Key Points
-
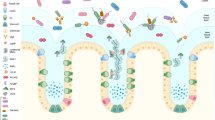
The gastrointestinal tract presents a continuous secreted and cell surface barrier to potential enteric pathogens. Specialized gastrointestinal epithelial cells secrete large amounts of mucin glycoproteins and antimicrobial molecules that, together, form the mucus barrier to infection. Although the lumen of the gastrointestinal tract contains large numbers of commensal microorganisms, the inner layers of mucus are sterile.
-
Secreted mucins are large, heavily O-glycosylated glycoproteins that are produced by goblet cells. During their biosynthesis, mucins homo-oligomerize into complex polymeric networks that, when secreted, give mucus its viscoelastic properties.
-
Antimicrobial molecules are produced throughout the gastrointestinal tract but particularly by the specialized Paneth cells in the small intestine. These molecules target different classes of pathogens and help keep the inner mucus layer sterile.
-
Cell surface mucins are heavily O-glycosylated transmembrane glycoproteins that are present on the apical surface of all gastrointestinal epithelial cells. These mucins limit binding of pathogens to epithelial cells by steric hindrance and by acting as releasable decoys for microbial adhesins.
-
Deficiencies in secreted or cell surface mucins in animal models lead to increased pathology during infection.
-
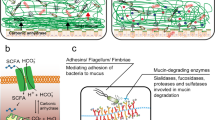
Pathogens have evolved multiple strategies to penetrate the mucosal barrier, including: disruption and penetration of the mucus, avoidance of the mucus barrier, and disruption of epithelial integrity and epithelial production of barrier components.
-
The production of components of the mucus barrier is influenced by the normal microbiota and by both innate and adaptive immune responses to pathogens. There are changes in the rate of mucus production and the content of mucus in response to infection; these factors are components of the mechanism of clearance of enteric pathogens and parasites.
Abstract
The extracellular secreted mucus and the cell surface glycocalyx prevent infection by the vast numbers of microorganisms that live in the healthy gut. Mucin glycoproteins are the major component of these barriers. In this Review, we describe the components of the secreted and cell surface mucosal barriers and the evidence that they form an effective barricade against potential pathogens. However, successful enteric pathogens have evolved strategies to circumvent these barriers. We discuss the interactions between enteric pathogens and mucins, and the mechanisms that these pathogens use to disrupt and avoid mucosal barriers. In addition, we describe dynamic alterations in the mucin barrier that are driven by host innate and adaptive immune responses to infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Johansson, M. E. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008). Demonstration of the spatial relationship between the intestinal epithelium, the inner and outer mucus layers and the microbiota.
Johansson, M. E., Holmen Larsson, J. M. & Hansson, G. C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc. Natl Acad. Sci. USA 25 Jun 2010 (doi:10.1073/pnas.1006451107).
Atuma, C., Strugala, V., Allen, A. & Holm, L. The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am. J. Physiol. Gastrointest. Liver Physiol. 280, G922–G929 (2001). In situ measurement of the mucus thickness throughout the rodent gastrointestinal tract and observations on the replacement of mucus following its physical removal.
Stappenbeck, T. S. Paneth cell development, differentiation, and function: new molecular cues. Gastroenterology 137, 30–33 (2009).
Heazlewood, C. K. et al. Aberrant mucin assembly in mice causes endoplasmic reticulum stress and spontaneous inflammation resembling ulcerative colitis. PLoS Med. 5, e54 (2008).
Park, S. W. et al. The protein disulfide isomerase AGR2 is essential for production of intestinal mucus. Proc. Natl Acad. Sci. USA 106, 6950–6955 (2009).
Zhao, F. et al. Disruption of Paneth and goblet cell homeostasis and increased endoplasmic reticulum stress in Agr2−/− mice. Dev. Biol. 338, 270–279 (2009).
Kaser, A. et al. XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134, 743–756 (2008).
Brandl, K. et al. Enhanced sensitivity to DSS colitis caused by a hypomorphic Mbtps1 mutation disrupting the ATF6-driven unfolded protein response. Proc. Natl Acad. Sci. USA 106, 3300–3305 (2009).
Cadwell, K. et al. A key role for autophagy and the autophagy gene Atg16l1 in mouse and human intestinal Paneth cells. Nature 456, 259–263 (2008).
Cadwell, K. et al. Virus-plus-susceptibility gene interaction determines Crohn's disease gene Atg16L1 phenotypes in intestine. Cell 141, 1135–1145 (2010).
McGuckin, M. A., Eri, R. D., Das, I., Lourie, R. & Florin, T. H. ER stress and the unfolded protein response in intestinal inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G820–G832 (2010).
Thornton, D. J., Rousseau, K. & McGuckin, M. A. Structure and function of the polymeric mucins in airways mucus. Annu. Rev. Physiol. 70, 459–486 (2008).
Davis, C. W. & Dickey, B. F. Regulated airway goblet cell mucin secretion. Annu. Rev. Physiol. 70, 487–512 (2008).
Rogers, D. F. Mucoactive agents for airway mucus hypersecretory diseases. Respir. Care 52, 1176–1193; discussion 1193–1177 (2007).
Larsson, J. M., Karlsson, H., Sjovall, H. & Hansson, G. C. A complex, but uniform O-glycosylation of the human MUC2 mucin from colonic biopsies analyzed by nanoLC/MSn. Glycobiology 19, 756–766 (2009).
Matsuo, K., Ota, H., Akamatsu, T., Sugiyama, A. & Katsuyama, T. Histochemistry of the surface mucous gel layer of the human colon. Gut 40, 782–789 (1997).
Henrissat, B., Surolia, A. & Stanley, P. in Essentials of Glycobiology. 2nd edn (eds Varki, A. et al.) (Cold Spring Harbor Laboratory Press, New York, 2009).
Ouellette, A. J. Paneth cells and innate mucosal immunity. Curr. Opin. Gastroenterol. 26, 547–553 (2010).
Porter, E. M., Bevins, C. L., Ghosh, D. & Ganz, T. The multifaceted Paneth cell. Cell. Mol. Life Sci. 59, 156–170 (2002).
White, S. H., Wimley, W. C. & Selsted, M. E. Structure, function, and membrane integration of defensins. Curr. Opin. Struct. Biol. 5, 521–527 (1995).
Hristova, K., Selsted, M. E. & White, S. H. Critical role of lipid composition in membrane permeabilization by rabbit neutrophil defensins. J. Biol. Chem. 272, 24224–24233 (1997).
Bruno, L. S. et al. Two-hybrid analysis of human salivary mucin MUC7 interactions. Biochim. Biophys. Acta 1746, 65–72 (2005).
Iontcheva, I., Oppenheim, F. G. & Troxler, R. F. Human salivary mucin MG1 selectively forms heterotypic complexes with amylase, proline rich proteins, statherin, and histatins. J. Dent. Res. 76, 734–743 (1997).
Kawakubo, M. et al. Natural antibiotic function of a human gastric mucin against Helicobacter pylori infection. Science 305, 1003–1006 (2004). Evidence that a mucin carbohydrate can directly inhibit the growth of a mucus-residing pathogen.
Gururaja, T. L. et al. Candidacidal activity prompted by N-terminus histatin-like domain of human salivary mucin (MUC7). Biochim. Biophys. Acta 1431, 107–119 (1999).
Strugnell, R. A. & Wijburg, O. L. The role of secretory antibodies in infection immunity. Nature Rev. Microbiol. 8, 656–667 (2010).
Phalipon, A. et al. Secretory component: a new role in secretory IgA-mediated immune exclusion in vivo. Immunity 17, 107–115 (2002).
Wilson, C. L. et al. Regulation of intestinal α-defensin activation by the metalloproteinase matrilysin in innate host defense. Science 286, 113–117 (1999).
Salzman, N. H., Ghosh, D., Huttner, K. M., Paterson, Y. & Bevins, C. L. Protection against enteric salmonellosis in transgenic mice expressing a human intestinal defensin. Nature 422, 522–526 (2003). In vivo evidence that human intestinal defensins can protect against enteric pathogens.
Salzman, N. H. et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nature Immunol. 11, 76–83 (2010).
Salzman, N. H. Microbiota–immune system interaction: an uneasy alliance. Curr. Opin. Microbiol. 14, 99–105 (2011).
Johansson, M. E., Thomsson, K. A. & Hansson, G. C. Proteomic analyses of the two mucus layers of the colon barrier reveal that their main component, the Muc2 mucin, is strongly bound to the Fcgbp protein. J. Proteome Res. 8, 3549–3557 (2009).
Lai, S. K., Wang, Y. Y., Cone, R., Wirtz, D. & Hanes, J. Altering mucus rheology to “solidify” human mucus at the nanoscale. PLoS ONE 4, e4294 (2009).
Lai, S. K. et al. Rapid transport of large polymeric nanoparticles in fresh undiluted human mucus. Proc. Natl Acad. Sci. USA 104, 1482–1487 (2007).
Tomasetto, C. et al. pS2/TFF1 interacts directly with the VWFC cysteine-rich domains of mucins. Gastroenterology 118, 70–80 (2000).
Newton, J. L., Allen, A., Westley, B. R. & May, F. E. B. The human trefoil peptide, TFF1, is present in different molecular forms that are intimately associated with mucus in normal stomach. Gut 46, 312–320 (2000).
Ruchaud-Sparagano, M. H., Westley, B. R. & May, F. E. The trefoil protein TFF1 is bound to MUC5AC in human gastric mucosa. Cell. Mol. Life Sci. 61, 1946–1954 (2004).
Kindon, H., Pothoulakis, C., Thim, L., Lynchdevaney, G. & Podolsky, D. K. Trefoil peptide protection of intestinal epithelial barrier function: cooperative interaction with mucin glycoprotein. Gastroenterology 109, 516–523 (1995).
Thim, L., Madsen, F. & Poulsen, S. S. Effect of trefoil factors on the viscoelastic properties of mucus gels. Eur. J. Clin. Invest. 32, 519–527 (2002).
Raynal, B. D., Hardingham, T. E., Sheehan, J. K. & Thornton, D. J. Calcium-dependent protein interactions in MUC5B provide reversible cross-links in salivary mucus. J. Biol. Chem. 278, 28703–28710 (2003).
Garcia, M. A., Yang, N. & Quinton, P. M. Normal mouse intestinal mucus release requires cystic fibrosis transmembrane regulator-dependent bicarbonate secretion. J. Clin. Invest. 119, 2613–2622 (2009).
Hattrup, C. L. & Gendler, S. J. Structure and function of the cell surface (tethered) mucins. Annu. Rev. Physiol. 70, 431–457 (2008).
Henry, S. et al. Structural and immunochemical identification of Le(a), Le(b), H type 1, and related glycolipids in small intestinal mucosa of a group O Le(a-b-) nonsecretor. Glycoconj. J. 14, 209–223 (1997).
Wreschner, D. H. et al. Generation of ligand-receptor alliances by “SEA” module-mediated cleavage of membrane-associated mucin proteins. Protein Sci. 11, 698–706 (2002).
Macao, B., Johansson, D. G., Hansson, G. C. & Hard, T. Autoproteolysis coupled to protein folding in the SEA domain of the membrane-bound MUC1 mucin. Nature Struct. Mol. Biol. 13, 71–76 (2006).
Linden, S. K. et al. MUC1 limits Helicobacter pylori infection both by steric hindrance and by acting as a releasable decoy. PLoS Pathog. 5, e1000617 (2009). Elucidation of the mechanisms by which cell surface mucins limit pathogen adhesion to the cell surface.
Thathiah, A., Blobel, C. P. & Carson, D. D. Tumor necrosis factor-α converting enzyme/ADAM 17 mediates MUC1 shedding. J. Biol. Chem. 278, 3386–3394 (2003).
Thathiah, A. & Carson, D. D. MT1-MMP mediates MUC1 shedding independently of TACE/ADAM17. Biochem. J. 382, 363–373 (2004).
McGuckin, M. A. et al. Muc1 mucin limits both Helicobacter pylori colonization of the murine gastric mucosa and associated gastritis. Gastroenterology 133, 1210–1218 (2007). First demonstration that the deficiency of a cell surface mucin predisposes the host to more severe chronic inflammation during a chronic infection.
Guang, W. et al. Muc1 cell surface mucin attenuates epithelial inflammation in response to a common mucosal pathogen. J. Biol. Chem. 285, 20547–20557 (2010).
McAuley, J. L. et al. MUC1 cell surface mucin is a critical element of the mucosal barrier to infection. J. Clin. Invest. 117, 2313–2324 (2007). First demonstration that a cell surface mucin limits both the translocation of enteric pathogens and damage to the gut.
Schreiber, S. et al. The spatial orientation of Helicobacter pylori in the gastric mucus. Proc. Natl Acad. Sci. USA 101, 5024–5029 (2004).
Mahdavi, J. et al. Helicobacter pylori SabA adhesin in persistent infection and chronic inflammation. Science 297, 573–578 (2002).
Aspholm-Hurtig, M. et al. Functional adaptation of BabA, the H. pylori ABO blood group antigen binding adhesin. Science 305, 519–522 (2004).
Carvalho, F. et al. MUC1 gene polymorphism and gastric cancer–an epidemiological study. Glycoconj. J. 14, 107–111 (1997).
Vinall, L. E. et al. Altered expression and allelic association of the hypervariable membrane mucin MUC1 in Helicobacter pylori gastritis. Gastroenterology 123, 41–49 (2002).
Silva, F. et al. MUC1 polymorphism confers increased risk for intestinal metaplasia in a Colombian population with chronic gastritis. Eur. J. Hum. Genet. 11, 380–384 (2003).
Magalhaes, A. et al. Fut2-null mice display an altered glycosylation profile and impaired BabA-mediated Helicobacter pylori adhesion to gastric mucosa. Glycobiology 19, 1525–1536 (2009).
Linden, S. et al. Role of ABO secretor status in mucosal innate immunity and H. pylori infection. PLoS Pathog. 4, e2 (2008).
Cooke, C. L. et al. Modification of gastric mucin oligosaccharide expression in rhesus macaques after infection with Helicobacter pylori. Gastroenterology 137, 1061–1071 (2009).
Lindesmith, L. et al. Human susceptibility and resistance to Norwalk virus infection. Nature Med. 9, 548–553 (2003).
Ueno, K. et al. MUC1 mucin is a negative regulator of Toll-like receptor signaling. Am. J. Respir. Cell. Mol. Biol. 38, 263–268 (2007).
Ahmad, R. et al. MUC1 oncoprotein activates the IκB kinase β complex and constitutive NF-κB signalling. Nature Cell Biol. 9, 1419–1427 (2007). Evidence that signalling by cell surface mucins modulates inflammatory signalling in epithelial cells.
Franke, A. et al. Genome-wide meta-analysis increases to 71 the number of confirmed Crohn's disease susceptibility loci. Nature Genet. 42, 1118–1125 (2010).
Bergstrom, K. S. et al. Muc2 protects against lethal infectious colitis by disassociating pathogenic and commensal bacteria from the colonic mucosa. PLoS Pathog. 6, e1000902 (2010). Finding that there is more severe pathology during bacterial infection in the intestine in the absence of the secreted mucin MUC2.
Hasnain, S. Z. et al. Mucin gene deficiency in mice impairs host resistance to an enteric parasitic infection. Gastroenterology 138, 1763–1771 (2010). First evidence that secreted mucins are important components of the T H 2-type immune response that mediates expulsion of nematode parasites.
Van der Sluis, M. et al. Muc2-deficient mice spontaneously develop Colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 131, 117–129 (2006). Finding that a deficiency in intestinal secreted mucins leads to spontaneous intestinal inflammation.
Hugdahl, M. B., Beery, J. T. & Doyle, M. P. Chemotactic behavior of Campylobacter jejuni. Infect. Immun. 56, 1560–1566 (1988).
Tu, Q. V., McGuckin, M. A. & Mendz, G. L. Campylobacter jejuni response to human mucin MUC2: modulation of colonization and pathogenicity determinants. J. Med. Microbiol. 57, 795–802 (2008).
Ottemann, K. M. & Lowenthal, A. C. Helicobacter pylori uses motility for initial colonization and to attain robust infection. Infect. Immun. 70, 1984–1990 (2002).
Ramos, H. C., Rumbo, M. & Sirard, J. C. Bacterial flagellins: mediators of pathogenicity and host immune responses in mucosa. Trends Microbiol. 12, 509–517 (2004).
Celli, J. P. et al. Helicobacter pylori moves through mucus by reducing mucin viscoelasticity. Proc. Natl Acad. Sci. USA 106, 14321–14326 (2009). Demonstration that H. pylori reduces mucus visocosity in its microenvironment to promote bacterial motility.
Lidell, M. E., Moncada, D. M., Chadee, K. & Hansson, G. C. Entamoeba histolytica cysteine proteases cleave the MUC2 mucin in its C-terminal domain and dissolve the protective colonic mucus gel. Proc. Natl Acad. Sci. USA 103, 9298–9303 (2006). Finding that there is specific enzymatic destruction of MUC2 polymers in mucus by an enteric amoebic parasite.
Silva, A. J., Pham, K. & Benitez, J. A. Haemagglutinin/protease expression and mucin gel penetration in El Tor biotype Vibrio cholerae. Microbiology 149, 1883–1891 (2003).
Deplancke, B. et al. Selective growth of mucolytic bacteria including Clostridium perfringens in a neonatal piglet model of total parenteral nutrition. Am. J. Clin. Nutr. 76, 1117–1125 (2002).
Sonnenburg, J. L., Angenent, L. T. & Gordon, J. I. Getting a grip on things: how do communities of bacterial symbionts become established in our intestine? Nature Immunol. 5, 569–573 (2004).
Sonnenburg, J. L. et al. Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science 307, 1955–1959 (2005). Exploration of the altered utilization of mucin carbohydrates by an individual commensal bacterium under different host dietary conditions.
Png, C. W. et al. Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am. J. Gastroenterol. 105, 2420–2428 (2010).
Law, G. K., Bertolo, R. F., Adjiri-Awere, A., Pencharz, P. B. & Ball, R. O. Adequate oral threonine is critical for mucin production and gut function in neonatal piglets. Am. J. Physiol. Gastrointest. Liver Physiol. 292, G1293–G1301 (2007).
Siebers, A. & Finlay, B. B. M cells and the pathogenesis of mucosal and systemic infections. Trends Microbiol. 4, 22–29 (1996).
Neutra, M. R., Mantis, N. J., Frey, A. & Giannasca, P. J. The composition and function of M cell apical membranes: implications for microbial pathogenesis. Semin. Immunol. 11, 171–181 (1999).
Macpherson, A. J. & Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665 (2004).
Macpherson, A. J., McCoy, K. D., Johansen, F. E. & Brandtzaeg, P. The immune geography of IgA induction and function. Mucosal Immunol. 1, 11–22 (2008).
Endt, K. et al. The microbiota mediates pathogen clearance from the gut lumen after non-typhoidal Salmonella diarrhea. PLoS Pathog. 6, e1001097 (2010).
Lelouard, H., Reggio, H., Mangeat, P., Neutra, M. & Montcourrier, P. Mucin-related epitopes distinguish M cells and enterocytes in rabbit appendix and Peyer's patches. Infect. Immun. 67, 357–367 (1999).
Lelouard, H. et al. Glycocalyx on rabbit intestinal M cells displays carbohydrate epitopes from Muc2. Infect. Immun. 69, 1061–1071 (2001).
Vazquez-Torres, A. & Fang, F. C. Cellular routes of invasion by enteropathogens. Curr. Opin. Microbiol. 3, 54–59 (2000).
Jones, B., Pascopella, L. & Falkow, S. Entry of microbes into the host: using M cells to break the mucosal barrier. Curr. Opin. Immunol. 7, 474–478 (1995).
Walk, S. T., Blum, A. M., Ewing, S. A., Weinstock, J. V. & Young, V. B. Alteration of the murine gut microbiota during infection with the parasitic helminth Heligmosomoides polygyrus. Inflamm. Bowel Dis. 16, 1841–1849 (2010).
Frank, D. N. et al. Disease phenotype and genotype are associated with shifts in intestinal-associated microbiota in inflammatory bowel diseases. Inflamm. Bowel Dis. 17, 189–194 (2010).
Willing, B. P. et al. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology 139, 1844–1854 (2010).
Hoffmann, C. et al. Community-wide response of the gut microbiota to enteropathogenic Citrobacter rodentium infection revealed by deep sequencing. Infect. Immun. 77, 4668–4678 (2009).
Dalby, A. B., Frank, D. N., St. Amand, A. L., Bendele, A. M. & Pace, N. R. Culture-independent analysis of indomethacin-induced alterations in the rat gastrointestinal microbiota. Appl. Environ. Microbiol. 72, 6707–6715 (2006).
Guttman, J. A. & Finlay, B. B. Tight junctions as targets of infectious agents. Biochim. Biophys. Acta 1788, 832–841 (2009).
Sakaguchi, T., Kohler, H., Gu, X., McCormick, B. A. & Reinecker, H. C. Shigella flexneri regulates tight junction-associated proteins in human intestinal epithelial cells. Cell. Microbiol. 4, 367–381 (2002).
Mengaud, J., Ohayon, H., Gounon, P., Mege, R. M. & Cossart, P. E-cadherin is the receptor for internalin, a surface protein required for entry of L. monocytogenes into epithelial cells. Cell 84, 923–932 (1996).
Goosney, D. L., Gruenheid, S. & Finlay, B. B. Gut feelings: enteropathogenic E. coli (EPEC) interactions with the host. Annu. Rev. Cell Dev. Biol. 16, 173–189 (2000).
Katz, J., Sambandam, V., Wu, J. H., Michalek, S. M. & Balkovetz, D. F. Characterization of Porphyromonas gingivalis-induced degradation of epithelial cell junctional complexes. Infect. Immun. 68, 1441–1449 (2000).
Amieva, M. R. et al. Disruption of the epithelial apical-junctional complex by Helicobacter pylori CagA. Science 300, 1430–1434 (2003).
Nava, P., Lopez, S., Arias, C. F., Islas, S. & Gonzalez-Mariscal, L. The rotavirus surface protein VP8 modulates the gate and fence function of tight junctions in epithelial cells. J. Cell Sci. 117, 5509–5519 (2004).
Meyerholz, D. K. et al. Early epithelial invasion by Salmonella enterica serovar Typhimurium DT104 in the swine ileum. Vet. Pathol. 39, 712–720 (2002).
Ren, J. et al. Human MUC1 carcinoma-associated protein confers resistance to genotoxic anticancer agents. Cancer Cell 5, 163–175 (2004). First demonstration that cell surface mucins modulate apoptosis in epithelial cells.
Wei, X., Xu, H. & Kufe, D. Human MUC1 oncoprotein regulates p53-responsive gene transcription in the genotoxic stress response. Cancer Cell 7, 167–178 (2005).
Kim, K. C., Lee, B. C., Pou, S. & Ciccolella, D. Effects of activation of polymorphonuclear leukocytes on airway goblet cell mucin release in a co-culture system. Inflamm Res. 52, 258–262 (2003).
Fischer, B. M., Krunkosky, T. M., Wright, D. T., Dolanokeefe, M. & Adler, K. B. Tumor necrosis factor-alpha (TNF-α) stimulates mucin secretion and gene expression in airway epithelium in vitro. Chest 107, S133–S135 (1995).
Hollande, E., Fanjul, M., Claret, S., Forguelafitte, M. E. & Bara, J. Effects of VIP on the regulation of mucin secretion in cultured human pancreatic cancer cells (Capan-1). In Vitro Cell. Dev. Biol. 31, 227–233 (1995).
Smirnova, M. G., Guo, L., Birchall, J. P. & Pearson, J. P. LPS up-regulates mucin and cytokine mRNA expression and stimulates mucin and cytokine secretion in goblet cells. Cell. Immunol. 221, 42–49 (2003).
Kishioka, C., Okamoto, K., Kim, J. & Rubin, B. K. Regulation of secretion from mucous and serous cells in the excised ferret trachea. Respir. Physiol. 126, 163–171 (2001).
Smirnova, M. G., Birchall, J. P. & Pearson, J. P. TNF-alpha in the regulation of MUC5AC secretion: some aspects of cytokine-induced mucin hypersecretion on the in vitro model. Cytokine 12, 1732–1736 (2000).
Enss, M. L. et al. Proinflammatory cytokines trigger MUC gene expression and mucin release in the intestinal cancer cell line LS180. Inflamm Res. 49, 162–169 (2000).
Klinkspoor, J. H. et al. Mucin secretion by the human colon cell line LS174T is regulated by bile salts. Glycobiology 9, 13–19 (1999).
Voynow, J. A. et al. Neutrophil elastase increases MUC5AC mRNA and protein expression in respiratory epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 20, 835–843 (1999).
Jarry, A., Vallette, G., Branka, J. E. & Laboisse, C. Direct secretory effect of interleukin-1 via type I receptors in human colonic mucous epithelial cells (HT29-C1.16E). Gut 38, 240–242 (1996).
Kim, K. C., Park, H. R., Shin, C. Y., Akiyama, T. & Ko, K. H. Nucleotide-induced mucin release from primary hamster tracheal surface epithelial cells involves the P2u purinoceptor. Eur. Resp. J. 9, 542–548 (1996).
Gottke, M. & Chadee, K. Exogenous nitric oxide stimulates mucin secretion from LS174T colonic adenocarcinoma cells. Inflamm. Res. 45, 209–212 (1996).
Tam, P. Y. & Verdugo, P. Control of mucus hydration as a Donnan equilibrium process. Nature 292, 340–342 (1981).
Verdugo, P. Mucin exocytosis. Am. Rev. Respir. Dis. 144, S33–S37 (1991).
Hill, R. R., Cowley, H. M. & Andremont, A. Influence of colonizing micro-flora on the mucin histochemistry of the neonatal mouse colon. Histochem. J. 22, 102–105 (1990).
Enss, M. L. et al. Response of germfree rat colonic mucous cells to peroral endotoxin application. Eur. J. Cell Biol. 71, 99–104 (1996).
Kandori, H., Hirayama, K., Takeda, M. & Doi, K. Histochemical, lectin-histochemical and morphometrical characteristics of intestinal goblet cells of germfree and conventional mice. Exp. Anim. 45, 155–160 (1996).
Fukushima, K. et al. Colonization of microflora in mice: mucosal defense against luminal bacteria. J. Gastroenterol. 34, 54–60 (1999).
Cash, H. L., Whitham, C. V., Behrendt, C. L. & Hooper, L. V. Symbiotic bacteria direct expression of an intestinal bactericidal lectin. Science 313, 1126–1130 (2006). Finding that the production of specific antimicrobial molecules is regulated by the commensal microbiota.
Hooper, L. V., Stappenbeck, T. S., Hong, C. V. & Gordon, J. I. Angiogenins: a new class of microbicidal proteins involved in innate immunity. Nature Immunol. 4, 269–273 (2003).
Sharma, R. & Schumacher, U. Carbohydrate expression in the intestinal mucosa. Adv. Anat. Embryol. Cell Biol. 160, III–IX, 1–91 (2001).
Freitas, M., Axelsson, L. G., Cayuela, C., Midtvedt, T. & Trugnan, G. Microbial-host interactions specifically control the glycosylation pattern in intestinal mouse mucosa. Histochem. Cell Biol. 118, 149–161 (2002).
George, S. et al. Lectin binding profile of the small intestine of five-week-old pigs in response to the use of chlortetracycline as a growth promotant and under gnotobiotic conditions. J. Anim. Sci. 85, 1640–1650 (2007).
Whittaker, L. et al. Interleukin-13 mediates a fundamental pathway for airway epithelial mucus induced by CD4 T cells and interleukin-9. Am. J. Respir. Cell. Mol. Biol. 27, 593–602 (2002).
Artis, D. et al. Tumor necrosis factor α is a critical component of interleukin 13-mediated protective T helper cell type 2 responses during helminth infection. J. Exp. Med. 190, 953–962 (1999).
Park, K. S. et al. SPDEF regulates goblet cell hyperplasia in the airway epithelium. J. Clin. Invest. 117, 978–988 (2007). First demonstration of the importance of the T H 2 cytokine mediated production of the SPDEF transcription factor on goblet cell differentiation.
Gregorieff, A. et al. The Ets-domain transcription factor Spdef promotes maturation of goblet and paneth cells in the intestinal epithelium. Gastroenterology 137, 1333–1345.e3 (2009).
Noah, T. K., Kazanjian, A., Whitsett, J. & Shroyer, N. F. SAM pointed domain ETS factor (SPDEF) regulates terminal differentiation and maturation of intestinal goblet cells. Exp. Cell Res. 316, 452–465 (2009).
Chen, G. et al. SPDEF is required for mouse pulmonary goblet cell differentiation and regulates a network of genes associated with mucus production. J. Clin. Invest. 119, 2914–2924 (2009).
Sugimoto, K. et al. IL-22 ameliorates intestinal inflammation in a mouse model of ulcerative colitis. J. Clin. Invest. 118, 534–544 (2008).
Chen, Y. et al. Stimulation of airway mucin gene expression by interleukin (IL)-17 through IL-6 paracrine/autocrine loop. J. Biol. Chem. 278, 17036–17043 (2003).
Andrianifahanana, M. et al. IFN-γ-induced expression of MUC4 in pancreatic cancer cells is mediated by STAT-1 upregulation: a novel mechanism for IFN-γ response. Oncogene 26, 7251–7261 (2007).
Ahn, D. H. et al. TNF-alpha activates MUC2 transcription via NF-kappaB but inhibits via JNK activation. Cell Physiol. Biochem. 15, 29–40 (2005).
Dabbagh, K. et al. IL-4 induces mucin gene expression and goblet cell metaplasia in vitro and in vivo. J.Immunol. 162, 6233–6237 (1999).
Longphre, M. et al. Allergen-induced IL-9 directly stimulates mucin transcription in respiratory epithelial cells. J. Clin. Invest. 104, 1375–1382 (1999).
Kim, Y. D. et al. Regulation of IL-1β-mediated MUC2 gene in NCI-H292 human airway epithelial cells. Biochem. Biophys. Res. Commun. 274, 112–116 (2000).
Shim, J. J. et al. IL-13 induces mucin production by stimulating epidermal growth factor receptors and by activating neutrophils. Am. J. Physiol. Lung Cell. Mol. Physiol. 280, L134–L140 (2001).
Smirnova, M. G., Kiselev, S. L., Birchall, J. P. & Pearson, J. P. Up-regulation of mucin secretion in HT29-MTX cells by the pro-inflammatory cytokines tumor necrosis factor-α and interleukin-6. Eur. Cytokine Netw. 12, 119–125 (2001).
Kim, Y. D. et al. Interleukin-1β induces MUC2 gene expression and mucin secretion via activation of PKC-MEK/ERK, and PI3K in human airway epithelial cells. J. Korean Med. Sci. 17, 765–771 (2002).
Song, J. S. et al. Nitric oxide induces MUC5AC mucin in respiratory epithelial cells through PKC and ERK dependent pathways. Respir. Res. 8, 28 (2007).
Wu, Y. M., Nowack, D. D., Omenn, G. S. & Haab, B. B. Mucin glycosylation is altered by pro-inflammatory signaling in pancreatic-cancer cells. J. Proteome Res. 8, 1876–1886 (2009).
Kanoh, A. et al. Interleukin-4 induces specific pp-GalNAc-T expression and alterations in mucin O-glycosylation in colonic epithelial cells. Biochim. Biophys. Acta 1780, 577–584 (2008).
Groux-Degroote, S. et al. IL-6 and IL-8 increase the expression of glycosyltransferases and sulfotransferases involved in the biosynthesis of sialylated and/or sulfated LewisX epitopes in the human bronchial mucosa. Biochem. J. 410, 213–223 (2008).
Beum, P. V., Basma, H., Bastola, D. R. & Cheng, P. W. Mucin biosynthesis: upregulation of core 2 β1,6 N-acetylglucosaminyltransferase by retinoic acid and Th2 cytokines in a human airway epithelial cell line. Am. J. Physiol. Lung Cell. Mol. Physiol. 288, L116–L124 (2005).
Yamauchi, J. et al. Altered expression of goblet cell- and mucin glycosylation-related genes in the intestinal epithelium during infection with the nematode Nippostrongylus brasiliensis in rat. APMIS 114, 270–278 (2006).
Takeda, K. et al. Direct effects of IL-4/IL-13 and the nematode Nippostrongylus brasiliensis on intestinal epithelial cells in vitro. Parasite Immunol. 32, 420–429 (2010).
Gewirtz, A. T., Navas, T. A., Lyons, S., Godowski, P. J. & Madara, J. L. Cutting edge: bacterial flagellin activates basolaterally expressed TLR5 to induce epithelial proinflammatory gene expression. J. Immunol. 167, 1882–1885 (2001).
Yanagihara, K., Seki, M. & Cheng, P. W. Lipopolysaccharide induces mucus cell metaplasia in mouse lung. Am. J. Respir. Cell. Mol. Biol. 24, 66–73 (2001).
Lemjabbar, H. & Basbaum, C. Platelet-activating factor receptor and ADAM10 mediate responses to Staphylococcus aureus in epithelial cells. Nature Med. 8, 41–46 (2002).
Dohrman, A. et al. Mucin gene (MUC2 and MUC5AC) upregulation by Gram-positive and Gram-negative bacteria. Biochim. Biophys. Acta 1406, 251–259 (1998).
Caballero-Franco, C., Keller, K., De Simone, C. & Chadee, K. The VSL#3 probiotic formula induces mucin gene expression and secretion in colonic epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 292, G315–G322 (2007).
Lievin- Le Moal, V., Servin, A. L. & Coconnier-Polter, M. H. The increase in mucin exocytosis and the upregulation of MUC genes encoding for membrane-bound mucins induced by the thiol-activated exotoxin listeriolysin O is a host cell defence response that inhibits the cell-entry of Listeria monocytogenes. Cell. Microbiol. 7, 1035–1048 (2005).
Slomiany, B. L. & Slomiany, A. Cytosolic phospholipase A2 activation in Helicobacter pylori lipopolysaccharide-induced interference with gastric mucin synthesis. IUBMB Life 58, 217–223 (2006).
Linden, S. K., Florin, T. H. & McGuckin, M. A. Mucin dynamics in intestinal bacterial infection. PLoS ONE 3, e3952 (2008). Description of progressive changes in cell surface and secreted intestinal mucins during infection with an attaching and effacing pathogen.
Boshuizen, J. A. et al. Homeostasis and function of goblet cells during rotavirus infection in mice. Virology 337, 210–221 (2005).
Hasnain, S. Z., Thornton, D. J. & Grencis, R. K. Changes in the mucosal barrier during acute and chronic Trichuris muris infection. Parasite Immunol. 33, 45–55 (2011).
Guk, S. M. et al. CD4+ T-cell-dependent goblet cell proliferation and expulsion of Gymnophalloides seoi from the intestine of C57BL/6 mice. J. Parasitol. 95, 581–590 (2009).
Hoang, V. C., Williams, M. A. & Simpson, H. V. Effects of weaning and infection with Teladorsagia circumcincta on mucin carbohydrate profiles of early weaned lambs. Vet. Parasitol. 171, 354–360 (2010).
Li, R. W. et al. Mucin biosynthesis in the bovine goblet cell induced by Cooperia oncophora infection. Vet. Parasitol. 165, 281–289 (2009).
Fontaine, O. et al. Setting research priorities to reduce global mortality from childhood diarrhoea by 2015. PLoS Med. 6, e41 (2009).
Linden, S. K., Driessen, K. M. & McGuckin, M. A. Improved in vitro model systems for gastrointestinal infection by choice of cell line, pH, microaerobic conditions and optimization of culture conditions. Helicobacter 12, 341–353 (2007).
Wickstrom, C., Herzberg, M. C., Beighton, D. & Svensater, G. Proteolytic degradation of human salivary MUC5B by dental biofilms. Microbiology 155, 2866–2872 (2009).
Tsai, H. H., Dwarakanath, A. D., Hart, C. A., Milton, J. D. & Rhodes, J. M. Increased faecal mucin sulphatase activity in ulcerative colitis: a potential target for treatment. Gut 36, 570–576 (1995).
Corfield, A. P. et al. The roles of enteric bacterial sialidase, sialate O-acetyl esterase and glycosulfatase in the degradation of human colonic mucin. Glycoconj. J. 10, 72–81 (1993).
Roberton, A. M. et al. A novel bacterial mucinase, glycosulfatase, is associated with bacterial vaginosis. J. Clin. Microbiol. 43, 5504–5508 (2005).
Berry, M., Harris, A., Lumb, R. & Powell, K. Commensal ocular bacteria degrade mucins. Br. J. Ophthalmol. 86, 1412–1416 (2002).
Martens, E. C., Roth, R., Heuser, J. E. & Gordon, J. I. Coordinate regulation of glycan degradation and polysaccharide capsule biosynthesis by a prominent human gut symbiont. J. Biol. Chem. 284, 18445–18457 (2009).
Falk, P., Hoskins, L. C. & Larson, G. Bacteria of the human intestinal microbiota produce glycosidases specific for lacto-series glycosphingolipids. J. Biochem. 108, 466–474 (1990); erratum 109, 798 (1991).
Vanderhoeven, J. S. & Camp, P. J. M. The use of lectins in monitoring degradation of oligosaccharide chains in mucin by oral streptococci. Caries Res. 28, 257–261 (1994).
Jansen, H. J., Hart, C. A., Rhodes, J. M., Saunders, J. R. & Smalley, J. W. A novel mucin-sulphatase activity found in Burkholderia cepacia and Pseudomonas aeruginosa. J. Med. Microbiol. 48, 551–557 (1999).
Henderson, I. R., Czeczulin, J., Eslava, C., Noriega, F. & Nataro, J. P. Characterization of Pic, a secreted protease of Shigella flexneri and enteroaggregative Escherichia coli. Infect. Immun. 67, 5587–5596 (1999).
Slomiany, B. L. et al. Campylobacter pyloridis degrades mucin and undermines gastric mucosal integrity. Biochem. Biophys. Res. Commun. 144, 307–314 (1987).
Terra, V. S., Homer, K. A., Rao, S. G., Andrew, P. W. & Yesilkaya, H. Characterization of novel β-galactosidase activity that contributes to glycoprotein degradation and virulence in Streptococcus pneumoniae. Infect. Immun. 78, 348–357 (2010).
Yesilkaya, H., Manco, S., Kadioglu, A., Terra, V. S. & Andrew, P. W. The ability to utilize mucin affects the regulation of virulence gene expression in Streptococcus pneumoniae. FEMS Microbiol. Lett. 278, 231–235 (2008).
Mantle, M. & Rombough, C. Growth in and breakdown of purified rabbit small intestinal mucin by Yersinia enterocolitica. Infect. Immun. 61, 4131–4138 (1993).
Ashida, H. et al. Characterization of two different endo-α-N-acetylgalactosaminidases from probiotic and pathogenic enterobacteria, Bifidobacterium longum and Clostridium perfringens. Glycobiology 18, 727–734 (2008).
Szabady, R. L., Yanta, J. H., Halladin, D. K., Schofield, M. J. & Welch, R. A. TagA is a secreted protease of Vibrio cholerae that specifically cleaves mucin glycoproteins. Microbiology 157, 516–525 (2011).
Grys, T. E., Walters, L. L. & Welch, R. A. Characterization of the StcE protease activity of Escherichia coli O157:H7. J. Bacteriol. 188, 4646–4653 (2006).
Cervantes-Sandoval, I., Serrano-Luna Jde, J., Garcia-Latorre, E., Tsutsumi, V. & Shibayama, M. Mucins in the host defence against Naegleria fowleri and mucinolytic activity as a possible means of evasion. Microbiology 154, 3895–3904 (2008).
Acknowledgements
M.A.M. and T.H.F. are supported by an Australian National Health and Medical Research Council (NHMRC) Senior Research Fellowship and an NHMRC Senior Practitioner Fellowship, respectively. This work is supported by NHMRC project grant 543704.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Commensal microorganism
-
A microorganism living in a mutually advantageous relationship with a mammalian host (for example, in the lumen of the gastrointestinal tract).
- Mucus
-
A viscous, highly hydrated fluid layer that overlies the mucosal surface and is rich in secreted mucin glycoproteins and other molecules involved in host defence against infection.
- Mucin
-
A cell surface or secreted glycoprotein that is characterized by the presence of a large filamentous domain which is rich in complex O-linked oligosaccharides.
- Antimicrobial molecule
-
One of a group of molecules that are produced by Paneth cells and other epithelial cells and that have a wide variety of structures. Many are small, cationic and amphipathic peptides or lectins, and are microbicidal because they interact with and disrupt microbial cell membranes. Many of these molecules can also be produced by granulocytes.
- Secreted mucin
-
A secreted glycoprotein with a central domain containing a dense array of O-linked oligosaccharides, and amino- and carboxy-terminal cysteine-rich domains that oligomerize the mucins into a large macromolecular complex, giving mucus its viscous properties.
- Goblet cell
-
A specialized secretory cell that manufactures secreted mucin glycoproteins and other components of the mucus.
- Paneth cell
-
A specialized intestinal secretory cell that manufactures antimicrobial molecules for secretion into the mucus.
- Theca
-
An intracellular collection of mucin granules that are packaged for secretion; the theca is responsible for the classical goblet cell morphology that is seen following conventional cell fixation.
- Lewis blood group antigen
-
A carbohydrate antigen that is found on the cell surface of red blood cells and also on epithelial glycolipids and glycoproteins, including mucins. Polymorphisms in glycosyltransferases determine the presence or absence of Lewis carbohydrates and also the precise isoforms of carbohydrate that are expressed on blood cells and the mucosae.
- Lamina propria
-
The tissue that underlies the mucosal epithelium and contains immune cells, connective tissue, and blood and lymphatic vessels.
- Rheological
-
Pertaining to the study of the properties of flow of matter.
- Cell surface mucin
-
A transmembrane glycoprotein with a dense array of O-linked oligosaccharides in its extracellular domain and with complex cytoplasmic domains that are involved in intracellular signalling. The extracellular domain of these mucins can be shed from the cell surface.
- Glycocalyx
-
An extracellular zone on the apical surface of mucosal epithelial cells that is composed of carbohydrate-rich transmembrane and secreted molecules, including cell surface mucin glycoproteins.
- Peyer's patch
-
An organized collection of tertiary lymphoid tissue directly underlying the intestinal epithelium; mainly found in the small intestine.
- Transcytose
-
To pass something (such as a microorganism) through a cell.
Rights and permissions
About this article
Cite this article
McGuckin, M., Lindén, S., Sutton, P. et al. Mucin dynamics and enteric pathogens. Nat Rev Microbiol 9, 265–278 (2011). https://doi.org/10.1038/nrmicro2538
Published:
Issue date:
DOI: https://doi.org/10.1038/nrmicro2538
This article is cited by
-
Curcumin attenuates aflatoxin B1-induced ileum injury in ducks by inhibiting NLRP3 inflammasome and regulating TLR4/NF-κB signaling pathway
Mycotoxin Research (2024)
-
State of the art in research on the gut-liver and gut-brain axis in poultry
Journal of Animal Science and Biotechnology (2023)
-
miR-125a-5p regulates the sialyltransferase ST3GAL1 in murine model of human intestinal campylobacteriosis
Gut Pathogens (2023)
-
The protective role of conjunctival goblet cell mucin sialylation
Nature Communications (2023)
-
Basal stem cell progeny establish their apical surface in a junctional niche during turnover of an adult barrier epithelium
Nature Cell Biology (2023)