Abstract
The Saqqara archaeological area is rich in ancient Egyptian artefacts that have been discovered periodically. In the context of preserving these excavated relics, a cartonnage mask, which could be dated to the Graeco-Roman period, was studied to provide information about the material composition of its pictorial layers (substrate, gesso layer and polychrome layer), to explore its manufacturing techniques, and to evaluate the impact of the surrounding environmental conditions on it. Multi-analytical methods were utilised: OM, SEM-EDS, RS, and ATR-FTIR. The results revealed that the mask’s colour palette included realgar, Egyptian blue, red ochre, lead white, carbon black, orpiment, and pararealgar. The binding medium used with the gesso layer and most polychrome layers was animal glue. Some changes in the molecular structure of the linen were observed, indicating the natural ageing process and chemical reactions with the surrounding environment. These outcomes provide useful information for appropriate restoration and conservation processes.
Similar content being viewed by others
Introduction
The ancient Egyptians were always concerned about the loss of the deceased’s head or the deprivation of their physical abilities. Thus, the main purpose of the mummy masks was to protect the head and facial features of the deceased to ensure a safe passage into the afterlife1,2,3. Mummy masks were often made of two- or three-dimensional cartonnage, layers of linen or papyrus covered with a thin layer of gesso (render), much like papier-mâché. Once it dried, it was decorated with paint and gilding4,5,6. In the Graeco-Roman period, the art of the cartonnage flourished and was widely utilised when it became a combination of Hellenistic and Egyptian arts as a result of the Greeks and Romans’ belief in the Egyptian faith7,8.
Egyptians have used colours throughout the ages to express their culture, traditions, and religious beliefs. In the pre-dynastic period, the ancient Egyptians utilised natural minerals available locally, such as red and yellow ochre, calcium carbonate, malachite, and black carbon. In the Fourth Dynasty, with the advancement of Egyptian civilisation, the first synthetic colour, Egyptian blue, was produced. As trade expanded in the Middle and New Kingdoms, some colours, such as orpiment and realgar, were imported from foreign countries and incorporated into the Egyptian palette9. A significant development in colouring materials occurred when the Romans invaded Egypt in 30 BC, as a wide range of materials and techniques were added to the ancient Egyptian palette to achieve various colours. Pliny, Vitruvius, Dioscorides, and Theophrastus mentioned that the coloured materials used by artists in the Roman Empire numbered around 3510,11. Colour manufacturers in the Graeco-Roman era expanded the variety of synthetic colours using mineral sources and pigments and unveiled the secret of making Egyptian blue. Among the interesting pigments that Greek and Roman artists introduced to the ancient Egyptian palette were white lead, cinnabar, and red lead (minium)12,13.
Some the literature addressed the Greco-Roman Egyptian polychrome artefacts. For example, Kasso et al.14 revealed the rare use of indigo dye in a cartonnage mask to create a purple colour. The study also noted the use of red and yellow ochre, cuprorivaite, and carbon as red, yellow, blue, and black colours, respectively. Kamel et al.15 mentioned that hematite, a reddish-orange colour, and hematite with magnetite, a brown colour, were used to decorate a stucco mask from the National Museum of Egyptian Civilization. Similarly, Abdel-Ghani et al.16 examined a cartonnage mask and found that madder, Egyptian blue, magnetite, malachite and atacamite, orpiment and yellow ochre were used to make pink, blue, black, green and yellow colours, respectively. Furthermore, Abdullah et al.17 found the hematite, lead white, Egyptian blue, earth green, and orpiment pigments in the palette of a wooden naos from the Grand Egyptian Museum and revealed that the mixture of black ivory and vermilion was introduced to obtain a brown colour. Scott et al.18 found that the green colours of cartonnage fragments in the Petrie Museum, London, were green earth or a mixture of Egyptian blue and yellow. While examining the green colour of a cartonnage collar in the School of Religion, University of California, Scott et al.19 found it was an organic copper carbohydrate. Sherbiny et al.20 examined a decorated wooden panel from Saqqara and highlighted the use of carbon, hematite, and goethite.
The identification of the pigments and binder is one of the most significant goals in the scientific study of ancient polychrome objects. It provides historical information about the artefact and solves the problems of dating and attributing the work to its creator. Furthermore, such information is crucial for restorers to select the most effective restoration and conservation techniques21,22. The pigments, binder, and ground layer of the ancient polychrome objects are identified using various multi-analytical techniques, including optical microscopy, scanning electron microscopy combined with energy dispersive X-ray microanalysis (SEM/EDS), X-ray diffraction (XRD), Raman spectroscopy, and Fourier transform infrared spectroscopy (FTIR)23,24,25,26,27. For instance, SEM-EDS can provide details about the micro-morphology of the surface and the elemental composition of the materials throughout micron or even sub-micron regions. The XRD technique gives information about the chemical composition of crystalline materials. Raman spectroscopy and FTIR detail the molecular structure of the organic and inorganic components found in the paint and ground layers28.
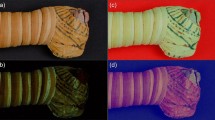
In this research, the object of study was a cartonnage mask discovered by the Egyptian Ministry of Antiquities mission in the south of the Saqqara site-Egypt, Egypt, in 2019. The mummy was badly deteriorated; therefore, neither the owner of this object nor the period could be identified. The mask was currently kept at the conservation lab under the excavation number (S3.19). The funeral mask (Fig. 1a, b), is for a lady with a calm smile, wide almond-shaped eyes, and thick blue eyebrows with a black outline. The wig extends to the neck and forms parallel broad lines decorated in orange and blue. The neck is decorated with a broad collar consisting of several rows of different geometric designs and is coloured yellow, brown, light green, and blue. The approximate dimensions of the cartonnage are 33 cm in length, 24 cm in width, and 11 cm in height.
a the front, and b the reverse, showing various sampling areas: (a1) orange colour, (a2) blue colour, (a3) light red colour, (a4) black colour, (a5) yellow colour, (a6) green colour, and (b1, 2) textile samples.
Due to the dry sandy burial environment and subsequent poor preservation conditions, the cartonnage was subjected to natural deterioration, resulting in the weakness and degradation of the fabric layer, separation and discolouration of some parts, as well as the loss of some paint layers.
The aim of this current work to investigate the layered structure of the cartonnage (support, ground, and paint layers), understand its manufacturing method, and study the degradation and changes caused by the surrounding environmental conditions through integrated analytical techniques. The results accurately characterise the chemical composition of the cartonnage materials and indicate that the cartonnage dates to the Graeco-Roman period compared to those from various periods in ancient Egypt. Following that, the restorer can develop a strategy for the treatment and conservation of the mask.
Results
Cross-sectional images of the mask showed that the structure of the cartonnage consists of four layers: Two textile layers, a ground layer, and a paint layer (Fig. 2). The approximate thickness of all cartonnage layers is 1.5 mm. The thickness of the textile support measures about 290 μm, while the ground layer is irregular and ranges from 475 to 516 μm.
a Image by optical microscopy, and b scheme illustrating all the layers of cartonnage mask.
Textile layer
The visual inspection of the fabric support of the cartonnage revealed the presence of dark and light areas. The light fabric sample was taken from the left edges of the mask, while the dark fabric was taken from the right edges of the mask (Fig. 1. b1, 2).
Under optical microscopy (Fig. 3a, b), the threads of the b1, 2 samples appear completely covered with the adhesive matter, and the colour of the threads in the b1 sample is bright yellow. In contrast, in the b2 sample, the threads range from dark brown to burnt in some areas. Also, a microscopic examination of both samples showed that the fabric type is linen and that the weave pattern of the linen support is plain weave 1/1. The warp and weft yarns of the textile take the shape of S-twists, and the diameter of one linen thread is approximately 215 µm, while the thickness of one fibril is around 17 µm. SEM photomicrographs (Fig. 3c) of two fabric samples revealed that the fibres are cylindrical bundles with transverse nodes, characteristic of linen29,30. On the other hand, SEM photos showed that the dark fabric sample was more damaged and rougher than the lighter sample, and its fibres were drier, more brittle and prone to breakage, with more longitudinal splitting and transverse cracking.
a Optical microscopy of the light thread, b optical microscopy of the dark thread sample, c SEM micrograph of the fibre, d FTIR-ATR analysis of the light fabric, and e the dark fabric.
FTIR spectrum confirmed that the textile support is linen. To understand the distinctions between the two samples, FTIR spectra were employed. By comparing the infrared spectra of b1 and b2 samples (Fig. 3d, e and Table 1), changes in the relative intensity and positions of some bands and the disappearance of some bands were observed. This result might indicate that the molecular structure of cellulose underwent chemical changes31. A strong broad (OH) stretching vibration band appeared at 3334 and 3281 cm−1 in the b1 sample, while the b2 sample peaks seemed to be displaced at low intensity to 3328 and 3290 cm−1, respectively. This shift might be related to the b2 sample’s high degree of dehydration due to the alteration of cellulose crystallinity32,33. The (νsCH2) band of methyl groups (cellulose compounds) shifted from 2852 cm−1 of the b1 sample to 2891 cm−1 of the b2 sample. Two absorption bands at 1371 and 1318 cm−1 were attributed to C-H and C-O-H bending vibrations34 associated with the composition of cellulose and hemicelluloses35. The peak at 1371 cm−1 of the b1 sample shifted to a low intensity at 1369 cm−1 in the b2 sample, which was perhaps a result of depolymerisation and cellulose’s supramolecular alterations36,37. The peak at 1318 cm−1 of the b1 sample shifted to a higher intensity at 1321 cm−1 in the b2 sample. Additionally, the increase in intensity of this band indicated the high crystallinity of cellulose; this could be due to a deficiency of water38. Garside and Wyeth reported that the strength of fibres is influenced by the relative crystallinity of cellulose39.
Polysaccharides in cellulose in the 1b sample are indicated by the (C-C) ring breathing band40 at 1206 and 1147 cm−1, and the asymmetric vibrations of the (C-O-C) glycosidic ether band41,42,43 at 1109 cm−1. These bands shifted to a relatively low intensity at 1204, 1157 and 1107 cm−1, respectively, in the b2 sample; this suggests that the cellulose chains broke44.
The stretching (C-OH) vibration band45 at 1024 cm−1 in the b1 sample shifted to a relatively less intense band at 1028 cm−1 in the b2 sample; this band’s change might be attributed to the hydrolysis of cellulose, which resulted in elevated carbonyl group concentrations46. The (C-O-C) stretching vibration band of the cellulose structure47 appeared at 908 cm−1 in sample b1 and disappeared in the b2 sample. The peaks at 699, 668, 605, 555, and 524 cm−1 in the b1 sample shifted to high intensity at 698, 660, 607, 556, and 521 cm−1 in the b2 sample, respectively.
Gum Arabic, as an adhesive to the two textile layers, was recognised by FTIR spectra. The b1 sample showed a prominent peak at 2917 cm−1, which was ascribed to the symmetric and asymmetric stretching vibrations of aliphatic (CH2, CH3)48, suggesting the presence of arabinose, rhamnose, and galactose49. In the b2 sample, the peak shifted to 2942 cm−1.
The (C = O) stretching vibration band appeared at 1633 cm−1 in the b1 sample, indicating the existence of carboxylate groups and intramolecularly bound water50. This peak shifted to a narrower and higher intensity band at 1640 cm−1 in the b2 sample, which might indicate a decomposition of gum in the b2 sample. The band at 1423 cm−1 of the b1 sample attributed to v3 antisymmetric CO32- stretching vibrations51 shifted to a low intensity at 1424 cm−1 in the b2 sample, which could be linked to a partial degradation of the units of polysaccharides of gum Arabic52.
Thombare et al.48 reported that the fingerprint of gum Arabic is in the region between 950 and 700 cm−1 and is mainly caused by the stretching vibration of the polymeric skeleton and the formation of α and β of the monomeric unit. In the b1 sample, the peaks at 874 and 840 cm−1 of β-glycosidic bonds of gum Arabic disappeared in the b2 sample.
Ground layer
SEM-BS micrographs (Fig. 4a) of the ground layer showed fine-grained calcite crystals with fairly equal sizes ranging from 5 to 12 µm. They also revealed the disintegration and incoherence of the ground layer and some voids between calcite grains.
a SEM micrograph displays fine-grained crystals of calcite, and b EDS spectrum of the layer.
EDS analysis (Fig. 4b) showed that the gesso layer is composed of Ca and C as major elements, indicating the use of calcite (CaCO3) to prepare the underlying plaster. A few traces of Al and Cl were also detected. Aluminium could be attributed to impurities of clay minerals in the limestone rock, which were used as a source of the ground layer, while chlorine was probably due to the presence of halite salts.
Raman spectra confirmed the presence of calcite. The distinctive strong band at 1086 cm−1 corresponded to the C-O symmetric stretch vibration, the band of medium intensity at 282 cm−1 attributed to the relative motion vibration between CO32− 53 and its bands at 153 and 710 cm−1 54,55. Calcium carbonate (CaCO3) has been commonly used as a preparation ground for cartonnage and wood since ancient Egyptian times12
The FTIR analysis of the gesso layer sample (Fig. 5a) showed calcite and animal glue spectra compared with reference samples. A database of IR reference spectra is available online at http://www.irug.org/. Calcite was identified as the main component, probably from the underlying ground layer by the characteristic band at 710 cm−1, which was attributed to the C-O in-plane bending vibration, a peak at 872 cm−1, assigned to the CO32– out-of-plane bending vibration and a peak at 1394 cm−1, ascribed to the CO32− stretching band53,56,57.
a The ground layer, b the orange layer, c the blue layer, and d the light red layer.
Animal glue was identified by an intense characteristic band at 3319 cm−1, attributed to the N-H stretching vibration58, and the bands at 2983–2870 cm−1, assigned to the C-H stretching vibrations (methylene groups), and a weak band at 1646 cm−1, which was due to the C = O stretching of amide I group27,59. Additionally, the amide II band at 1543 cm−1 was attributed to C-N-H bending vibrations60,61,62.
Orange colour
The optical microscopy of the orange sample taken from the wig (Fig. 1. a1) displayed the roughness and brightness of the surface and crystals of different shades, ranging from reddish-orange to bright yellow (Fig. 6a). It also showed the binding medium between the colour granules in some areas. The cross-section examination (Fig. 6b) illustrated that the thin orange layer ranged from 24 to 30 µm.
a Optical microscopy of the surface, b cross-section of the sample, and c Raman spectrum of the colour.
The SEM micrograph exhibited the consistency and interdependence of the colour grains in some areas, but there were fine cracks and voids on the surface. EDS analysis showed the existence of arsenic and sulphur, which might indicate the use of realgar (As4S4). Calcium and carbon, indicating the underlying ground layer, were also revealed.
The micro-Raman spectrometer (Fig. 6c) confirmed the presence of realgar, which exhibited its characteristic bands at 142, 166, 183, 190, 219, 341, 352, and 370 cm−1 63,64.
The FTIR spectra of the sample (Fig. 5b) revealed the presence of animal glue as a binder of the colour, as demonstrated by its characteristic bands. An intense band at 3276 cm−1 was attributed to N-H stretching, the bands at 2943 and 2837 cm−1 were characteristic of C-H stretching (methylene groups), and the band at 1639 cm−1 (C = O stretching band) was assigned to amide I groups27. There were also bands at 1521 and 1422 cm−1, indicating amide II and III groups, respectively.
FTIR confirmed the presence of calcite in the ground layer, as shown by the existence of distinctive bands at 1398, 873 and 711 cm−1 65.
Blue colour
The optical microscopy of the surface of the blue paint sample taken from the wig (Fig. 1. a2) showed coarse grains of heterogeneous sizes and different shades, ranging from glassy and pale to dark blue (Fig. 7a). The cross-section revealed that the thickness of the paint layer ranged from 114 to 122 µm.
a Optical microscopy of the surface, and b Raman spectrum of the paint.
SEM image of the blue pigment exhibited the distinctive shape of the crystals of the mineral cuprorivaite, the major constituent of Egyptian blue. EDS microanalysis of a single crystal in the sample indicated that the predominant elements were calcium, copper, silicon, and oxygen, forming the chemical formula of cuprorivaite (CaCuSi4O10). EDS also revealed carbon and aluminium as major elements in the ground layer. Chlorine was found in relatively small amounts, which could be attributed to the sandy soil where the mask was buried, characterised by a high concentration of sodium chloride salt.
Egyptian blue was also detected by the micro-Raman spectrometer (Fig. 7b) with its characteristic strong band at 1084 cm−1, bands of medium intensity at 582, and 991 and weak bands at 157, 222, 372, and 470 cm−1 66,67. The Raman band at 1049 cm−1 is not assignable.
FTIR spectrum (Fig. 5c) confirmed the EDS and Raman results, showing the absorption bands of Egyptian blue and calcite. Egyptian blue was identified by the characteristic silicate bands at 1111, 1030 cm−1 and 669, 597 cm−1, which were attributed to the anti-symmetrical and symmetrical Si-O-Si stretching bands, respectively68,69. The characteristic bands of calcite and animal glue were also observed.
Light red colour
The light red paint was sampled from the right ear of the mask (Fig. 1. a3). The surface of the sample revealed the difference in the degree of light red colour from one area to another, where some areas appeared dark and others light (Fig. 8a). This result might be because the artist did not mix red and white paint well. The cross-sectional image showed a very thin and regular thickness of about 17 μm.
a Optical microscopy of the surface, b Raman spectrum, c EDS spectrum.
Micro-Raman spectrometer (Fig. 8b) revealed that the light red pigment consisted of a mixture of red ochre (Fe2O3 (hematite) + clay minerals) as a red pigment and lead white as a white pigment. The characteristic bands of red ochre were detected at 227, 242, 304, 492, and 601 cm−1 70,71, in addition to the bands assigned to lead white at 106, 329, 419, 678, 964, and 1059 cm−1 64,67,72. Also, calcite was identified as the main component, probably from the ground layer at distinctive bands of 154, 281, 719, and 1084 cm−1.
EDS spectrum (Fig. 8c) showed the predominant presence of lead, accompanied by small amounts of iron, silicon, aluminium, and magnesium. The peaks of lead and iron indicated lead white, basic lead (II) carbonate (2PbCO3·Pb(OH)2), and iron oxide (Fe2O3), respectively. The presence of silicon, aluminium, and magnesium might be due to the use of clay minerals with iron oxide or as impurities from the ground layer. Moreover, EDS spectra detected traces of Na and Cl, which might be attributed to halite salts, as well as Ca and C, which are distinctive of calcite.
FTIR spectrum (Fig. 5d) confirmed red ochre in the light red layer and recognised the distinctive bands of iron oxide, kaolin, and quartz. The bands of iron oxide at 530 and 468 cm−1 were assigned to Fe-O stretching vibrations73. Kaolin was recognised by the strong band at 1026 cm−1, which was attributed to asymmetric Si-O-Si stretching vibrations57 and the bands at 3692, 3671, and 3619 cm−1, which were ascribed to the O-H stretching vibrations60,74. No distinctive absorption for quartz was detected because it probably overlapped with the intense bands of iron oxide and kaolin.
Calcite was identified by the characteristic band at 710 cm−1, which was attributed to the C-O in-plane bending vibration, and the band at 872 cm−1 was assigned to the CO32– out-of-plane bending vibration. Lead white, which could be used as a white pigment to produce a light red tone, was also recognised at 669 cm−1 by its characteristic bands75,76. The CO32- stretching vibration band at 1411 cm−1 made it difficult to distinguish whether it belonged to lead white or calcium carbonate because the asymmetric stretching bands were very close77. FTIR data also showed the characteristic peaks of animal glue at 3337, 3296, 2917, 2849, 1628, 1576, and 1539 cm−1.
Black colour
The optical image of the black layer (Fig. 1. a4) taken from the black vertical lines separating the blue and orange colours of the wig revealed fine homogeneous black grains, often circular. The thickness of the paint appeared regular, ranging from 23 to 28 µm.
An SEM image (Fig. 9a) showed a rough, irregular surface with some microcracks and gaps. EDS analysis (Fig. 9b) revealed that the black sample mainly consisted of carbon, indicating that carbon black was used in addition to small amounts of Si and Al as impurities.
a SEM micrograph of the surface, b EDS spectrum, c Raman spectrum.
The results of Raman analyses (Fig. 9c) confirmed that the black paint was carbon; its characteristic broad bands at 1349 and 1595 cm−1 were detected55,78.
Yellow colour
The optical microscopy image of the yellow paint from the broad collar (Fig. 1. a5) displayed an uneven and rough texture with crystals of different hues ranging from dark yellow to brown (Fig. 10a).
a Optical microscopy of the crystals of different hues, ranging from dark yellow to brown; b Raman spectrum of particles of yellow pigment, (blue) yellow particles, (green) light brown particles, (red) dark brown particles, and c EDS spectrum of the sample.
Owing to the heterogeneity in yellow hues, Raman spectroscopy was utilised to analyse the yellow, light brown, and dark brown particles separately (Fig. 10b). The obtained spectrum of the yellow particle indicated only orpiment with characteristic bands at 105, 136, 153, 179, 200, 290, 309, 354, and 382 cm−1 63,79. In contrast, the Raman spectrum of the light brown and dark brown particles revealed both orpiment and pararealgar. The characteristic bands of pararealgar appeared at 117, 142, 184, 192, 222, 231, 274, 333, 345, and 363 cm−1 80,81.
The results were confirmed by EDS (Fig. 10c), which showed a high intensity of arsenic and carbon and a low intensity of calcium and sulphur. Arsenic and sulphur suggested the utilisation of arsenic sulphide pigments.
Green colour
The green pigment was sampled from the broad collar (Fig. 1. a6). The microscopic examination (Fig. 11a) of the green paint showed a coarse and saturated layer with a mixture of yellow and blue pigment grains.
a Optical microscopy of the surface, and b Raman spectrum of the yellow particles.
Raman analyses (Fig. 11b) indicated that the yellow grains were composed of orpiment with a characteristic prominent band at 353 cm−1 64, a medium band at 308 cm−1, and weak bands at 135, 154, 200, 289, and 381 cm−1 67. On the contrary, the blue grains did not exhibit any Raman signal due to significant fluorescence emission spectra. This luminescence might be evidence of the existence of cuprorivaite82,83,84.
The EDS spectrum revealed copper, calcium, silicon, and oxygen, indicating the presence of Egyptian blue (a cuprorivaite mineral) and arsenic and sulphur, possibly due to the use of orpiment. Carbon was also detected, which is distinctive to calcite. In addition, small amounts of Al and Cl were found. These results confirmed that a mixture of blue and yellow pigments was used to produce a green pigment.
Discussion
The integrated scientific analysis of the studied cartonnage confirmed that it dates to the Graeco-Roman period. This study secured fundamental information on the type of support, the materials of the ground layer and the pigments used in this cartonnage. Table 2 summarises the results of the scientific analyses of (EDS, Raman spectroscopy, and FTIR-ATR)of cartonnage samples.
The results clarified that the mask was made of a double-layer woven fabric used as a support, covered with a single layer of gesso on one side only, which was used to apply the paint layer. This structure is similar to a broad collar of cartonnage from an archaeological research collection in California dating back to the Late Period19. In Graeco-Roman cartonnage, the use of a single ground layer is relatively widespread. It has been previously detected in cartonnages from the Egyptian Museum85 and the Fitzwilliam Museum86 and cartonnage fragments from the Petrie Museum, London18. Using calcite with an organic medium as a ground layer of the paints was previously detected on cartonnage fragments from El-Lisht site, Egypt87, and on Graeco-Roman cartonnage from the Ismailia Museum, Egypt88.
The desert burial environment, high temperatures, and poor preservation of the mask encouraged the oxidation and re-crystallisation of the fabric of the substrate. OM and SEM examination revealed that the textile support suffered from deterioration; this could be attributed to the decomposition of gum Arabic89 or the thermal deterioration of the textile fibres90. In addition, an ATR-FTIR analysis of two different support samples (light and dark) revealed that the dark parts were more fragile and damaged than the light areas; this was observed when some bands disappeared from the dark sample. The disappearance of the band at 908 cm−1, which indicated the glycosidic bonds among the glucose units in the cellulose II system, is evidence of the cellulose degradation process44. Also, the disappearance of the fingerprint of gum Arabic in the dark sample was a sign of the decomposition of gum. The exact cause of the damage to these parts was unknown, but it might be due to the use of a larger amount of adhesive material in these areas or because these parts had more contact with the mummy. Similar results were reported by Afifi et al.87.
The palette of this cartonnage matched those identified in ancient Egyptian polychrome during the Roman era. It also gave us new insights into the colouring materials used in ancient Egyptian cartonnage. Among these highlights was the use of realgar as an orange colour, as its use alone has never been documented in the pigment palette of the Egyptian cartonnage previously studied. In general, orange was not widely used to decorate Egyptian cartonnage. It was identified in two cases: the first on Ptolemaic gilded cartonnage91 was a mixture of realgar with orpiment and uzonite; the second case was on a Graeco-Roman mask, was made from orpiment and hematite16.
In the wide context, realgar was used to produce different shades on Egyptian cartonnage from the Roman period. For example, it was used with goethite to create a yellow hue92 and mixed with Egyptian blue, orpiment, and hematite to achieve a purple hue93. To make a pink hue, realgar was found combined with red ochre and white pigment94.
Realgar α-As4S4/β-As4S4, an orange-red natural mineral, was used as a pigment in ancient Egypt from the New Kingdom (1550–1069 BC) to the Roman Period (30 B.C.–395 A.D.) in all contexts, but it was not found before this time12,95,96. Realgar is common and abundant in many arsenic-bearing sediments and is often associated with volcanic springs with hydrothermal-related mineralisation, where it occurs as an oxidised product97,98.
Another highlight in the palette of this cartonnage was the use of lead white in combination with ochre red to create a light red shade. Lead white has not been documented before as a mixture with other pigments in the decoration of Egyptian cartonnage; however, it was only found as a pure white colour on cartonnage dating back to the Graeco-Roman period99. Lead white was used in Egypt from the Graeco-Roman Period until the 19th century100. It was manufactured by exposing lead strips to vinegar or acetic acid vapours in a closed, carbon dioxide-rich environment. Carbon dioxide was provided by fermentation materials, such as animal manure and waste grape skins, which provided the high temperature required to speed up the reactions101,102. In the previously studied cartonnages, gypsum, calcite, and huntite were the most used white pigments combined with other colours to produce lighter shades90,103.
The ochre red was widely used from the 5th Dynasty to the Roman period in Egypt104, as documented in many cartonnages16,94,105. The primary ingredient of red earth pigments is octahedral iron oxide, also known as haematite, in addition to varying amounts of quartz and clay minerals106,107. Red ochres are formed in the weathered, oxidised areas of iron-rich mineral sediment, particularly those connected to huge volcanic sulphide deposits108,109. The light red or pink colour was detected on Late period foot case cartonnage, which was red ochre mixed with gypsum103 and on Roman cartonnage, it was madder dyestuff16,92,110.
The Egyptian blue was revealed in two areas in this cartonnage: the first in the blue area and the second in the green area. Egyptian blue was used extensively from the 4th Dynasty in Egypt until the late Roman era111. It was earlier recorded as a blue pigment on Graeco-Roman cartonnages from the Egyptian Museum16,105, Hawara (Egypt)112 and the University of Southern California99, a Ptolemaic gilded cartonnage from La Plata Museum, Argentina91, and on a Late Period cartonnage from Cairo Museum105. A few exceptions indicated the use of indigo and cobalt blue as blue colours on Roman cartonnage from the Petrie excavations in Hawara86 and on cartonnage from the Late Period at Saqqara8, respectively.
The blue colour (Egyptian blue) was mixed with the yellow colour (orpiment) to produce the green hue in this cartonnage. This artistic style was used in previous cartonnages, but the yellow component used might either be iron oxide99, goethite18, or orpiment90. This method of producing the green colour appeared sporadically during the Twelfth Dynasty and spread widely during the late Eighteenth Dynasty and the Roman period113. In a broader context, the most common green colourants in Egyptian cartonnage were earth green87,99,105,114, malachite85, and a mixture of malachite and atacamite16.
Orpiment was identified in the yellow pigment in this cartonnage along with pararealgar. It is an arsenic sulphide (As2S3) that ranges from yellow to greenish yellow and contains 60% arsenic. It frequently occurs as granular or massive deposits109. The use of orpiment was previously documented on cartonnages fron the Late Period85,105,115, the Ptolemaic period116,117, and the Roman period86,99.
The existence of pararealgar raises the question of whether the ancient Egyptian artist used realgar intentionally or mistakenly; with natural ageing, it turned into pararealgar as a degradation product subsequently. This suggestion may be likely because the ancient Egyptian used realgar in the cartonnage mask understudy. The second possibility is the ancient Egyptian artist deliberately used pararealgar. Mineral pararealgar is often found accompanied by orpiment and realgar98, and it is a product of the photo-induced degradation of realgar118. For instance, Hussein et al.103 documented the use of pararealgar aside from orpiment and yellow ochre as a yellow pigment in foot case cartonnage dating to the Late Period.
The black colour in this cartonnage was identified as carbon black, previously detected on cartonnages from the Late Period8,85,119 and the Graeco-Roman period94,99,112. According to its crystallisation form, carbon black is classified into crystalline carbon and amorphous carbon. It is considered the most common black pigment in the palette of ancient Egyptian cartonnage. There are several sources of carbon black, such as charcoal black, lamp carbon, and bone (ivory) carbon. Carbon black was found on Egyptian wall paintings dating back to the 4th Dynasty held by the British Museum66, on a polychrome wooden coffin from the Middle Kingdom120, and on the lid of a male adult coffin dating to the Ptolemaic period121.
This study highlights important information about ancient Egyptian cartonnage and emphasises the significance of utilising integrated analytical techniques to characterise cultural relics from ancient times thoroughly.
Methods
Samples
Eight micro-samples were carefully collected from the various structural layers of the polychromic cartonnage mask according to the method mentioned by Teutonico122. Six samples (approximately 2 × 3 to 5 × 10 mm) representing the pigment palette (orange, blue, light red, black, yellow, and green) were used, in addition to two samples from the cartonnage support layer (approximately 1 × 2 cm). Fragments of paint samples 1 mm in size were embedded into transparent epoxy resin (low viscosity No. 610773-4853). After the resin dried, they were grounded and then polished with silicon carbide papers (800, 1000, 1300 grit) until the cross-section surface was smooth123,124.
Optical microscopy (OM)
An optical microscope (model BTC BIM 313 T) coupled with a digital camera (ToupCam) was used to examine the samples. The microscope observations in the reflected light at magnifications from 100x to 500x allowed for the identification of the topography of the paint (the colour of certain individual pigment grains and the presence of pigment mixtures) and the cross-sectional strata of the cartonnage. Additionally, the type of fibre of the textile was identified using transmitted light modes.
Scanning electron microscopy with energy dispersive x-ray spectrometry (SEM-EDS)
A JEOL JSM-IT200 (Tokyo, Japan) SEM instrument fitted out with an energy-dispersive X-ray spectrometer (EDS) unit was used for the SEM-EDS analysis. SEM images and EDS spectra of the samples were taken under low-vacuum mode, which did not require coating the samples with gold or graphite to avoid any potential damage to the samples. The samples were prepared for imaging under the secondary electron (SE) and backscattered (BSE) modes of the SEM. The elemental chemical composition of the samples was determined using an EDS unit with operating conditions of a 20, 25 kV accelerating voltage, 11 scans, and 50 Pa pressure. The SEM-EDS measurements were conducted at the Central Laboratory for Microanalysis and Nanotechnology, Minia University, Egypt.
Raman spectroscopy(RS)
The Raman spectra were collected by a Senterra spectrometer (Bruker) instrument coupled with an Olympus microscope with a 10x, 20x and 50x objective lens. Samples were excited using a red He/Ne laser with a wavelength of 785 nm. The spectra were recorded in a spectral region from 100 to 2000 cm–1 at 4 cm–1 spectral resolution. The laser power was between 0.2 and 5 mW, and the collection time was around 30 s.
Fourier transform infrared spectroscopy coupled with attenuated total reflection (ATR-FTIR)
FTIR spectra were recorded using a Bruker spectrometer VERTEX 70, equipped with an ATR crystal, working in absorbance mode. To obtain distinct absorption spectra for each sample, spectra were collected in the range of 400 to 4000 cm–1, at a spectral resolution of 4 cm−1, with 20 scans. Sample spectra were contrasted with reference spectra125,126. FTIR analysis allowed the identification of the type of textile fibres and binding media of the ground layer and pigments and the study of the changes to the molecular structure of the fibres due to degradation and natural ageing processes.
Data availability
The data generated or analysed during this study are included in this published article.
Abbreviations
- OM:
-
Optical microscopy
- SEM-EDS:
-
Scanning electron microscopy with energy dispersive x-ray spectrometry
- SE:
-
Secondary electron
- BSE:
-
Backscattered electron
- RS:
-
Raman Spectroscopy
- ATR-FTIR:
-
Fourier transform infrared spectroscopy coupled with attenuated total reflection
References
Derbala, A. Unpublished Cartonnage Mummy-Mask from El-Ashmounin Museum Magazine. BCPS 39, 105–120 (2022).
DeMello M. Faces Around the World: A Cultural Encyclopedia of the Human Face. USA: Bloomsbury Publishing; (2012).
Cooney K. M. Coffins, Cartonnage, and Sarcophagi. In A Companion to Ancient Egyptian art. (ed.Hartwig M. K.) 269–292 (John Wiley & Sons Ltd., 2015).
Andrews C. Egyptian Mummies. London: British Museum Press; (1998).
Filer J. The Mystery of the Egyptian Mummy. Oxford University Press; (2003).
Gibson, A. et al. An Assessment of Multimodal Imaging of Subsurface Text in Mummy Cartonnage Using Surrogate Papyrus Phantoms. Herit. Sci. 6, 1–13 (2018).
Root M. C. Faces of Immortality: Egyptian Mummy Masks, Painted Portraits, and Canopic Jars in the Kelsey Museum of Archaeology. Kelsey Museum of ancient and mediaeval archaeology; (1979).
Afifi, H. A., Galal, H. S. & Hassan, R. R. Characterisation of Pharaonic Cartonnage from a Late Period, Saqqara Excavations. Pigment Resin Technol. 49, 255–264 (2020).
Becker, H. Pigment Nomenclature in the Ancient Near East, Greece, and Rome. Archaeol. Anthropol. Sci. 14, 1–25 (2022).
Davy, H. Some experiments and observations on the colours used in painting by the ancients. Philos. Trans. R. Soc. Lond. 105, 97–124 (1815).
Jorge-Villar, S. E. & Edwards, H. G. Green and Blue Pigments in Roman Wall Paintings: A Challenge for Raman Spectroscopy. J. Raman Spectrosc. 52, 2190–2203 (2021).
Scott, D. A. A Review of Ancient Egyptian Pigments and Cosmetics. Stud. Conserv 61, 185–202 (2016).
Becker H. Technology and trade. In: A Cultural History of Colour in Antiquity. Wharton D., editor. Bloomsbury Publishing. 35–39 (2022)
Kasso, T. et al. Facing Death: A Multidisciplinary Analysis of a Romano-Egyptian Mummy Mask at the Ny Carlsberg Glyptotek, Copenhagen. Herit. Sci. 12, 1–22 (2024).
Kamel, A., Ali, M. F., Khedr, M. S. & Mahmoud, H. H. Characterisation of a Greco-Roman Stucco Mask (REG. NO. 229) Preserved at the National Museum of Egyptian Civilisation, Cairo, Egypt. Sci. Arts 23, 773–782 (2023).
Abdel-Ghani, M., Afifi, H. A., Mahmoud, R. & Osman, M. Graeco-Roman Egyptian cartonnage from the Egyptian Museum in Cairo, Egypt: technical and analytical investigation. J. Archaeol. Sci. Rep. 31, 1–13 (2020).
Abdallah, M., Abdrabou, A. & Kamal, H. M. Multiscientific analytical approach of polychrome Greco-Roman palette applied on a wooden model naos: case study. Mediterranean Archaeol. Archaeom. 20, 1–21 (2020).
Scott, D. A., Warmlander, S., Mazurek, J. & Quirke, S. Examination of some Pigments, Grounds and Media from Egyptian Cartonnage Fragments in the Petrie Museum, University College London. J. Archaeol. Sci. 36, 923–932 (2009).
Scott, D. A. et al. An Ancient Egyptian Cartonnage Broad Collar-Technical Examination of Pigments and Binding Media. Stud. Conserv 49, 177–192 (2004).
El Sherbiny, D., Ali, N. M., Abd El Aal, S. & Vallet, J. M. A Multi-Analytical Approach Applied to the Archaeometric Study of a Greco-Roman Decorated Wooden Panel from Egypt. JAEI 7, 27–37 (2015).
Doménech-Carbó, A., Doménech-Carbó M. T., & Costa, V. Electrochemical Methods in Archaeometry, Conservation, and Restoration. Springer Science & Business Media; (2009).
Koochakzaei, A., Alizadeh Gharetapeh, S. & Jelodarian Bidgoli, B. Identification of pigments used in a Qajar manuscript from Iran by using atomic and molecular spectroscopy and technical photography methods. Herit. Sci. 10, 1–16 (2022).
Kovala-Demertzi, D. et al. Pigment Identification in a Greek Icon by Optical Microscopy and Infrared Microspectroscopy. J. Cult. Herit. 13, 107–113 (2012).
Edwards, H. G., Vandenabeele, P. & Benoy, T. J. Raman Spectroscopic Study of “The Malatesta”: A Renaissance Painting? Spectrochim. Acta, Part A 137, 45–49 (2015).
Carbó, M. D., Martínez, V. P., Adelantado, J. G., Reig, F. B. & Moreno, M. M. Fourier Transform Infrared Spectroscopy and the Analytical Study of Sculptures and Wall Decoration. J. Mol. Struct. 410, 559–563 (1997).
Damiani, D., Gliozzo, E. & Memmi, I. T. The ‘Madonna and Child Enthroned with Saints’ of Ambrogio Lorenzetti in the St. Augustine Church (Siena, Italy): Raman Microspectroscopy and SEM-EDS Characterisation of the Pigments. Archaeol. Anthropol. Sci. 6, 363–371 (2014).
Sarmiento, A. et al. Classification and Identification of Organic Binding Media in Artworks by Means of Fourier Transform Infrared Spectroscopy and Principal Component Analysis. Anal. Bioanal. Chem. 399, 3601–3611 (2011).
Zeng, Q. G., Zhang, G. X., Leung, C. W. & Zuo, J. Studies of Wall Painting Fragments from Kaiping Diaolou by SEM/EDX, Micro Raman, and FT-IR Spectroscopy. Microchem. J. 96, 330–336 (2010).
Markova I. Textile Fibre Microscopy: A Practical Approach. John Wiley & Sons (2019)
Marouf M., Saber M. Treatment and Conservation of Archaeological Garment from Greco-Roman Period–The Egyptian Museum, Cairo. 4th International Congress on Science and Technology for the Safeguard of Cultural Heritage in the Mediterranean Basin (2009).
Mansour, M. M., Akarish, A. I., Hassan, R. R. & Shoeib, A. S. Analytical Study of Deteriorated Pharaonic Textile from Mummified Burials-Saqqara. JCHC 5, 17–26 (2018).
Abdel-Maksoud, G., Abdel-Hamied, M. & El-Shemy, H. A. Analytical Techniques Used for Condition Assessment of a Late Period Mummy. J. Cult. Herit. 48, 83–92 (2021).
Esteves, B., Velez Marques, A., Domingos, I. & Pereira, H. Chemical Changes of Heat Treated Pine and Eucalypt Wood Monitored by FTIR. Maderas-Cienc. Tecnol. 15, 245–258 (2013).
Nelson, M. L. & O’Connor, R. T. Relation of Certain Infrared Bands to Cellulose Crystallinity and Crystal Lattice Type. Part II. A New Infrared Ratio for Estimation of Crystallinity in Celluloses I and II. J. Appl. Polym. Sci. 8, 1325–1341 (1964).
Chen, C., Luo, J., Qin, W. & Tong, Z. Elemental Analysis, Chemical Composition, Cellulose Crystallinity, and FT-IR Spectra of Toona Sinensis Wood. Monatsh. Chem. 145, 175–185 (2014).
Kavkler, K. & Frelih, M. Analysis of a Ptolemaic Mummy Foot from Slovene Ethnographic Museum. Procedia Chem. 8, 159–164 (2013).
Kavkler, K., Gunde-Cimerman, N., Zalar, P. & Demšar, A. FTIR Spectroscopy of Biodegraded Historical Textiles. Polym. Degrad. Stab. 96, 574–580 (2011).
Boukir, A., Hajji, L. & Zghari, B. Effect of Moist and Dry Heat Weathering Conditions on Cellulose Degradation of Historical Manuscripts Exposed to Accelerated Ageing: 13C NMR and FTIR Spectroscopy as a Non-Invasive Monitoring Approach. J. Mater. Environ. Sci. 9, 641–654 (2018).
Garside, P. & Wyeth, P. Polarised ATR-FTIR Characterisation of Cellulosic Fibres in Relation to Historic Artefacts. Restaurator 25, 249–259 (2004).
Garside, P. & Wyeth, P. Identification of Cellulosic Fibres by FTIR Spectroscopy-Thread and Single Fibre Analysis by Attenuated Total Reflectance. Stud. Conserv 48, 269–275 (2003).
Coletti, F., Romani, M., Ceres, G., Zammit, U. & Guidi, M. C. Evaluation of Microscopy Techniques and ATR-FTIR Spectroscopy on Textile Fibres from the Vesuvian Area: A Pilot Study on Degradation Processes that Prevent the Characterisation of Bast Fibers. J. Archaeol. Sci. Rep. 36, 102794 (2021).
Esmaeilzadeh, M. J. & Rashidi, A. Evaluation of the Disintegration of Linen Fabric under Composting Conditions. Environ. Sci. Pollut. Res. 25, 29070–29077 (2018).
Margariti, C. The Application of FTIR Microspectroscopy in a Non-Invasive and Non-destructive Way to the Study and Conservation of Mineralised Excavated Textiles. Herit. Sci. 7, 1–14 (2019).
Cao, Q., Zhu, S., Zhao, H. & Tu, H. Application of Fourier Transform Attenuated Total Reflection Infrared Spectroscopy to Identifying Archaeological Fibres. Res. J. Text. Appar. 14, 38–41 (2010).
Liang, C. Y. & Marchessault, R. H. Infrared Spectra of Crystalline Polysaccharides. II. Native Celluloses in the Region from 640 to 1700 cm− 1. J. Polym. Sci. 39, 269–278 (1959).
High, K. E. & Penkman, K. E. A Review of Analytical Methods for Assessing Preservation in Waterlogged Archaeological Wood and Their Application in Practice. Herit. Sci. 83, 1–33 (2020).
Melelli, A. et al. Chemical, Morphological, and Mechanical Study of the Ageing of Textile Flax Fibres from 17th/18th-Century Paintings on Canvas. J. Cult. Herit. 52, 202–214 (2021).
Thombare, N., Mahto, A., Singh, D., Chowdhury, A. R. & Ansari, M. F. Comparative FTIR Characterisation of Various Natural Gums: A Criterion for Their Identification. J. Polym. Environ. 8, 3372–3380 (2023).
Čiuladienė, A., Kareiva, A. & Raudonis, R. From model to artefact: Versatile Characterisation of Cinnabar, Red Lead and Realgar Red Paints for Rubrics and Miniatures. Chemija. 31–34 (2020).
Bashir, M. & Haripriya, S. Assessment of Physical and Structural Characteristics of Almond Gum. Int. J. Biol. Macromol. 93, 476–482 (2016).
Zaffino, C., Guglielmi, V., Faraone, S., Vinaccia, A. & Bruni, S. Exploiting External Reflection FTIR Spectroscopy for the in-situ Identification of Pigments and Binders in Illuminated Manuscripts. Brochantite and Posnjakite as a Case Study. Spectrochim. Acta, Part A 136, 1076–1085 (2015).
Nodari, L. & Ricciardi, P. Non-invasive Identification of Paint Binders in Illuminated Manuscripts by ER-FTIR Spectroscopy: A Systematic Study of the Influence of Different Pigments on the Binders’ Characteristic Spectral Features. Herit. Sci. 7, 1–13 (2019).
Zou W. & Yeo S. Y. Investigation on the Painting Materials and Profile Structures Used in Ancient Chinese Folk Architectural Paintings by Multiple Analytical Methods. Coatings. 12–13 (2022).
Mazzocchin, G. A., Agnoli, F. & Salvadori, M. Analysis of Roman age Wall Paintings found in Pordenone, Trieste and Montegrotto. Talanta 64, 732–741 (2004).
Edwards, H. G. M., Newton, E. M. & Russ, J. Raman Spectroscopic Analysis of Pigments and Substrata in Prehistoric Rock Art. J. Mol. Struct. 550, 245–256 (2000).
Bracci, S. et al. Multidisciplinary Approach for the Study of an Egyptian Coffin (late 22nd/early 25th dynasty): Combining Imaging and Spectroscopic Techniques. Spectrochim. Acta, Part A 145, 511–522 (2015).
Miliani, C., Rosi, F., Daveri, A. & Brunetti, B. G. Reflection Infrared Spectroscopy for the Non-invasive in Situ Study of Artists’ Pigments. J. Appl. Phys. 106, 295–307 (2012).
Carbó, M. D., Reig, F. B., Martínez, V. P. & Adelantado, J. G. Analytical Study of Wall Paintings of the Basilica de La Virgen de Los Desamparados de Valencia with FT-IR Spectroscopy. Quim. Anal. 16, 273–279 (1997).
Pellegrini, D. et al. Fourier Transform Infrared Spectroscopic Study of Rabbit Glue/Inorganic Pigments Mixtures in Fresh and Aged Reference Paint Reconstructions. Microchem J. 124, 31–35 (2016).
Čiuladienė, A., Luckutė, A., Kiuberis, J. & Kareiva, A. Investigation of the Chemical Composition of Red Pigments and Binding Media. Chemija 29, 243–256 (2018).
Manzano, E., Navas, N., Checa-Moreno, R., Rodriguez-Simón, L. & Capitán-Vallvey, L. F. Preliminary Study of UV Ageing Process of Proteinaceous Paint Binder by FT-IR and Principal Component Analysis. Talanta 77, 1724–1731 (2009).
Invernizzi, C., Rovetta, T., Licchelli, M. & Malagodi, M. Mid and Near-infrared Reflection Spectral Database of Natural Organic Materials in the Cultural Heritage Field. Int. J. Anal. Chem. 1, 16 (2018).
Campos, M. M. V. & Aguayo, T. A. Vibrational Spectroscopy for the Study of Chilean Cultural Heritage. Herit. Sci. 3, 1–10 (2015).
Marucci, G., Beeby, A., Parker, A. W. & Nicholson, C. E. Raman Spectroscopic Library of Mediaeval Pigments Collected with Five Different Wavelengths for Investigation of Illuminated Manuscripts. Anal. Methods 10, 1219–1236 (2018).
Glavcheva, Z. I., Yancheva, D. Y., Kancheva, Y. K., Velcheva, E. A. & Stamboliyska, B. A. Development of FTIR Spectra Database of Reference Art and Archaeological Materials. Bulg. Chem. Commun. 46, 164–169 (2014).
Ambers, J. Raman Analysis of Pigments from the Egyptian Old Kingdom. J. Raman Spectrosc. 35, 768–773 (2004).
Caggiani, M. C., Cosentino, A. & Mangone, A. Pigments Checker Version 3.0, a Handy Set for Conservation Scientists: A Free Online Raman Spectra Database. Microchem. J. 129, 123–132 (2016).
Mirti, P. et al. Spectrochemical and Structural Studies on a Roman Sample of Egyptian Blue. Spectrochim. Acta, Part A 51, 437–446 (1995).
Bruni, S., Cariati, F., Casadio, F. & Toniolo, L. Spectrochemical Characterisation by Micro-FTIR Spectroscopy of Blue Pigments in Different Polychrome Works of art. Vib. Spectrosc. 20, 15–25 (1999).
Perardi, A., Appolonia, L. & Mirti, P. Non-destructive in Situ determination of Pigments in 15th Century Wall Paintings by Raman Microscopy. Anal. Chim. Acta 480, 317–325 (2003).
Poliszuk, A. & Ybarra, G. Analysis of Cultural Heritage Materials by Infrared Spectroscopy. In: Infrared Spectroscopy: Theory, Developments, and Applications. Cozzolino D. editor. Nova Science Publishers. p. 519–536 (2014)
Halac, E. B., Reinoso, M., Luda, M. & Marte, F. Raman Mapping Analysis of Pigments from Proas Iluminadas by Quinquela Martín. J. Cult. Herit. 13, 469–473 (2012).
Guglielmi, V., Andreoli, M., Comite, V., Baroni, A. & Fermo, P. The Combined Use of SEM-EDX, Raman, ATR-FTIR, and Visible Reflectance Techniques for the Characterisation of Roman Wall Painting Pigments from Monte d’Oro Area (Rome): An Insight into Red, Yellow, and Pink Shades. Environ. Sci. Pollut. Res. 29, 29419–29437 (2022).
Genestar, C. & Pons, C. Earth Pigments in Painting: Characterisation and Differentiation by means of FTIR Spectroscopy and SEM-EDS Microanalysis. Anal. Bioanal. Chem. 382, 269–274 (2005).
Campani, E., Casoli, A., Darecchio, M. E., Paccagnella, F. & Zannini, P. Saint Petronio Basilica in Bologna (Italy): A Case Study on a XVI Century Mural painting. Nuovo Cim. Della Soc. Ital. Di Fis. B-Basic Top. Phys. 125, 219–241 (2010).
Carbó, M. D., Reig, F. B., Adelantado, J. G. & Martinez, V. P. Fourier Transform Infrared Spectroscopy and the Analytical Study of Works of Art for Purposes of Diagnosis and Conservation. Anal. Chim. Acta 330, 207–215 (1996).
Centeno, S. A., Guzman, M. I., Yamazakikleps, A. & Védova, C. Characterisation by FTIR of the Effect of Lead White on Some Properties of Proteinaceous Binding Media. JAIC 43, 139–150 (2004).
Tomasini, E. P., Halac, E. B., Reinoso, M., Di Liscia, E. J. & Maier, M. S. Micro‐Raman Spectroscopy of Carbon-based Black Pigments. J. Raman Spectrosc. 43, 1671–1675 (2012).
Nastova, I. et al. Micro‐Raman Spectroscopic Analysis of Inks and Pigments in Illuminated Mediaeval Old‐Slavonic Manuscripts. J. Raman Spectrosc. 43, 1729–1736 (2012).
Ogalde, J. P. et al. Multi-Instrumental Identification of Orpiment in Archaeological Mortuary Contexts. J. Chil. Chem. Soc. 59, 2571–2573 (2014).
Vermeulen, M., Saverwyns, S., Coudray, A., Janssens, K. & Sanyova, J. Identification by Raman Spectroscopy of Pararealgar as a Starting Material in the Synthesis of Amorphous Arsenic Sulphide Pigments. Dyes Pigments 149, 290–297 (2018).
Mahmoud, H. H. M. Investigations by Raman Microscopy, ESEM and FTIR-ATR of Wall Paintings from Qasr el-Ghuieta Temple, Kharga Oasis, Egypt. Herit. Sci. 2, 1–11 (2014).
Malegiannaki, I. & Anglos, D. Open-air Laser-induced Breakdown Spectroscopy (LIBS), In: Analytical Strategies for Cultural Heritage Materials and Their Degradation. Madariaga J. M., editor. London: Royal Society of Chemistry. 45–74 (2021).
Badr, N. M., Ali, M. F., El Hadidi, N. M. & Naeem, G. A. Identification of Materials Used in a Wooden Coffin Lid Covered with Composite Layers dating back to the Ptolemaic Period in Egypt. Conservar Patrim.ónio 29, 11–24 (2018).
Abd El-Tawab, N. et al. Analytical Investigation of Cartonnage Fragment from Late Period. Egypt. EJARS 2, 69–78 (2012).
Rowe, S., Siddall, R., & Stacey, R. Roman Egyptian Gilded Cartonnage: Technical Study and Conservation of a Mummy Mask from Hawara, In: Decorated Surfaces Ancient Egyptian Objects, Technology, Deterioration, and Conservation. Dawson J., Rozeik C., Wright M. M., editors. London: Archetype Publications. p.106–121 (2010).
Afifi, H. A., Hassan, R. R. A., Abd-EL-Fattah, M. A. & Menofy, S. M. Spectroscopic Characterisation of Preparation, Pigment, and Binding Media of Archaeological Cartonnage from Lisht, Egypt. Sci. Cult. 6, 9–23 (2020).
Alawneh, F., Elserogy, A. R. & Almasri, E. Using Interdisciplinary Studies and Analyses in the Conservation of Greco-Roman Cartonnage. Conserv. Sci. Cultural Herit. 18, 219–238 (2018).
Kuan, Y. H., Bhat, R., Senan, C., Williams, P. A. & Karim, A. A. Effects of Ultraviolet Irradiation on the Physicochemical and Functional Properties of Gum Arabic. J. Agric. Food Chem. 57, 9154–9159 (2009).
Ali, M. F., Mansour, M. M. A., Badr, N. M. & Salem, M. Z. M. A Study of Biodeterioration and Chro-matic Alterations of Painted and Gilded Mummy Cartonnage at the Saqqara Museum Storeroom, Egypt, Archaeometry. 60, 845–858 (2017).
Gard, F. S. et al. A Noninvasive Complementary Study of an Egyptian Polychrome Cartonnage Pigments Using SEM, EPMA, and Raman Spectroscopy. Surf. Interface Anal. 52: 755–769. (2020)
Sanches, F. et al. Multi-Technique Characterisation of Cartonnage and Linen Samples of an Egyptian Mummy from the Roman Period. Quantum Beam Sci. 8, 1–22 (2024).
Dujmušić, D., Desnica, V., Šatović, D. & Fazinić, S. IBA and Complementary Spectroscopic Methods for Identification of Zagreb Mummy’s Pigments. Nucl. Inst. Methods Phys. Res. B. 465, 53–58 (2020).
Calza, C., Anjos, M. J., de SouzaSM, Brancaglion, J. R. & Lopes, R. T. X-ray Microfluorescence Analysis of Pigments in Decorative Paintings from the Sarcophagus Cartonnage of an Egyptian Mummy. Nucl. Inst. Methods Phys. Res. B. 263, 249–252 (2007).
Vandenbeele, P. et al. Spectroscopic Examination of Two Egyptian Masks: A Combined Approach. Anal. Lett. 33, 3315–3332 (2001).
Siddall, R. Mineral pigments in archaeology: Their Analysis and the Range of Available Materials. Miner. J. 8, 201 (2018).
Eastaugh, N., Walsh, V., Chaplin, T. & Siddall, R. Pigment Compendium Set: Pigment Compendium: A Dictionary of Historical Pigments. Oxford: Butterworth-Heinemann; (2005).
Roberts, A. C., Ansell, H. G. & Bonardi, M. Pararealgar, a New Polymorph of AsS, from British Columbia. Can. Mineral. 18, 525–527 (1980).
Scott, D. A. et al. An Egyptian Cartonnage of the Graeco-Roman Period. Stud. Conserv 48, 41–56 (2003).
Rapp, G. R. Archaeomineralogy. Berlin: Springer; (2013).
Barnett, J. R., Miller, S. & Pearce, E. Colour and Art: A Brief History of Pigments. Opt. Laser Technol. 38, 445–453 (2006).
Gettens, R. J., Kuhn, H. & Chase, W. T. Lead White. In: Artistis‘ Pigments: A Handbook of Their History and Characteristics. Roy A. editor. New York: Oxford University Press. p. 67-81; (1993)
Hussein, A. M., Madkour, F. S., Afifi, H. M., Abdel-Ghani, M. & Abd Elfatah, M. Comprehensive Study of an Ancient Egyptian Foot Case Cartonnage using Raman, ESEM-EDS, XRD, and FTIR. Vib. Spectrosc. 106, 102987 (2020).
Di Stefano, L. M. & Fuchs, R. Characterisation of the Pigments in a Ptolemaic Egyptian Book of the Dead Papyrus. Archaeol. Anthr. Sci. 3, 229–244 (2011).
Ali, M., Abdel-Ghani, M. & Abou Seif, N. An analytical study of a late period multi-piece cartonnage from the Egyptian museum in Cairo. EJARS 12, 29–40 (2022).
Elias, M., Chartier, C., Prévot, G., Garay, H. & Vignaud, C. The Colour of Ochres Explained by Their Composition. Mater. Sci. Eng., B. 127, 70–80 (2006).
Bikiaris, D. et al. Ochre-differentiation through Micro-Raman and Micro-FTIR Spectroscopies: Application on Wall Paintings at Meteora and Mount Athos, Greece. Spectrochim. Acta, Part A 56, 3–18 (2000).
Nicola, M., Mastrippolito, C. & Masic, A. Iron Oxide-Based Pigments and Their Use in History. In: Iron Oxides: From Nature to Applications. Faivre D. Editor. Germany: Wiley. p. 545–566 (2016)
Hedegaard, S. B., Delbey, T., Brøns, C. & Rasmussen, K. L. Painting the Palace of Apries II: Ancient Pigments of the Reliefs from the Palace of Apries, Lower Egypt. Herit. Sci. 7, 1–32 (2019).
Ali, M. F., El Sheikha, A. M. & Ali, A. E. Analytical Study and Conservation of Gilded Mummiform Cartonnage from the Greco-Roman Period in Cairo Museum. Mediterr. Archaeol. Ar. 16, 127–137 (2016).
Tite, M. S., Bimson, M. & Meeks, N. D. Technological Characterisation of Egyptian Blue. Arch.éoSciences, Rev. d.’Arch.éom.étrie 1, 297–301 (1981).
Afifi, H. A. Analytical Investigation of Pigments, Grounds Layer, and Media of Cartonnage Fragments from Greek Roman Period. J. Mediterr. Archaeol. 11, 91–98 (2011).
Ragai, J. Colour: Its Significance and Production in Ancient Egypt. Endeavour 10, 74–79 (1986).
Salim, E. & Abouseif, N. R. Multi-Analtical Characterisation of an Ancient Cartonnage Fragment from the American University Excavations: A Case Study. Int. J. Conserv. Sci. 13, 1006–1018 (2023).
Abd El Aal, S. Characterisation and Examination of Pigments, Grounds, and Media from Ancient Egyptian Cartonnage. EJARS 4, 35–46 (2014).
Gard, F. S. et al. Pigments Analysis of an Egyptian Cartonnage by Means of XPS and Raman Spectroscopy. Appl. Phys. A. 126, 1–12 (2020).
Vandenabeele, P. et al. Spectroscopic Examination of two Egyptian Masks: A Combined Method Approach. Anal. Lett. 33, 3315–3332 (2000).
Corbeil, M. C. & Helwig, K. An Occurrence of Pararealgar as an Original or Altered Artists’ Pigment. Stud. Conserv. 40, 133–138 (1995).
Sayed, H., Sadek, H., Abdel-Aziz, M. S., Mahmoud, N. & Radi, R. The Use of Miscellaneous Scientific Methods in Characterisation and Analysing the Gilding Cartonnage Tape for the Late Period. Egypt. J. Chem. 64, 2579–2592 (2021).
Abdrabou, A., Hussein, A. & Sultan, G. M. New Insights into a Polychrome Middle Kingdom Palette Applied to a Wooden Coffin: A Multidisciplinary Analytical Approach. J. Cult. Herit. 54, 118–129 (2022).
Edwards, H. G., Jorge-Villar, S. E. & Eremin, K. A. Raman Spectroscopic Analysis of Pigments from Dynastic Egyptian Funerary Artefacts. J. Raman Spectrosc. 35, 786–795 (2004).
Teutonico, J. M. A Laboratory Manual for Architectural Conservators. Rome: ICCROM; (1988).
Wachowiak, M. J. Efficient New Methods for Embedding Paint and Varnish Samples for Microscopy. JAIC 43, 205–226 (2004).
Ullrich, W. Cross-Section Analysis of Paint Layers–Materials, Methodology, and Examples, Int. J. Cult. Prop. 4, 49–56 (2008).
Derrik, R., Stulik, D. & Landy, M. Infrared Spectroscopy in Conservation Science. Los Angeles: The Getty Conservation Institute (1999).
IRUG Spectral Database: http://www.irug.org/search-spectral-database?reset=Reset.
Acknowledgements
The authors are grateful to Prof. Dr. Omar Mohamed Abdel Aziz, Department of Geology, Faculty of Science, Minia University, for his assistance with the light microscope examination. We also thank Prof. Dr. Hamdy Farghaly, Department of Physics, Faculty of Science, Minia University, Egypt, for the collaboration.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
A.M.H. led the overall manuscript and methodology, conducted analysis on the samples, result interpretation and drafted the paper. A.Y.E. conducted the archaeological analysis of the mask, extracted the samples for analysis and revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hussein, A.M., Ewais, A.Y. An integrated scientific analysis of a Graeco-Roman Cartonnage mask from Saqqara, Egypt. npj Herit. Sci. 13, 104 (2025). https://doi.org/10.1038/s40494-025-01655-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s40494-025-01655-5
This article is cited by
-
Study of Ink stick unearthed from the tomb of Fan Xiaochong in Shaanxi Province
npj Heritage Science (2025)