Abstract
The study investigates acetic salt efflorescence, a common issue in museums that damages artifacts exposed to acetic acid vapors. White efflorescent crystals on two Han Dynasty Eaves Tiles from the National Museum of China were analyzed using optical microscopy, SEM, Raman spectroscopy, and FTIR. The analysis identified thecotrichite (Ca₃(CH₃COO)₃Cl(NO₃)₂·6H₂O), formed through the reaction of acetic acid with residual chlorides, nitrates, and calcareous components in the tiles. A tailored desalination approach was employed: the cracked “Hanbingtianxia” tile was treated with a poultice method, while the intact “Changlewuji” tile underwent static water immersion. The process was meticulously monitored using conductivity measurements and ion chromatography to ensure optimal results. Acid-free storage cartons and humidity control were implemented to prevent efflorescence recurrence, confirming the success of the preservation strategies.
Similar content being viewed by others
Introduction
Acetic salt efflorescence is derived from the reaction of acetic acid with residual chlorides, nitrates et al in calcareous heritage objects, as well as with sodium, carbonates, and similar residues in metal objects. This phenomenon is frequently observed in museums, where prolonged exposure to organic acid vapors leads to the deterioration of sensitive artifacts. The most prevalent source of organic acids is wood and other organic materials used in the construction of display and storage cases, such as wood and wood products, adhesives, sealants, paints, polyester and rubber, which can off-gas acetic and formic acid and so on1,2. In particular, acetic acid is known to emit from all natural woods with hardwoods, e.g. oak, being thought to emit the highest concentrations of acetic acid2,3,4,5, with as much as 7% of the original weight of wood being released as acetic acid vapor over a period of 2 years at 48 °C6. The source of acetic acid is, in part, due to the hydrolysis of acetyl group esters in the hemicellulose7. Oak is now generally avoided as a material for museum furniture, however other woods such as mahogany, have been reported to be innocuous and are still recommended for museum use8. The acetate-based efflorescence is often found on susceptible items held in museum enclosures contaminated with acetic acid vapors.
Moreover, acetate compounds that have been identified on various materials in previous years include those found on calcareous substrates, copper, copper alloy objects, and litharge (PbO) pigments. Examples of such compounds include calclacite (Ca(CH3COO)Cl·5H2O)9,10, Ca2(CH3COO)(HCOO)(NO3)2·4H2O11, Ca2(CH3COO)3(NO3)·2H2O12,13, thecotrichite12,13,14,15,16,17, calcium acetate hemihydrate (Ca(CH3COO)2·0.5H2O)10,18,19, calcium acetate formate (Ca(CH3COO)(CHOO)·H2O)19,20, calcium acetate hydrate (Ca(CH3COO)2·H2O)19, Ca3(CH3COO)4(HCOO)2·4H2O)20; sodium copper formate21, sodium acetate hydrate, sodium copper carbonate acetate21, lead formate22 and so on.
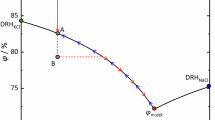
The formation mechanisms of efflorescent salts are complex and depend on the nature of soluble salts within the pores, or surfaces of the objects. Taking acetates as an example, solutions of acetic acid react with alkalis, oxides, hydroxides, carbonates, and electropositive metals to form acetates. Regardless of the salt combinations present, crystallization occurs when a supersaturated solution of the salt exists and the relative humidity (RH) of the surrounding environment supports solid-phase formation. For instance, analysis of thecotrichite’s phase diagram (Fig. 1)12 reveals why it is such an incongruent product: the addition of even a small amount of acetate to a solution saturated with calcium, nitrate, and chloride ions rapidly shifts the system into the stability field of thecotrichite (Ca3(CH3COO)3Cl(NO3)2·6H2O). With a deliquescent point in the range of 80–90% RH12, thecotrichite will invariably precipitate in museum display or storage environments maintained at 50–60% RH. Consequently, due to the water-soluble nature of these salts, the repeated cycle of deliquescence and crystallization can exert significant stress within the pores of the object, ultimately leading to its disintegration.
Solid phases corresponding to the stability fields are: (1) CaCl2·6H2O, (2) CaCl2·4H2O, (3) CaCl(NO3)·2H2O, (4) Ca(NO3)2·3H2O, (5) Ca(NO3)2·4H2O, (6) Ca(CH3COO)(NO3)·3H2O, (7) Ca2(CH3COO)3(NO3)·2H2O, (8) Ca(CH3COO)2·H2O, (9) Ca(CH3COO)CI·5H2O, (10) Ca3(CH3COO)3CI(NO3)2·6H2O12
In this study, we identified needle-like, white salt efflorescence on two Han Dynasty Eaves Tiles collected in the National Museum of China, both of which were stored in wooden cabinets. The crystalline composition and nature of the efflorescence were determined through direct sampling and analysis using Raman spectroscopy, FTIR spectroscopy, optical microscopy and SEM. And desalination processes, including poulticing and immersion in static water, were applied to the two eaves tiles respectively, and their efficacy was thoroughly evaluated and discussed. Additionally, the formation of the salt efflorescence will be discussed, and recommendations for the preservation of calcareous materials were proposed.
Methods
Materials
Two samples of white efflorescence crystal were collected from two different objects, which were stored in National Museum of China. The first object is a Han Dynasty eaves tile named as Hanbingtianxia (汉并天下). It was donated to National Museum of China since 2001. Before that, it was excavated from the Han Dynasty Great Wall Site in Xi’an Shaanxi, China. A crack on the Hanbingtianxia artifact, which had been repaired using adhesives and subsequently recolored following reattachment, displays the formation of white efflorescence crystals with a distinct needle-like morphology (Fig. 2a–c). The second artifact is a Han Dynasty eaves tile named Changlewuji (长乐无极), which has been in the collection since its donation in 1949. Needle-like crystals were observed on the bottom surface of the eaves tile, intermixed with traces of soil (Fig. 2d–f).
a–c Eaves tiles named as Hangbingtianxia, 16 cm in diameter, 4.2 cm of thickness, 863 g, with needle like crystals growing on the crack, which has been repaired with adhesives. d–f Eaves tiles named as Changlewuji, 18.2 cm in diameter, high 54.7 cm, 3925 g, with needle like crystals growing at the bottom mixed with some soil.
Characterization
The white efflorescence crystals were analyzed using a combination of advanced techniques. Scanning electron microscopy (SEM) and energy dispersive X-ray spectroscopy (EDS) were performed using a Phenom XL instrument (Netherlands) at an accelerating voltage of 15 kV. Optical characterization was conducted with a Keyence VHX-2000 digital microscope (Japan) at magnifications of 20–200× and 100–1000×. Micro-Raman spectroscopy was carried out using a LabRAM HR Evolution spectrometer (HORIBA, France) equipped with a 473 nm laser, maintaining a power below 400 μW on the sample surface. Fourier transform infrared (FTIR) spectra were acquired with a Thermo Fisher® Nicolet iS50 spectrometer (USA) in Attenuated Total Reflection (ATR) mode, where samples were pressed onto a diamond ATR crystal for optimal contact. Spectra were recorded in the range of 4000–400 cm⁻¹ with 32 scans at a resolution of 4 cm⁻¹.
Desalination procedures and effectiveness evaluation
Two desalination procedures were employed for the treatment of the Han Dynasty Eaves Tiles, with the cracked “Hanbingtianxia” tile treated using a poultice method and the intact “Changlewuji” tile subjected to static water immersion.
For the “Hanbingtianxia” tile, a poulticing method was used. A commercially available desalting pulp (BioLine® DP-02, pH 8.0 ± 0.5, density 0.85–0.90 g/cm3) from Shanghai Desaibao Building Materials Co., Ltd. (上海德赛堡建筑材料有限公司), was applied. This pulp, composed of natural wood fibers and compliant with the WTA “4-4-04/D” standard, exhibits high porosity, low shrinkage, and a large internal surface area. Firstly, prewet the object using water sprays, and the wet poultice material was applied to the surface of the object; Then it was maintained in place for a specified duration, typically ranging from one to five days, prior to its removal; Take a certain area of poultice, and measure the conductivity (DDSJ-308F, SCRPF, China) and ion concentration (Aquion-1200, Thermo Fisher, USA) of the supernate by soaking the taken dried poultice in the deionized water for ~3 days; Renew the poultice until the measured conductivity no longer changed, the object was removed and air-dried (Fig. 3). The entire procedure was conducted under ambient temperature conditions.
The desalination procedure of poulticing.
For the “Changlewuji” tile, immersion in static deionized water (37.8 L) at ambient temperature was conducted. The tile was submerged for 3–5 days, allowing soluble salts to dissolve into the water. The bath water was replaced with clean water; Extraction of salts in bath was monitored by conductivity and ion concentrations; Once the conductivity readings stabilize and no further changes are observed, the object is removed from the bath and air-dried. Both methods were performed under ambient temperature conditions, ensuring effective and controlled salt extraction.
Ion chromatography was performed using an Aquion-1200 system (Thermo Fisher, USA) equipped with a conductivity detector, an IonPac AS11-HC 4 × 250 mm analytical column, an IonPac AG11-HC 4 × 250 mm guard column, and a Dionex anion electrolytically regenerated suppressor (AERS 300). The eluent used was potassium hydroxide with a gradient ranging from 2 to 30 mM at a flow rate of 1.2 mL·min−1 over a runtime of 35 minutes. Eluent concentrations were established using a RFC-30 eluent generator cartridge. This setup allows for the analysis of fluoride, acetate, formate, chloride, nitrite, bromide, nitrate, sulfate and phosphate ions in concentrations below parts per million levels.
Results
Accurate identification of salt efflorescence
Firstly, the microscope (Fig. 4a, b) and SEM (Fig. 4c, d), SEM-EDS (Fig. 4e–g) were analyzed. The similar, needle like, efflorescence phase was found on the two eaves tiles, that was visible with the naked eye or using an optical (Fig. 4a, b) or SEM employing a low magnification (Fig. 4c, d). And according the SEM-EDS semi-quantative analysis (Table 1), both the needle like samples in region 1 and region 2 (Fig. 4c, d) was primarily composed of Ca, N, Cl, O, C elements, with calcium being the main cation detected. Calcium and chloride were found approximately with a ratio of 3:1. But due to the limitation of the EDS method, the given content of C, N, O as a light element rather represents a rough estimate than an exact determination. What’s more, the sample in region 3 shows a clay like impurities containing elements such as Ca, Si, Al, Mg, K, Na, O, N and C.
a-b The microphotographs of white efflorescence crystals from Hanbingtianxia (a), and Changlewuji (b); c-d SEM-BSE images of white efflorescence crystals from Hanbingtianxia (c) and Changlewuji (d); e-g The energy spectra in the region 1 (e), region 2 (f) and region 3 (g).
The structure of the white needle like efflorescence crystals on the two tiles was investigated using both μ-Raman spectroscopy and FTIR spectroscopy. The complete Raman spectra are presented in Fig. 3a, with tentative band assignments provided in Table 2. In the low Raman shift region, or the fingerprint region (500–1500 cm⁻¹), the spectra are dominated by bands attributable to acetate and nitrate groups. In the high Raman shift region (2800–3600 cm⁻¹), several Raman bands corresponding to C–H stretching vibrations (2900–3050 cm⁻¹) and low-intensity bands associated with O–H vibrations (centered at 3372 and 3470 cm⁻¹) are observed. These vibrational modes are consistent with the presence of acetate, nitrate, and hydrate water, aligning with spectra reported in a poster presented at IRUG 723 and thecotrichite identified on a glazed tile from the collection of Landesmuseum Württemberg24.
Additionally, FTIR spectroscopy in ATR mode (Fig. 5b and Table 3) was employed, providing further confirmation of the thecotrichite structure within the white efflorescence25. The relatively small splitting (Δν ≈ 170 cm⁻¹) between the most intense symmetrical (Tables 3, band 3) and antisymmetrical (Table 3, band 5) carboxylate-related C-O stretching modes suggests that the majority of the acetate anions exhibit bidentate and bridging coordination behavior20,26.
The structure of thecotrichite in Hanbingtianxia has been validated by XRD spectra in previous literature27. In addition, the efflorescence contains minor impurities, including small amounts of soluble salts such as NaCl, as well as insoluble salts like calcite (CaCO₃), quartz, and trace quantities of albite and microcline (K(AlSi₃)O₈). These findings are consistent with the results obtained from SEM-EDS analysis (Table 1). Specifically, the Ca:Cl ratio exceeding 3 indicates the presence of calcite, whereas an N:Cl ratio of less than 2 signifies the existence of NaCl.
Desalination of eaves tiles
From the phase diagram of thecotrichite (Fig. 1)12, it is evident that a complex quaternary system comprising one cation (calcium) and three anions (chloride, nitrate, and acetate) contributes to its intricate crystallization-deliquescence behavior due to its water-soluble nature. This cycle of deliquescence and crystallization can generate significant stress within the pores of the object, ultimately leading to its disintegration. Therefore, the extraction of these efflorescent salts and the prevention of thecotrichite regrowth are critically urgent tasks. As we know, for thecotrichite to form, soluble chloride and nitrate salt contaminants must be present as well as volatile acetic acid. So, extracting soluble chlorides and nitrates, along with preventing the release of acetic acid, is an effective approach to addressing these issues.
Given the presence of cracks, evidence of prior repairs, and a recolored layer on Hanbingtianxia, the poulticing method was chosen for desalination for Hanbingtianxia eaves tile, as it provides a significantly gentler and more controlled approach. On the other hand, for the structurally intact and unbroken Changlewuji eaves tile, immersion in static water was selected as the optimal desalination technique.
Prolonged desalination can cause the surface of low-temperature pottery materials to soften and deteriorate due to prolonged contact with water. Therefore, the shorter the desalination process, the lower the risk of material degradation. To determine the endpoint for desalination, the conductivity of the soaking solution or the supernatant obtained from soaking the dried poultice was measured. Since conductivity is positively correlated with the amount of salt dissolved from the sample, a higher conductivity indicates a greater concentration of dissolved salts.
As for the desalination of Hanbingtianxia, the conductivity of poultice is measured by the following procedure: Take a certain area of poultice, and dried it for about 3 days. Soak the dried poultice in the deionized water (35 ml) for ~3 days to dissolve the salt, measure the conductivity of the supernate.
The adjusted conductivity was calculated by the resulting Eq. (1):
where k represents the conductivity in μS·cm⁻¹, and M is the weight of the dried poultice in grams. Since the volume of water used for soaking the dried poultice was consistently 35 ml, it was not factored into the formula. The term ‘kadj’, or “adjusted conductivity,” is a numerical value that standardizes conductivity readings by accounting for the mass of the dried poultice involved. This Eq. (1) and the ‘kadj’ term allow for the direct comparison of conductivity measurements across different desalination scenarios by normalizing them with respect to the amount of poultice used. As illustrated in Fig. 6a, the desalination process continued over 18 days. From the first poultice change to the fifth poultice change, the slope of conductivity versus time gradually decreases, reflecting a progressive reduction in the desalination rate. The final conductivity measurement (5th) was recorded at 854.3 μS/(cm·g), which was significantly lower than that of the pure poultice (943 μS/(cm·g)), indicating a substantial reduction in salt content.
a The desalination process is poulticing for Hanbingtianxia, for example, 1st means that the first time to renew the poultice for the time interval of 1 day between poultice changes. b The desalination process is immersing in the deioned water for Changlewuji, for example, 5th means the fifth time to replace with the clean water for the time interval of 5 day between water changes (soaking at ambient temperature)
Regarding the Changlewuji, the desalination experiment involving immersion in static water was carried out over a period of 13 days, with the progress systematically monitored through conductivity measurements, as illustrated in Fig. 6b. Since the volume of water used for immersing the object remained constant at 37.8 L, it was not factored into the conductivity adjustment. Consequently, the conductivity readings from the soaking solution could be directly compared throughout the desalination process for this specific object. As shown in Fig. 6b, as the times of deionized water changes increases during the immersion experiment, the desalination rate gradually declines. Notably, the desalination rate in the second time is significantly higher than in the first. This phenomenon may be explained by the fact that the initial immersion primarily involves the gradual permeation of water into the Hanbingtianxia tile ends, whereas during the second immersion, the salts within the object begin to diffuse into the water more extensively. The conductivity rates of the water solution following the fourth and fifth water changes are comparable, with the conductivity values being closely aligned (34 μS/cm for the fourth and 43 μS/cm for the fifth). This suggests that the desalination experiment may be deemed complete, as performing additional desalination cycles is unlikely to yield a substantial additional reduction in salt content.
Then the effectiveness of the desalination process was subsequently evaluated by comparing the concentrations of chloride ions, nitrite ions, and nitrate ions throughout the desalination process using ion chromatography. The ion concentration was determined by analyzing the supernatant obtained after soaking the dried poultice (Hanbingtianxia) and the soaking solution (Changlewuji), respectively.
For Hanbingtianxia, the ion concentration was measured and calculated using the following Eq. (2):
where c represents the concentration in µg/ml, which is directly by ion chromatography, V is the volume of water used for soaking the dried poultice (35 ml), and M is the weight of the dried poultice in grams. The term cadj represents the mass of ion concentration per gram of dry poultice.
For Changlewuji, since the volume of water used for immersing the object remained constant at 37.8 L and the weight of the object was 3925 g, the ion concentrations were directly measured using ion chromatography without requiring additional calculations.
As shown in Fig. 7, the concentrations of acetate ions, chloride ions, nitrate ions, and sulfate ions in the Hanbingtianxia artifact were quantified using the formula mentioned earlier (Eq. (2)). The results indicate that the release rates of these ions exhibit a gradual decline with each successive poultice replacement, signifying a progressive reduction in the desalination rate over time. This trend aligns closely with the variations in conductivity illustrated in Fig. 6a. After the fifth poultice replacement, even following a five-day application period, the ion concentrations such as Cl−, NO3−, and SO42− within the poultice supernate showed no change, confirming that the poultice desalination experiment has reached its terminal point. By this stage, all the ion concentrations had decreased to a very low level.
a Acetate ions, b Cl−, c NO3−, d SO42−
For Changlewuji eaves tile (Fig. 8), the concentrations of chloride ions, nitrate ions, and sulfate ions were measured, revealing a consistent decline in their release rates with each successive water replacement. Notably, the ion release rate during the second replacement was significantly higher than during the first. This phenomenon can be attributed to the fact that the initial water replacement primarily facilitated the permeation of water into the object, whereas during the second immersion, the salts within the object began to diffuse into the water more extensively. Unlike Hanbingtianxia, no acetate ions were detected in Changlewuji. This absence is likely due to the fact that acetic acid is predominantly emitted from wooden materials in the storage environment rather than originating from the objects themselves. Furthermore, any externally derived acetic acid had already transformed into salt efflorescence and was effectively removed through physical cleaning prior to the desalination treatment. In contrast, the detection of trace amounts of residual acetate ions in the Hanbingtianxia artifacts suggests that these ions are intrinsic to the objects. This finding implies that the residual acetate ions may not solely originate from wooden materials in the storage environment but could also result from the degradation of adhesives used in previous restoration efforts, which gradually release acetic acid vapor over time.
a Cl−, b NO3−, c SO42−
In summary, ion chromatography analysis demonstrated a significant reduction in the concentrations of chloride ions, nitrate ions, sulfate ions and acetate ions, which decreased to baseline levels. This result underscores the high efficacy of the desalination process.
Discussion
The calcium source was further discussed. The objects of eaves tiles were composed of quartz (SiO2), feldspar ((Na,Ca)Al(Si,Al)3O8), plagioclase(K(AlSi3)O8)27, there. Regarding the source of the thecotrichite components, calcium undoubtedly derives from the calcareous component of the object (eaves tiles) as either calcareous clay or calcite inclusions or both. In addition, other insoluble effloresce such as calcite (CaCO₃)27, also serve as one of the sources of calcium.
The source of acetate was also investigated. The eaves tiles were stored in collections for several decades. For such objects, the treatment on the excavation site and later in the museums is nearly never documented. Acetate is known to result from the reaction of acetic acid emissions from wooden storage materials with the objects, and the presence of soluble salts in the pores and on the surface of the ceramic encourages the reaction12. The acetic acid in other wooden storage cases in National Museum of China were identified in which acetate efflorescence has been identified on potteries28. So, we inferred the source of acetic acid of Changlewuji is probably wood storage cabinet. While, the salt efflorescences on the Hanbingtianxia artifacts are primarily observed along cracks containing adhesive materials and their surrounding areas. Ion chromatography analysis detected acetate ions in these regions, confirming that the sources of acetic acid emissions are not limited to the wooden storage cabinet but are significantly attributed to the degradation of adhesive compounds29 used in previous restoration efforts.
For the source of nitrate and chloride, we propose that chloride may derive from saline soil during burial10. Nitrate can be formed in the soil by nitrifying bacteria but is very mobile11 or from the atmospheric dust30. Groundwater chlorides/nitrates31 can accumulate through continuous infiltration of water and evaporation, reaching salt concentrations greater than that found during this isolated test and sufficient to contribute to the formation of thecotrichite.
Finally, we elucidate the formation pathways of Ca₃(CH₃COO)₃Cl(NO₃)₂·6H₂O crystallites on calcareous heritage surfaces. The highly soluble calcium nitrate and calcium chloride were in the object body that reacted with volatile acetic acid emissions forming crystallization of thecotrichite on the surface of the eaves tiles13. At typical museum conditions (about 55% RH) thecotrichite can crystallize out while the hygroscopic salts, calcium nitrate, and calcium chloride in the pores promote further dissolution of acetic acid, serving as a reservoir for more thecotrichite crystallization and continued deterioration of the object. Thecotrichite derives from a complex quaternary system consisting of one cation (calcium) and three anions (chloride, nitrate, and acetate) that contribute to its complicated crystallization deliquescence behavior. A phase diagram has been developed for thecotrichite that may be used to determine which solid phase or phases ‘would precipitate out from a mixed solution of any given composition12. However, controlling the relative humidity (RH) is not a straightforward task, as hygroscopic components such as calcium chloride and calcium nitrate remain active even at RH levels as low as 20%12. On the other hand, in the absence of chloride ions and nitrate residues in these artifacts, the formation of acetate salts can be effectively prevented under suitable humidity and temperature conditions12.
As a summary, the needle like efflorescence was found on two eaves tiles in the National Museum of China, which was identified as acetic salts mainly composed with thecotrichite (Ca3(CH3COO)3Cl(NO3)2·6H2O). It derived from the reaction of acetic acid with residual chloride, nitrate and calcareous component in the eaves tiles. The primary source of acetic acid was traced to the wooden storage cabinet, while the spatial distribution of efflorescence patterns, particularly along cracks containing adhesive materials, suggested that the adhesive compounds might represent a secondary acetic acid source. Based on comprehensive condition assessments, tailored desalination protocols were implemented, incorporating both poulticing techniques and static water immersion methods. The desalination process was meticulously monitored through conductivity measurements and ion chromatography analysis, ensuring optimal extraction efficiency. To establish long-term preservation conditions, acid-free storage cartons and relative humidity controlling (a stable relative humidity of 50% and room temperature) were fabricated. Over a twelve-month monitoring period, this conservation strategy proved highly effective, as no reformation of crystalline deposits was observed, demonstrating the long-term success of the measures taken. This study contributes to the scientific understanding and treatment of crystalline salt deterioration in calcareous collections, while providing practical recommendations for the conservation of calcium-based cultural heritage materials.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request. The data analysis in this study can be obtained from the corresponding author upon reasonable request.
References
Gibson, L. T. & Watt, C. M. Acetic and formic acids emitted from wood samples and their effect on selected materials in museum environments. Corros. Sci. 52, 172–178 (2010).
Arni, P. C., Cochrane, C. G. & Gray, J. D. The emission of corrosive vapours by wood. I. Survey of the acid-release properties of certain freshly felled hardwoods and softwoods. J. Appl. Chem. 15, 305–313 (1965).
Arni, P. C., Cochrane, C. G. & Gray, J. D. The emission of corrosive vapours by wood. II. The analysis of the vapours emitted by certain freshly felled hardwoods and softwoods by gas chromato-graphy and spectrophotometry. J. Appl. Chem 15, 463–468 (1965).
Farmer, R. H. Corrosion of metals in association with wood. Part 2: corrosion by acidic vapours from wood. Wood 27, 326–328 (1962).
Farmer, R. H. Corrosion of metals in association with wood. Part 2: corrosion of metals in contact with wood. Wood 27, 443–446 (1962).
Budd, M. K. Corrosion of metals in association with wood. Appl. Mater. Res. 124, 125 (1965).
Farmer, R. H. Chemistry in the utilization of wood (Pergamon Press, 1967).
Shen, J., Shen, Y., Xu, F., Zhou, X. & Wu, L. Evaluating the suitability of museum storage or display materials for the conservation of metal objects: a study on the conformance between the deposited metal film method and the Oddy test. Environ. Sci. Pollut. Res. 25, 35109–35129 (2018).
Van Tassel, R. On the crystallography of calclacite, Ca(CH3COO)Cl.5H2O. Acta Crystallogr. 11, 745–746 (1958).
Boccia Paterakis, A. & Steiger, M. Salt efflorescence on pottery in the Athenian Agora: a closer look. Stud. Conserv. 60, 172–184 (2015). 2015; 60: 172-184.
Bette, S., Eggert, G., Fischer, A., Stelzner, J. & Dinnebier, R. E. Characterization of a new efflorescence salt on calcareous historic objects stored in wood cabinets: Ca2(CH3COO)(HCOO)(NO3)2·4H2O. Corros. Sci. 132, 68–78 (2018).
Gibson, L. T. et al. The mode of formation of thecotrichite, a widespread calcium acetate chloride nitrate efflorescence. Stud. Conserv. 50, 284–294 (2005).
Linnow, K., Halsberghe, L. & Steiger, M. Analysis of calcium acetate efflorescences formed on ceramic tiles in a museum environment. J. Cult. Herit. 8, 44–52 (2007).
Gibson, L. T., Cooksey, B. G., Littlejohn, D. & Tennent, N. H. Characterisation of an unusual crystalline efflorescence on an Egyptian limestone relief. Anal. Chim. Acta. 337, 151–164 (1997).
Bette, S., Eggert, G. & Dinnebier, R. E. The variety of calcium bearing efflorescence phases - an explanation by crystal chemistry. Acta Crystallogr. Sect. A. 7, C540–C541 (2021).
Dinnebier, R. E., Fischer, A., Eggert, G., Runčevski, T. & Wahlberg, N. X-ray powder diffraction in conservation science: towards routine crystal structure determination of corrosion products on heritage art objects. J. Vis. Exp. 112, 54109 (2016).
Halsberghe, L., Gibson, L. T. & Erhardt, D. A collection of ceramics damaged by acetate salts: conservation and investigation into the causes. In Preprints of the 14th Triennial Meeting ICOM Committee for Conservation, 1, 131–138 (The Hague, 2005).
Paterakis, A. B. The influence of conservation treatments and environmental storage factors on corrosion of copper alloys in the ancient Athenian Agora. J. Am. Inst. Conserv. 42, 313–339 (2003).
Tennent, N. H. & Baird, T. The deterioration of mollusca collections: identification of shell efflorescence. Stud. Conserv. 30, 73–85 (1985).
Bette, S., Müller, M. X., Eggert, G., Schleid, T. & Dinnebier, R. E. Efflorescence on calcareous objects in museums: crystallisation, phase characterisation and crystal structures of calcium acetate formate phases. Dalton Trans. 48, 16062–16073 (2019).
Eggert, G. & Fischer, A. Curious corrosion compounds caused by contact: a review of glass-induced metal corrosion on museum exhibits (GIMME). Corros. Mater. Degrad. 3, 553–565 (2022).
Švarcová, S. et al. Uncovering lead formate crystallization in oil-based paintings. Dalton Trans 49, 5044–5054 (2020).
Schönemann, A., Kutzke, H., Lendl, B. & Eggert, G. Combined Raman and FTIR study on thecotrichite and related efflorescence. In IRUG 7: the seventh biennial gathering of the Infrared and Raman User’s Group, 88–93 (2006).
Wahlberg, N. et al. Crystal structure of thecotrichite, an efflorescent salt on calcareous objects stored in wooden cabinets. Cryst. Growth Des. 15, 2795–2800 (2015).
Eggert, G. et al. Efflorescence X? Case solved: Ca3(CH3COO)3Cl(NO3)2·6H2O! The research history, identification, and crystal structure of thecotrichite. In ICOM Committee for Conservation: Glass and Ceramics Working Group Interim Meeting, 135–144 (ICOM, 2016).
Bette, S. et al. Crystal structure, polymorphism, and anisotropic thermal expansion of α-Ca(CH3COO)2. Cryst. Growth Des. 20, 5346–5355 (2020).
Wu, N., Wang, K., Yan, D. & Lei, L. Spectrometric analysis of crystalline efflorescence in one Han Dynasty Eaves Tile. J. Light Scatt. 34, 95–102 (2022). (In Chinese).
Li, M., Yang, Q., Wu, N., Ding, L. & Yan, D. Desalting protection of painted pottery drum collection in Neolithic period. Conservation and Restoration Reports on Cultural Relics from the National Museum of China. 36–44 (2019). (In Chinese).
Tetreault, J. Adhesive testing at the Canadian conservation institute: an evaluation of selected poly(vinyl acetate) and acrylic adhesives. Stud. Conserv. 41, 19–44 (1996).
Karydis, V. A., Tsimpidi, A. P., Pozzer, A., Astitha, M. & Lelieveld, J. Effects of mineral dust on global atmospheric nitrate concentrations. Atmos. Chem. Phys. 16, 1491–1509 (2016).
Dun, Y., Tang, C. & Shen, Y. Identifying interactions between river water and groundwater in the North China Plain using multiple tracers. Environ. Earth Sci. 72, 99–110 (2014).
Acknowledgements
This work was supported by the Cultural Relics Science and Technology Research Project of National Cultural Heritage Administration (self-funded) (2023ZCK003) and the author is indebted to associate research librarian Na Wu (National Museum of China) for her help in the laboratory tests and helpful advice.
Author information
Authors and Affiliations
Contributions
Na Zhao: Conceptualization, Methodology, Validation, Investigation, Data curation, Writing - original draft, Writing - review & editing, Visualization.Ran Zhang: Conceptualization, Methodology and Writing - review & editing and Funding acquisition. Lei Lei: Dating methodology, data collection and analysis. Qin Yang: Sampling and data collection, Writing - review & editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, N., Zhang, R., Lei, L. et al. Accurate identification and extraction of acetate salts on Han Dynasty Eaves Tiles in museum environment. npj Herit. Sci. 13, 334 (2025). https://doi.org/10.1038/s40494-025-01885-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s40494-025-01885-7