Abstract
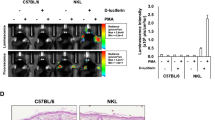
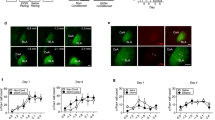
Nuclear factor κ-light chain enhancer of activated B cells (NF-κB) is a transcription factor commonly associated with innate immunity and is activated by infection and inflammation. NF-κB has recently gained attention as a mediator of complex psychiatric phenomena such as stress and addiction. In regards to alcohol, most research on NF-κB has focused on neurotoxicity and few studies have explored the role of NF-κB in alcohol reward, reinforcement, or consumption. In these studies, we used conditioned place preference to assess the activity of NF-κB in response to rewarding doses of alcohol. To measure NF-κB activity we used a line of transgenic mice that express the LacZ gene under the control of an NF-κB-regulated promoter. In these animals, staining for β-galactosidase (β-gal) identifies cells in which NF-κB has been activated. We then used the Daun02 inactivation method to specifically silence NF-κB-expressing cells during place preference conditioning. Daun02 is an inactive prodrug that is converted to the inhibitory molecule daunorubicin by β-gal. After alcohol place conditioning, we observed increased β-gal staining in the nucleus accumbens (NAC) shell and dorsal raphe nucleus, and found that disruption of NF-κB-expressing cells using Daun02 attenuated the development of alcohol place preference when infused into the NAC shell following conditioning sessions. We found this effect to be regionally and temporally specific. These results suggest that, in addition to its role in alcohol-induced neurotoxicity, NF-κB mediates the development of alcohol place preference via its actions in the NAC shell.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Bhakar AL, Tannis LL, Zeindler C, Russo MP, Jobin C, Park DS et al (2002). Constitutive nuclear factor-kappa B activity is required for central neuron survival. J Neurosci 22: 8466–8475.
Blednov YA, Benavidez JM, Geil C, Perra S, Morikawa H, Harris RA (2011). Activation of inflammatory signaling by lipopolysaccharide produces a prolonged increase of voluntary alcohol intake in mice. Brain Behav Immun 25 (Suppl 1): S92–S105.
Boileau I, Assaad JM, Pihl RO, Benkelfat C, Leyton M, Diksic M et al (2003). Alcohol promotes dopamine release in the human nucleus accumbens. Synapse 49: 226–231.
Bossert JM, Stern AL, Theberge FR, Cifani C, Koya E, Hope BT et al (2011). Ventral medial prefrontal cortex neuronal ensembles mediate context-induced relapse to heroin. Nat Neurosci 14: 420–422.
Christoffel DJ, Golden SA, Dumitriu D, Robison AJ, Janssen WG, Ahn HF et al (2011). IkappaB kinase regulates social defeat stress-induced synaptic and behavioral plasticity. J Neurosci 31: 314–321.
Christoffel DJ, Golden SA, Heshmati M, Graham A, Birnbaum S, Neve RL et al (2012). Effects of inhibitor of kappaB kinase activity in the nucleus accumbens on emotional behavior. Neuropsychopharmacology 37: 2615–2623.
Crews F, Nixon K, Kim D, Joseph J, Shukitt-Hale B, Qin L et al (2006). BHT blocks NF-kappaB activation and ethanol-induced brain damage. Alcohol Clin Exp Res 30: 1938–1949.
Crews FT, Nixon K (2009). Mechanisms of neurodegeneration and regeneration in alcoholism. Alcohol Alcohol 44: 115–127.
Crews FT, Sarkar DK, Qin L, Zou J, Boyadjieva N, Vetreno RP (2015). Neuroimmune function and the consequences of alcohol exposure. Alcohol Res 37: 344–351.
Cruz FC, Babin KR, Leao RM, Goldart EM, Bossert JM, Shaham Y et al (2014). Role of nucleus accumbens shell neuronal ensembles in context-induced reinstatement of cocaine-seeking. J Neurosci 34: 7437–7446.
Cruz FC, Koya E, Guez-Barber DH, Bossert JM, Lupica CR, Shaham Y et al (2013). New technologies for examining the role of neuronal ensembles in drug addiction and fear. Nat Rev Neurosci 14: 743–754.
de Guglielmo G, Crawford E, Kim S, Vendruscolo LF, Hope BT, Brennan M et al (2016). Recruitment of a neuronal ensemble in the central nucleus of the amygdala is required for alcohol dependence. J Neurosci 36: 9446–9453.
Fanous S, Goldart EM, Theberge FR, Bossert JM, Shaham Y, Hope BT (2012). Role of orbitofrontal cortex neuronal ensembles in the expression of incubation of heroin craving. J Neurosci 32: 11600–11609.
Farquhar D, Pan BF, Sakurai M, Ghosh A, Mullen CA, Nelson JA (2002). Suicide gene therapy using E. coli beta-galactosidase. Cancer Chemother Pharmacol 50: 65–70.
Funk D, Coen K, Tamadon S, Hope BT, Shaham Y, Le AD (2016). Role of central amygdala neuronal ensembles in incubation of nicotine craving. J Neurosci 36: 8612–8623.
Gerondakis S, Fulford TS, Messina NL, Grumont RJ (2014). NF-kappaB control of T cell development. Nat Immunol 15: 15–25.
Kaltschmidt B, Ndiaye D, Korte M, Pothion S, Arbibe L, Prullage M et al (2006). NF-kappaB regulates spatial memory formation and synaptic plasticity through protein kinase A/CREB signaling. Mol Cell Biol 26: 2936–2946.
Koo JW, Russo SJ, Ferguson D, Nestler EJ, Duman RS (2010). Nuclear factor-kappaB is a critical mediator of stress-impaired neurogenesis and depressive behavior. Proc Natl Acad Sci USA 107: 2669–2674.
Koya E, Golden SA, Harvey BK, Guez-Barber DH, Berkow A, Simmons DE et al (2009). Targeted disruption of cocaine-activated nucleus accumbens neurons prevents context-specific sensitization. Nat Neurosci 12: 1069–1073.
Nennig SE, Schank JR (2017). The role of NFκB in drug addiction: beyond inflammation. Alcohol Alcohol 52: 172–179.
Oeckinghaus A, Ghosh S (2009). The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol 1: a000034.
Pfarr S, Meinhardt MW, Klee ML, Hansson AC, Vengeliene V, Schonig K et al (2015). Losing control: excessive alcohol seeking after selective inactivation of cue-responsive neurons in the infralimbic cortex. J Neurosci 35: 10750–10761.
Qin L, Crews FT (2012). NADPH oxidase and reactive oxygen species contribute to alcohol-induced microglial activation and neurodegeneration. J Neuroinflam 9: 5.
Qin L, He J, Hanes RN, Pluzarev O, Hong JS, Crews FT (2008). Increased systemic and brain cytokine production and neuroinflammation by endotoxin following ethanol treatment. J Neuroinflam 5: 10.
Qin L, Wu X, Block ML, Liu Y, Breese GR, Hong JS et al (2007). Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 55: 453–462.
Russo SJ, Wilkinson MB, Mazei-Robison MS, Dietz DM, Maze I, Krishnan V et al (2009). Nuclear factor kappa B signaling regulates neuronal morphology and cocaine reward. J Neurosci 29: 3529–3537.
Salgado S, Kaplitt MG (2015). The nucleus accumbens: a comprehensive review. Stereotact Funct Neurosurg 93: 75–93.
Snow WM, Stoesz BM, Kelly DM, Albensi BC (2014). Roles for NF-kappaB and gene targets of NF-kappaB in synaptic plasticity, memory, and navigation. Mol Neurobiol 49: 757–770.
Truitt JM, Blednov YA, Benavidez JM, Black M, Ponomareva O, Law J et al (2016). Inhibition of IKKbeta reduces ethanol consumption in C57BL/6J Mice. eNeuro 3.
Wang X, Douglas SD, Commons KG, Pleasure DE, Lai J, Ho C et al (2004). A non-peptide substance P antagonist (CP-96,345) inhibits morphine-induced NF-kappa B promoter activation in human NT2-N neurons. J Neurosci Res 75: 544–553.
Zhang X, Cui Y, Jing J, Cui Y, Xin W, Liu X (2011). Involvement of p38/NF-kappaB signaling pathway in the nucleus accumbens in the rewarding effects of morphine in rats. Behav Brain Res 218: 184–189.
Zou J, Crews F (2006). CREB and NF-kappaB transcription factors regulate sensitivity to excitotoxic and oxidative stress induced neuronal cell death. Cell Mol Neurobiol 26: 385–405.
Zou J, Crews F (2010). Induction of innate immune gene expression cascades in brain slice cultures by ethanol: key role of NF-kappaB and proinflammatory cytokines. Alcohol Clin Exp Res 34: 777–789.
Zou JY, Crews FT (2005). TNF alpha potentiates glutamate neurotoxicity by inhibiting glutamate uptake in organotypic brain slice cultures: neuroprotection by NF kappa B inhibition. Brain Res 1034: 11–24.
Acknowledgements
We thank Dr Scott Russo (Mt. Sinai Medical School) for providing NF-κB-LacZ breeder mice and Dr Georgia Hodes (Virginia Polytechnic Institute and State University) for breeding/genotyping advice. We thank Dr Nikolay Filipov (University of Georgia) for sharing his expertise in mouse cannulation surgery. We also thank Dr Yavin Shaham (National Institute on Drug Abuse) for thoughtful comments on our data.
Author contributions
SEN and JRS analyzed the data, prepared figures, and wrote the manuscript. SEN, JRS, CK, HDF, SHC, BMS, MKS, and JEE contributed to data collection. CL and JFC preformed the TUNEL assay. SEN, JRS, and MH contributed to the experimental design and drafting the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Neuropsychopharmacology website
Supplementary information
Rights and permissions
About this article
Cite this article
Nennig, S., Fulenwider, H., Chimberoff, S. et al. Selective Lesioning of Nuclear Factor-κB Activated Cells in the Nucleus Accumbens Shell Attenuates Alcohol Place Preference. Neuropsychopharmacol. 43, 1032–1040 (2018). https://doi.org/10.1038/npp.2017.214
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/npp.2017.214
This article is cited by
-
A systematic review and meta-analysis on the transcriptomic signatures in alcohol use disorder
Molecular Psychiatry (2025)
-
Increased Alcohol Consumption in Mice Lacking Sodium Bicarbonate Transporter NBCn1
Scientific Reports (2020)
-
Developing neuroscience-based treatments for alcohol addiction: A matter of choice?
Translational Psychiatry (2019)