Abstract
Introduction:
The precise temperature for optimal neuroprotection in infants with neonatal encephalopathy is unclear. Our aim was to assess systemic effects of whole-body cooling to 35 °C, 33.5 °C, and 30 °C in a piglet model of perinatal asphyxia.
Methods:
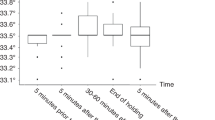
Twenty–eight anesthetized male piglets aged <24 h underwent hypoxia–ischemia (HI) and were randomized to normothermia or cooling to rectal temperature (Trec) 35 °C, 33.5 °C, or 30 °C during 2–26 h after insult (n = 7 in each group). HR, MABP, and Trec were recorded continuously.
Results:
Five animals cooled to 30 °C had fatal cardiac arrests. During 30 °C cooling, heart rate (HR) was lower vs. normothermia (P < 0.001). Although mean arterial blood pressure (MABP) did not vary between groups, more fluid boluses were needed at 30 °C than at normothermia (P < 0.02); dopamine use was higher at 30 °C than at normothermia or 35 °C (P = 0.005 and P = 0.02, respectively). Base deficit was increased at 30 °C at 12, 24, and 36 h vs. all other groups (P < 0.05), pH was acidotic at 36 h vs. normothermia (P = 0.04), and blood glucose was higher for the 30 °C group at 12 h vs. the normothermia and 35 °C groups (P < 0.05). Potassium was lower at 12 h in the 30 °C group vs. the 33.5 °C and 35 °C groups. There was no difference in cortisol level between groups.
Discussion:
Cooling to 30 °C led to metabolic derangement and more cardiac arrests and deaths than cooling to 33.5 °C or 35 °C. Inadvertent overcooling should be avoided.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Edwards AD, Brocklehurst P, Gunn AJ, et al. Neurological outcomes at 18 months of age after moderate hypothermia for perinatal hypoxic ischaemic encephalopathy: synthesis and meta-analysis of trial data. BMJ 2010;340:c363.
Colbourne F, Corbett D . Delayed postischemic hypothermia: a six month survival study using behavioral and histological assessments of neuroprotection. J Neurosci 1995;15:7250–60.
Azzopardi DV, Strohm B, Edwards AD, et al.; TOBY Study Group. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N Engl J Med 2009;361:1349–58.
Simbruner G, Mittal RA, Rohlmann F, Muche R; neo.nEURO.network Trial Participants. Systemic hypothermia after neonatal encephalopathy: outcomes of neo.nEURO.network RCT. Pediatrics 2010;126:e771–8.
Shankaran S, Laptook AR, Ehrenkranz RA, et al.; National Institute of Child Health and Human Development Neonatal Research Network. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med 2005;353:1574–84.
Gluckman PD, Wyatt JS, Azzopardi D, et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet 2005;365:663–70.
Zhou WH, Cheng GQ, Shao XM, et al.; China Study Group. Selective head cooling with mild systemic hypothermia after neonatal hypoxic-ischemic encephalopathy: a multicenter randomized controlled trial in China. J Pediatr 2010;157:367–72, 372.e1–3.
Gunn AJ, Gunn TR . The ‘pharmacology’ of neuronal rescue with cerebral hypothermia. Early Hum Dev 1998;53:19–35.
Iwata O, Thornton JS, Sellwood MW, et al. Depth of delayed cooling alters neuroprotection pattern after hypoxia-ischemia. Ann Neurol 2005;58:75–87.
Steen PA, Soule EH, Michenfelder JD . Deterimental effect of prolonged hypothermia in cats and monkeys with and without regional cerebral ischemia. Stroke 1979;10:522–9.
Schubert A . Side effects of mild hypothermia. J Neurosurg Anesthesiol 1995;7:139–47.
Shah PS . Hypothermia: a systematic review and meta-analysis of clinical trials. Semin Fetal Neonatal Med 2010;15:238–46.
National Institute for Health and Clinical Excellence. Therapeutic hypothermia with intracorporeal temperature monitoring for hypoxic perinatal brain injury. http://www.nice.org.uk/nicemedia/live/11315/48809/48809.pdf. May 2010.
Jacobs S, Hunt R, Tarnow-Mordi W, Inder T, Davis P . Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev 2007:CD003311.
Fairchild K, Sokora D, Scott J, Zanelli S . Therapeutic hypothermia on neonatal transport: 4-year experience in a single NICU. J Perinatol 2010;30:324–9.
Compagnoni G, Bottura C, Cavallaro G, Cristofori G, Lista G, Mosca F . Safety of deep hypothermia in treating neonatal asphyxia. Neonatology 2008;93:230–5.
Azzopardi D, Robertson NJ, Cowan FM, Rutherford MA, Rampling M, Edwards AD . Pilot study of treatment with whole body hypothermia for neonatal encephalopathy. Pediatrics 2000;106:684–94.
Hoque N, Chakkarapani E, Liu X, Thoresen M . A comparison of cooling methods used in therapeutic hypothermia for perinatal asphyxia. Pediatrics 2010;126:e124–30.
Robertson NJ, Kendall GS, Thayyil S . Techniques for therapeutic hypothermia during transport and in hospital for perinatal asphyxial encephalopathy. Semin Fetal Neonatal Med 2010;15:276–86.
Lazorthes G, Campan L . Moderate hypothermia in the treatment of head injuries. Clin Neurosurg 1964;12:293–9.
Yager J, Towfighi J, Vannucci RC . Influence of mild hypothermia on hypoxic-ischemic brain damage in the immature rat. Pediatr Res 1993;34:525–9.
Sahuquillo J, Vilalta A . Cooling the injured brain: how does moderate hypothermia influence the pathophysiology of traumatic brain injury. Curr Pharm Des 2007;13:2310–22.
Busto R, Globus MY, Dietrich WD, Martinez E, Valdés I, Ginsberg MD . Effect of mild hypothermia on ischemia-induced release of neurotransmitters and free fatty acids in rat brain. Stroke 1989;20:904–10.
Steen PA, Milde JH, Michenfelder JD . The detrimental effects of prolonged hypothermia and rewarming in the dog. Anesthesiology 1980;52:224–30.
Groban L, Zapata-Sudo G, Lin M, Nelson TE . Effects of moderate and deep hypothermia on Ca2+ signaling in rat ventricular myocytes. Cell Physiol Biochem 2002;12:101–10.
Battin MR, Thoresen M, Robinson E, Polin RA, Edwards AD, Gunn AJ; Cool Cap Trial Group. Does head cooling with mild systemic hypothermia affect requirement for blood pressure support? Pediatrics 2009;123:1031–6.
Clifton GL, Allen S, Berry J, Koch SM . Systemic hypothermia in treatment of brain injury. J Neurotrauma 1992;9: Suppl 2:S487–95.
Sprung J, Cheng EY, Gamulin S, Kampine JP, Bosnjak ZJ . Effects of acute hypothermia and beta-adrenergic receptor blockade on serum potassium concentration in rats. Crit Care Med 1991;19:1545–51.
Pulsinelli WA, Waldman S, Rawlinson D, Plum F . Moderate hyperglycemia augments ischemic brain damage: a neuropathologic study in the rat. Neurology 1982;32:1239–46.
Hattori H, Wasterlain CG . Posthypoxic glucose supplement reduces hypoxic-ischemic brain damage in the neonatal rat. Ann Neurol 1990;28:122–8.
Vannucci RC, Brucklacher RM, Vannucci SJ . The effect of hyperglycemia on cerebral metabolism during hypoxia-ischemia in the immature rat. J Cereb Blood Flow Metab 1996;16:1026–33.
Thoresen M, Satas S, Løberg EM, et al. Twenty-four hours of mild hypothermia in unsedated newborn pigs starting after a severe global hypoxic-ischemic insult is not neuroprotective. Pediatr Res 2001;50:405–11.
Falahati A, Sharkey SW, Christensen D, et al. Implementation of serum cardiac troponin I as marker for detection of acute myocardial infarction. Am Heart J 1999;137:332–7.
Liu X, Tooley J, Løberg EM, Suleiman MS, Thoresen M . Immediate hypothermia reduces cardiac troponin I after hypoxic-ischemic encephalopathy in newborn pigs. Pediatr Res 2011;70:352–6.
Mathur NB, Krishnamurthy S, Mishra TK . Evaluation of WHO classification of hypothermia in sick extramural neonates as predictor of fatality. J Trop Pediatr 2005;51:341–5.
Murray DJ, Forbes RB, Mahoney LT . Comparative hemodynamic depression of halothane versus isoflurane in neonates and infants: an echocardiographic study. Anesth Analg 1992;74:329–37.
Kurth CD, O’Rourke MM, O’Hara IB . Comparison of pH-stat and alpha-stat cardiopulmonary bypass on cerebral oxygenation and blood flow in relation to hypothermic circulatory arrest in piglets. Anesthesiology 1998;89:110–8.
Jonas RA, Bellinger DC, Rappaport LA, et al. Relation of pH strategy and developmental outcome after hypothermic circulatory arrest. J Thorac Cardiovasc Surg 1993;106:362–8.
Lorek A, Takei Y, Cady EB, et al. Delayed (“secondary”) cerebral energy failure after acute hypoxia-ischemia in the newborn piglet: continuous 48-hour studies by phosphorus magnetic resonance spectroscopy. Pediatr Res 1994;36:699–706.
Acknowledgements
We thank Paul Basset for statistical support; Neil Sebire and Elizabeth Powell for organ pathology assessment; David Cox, Alan Groves, Mark Busbridge, and Richard Chapman for serum troponin assessment.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kerenyi, A., Kelen, D., Faulkner, S. et al. Systemic effects of whole-body cooling to 35 °C, 33.5 °C, and 30 °C in a piglet model of perinatal asphyxia: implications for therapeutic hypothermia. Pediatr Res 71, 573–582 (2012). https://doi.org/10.1038/pr.2012.8
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2012.8
This article is cited by
-
Protective effects of delayed intraventricular TLR7 agonist administration on cerebral white and gray matter following asphyxia in the preterm fetal sheep
Scientific Reports (2019)
-
Therapeutic hypothermia translates from ancient history in to practice
Pediatric Research (2017)
-
Diagnose und Behandlung der perinatalen Asphyxie
Monatsschrift Kinderheilkunde (2014)
-
Comparison of Three Hypothermic Target Temperatures for the Treatment of Hypoxic Ischemia: mRNA Level Responses of Eight Genes in the Piglet Brain
Translational Stroke Research (2013)