Abstract
Goldwork is central to Akan cultural heritage and was a key commodity in European trade along the West African coast for centuries. Previous assessments of gold quality have relied mainly on speculative, biased and largely unquantified European written sources. Here, we present elemental composition data of Akan gold artifacts recovered from the wreck of the pirate shipwreck the Whydah Gally (1717), obtained using portable X-ray fluorescence (pXRF) and Scanning Electron Microscopy (SEM-EDS). The results are compared with contemporary written accounts and published compositional data from gold deposits in Ghana’s Ashanti Gold Belt. The observed elemental patterns do not support claims of systematic heavy adulteration of Akan gold; instead, most compositions fall within the natural variability of West African gold. However, due to variability in the ore data the applied methods cannot conclusively identify intentional additions of silver or copper, highlighting the need for fully quantitative analytical approaches.
Similar content being viewed by others
Introduction
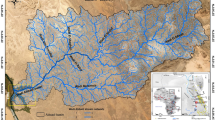
For over a millennium, West African gold has been a coveted commodity, initially as a key component of the trans-Saharan trade and later as a focus of European maritime trade. West African gold was likely reaching the Mediterranean world by the middle of the first millennium CE. The details of the trade and the sources of the gold involved are poorly known, as both documentary insights and archeological data are limited1,2,3,4,5,6,7,8,9,10,11,12. It is, however, likely that the trans-Saharan trade first exploited the Bouré and Bambuk gold fields in portions of what are now the countries of Guinea, Senegal and Mali. With the opening of maritime trade in the 15th century, Europeans focused on the Gold Coast—modern-day Ghana and the deposits of the Ashanti Gold Belt. The Ashanti Gold Belt is a series of Paleoproterozoic Birimian volcanic deposits that extend 300 km from the Ghanaian coast west of Takoradi, northeast to Konongo in the Ashanti Region (Fig. 1). It includes the locations of the principal modern mines around the towns of Konongo, Obuasi, Mampon, Bogosu and Prestea13,14,15.
The Ashanti Gold Belt is highlighted in tan, and the major mines are indicated: 1 = Konongo, 2 = Obuasi, 3 = Mampon, 4 = Bogosu, 5 = Prestea. The principal British forts of the early eighteenth century that the Whydah Gally may have visited are highlighted in red. Elmina, the Dutch headquarters in West Africa, is highlighted in blue.
The Portuguese were trading on the Ghanaian coast by the 1470s, and in 1482 founded Castelo de São Jorge da Mina—now known as Elmina Castle—specifically to secure the gold trade with the local Akan peoples5,7,16,17,18. In the following centuries, dozens of castles, forts and trading lodges were established by the Dutch, English, Swedes, Danes, Brandenburgers, and French along the 550-kilometer-long coastline to trade for gold, other commodities, and enslaved Africans19,20,21,22,23,24 (Fig. 1). While aspects of African-European trade are well documented and contemporary accounts provide descriptions of early Akan gold working, these sources reveal little regarding the composition of the gold traded and its relative purity11,25,26,27,28. Africanists have long underscored that early accounts are not neutral and the need for historians, archeologists, and anthropologists to critically evaluate these sources29,30,31,32,33,34,35,36. Some sources present biased and unsubstantiated perspectives, while others simply copy the work of earlier writers without reference.
A recurring theme in early European sources is the view that Akan traders often passed off adulterated materials of lesser value as high-karat gold. Writing in the early 17th century, Pieter de Marees stated that the Akan learned to fuse gold with silver from the Portuguese, and subsequently grossly debased the gold traded37. He further suggested ways “to guard against fraud and adulteration of the gold”. He urged traders to be sure the gold dust was not mixed with sand, and to cut or hammer gold nuggets to ensure their interiors were not hiding iron, stone or other materials. Kracaws (variously krakaws, kacorawns, krakras, crackra, kakra) used as medium of exchange on the coast, particularly around Elmina, was said to consist of brass, silver, and copper filings mixed with gold, at times being little more than brass25,26,28,37,38,39,40,41,42,43.
In the early 18th century, William Bosman further stated that nuggets of Akan gold were often an alloy of silver and copper, with only a portion of gold, while the gold dust traded might include copper or powdered glass26. Records of the Geoctrooieerde Westindische Compagnie (Dutch West India Company or WIC) express repeated concerns regarding the quality of the kracaws-gold42. Similar comments are made by writers through the 19th century5,44,45,46,47,48,49,50. Akan metal smiths worked in silver, copper, brass, and varied alloys, confirming both their access to raw materials and the ability to work them51,52,53. Considering the scarcity of silver deposits in West Africa, some contemporary sources suggested that European silver coins were the main source of silver on the Guinea coast25,26,27,28,51,54.
While Europeans were undoubtedly concerned with the quality of the gold traded, accounts of adulterated Akan gold cannot be taken at face value. European traders and African merchants alike would likely have carefully monitored the quality of the gold. Yet European accounts generally provide no substantive analyses, and accounts of adulterated gold may be based more on speculation and secondhand accounts than direct evidence55.Gold could be tested in varied ways, including physical examination for impurities and chemical tests such as aqua regia37,56,57,58. The claims of adulteration pose a historiographical problem that obscures assessment of the Akan gold trade. Scientific analysis of archaeological finds provides an unbiased approach and can shed light on the composition of Akan gold and Akan metallurgy.
A unique opportunity to assess the quality and composition of Akan gold is provided by over 300 items recovered from the wreck of the Whydah Gally, which sank off the coast of Massachusetts, near Cape Cod, on April 26th, 171759,60. Popularly simply referred to as the Whydah, the ship’s route can be partially traced through documentary records. The ship departed England for West Africa in 1716, possibly trading on the coast from the Senegambia to the Bight of Benin, including the Gold Coast: modern day Ghana. The ship then sailed to Jamaica, where it was captured off Long Key in the central Bahamas by the notorious pirate Samuel “Black Sam” Bellamy, who subsequently made the Whydah his flag ship61. The ship capsized, broke up, and was scattered due to bad weather at midnight on April 26, 1717, near modern site of Marconi Beach at Wellfleet, Massachusetts.
The wreck of the Whydah was discovered in 1984 by a team led by Barry Clifford60,62. Work at the site is undertaken under a permit issued by the U.S. Army Corps of Engineers (USACE) to the Whydah Project. The artifacts recovered from the wreck are currently housed in the Whydah Pirate Museum in West Yarmouth, Massachusetts, and at Real Pirates in Salem, Massachusetts. The assemblage represents one of the largest integrated collections of artifacts from a single shipwreck61,63.
The Akan gold artifacts from the Whydah are the largest and most closely dated assemblage of Akan gold artifacts in the world64,65,66. Most Akan gold artifacts in collections are ethnographically or privately collected with limited context and cannot be closely dated5,11,43. Few have been recovered from well-dated archaeological contexts. The Whydah finds thus afford a unique opportunity to study the material composition of the gold that was offered to traders on the Gold Coast in the early 18th century.
The gold Akan artifacts from the wreck of the Whydah consist of over 300 individual pieces. The Asante sociocultural traditions and metalworking technology are among the best documented of the Akan groups. It is likely that some, perhaps most, of the fine gold work and casting in other metals traded on the Ghanaian coast between the 15th and the 19th centuries was produced in the Asante interior5,52. However, the exact sources of the Whydah pieces and the gold used to make them cannot be determined. Other Akan groups cast metals and multiple sources are likely represented. However, the intact pieces are stylistically Akan, as are the casting methods employed52,64,65,66,67,68,69. In this study, the purpose of comparing the data is not to source the gold but to assess the quality of the Whydah gold finds.
Most of the Whydah finds are small millimeter-sized beads, gold nuggets and fragments, but pieces of jewelry are also represented, many with typical Akan threadwork (Figs. 2, 3) designs produced using the cire perdue—lost wax—casting method. There is also a direct cast fragment of a gastropod shell. Only two dozen pieces are intact. Most cast pieces are fragmentary or damaged, having been sold to Europeans for their material value and not their artistry. Some of the pieces have secondary drilled perforations enabling them to be strung.
A cire perdue cast gold bead in situ is visible in the upper left of the photo. Flakes of gold dust and a silver coin (upper right) are also visible.
All of these were examined in the study.
The evidence of clipping, snapping, and hammering likely indicates the testing of the pieces for adulteration, as well as the division of pieces into smaller weights. European writers suggested striking gold objects with a hammer or cutting them with a knife to ensure the gold pieces did not include bits of iron, glass beads or other material26,37,66.
For the present study, 70 pieces were examined (S1 table sheet 1), 27 of which were randomly selected for elemental analyses (Table 1 and Fig. 4), including eight fragments of cast gold artifacts and fifteen gold lumps or nuggets. On macroscopic examination, another four pieces (5784/25, 5786/25, 5795/25 and 5799/25) had unique characteristics and their manufacture is designated as “unknown” to indicate it is not certain whether they are anthropogenic or natural. Analyses showed that these are likely natural except 5784/25. Cast artifacts were produced using cire perdue (lost-wax-casting) and, in one case, direct casting. Nuggets, as used here, refer to lumps of gold that appear to be natural formations, precipitated geologically. Analyses show that these pieces match the geological data for Ag/Au ratios (see “Discussion” below).
They were photographed with a Keyence VHX-1000 digital microscope. The scale in the bottom, right corners is 1 mm.
The samples examined here were mostly taken out of iron concretions (Fig. 2), and some surface contamination can be expected. Most samples show surface corrosion, appearing tarnished, while others have a brown-reddish patina possibly from being deposited 300 years under water.
Methods
All samples have been cleaned in an ultrasonic bath for 1 h prior to any analysis.
Portable X-ray fluorescence analysis (pXRF)
A Thermo Scientific Niton Gold XL3t portable energy dispersive X-ray fluorescence spectrometer (pXRF) was used to determine the elemental composition of the samples. After 45 min of warm-up, the program “TestAllgeo” was used for the measurements. Gold metal standards NA-Au-30 and NA-Au-31 of the Norddeutsche Affinerie (today: Aurubis) have been interposed as standard reference material after every three measurements. The pXRF was set at 50 keV, 8 mm aperture, and a measurement time of 120 s. Raw data have been visually inspected for obvious spectral artefacts and peak shifts and data have been reduced to Pb, Au, Zn, Cu, Ni, Fe, Sn, Ag and Pd.
In metal samples, pXRF can detect bulk element composition, while the analysis of trace element compositions requires analytical instruments with more precise detection limits and higher analytical resolution such as ICP-MS. Energy dispersive analysis using pXRF poses several interpretive problems, including the overlap of element peaks and misidentifications owing to the small areas assessed. In addition, pXRF assessment may be complicated by the surface contamination typical for archaeological samples. However, it provides (semi-)qualitative analysis to assess the bulk composition of main elements in-situ when objects cannot be brought to a laboratory. The non-destructive analysis pXRF also makes it more suitable in many settings, including the present study.
Scanning electron microscopy coupled with energy dispersive X-ray spectroscopy (SEM-EDS)
A Zeiss Supra 40VP scanning electron microscope (SEM) was used to identify clean, uncontaminated loci suitable for analysis. Energy-dispersive X-ray spectroscopy (EDS) was carried out using a Noran System 7 silicon drift detector. EDS measurements were performed under high-vacuum conditions. Analyses were performed with an accelerating voltage of 20 kV using a small condenser aperture (30 µm) in order to limit beam diameter and reduce radiation background in the Au-rich matrix. The instrument was operated primarily in backscattered electron detector (BSD) mode, in which image contrast correlates with average atomic number; phases enriched in high-Z elements (e.g., Au) appear brighter than those with lower atomic numbers (e.g., Cu). This imaging mode proved effective for identifying compositional variations as well as surface features such as patinas and corrosion products.
Measurement time was set to 60 s per analysis. For each sample, two to four different loci were analyzed. Each locus was analyzed using area analyses covering 100 × 100 µm squares, and mean values were calculated from the resulting datasets. Quantification was achieved using the Au M line, the Ag L line, and the Cu Kα line, applying the default ZAF-type matrix correction algorithm implemented in the Noran System 7 software. Calibration and analytical accuracy were checked using the same certified reference materials NA-Au-30 and NA-Au-31 as in the pXRF measurements (details see above).
Results
All analytical results are available in the supplementary material (S1 table sheet 2-6).
pXRF
The 27 analyzed gold samples are not homogenous in composition (Fig. 5). Amounts of Au range from 73.5 to 96.7 wt% with a mean of 87.5 wt%. Ag is the main impurity and is present in amounts from 0.04 to 22.6 wt-% with a mean of 7 wt%. Cu is variable too and ranges from 0.04 to 5.8 wt% but the mean value is around 0.8 wt%. Fe was detected in all samples ranging from 0.4 to 10.8 wt% with a mean of 3.5 wt%. The same applies for Pb, which ranges from 0.1 to 3.2 wt% with a mean of 1 wt%. Zn occurs in five samples in amounts between 0.1 and 2.3 wt%. Ni was detected in a single sample at 0.1 wt% and Sn was detected in six samples 0.1 to 1.6 wt%. Pd is present in three samples and is below 0.1 wt%.
Box and Whisker Plot of the results of the pXRF measurements of all 27 samples (data from S1 table sheet 2). Note the variability of Ag, Fe and also Pb and Cu.
Repeated pXRF measurements of the gold standard reference materials NA-Au-30 and NA-Au-31 (S1 Table, sheet 5) show high analytical reproducibility across the measurement series. For NA-Au-30, most elements are consistently measured at or below detection limits, indicating that Pb, Zn, Ni, Fe, and Pd could not be reliably quantified at these low concentrations in an Au-rich matrix. Silver, with a mean concentration of approximately 1.05 wt-%, is in good agreement with the certified value of 1.00 wt%.
For NA-Au-31, Ag concentrations seem to be reproducible, with a mean of approximately 5.01 wt%, but slightly lower than the reference value of 5.45 wt%. Copper (mean ≈ 0.18 wt%) and iron (mean ≈ 0.11 wt%) are consistently measured and broadly consistent with their certified values. In contrast, Ni and Pd are systematically underestimated relative to the reference concentrations, whereas Sn is overestimated.
SEM-EDS
Area analysis on virtually clean surfaces gave mean results of Au ranging from 70.3 to 100 wt%, Ag = 0–26.5 wt-% and Cu = 0–3.8 wt%. Fe ranges from 0 to 4.1 wt%, Pb from 0 to 1.4 wt% deviating from the pXRF measurements (Fig. 6). The individual surface measurements on the respective samples do not yield homogeneous results; fluctuations are apparent. BSD imaging revealed dark patches in depressions, crevices, and cracks on all the samples examined (Fig. 7). EDS analysis of four of these dark areas (Fig. 8) revealed them to be heterogeneous in composition. Fe is the most dominant ranging from (19.5–95.8 wt%) but also commonly Cu (0.8–12 wt-%), Ag (0–7 wt-%). Pb can be present up to 19 wt% or completely absent. The same applies to Sn (up to 20 wt%) and Zn (0–2 wt%) and Au (0–37 wt%).
They were carried out at clean areas for Ag, Cu, Pb and Fe in wt-%. Note the deviation of Pb and Fe, which are mostly higher in pXRF.
Four samples are shown: A = 5779/25 (cast), B = 5790/25 (nugget), C = 5792/25 (nugget), D = 5793/25(nugget). They all show dark patches in depressions, crevices, and cracks. Note threadwork in casting fragment in the upper left and notable flattening of the nuggets at bottom.
These appear in the BSD-imaging and were measured by SEM-EDS in samples 5779/25 (cast), 5780/25 (cast), 5782/25 (nugget) and 5783/25 (cast) (data in S1Table sheet 4).
SEM-EDS measurements of the gold standard reference materials NA-Au-30 and NA-Au-31 (S1 Table, sheet 6) show highly reproducible values for Au and Ag, with mean concentrations of 98.9 and 1.1 wt-% for NA-Au-30, and 94.7 and 5.3 wt-% for NA-Au-31, respectively. Copper was below the detection limit in all analyses, consistent with its very low certified concentrations. Overall, these results confirm the reliability of SEM-EDS for major element quantification in Au-rich samples, while elements present at trace levels such as Cu (<0.2 wt-%) cannot be quantified accurately using this method.
Discussion
This study provides the first analysis of Akan gold artifacts from well-dated archaeological contexts by means of non-destructive pXRF and SEM-EDS analysis. While the semi-quantitative analysis of these methods did not allow the identification of specific gold sources, and the contamination from concretion material limited the significance of the data, it did allow a first cautious assessment of their bulk material composition and the purity of the gold that was traded on the West African coast in the early 18th century.
The deviation between the pXRF and the SEM-EDS data, especially regarding Fe, Pb and to a lesser extent Cu and Ag (Fig. 6), is likely from the elemental composition of the dark spots visible in the BSD-imaging (Fig. 7), which are composed mainly of Fe with some percentages of Pb and other elements usually not associated with gold (Fig. 8). We interpret the dark patches as being remnants of the concretional masses from which they have been extracted. Cleaning in ultrasonic bath could not remove all the concretional residue from the samples. This inclusive material limits the significance of the pXRF analyses, as the Pb and Fe contents do not correspond to the actual composition of the gold but reflect the contamination, or represent a mixed analysis between the gold and the contaminated surface. Measuring inhomogeneous samples with a big excitation spot may in this case lead to unreliable results. On closer inspection, it is noticeable that in some cases the Cu and Ag contents recorded by pXRF analysis are also higher than those recorded by SEM-EDS (Fig. 6). This is critical because these two elements also occur in the concretion. Figure 2 clearly shows that parts of the gold were often found in concretions together with silver coins, whereby silver can quickly react with salt water to form silver chloride. This means that even the Cu and Ag contents obtained by pXRF must be critically evaluated.
SEM-EDS measurements at optically pure, uncontaminated areas could be a solution to circumvent surface contamination, but these are not without limitations. The lower kV of the measurements compared to pXRF makes it particularly vulnerable to surface contamination as it often exclusively collects the lower energy lines. Elemental segregation on the gold surface which can lead to local enrichments of silver or copper, pose a serious problem for SEM-EDS analysis as the surface analyses may not correspond to the actual composition of the gold objects. These effects are to be expected with metal objects from maritime contexts. Although care has been taken here to select the largest possible fields (100×100 µm) for the EDS measurements, and in many cases the individual loci are indeed comparable with each other (S1 Table sheet 3), they can also be subject to strong fluctuations (e.g., 5789/25 (cast)). Figure 6 reveals that in some cases SEM-EDS analysis detected higher amounts of Ag and to a lesser extent also Cu. These deviations from the pXRF data may be interpreted as local enrichments rather than bulk composition. Therefore, SEM-EDS analysis does not provide a reliable determination of the actual composition of the gold objects either and must also be taken with caution. However, due to the lower level of contamination by Fe, Pb, and presumably also Cu and Ag in some cases (Fig. 6), the SEM-EDS data are preferable to the pXRF data. For this reason, these data will be used in the further discussion of the results, diagrams, and comparisons with geological deposits.
The study pieces designated “nuggets” are interpreted as naturally occurring material, without any metallurgical alteration. Their Au/Ag contents correspond closely with geological deposits naturally occurring gold from the Ashanti Belt. They also contain no more than 0.5 wt% Cu, which is also consistent with naturally occurring gold deposits.
Further quantitative analyses could further assess this interpretation. SEM-EDS analyses of sediments and minerals in small cracks in the gold nuggets could provide further information, but since the metallic concretion residues also appear in the cracks and crevices in all the gold objects examined, this does not provide useful assessment in this case.
The objectives of the analyses were to assess the quality and composition of Akan gold, and to compare the data with historical descriptions by contemporary European writers.
As discussed, early European accounts often refer to the adulteration of gold with silver. The SEM-EDS data detected silver in all but four samples (5784/25 (unknown), 5790/25 (nugget), 5795/25 (unknown), 5797/25 (nugget)), but no more than can be expected based on data for geological deposits in the Ashanti Belt (Figs. 9, 10)70,71,72,73. Only sample 5803/25 (cast) is a few wt-% above the maximum published silver content for the gold occurrence at Prestea, the deposit where Ag is highest.
A Au vs. Ag content, B Au vs. Cu content, C Ag vs. Cu content, D Fe vs. Cu content. Data for the Ashanti belt mining areas of Bogosu, Prestea and Obuasi have been added for comparison. The data used can be found in the supplementary material (S1 table sheet 3). Data for Bogosu and Prestea70, Obuasi71. All Data reduced to Au+Ag+Cu=100, except D.
The one on the right shows the influence of the elements gold, silver, and copper on coloration and marks the area (red rectangle) where the gold finds examined are located. In the left diagram, the Cu contents of the studied gold objects have been multiplied by 10 and the Au content divided by 10 to clearly show that the cast objects exceed the limit of the copper contents found in the Ashanti Gold Belt, while most of the gold is quite like the geological data. Ternary diagram data for gold hues by Metallos https://commons.wikimedia.org/wiki/File:Ag-Au-Cu-colours-english.svg.
In a similar way, there is limited evidence for the intentional addition of copper or brass. Zn contents only appear in the pXRF analysis (samples 5783/25 (cast), 5785/25 (cast), 5802/25 (nugget)) and are completely absent in the SEM-EDS measurement of the bright areas. Cu appears only in small quantities and is exclusively found in the cast objects above 0.5 wt.-%, the limit value set for Cu in the Ashanti Gold Belt at Prestea where Cu is highest. (Figs. 9, 10). The exception here is again 5803/25 (cast), which has a greater Cu content in addition to its very high silver content. However, this is a fragment of a casting or overflow, and the composition is consistent with the interpretation that most Akan gold casts have higher Cu amounts than potentially naturally occurring nuggets. The Cu amounts are relatively low and do not exceed 4 wt-%, indeed in most samples they are around 1 wt-% or below. Notably, under SEM magnification 5779/25 (cast) also displays dendritic structures, indicative of the re-crystallization during casting. These structures are often not visible on castings as they often disappear when the gold is polished or subject to wear and tear.
The Cu-content in the cast pieces is higher than in geological occurrences in the Ashanti Gold Belt, raising the question of whether the copper was added intentionally (Figs. 9, 10). However, the higher Cu-content does not likely derive from secondary contamination; If this was the case there should be a direct correlation between the SEM-EDS iron content (the main contaminant) and the copper content, but this is not the case (Fig. 9D). Possibly, the crucibles used by the Akan goldsmiths to melt gold were not completely clean and contained residues from previous castings involving other non-ferrous metals, which then unintentionally contaminated the gold during casting74. The Akan worked in silver, copper, and brass, thus contamination from non-ferrous materials used in the same workshop may have taken place. However, intentional addition must also be discussed, as it is known that even small amounts of Cu have a hardening effect on the mechanical properties of gold alloys74. Indeed, analyses of other archaeological gold work have revealed similar copper contents75,76,77. This hardening effect was probably desirable in finely detailed gold casts, some of which are wafer-thin. The addition of Cu to the gold would increase durability and general resistance to wear, although possibly not in the low percentage range reported here. It is unlikely that the copper was added in order to obtain a certain hue because the amounts detected here are much too low to have any effect on the coloration (Fig. 10).
On the other hand, the gold deposits within the Ashanti Gold Belt (Obuasi, Prestea, and Bogosu) have significantly different gold/silver ratios to each other70,71 and since semi-quantitative analysis does not allow the finds from the Whydah to be assigned to a specific deposit, it remains unclear whether deposits with lower silver content, such as Obuasi and Bogosu, have been used and then mixed with external silver. It is not clear whether the gold stems from the same ports of acquisition as the Akan material or whether it was acquired elsewhere on the West African coast.
Notably, non-African gold was also traded at the coast54. In 1690, the Brazilian gold rush in the Minas Gerais region placed the Portuguese ahead of their European rivals in West Africa78. It was believed that during the 18th century and the rise in gold circulation, the Akan demanded gold from the Europeans for the purchase of slaves and in turn used slaves in exchange for European goods5. As a result, the Akan states hoarded gold after around 17005, possibly in connection with the developments in Brazil and the global gold market. The reason for this development could be that the flow of gold from within the country declined for domestic, political reasons. Conversely, this would possibly mean that either more adulterated gold entered circulation or that more gold had to be imported from the Americas for barter with the Europeans. These points further complicate the question of the origins and composition of the gold that was traded on the coast. ICP-MS analysis of trace elements and platinum group elements74,75,76,79,80,81, as well as various isotope analysis75,77,82 in the Whydah’s gold could further clarify the source of the gold, but this goes beyond the scope of this study.
The intentional addition of silver for the purpose of diluting the gold content, as European traders often warned against, cannot be safely assumed in the case of the Whydah’s material. Rather the composition of the gold traded closely corresponds to the silver-rich geological deposits of the Ashanti Gold Belt, such as Prestea that could have been exploited.
It is also striking that, in the case of the cast objects, the copper content is almost always above the data reported for the geological deposits. The reasons for this have been discussed and range from intentional addition for the purpose of hardening the material to unintentional contamination from working with other materials in the same workshop. However, the copper contents are so low they do not justify the frequent warnings regarding the adulteration of the gold traded.
The finds from the Whydah provide insight into a narrow time frame and their composition cannot be generally applied to all the gold traded, considering the centuries-long gold trade on the Ghanaian coast, other sources and ports where gold was traded, and the many African groups involved in the gold trade. It is possible that the Europeans who traded for the Whydah gold were very careful or simply lucky to receive gold of high quality. However, the range of samples examined here reveals little indication of false gold. Nevertheless, analyses and data on geological deposits in the Ashanti Belt show that the gold is far from pure and that silver content can be as high as 22%. This would certainly have been noticed by European traders, which, along with the deliberate addition of silver, could explain the recurring allegations of alleged dilution with silver by the Akan.
The timing of the gold transaction is significant. The early 18th century was a period of sociopolitical and socioeconomic change on the coast, which included the growth of an African merchant class and formation of the Asante Kingdom. In the course of this, more gold is said to have been hoarded. This makes the findings considered here even more significant. Future studies will concentrate on quantitative ICP-MS trace-element and isotope data while adding samples of South and Central American gold and silver to assess whether the Akan had access to these sources when crafting gold artefacts such as those discussed in this paper.
Data availability
All data collected in this study are included in this paper and in the supplementary material (S1 table).
References
Austen, A. Trans-Saharan Africa in World History (Oxford, 2010).
Bovill, E. W. The Golden Trade of the Moors (Marcus Weiner, 1999).
Blake, J. W. West Africa: Quest for God and Gold, 1454–1578 (Curzon Press, 1977).
Curtin, P. D. The lure of Bambuk gold. J. Afr. Hist. 14, 623–631 (1973).
Garrard, T. F. Akan Weights and the Gold Trade (London, 1980).
Garrard, T. F. Myth and metrology: the early trans-Saharan gold trade. J. Afr. Hist. 23, 443–461 (1982).
Hair, P. E. H. The Founding of Castelo São Jorge Da Mina: an Analysis of the Sources (African Studies Center, University of Wisconsin, 1994).
Levtzion, N., Hopkins, J. F. Corpus of Early Arabic Sources for West African History (Cambridge University Press, 1981).
McIntosh, S. K. A reconsideration of Wangara/Palolus, Island of Gold. J. Afr. Hist. 22, 145–158 (1981).
Nixon, S., Rehren, T. & Guerra, M. F. New light on the early Islamic West African gold trade: coin moulds from Tadmekka, Mali. Antiquity 85, 353–368 (2011).
Phillipson, D. W. Trans-Saharan gold trade and Byzantine coinage. Antiquaries J. 97, 145–169 (2017).
Wilson, A. Saharan trade in the Roman period. Azania 47, 409–449 (2012).
Carranza, E. J. M., Owusu, E. A. & Hale, M. Mapping of prospectivity and estimation of number of undiscovered prospects for lode gold, southwestern Ashanti Belt, Ghana. Min. Depos. 44, 915–938 (2009).
Leube, A., Hirdes, W., Mauer, R. & Kesse, G. O. The early Proterozoic Birimian Supergroup of Ghana and some aspects of its associated gold mineralisation. Precambr. Res. 46, 139–165 (1990).
Milesi, J. P., Ledru, P., Feybesse, J.-L., Dommanget, A. & Marcoux, E. Early Proterozoic ore deposits and tectonics of the Birimian orogenic belt, West Africa. Precambr. Res. 58, 305–344 (1992).
Ballong-Wen-Mewuda, J. B. La Vie D’un Comptoir Portugais En Afrique Occidentale (École des Hautes Études en Sciences Sociales, Centre d’Études Portugaises: Fondation Calouste Gulbenkian/ Commission Nationale por les Commémorations des Décourvertes Portugaises 1993).
Elbl, I. The Portuguese Trade with West Africa, 1440-1521. Ph.D.-thesis, University of Toronto (1986).
Elbl, I. The volume of the early Atlantic Slave Trade, 1450-1521. J. Afr. Hist. 38, 31–75 (1997).
Daaku, K. Y. Trade And Politics On The Gold Coast (Clarendon Press, 1970).
DeCorse, C. R. Early trade posts and forts of West Africa. In First Forts: Essays on the Archaeology of Proto-colonial Fortifications (ed. Klingelhofer, E.) 209–233 (Leiden, 2010).
DeCorse, C. R. Tools of empire: trade, resources and the British Forts of West Africa. In Building the British Atlantic World, 1600-1850 (ed. Herman, B. L. Maudlin, D) 165–187 (2016).
Kea, R. A. Settlements, Trade, And Polities in the Seventeenth-Century Gold Coast (Johns Hopkins University Press, 1982).
Lawrence, A. W. Trade Castles and Forts of West Africa (Jonathan Cape, 1963).
Osei-Tutu, J. K. Forts, Castles, and Society in West Africa, 1450–1960 (Brill, 2019).
De Marees, P. Beschryvinge Ende Historische Verhael Vant Govt Koninckvijck Van Guinea. (Amsterdam, 1602; modern ed. with original pagination marked, S. P. L’Honore, Naber, The Hague, 1912).
Bosman, W. A New and Accurate Description of the Coast of Guinea (Utrecht, 1704; first English trans., London 1705; facsimile, with annotations, by John D. Fage and Ray Bradbury, 1967).
Ratelband, K. Vijf Dagregisters Van Het Kasteel São Jorge Da Mina (Elmina) Aan De Goudkust 1645–1647 (‘S-Gravenhage, 1953).
Swanzy, F. A French voyage to West Africa in 1666–1667. J. R. Afr. Soc. 7, 190–204 (1908).
Hair, P. E. H., Jones, A. Law, R. Barbot on Guinea: The Writings of Jean Barbot on West Africa 1678–1712. (Hakluyt Society, 1992).
Jones, A. German Sources for West African History, 1599–1669 (Franz Steiner Verlag, 1983).
Jones, A. Semper aliquid verteris: printed sources for the history of the Ivory and Gold Coasts, 1500-1750. J. Afr. Hist. 27, 215–235 (1986).
Jones, A. Raw, Medium, Well Done: a Critical Review of Editorial Work and Quasi-Editorial Work on pre-1885 European Sources for Sub-Saharan Africa, 1960–1986 (African Studies Program, University of Wisconsin, 1987).
Jones, A. Drink deep or taste not: thoughts on the use of early European records in the study of African material culture. Hist. Afr. 21, 349–370 (1994).
Jones, A. Written sources for the material culture of the Gold Coast before 1800. A provisional checklist. J. des. Afr. 75, 99–127 (2005).
Van Dantzig, A. Willem Bosman’s new and accurate description of the Coast of Guinea. How accurate is it? Hist. Afr. 1, 101–108 (1974).
Hair, P. E. H. Barbot, Dapper, Davity: A critique of sources on Sierra Leone and Cape Mount. Hist. Afr. 1, 25–54 (1974).
De Marees, P. Description And Historical Account of the Gold Kingdom of Guinea (1602). Translated and edited by A. Van Dantzig and A. Jones (Oxford University Press, 1987).
Hayes, R., The Negociator’s Magazine: or, the Most Authentick Account Yet Published of the Monies, Weights, and Measures of the Principal Places of Trade in the World. The Fourth Edition, With Many Alterations, and Great Additions (1739).
Atkins, J., A Voyage to Guinea, Brazil and the West Indies (1735).
Pietz, W. The problem of the fetish, IIIa: Bosman’s Guinea and the enlightenment theory of fetishism. RES: Anthropol. Aesthet. 16, 105–124 (1988).
Law, R. The ‘golden age’ in the history of the pre-colonial gold coast: the era of gold exports (15TH to 17TH centuries) re-examined. Trans. Hist. Soc. Ghana 17, 109–136 (2015).
Van Dantzig, A. The Ankobra gold interest. Trans. Hist. Soc. Ghana 14, 169–185 (1973).
DeCorse C. R. An Archaeology of Elmina. Africans And Europeans on the Gold Coast 1400–1900 (Perch. Press, 2021).
Adams, J. Sketches taken during ten voyages to Africa, Between the Years 1786 And 1800 (New York Reprint Corporation, 1970).
Brun, S. Samuel Brun’s voyages of 1611–1620. German Sources For West African History. 44–96 (Franz Steiner1983).
Hawthorne, N. J Afr. Cruiser (Wiley and Putnam, 1815).
Hemmersam, M. Description of the Gold Coast, 1639–1645. In German Sources For West African History 97–133 (Franz Steiner, 1983).
Müller, W. J. Description of the Fetu country, 1662–1669. In German Sources For West African History 134–259 (Franz Steiner, 1983).
Rømer, L. F. A Reliable Account of the Coast of Guinea (1760), translated and edited by S. Winsnes (University of Oxford Press, 2000).
Tilleman, E. A Short and Simple Account of the Country of Guinea and Its Nature, translated and edited by Selena Axelrod Winsnes (African Studies Program, University of Wisconsin, 1994).
Garrard, T. F. Akan silver. Afr. Arts 17, 48–89 (1984).
DeCorse, C. R., Brass Working and Mforowa Manufacture among the Akan of Coastal Ghana during the 17th–20th centuries, Afrique: Archéologie & Arts [En ligne], 18 | 2022, mis en ligne le 02 novembre 2022. http://journals.openedition.org/aaa/3707.
Silverman, R., Akan Metal Arts, Yesterday and Today, In Peace, Power And Prestige: Metal Arts in Africa (ed. Cooksey, S.) 39–61 (Samuel P. Harn Museum of Art, University of Florida, 2020).
Alpern, S. What Africans got for their slaves: a master list of European trade goods. Hist. Afr. 22, 5–43 (1995).
Van Dantzig, A. The Dutch and The Guinea Coast 1674–1742: A Collection of Documents from The General State Archive at the Hague, Compiled and Translated By A. Van Dantzig (Ghana Academy of Arts and Sciences, 1978).
Craddock, P. T., Cowell, M. R. & Guerra, M. F. Assay and refining of gold in the post-medieval Islamic world: potential and reality. Hist. Metall. 54, 9–20 (2023).
Oddy, A. Assaying in antiquity. Gold. Bull. 16, 52–59 (1983).
Smith, C. S., Forbes, R. J. Metallurgy and assaying in History of Technology, Vol. III: From The Renaissance to the Industrial Revolution, 1500–1750 (ed. Singer, C. et al.) 27–71 (Oxford University Press, 1957).
Hamilton C. E. Whydah Joint Venture, United States, Massachusetts Historical Commission, Massachusetts Board of Underwater Archaeological Resources. Final Report of Archaeological Data Recovery: The Whydah Shipwreck Site Wlf-Ha-1 (Whydah Joint Venture; 1992).
Clifford, B., Kinkor, K. J. Real Pirates: The Untold Story of the Whydah from Slave Ship to Pirate Ship (National Geographic, 2007).
Hamilton, C. E. The Pirate Ship Whydah. In X Marks the Spot. The Archaeology of Piracy (ed. Skowronek, R. K., Ewen, C. R.) 130–159 (University Press of Florida, 2006).
Webster, D. Pirates of the Whydah. National Geographic 195, 66 (1999).
Kinkor, K., J. Artefacts that talk like pirates: Jolly Roger iconography and archaeological sites in pieces of eight. In: More Archaeology Of Piracy (ed. Skowronek, R.K. and Ewen, C.R.) 228–238 (University Press of Florida 2016).
Ehrlich, M. J. Early Akan Gold from the Wreck of the Whydah. Afr. Arts 22, 52–57 (1989).
Ehrlich, M. J. African Gold from a Pirate Shipwreck. Minerva 2, 24–29 (1991).
Ehrlich, M. J. Two Akan Gold Ornaments from the Wreck of the Whydah. Afr. Arts 45, 32–41 (2012).
Cole, H. M., Ross, D. H. The Arts of Ghana (UCLA Museum of Cultural History, 1977).
Ross, D. H. Gold of the Akan in the Glassell Collection (Museum of Fine Arts, 2002).
Ross, D. H. Royal Arts of the Akan: West African Gold in Museum Liaunig (Museum Liaunig, 2008).
Mumin, A. H., Fleet, M. E. & Chryssoulis, S. L. Gold mineralization in As-rich mesothermal gold ores of the Bogosu-Prestea mining district of the Ashanti Gold Belt, Ghana: remobilization of“invisible” gold. Mineral. Depos. 29, 445–460 (1994).
Osae, S. D., Kase, K. & Yamamoto, M. A. Geochemical study of the Ashanti gold deposit at Obuasi, Ghana. Okayama Univ. Earth Sci. Rep. 1, 81–90 (1995).
Bowell, R. J. Supergene gold mineralogy at Ashanti, Ghana: Implications for the supergene behaviour of gold. Mineral. Mag. 56, 545–560 (1992).
Salvi, S. et al. The Pampe gold deposit (Ghana): constraints on sulfide evolution during gold mineralization. Ore Geol. Rev. 78, 673–686 (2016).
Pernicka, E. Possibilities and limitations of provenance studies of ancient silver and gold. In: Metalle Der Macht—Frühes Gold Und Silber/Metals of Power—Early Gold and Silver (Meller, H., Risch, R., Pernicka E. eds.) 2014, 153–164.
Jansen M., Geochemie Und Archäometallurgie Des Goldes Der Bronzezeit In Vorderasien. PhD. Dissertation (Ruhr-University of Bochum, 2019). https://hss-opus.ub.ruhr-uni-bochum.de/opus4/frontdoor/index/index/year/2019/docId/6686.
Magnavita, S. & Mertz-Kraus, R. XRF and LA-ICP-MS studies of gold and silver artefacts from a 12–13th century CE tumulus in Senegal: implications for the medieval African gold trade. J. Archaeol. Sci.: Rep. 23, 416–425 (2019).
Numrich, M. et al. Portable laser ablation sheds light on Early Bronze Age gold treasures in the Old World: new insights from Troy, Poliochni, and related finds. J. Archaeol. Sci. 149, 105694 (2023).
Law, R., The gold trade of Whydah in the seventeenth and eighteenth centuries. African Economic and Social History (Henige, D., McCaskie, T. C. eds.) 106–111 (Madison, 1990).
Guerra, F., Sarthre, C. O., Gondonneau, A. & Barrandon, J. N. Precious metals and provenance enquiries using LA-ICP-MS. J. Archaeol. Sci. 26, 1101–1110 (1999).
Guerra, M. F. The circulation of South American precious metals in Brazil at the end of the 17th century. J. Archaeol. Sci. 31, 1225–1236 (2004).
Guerra, M. F. Trace elements fingerprinting using accelerators and ICP-MS: circulation of gold from the 6th century BC to the 12th century AD (vol. 223) (eds.) In Cultural Heritage Conservation and Environmental Impact Assessment by Non-Destructive Testing and Micro-Analysis (van Grieken, R., Janssens, K. eds) 223–244 (Taylor and Francis, 2005).
De Palaminy, L. Development of Wet and In Situ Isotopic Measurements of Lead, Iron and Copper for the Provenance of Historical Gold: Application To Islamic Coins. PhD. Dissertation Geochemistry. Université de Toulouse, 2024. English. ffNNT : ff. fftel-05099967f.
Acknowledgements
We express our gratitude to the Whydah Pirate Museum in Yarmouth, MA, for giving us access to the gold finds and archival records, as well as to Meredith Katz for helping with sampling the objects on display. We also thank Michael Prange for providing access to the material science laboratory of the German Mining Museum in Bochum, Germany and the laboratory staff for helping with the analytical instruments. Last but not least, we thank the anonymous Reviewers for their feedback, which greatly improved the scientific approach of this study. This study received no funding.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
T.B.S. was primarily responsible for research supervision, study design, geochemical data collection, and writing. B.C. facilitated access to the material, organized research logistics, provided resources, took and edited photographs, and supplied information on the site. C.R.D.C. reviewed the entire work, placed the results in their archaeological and historical context, interpreted and expanded on the historical primary and secondary sources, and finalized the text. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Skowronek, T.B., Clifford, B. & DeCorse, C.R. Pirate gold provides new insights into West African trade using pXRF and SEM EDS analysis. npj Herit. Sci. 14, 169 (2026). https://doi.org/10.1038/s40494-026-02441-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s40494-026-02441-7