsychopharmacology website
Supplementary information
Rights and permissions
About this article
Cite this article
McClay, J., Adkins, D., Åberg, K. et al. Genome-Wide Pharmacogenomic Study of Neurocognition As an Indicator of Antipsychotic Treatment Response in Schizophrenia. Neuropsychopharmacol 36, 616–626 (2011). https://doi.org/10.1038/npp.2010.193
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/npp.2010.193
Keywords
This article is cited by
-
Exploring functional dysconnectivity in schizophrenia: alterations in eigenvector centrality mapping and insights into related genes from transcriptional profiles
Schizophrenia (2024)
-
Association of DRD2, DRD4 and COMT genes variants and their gene-gene interactions with antipsychotic treatment response in patients with schizophrenia
BMC Psychiatry (2023)
-
Targeted exome sequencing identifies five novel loci at genome-wide significance for modulating antidepressant response in patients with major depressive disorder
Translational Psychiatry (2020)
-
A functional HTR1A polymorphism, rs6295, predicts short-term response to lurasidone: confirmation with meta-analysis of other antipsychotic drugs
The Pharmacogenomics Journal (2020)
-
Genetics and Antipsychotic Response in Schizophrenia: an Update
Current Behavioral Neuroscience Reports (2017)