Abstract
Background
Emerging evidence suggests that digital therapies are effective for treating obesity; however, an evidence gap exists regarding the potency of stand-alone digital therapeutics with limited additional support from a health care professional. This randomized controlled trial examined the efficacy of a digital health application for weight management in people with obesity.
Methods
The decentralized digital randomized controlled trial included 164 adults with obesity (BMI 30–45 kg/m²) from Germany who were randomly assigned to either an intervention group (receiving a digital health application for weight management for 6 months) or a control group (receiving care as usual for 6 months). Data on weight (primary endpoint at 6 months), quality of life (WHOQOL-BREF), and food literacy (SFLQ) were collected at baseline, 3, and 6 months.
Results
Participants (n = 164; 42% female, 58% male) had a mean BMI of 37.82 ± 4.25 kg/m² and a mean age of 45.92 ± 10.66 years. At 6 months, the intervention group achieved a mean weight loss of 5.29% ([95% CI: −6.73% to −3.86%], p < 0.001) compared to 1.76% ([95% CI: −3.10% to −0.42%], p = 0.010) in the control group (estimated marginal mean difference (EMMD): −3.53% [95% CI: −5.16% to −1.91%]; p < 0.001) with a large effect size (d = 0.80, [95% CI: 0.43–1.19]). Compared to controls at 6 months, food literacy (EMMD: 2.77, d = 0.49, p < 0.001) and perceived overall quality of life (EMMD: 0.33, d = 0.40, p = 0.010) improved in the intervention group, though no differences in the four quality of life domains were found.
Conclusions
The digital health application for weight management demonstrated efficacy in achieving weight loss (>5%) in people with obesity at 6 months.
Trial registration
This study was registered in the German Clinical Trials Register (Registration number: DRKS00033045).
Similar content being viewed by others
Introduction
Obesity is a chronic, relapsing, multifactorial disease associated with numerous health risks and increased mortality [1]. The prevalence of obesity has risen significantly worldwide, and in Germany, more than half of the adult population are now overweight or live with obesity [2]. Despite medical necessity, access to treatment remains constrained due to limited treatment options and insufficient reimbursement. Consequently, only a fraction of patients with obesity receive treatment in Germany [3]. Effective therapies for obesity include lifestyle interventions that can be digitally implemented [4]. In 2019, Germany introduced digital health applications (“Digitale Gesundheitsanwendungen” abbreviated in German as DiGA) into its health care system. DiGAs are medical devices (e.g., apps) that help diagnose, monitor and treat diseases with a digital mode of action. The clinical efficacy of DiGA is evaluated by the Federal Institute for Drugs and Medical Devices (Bundesministerium für Arzneimittel und Medizinprodukte (BfArM)) prior to approval for full reimbursement in statutory health care [5]. To that end, Oviva Direkt for Obesity (Oviva AG, Potsdam, Germany), a DiGA for weight management, was evaluated in a randomized controlled trial, showing 3.2 ± 3.0% weight loss at 3 months for people with obesity with a body-mass-index (BMI) of 30–40 kg/m² and a statistically significant group difference compared to a control group [6]. This study led to the approval of Oviva Direkt for Obesity for the treatment of obesity in Germany and its endorsement in the updated German guideline for the prevention and treatment of obesity [7]. The aim of the present randomized controlled trial was to evaluate the efficacy of Oviva Direkt for Obesity at 6 months in people with obesity with a BMI of 30–45 kg/m². Moreover, the study sought to assess additional benefits such as quality of life and food literacy in a broader population, including those with a BMI of ≥40–45 kg/m².
Methods
Study design
This study was a prospective decentralized randomized controlled trial with technology-enabled, remote recruitment, care delivery, and data collection conducted in Germany. The study’s primary endpoint was defined as the change in body weight assessed at 6 months of intervention. The study had a crossover design with participants in the intervention group continuing to receive the intervention for an additional 6 months (after primary endpoint assessment), whilst participants in the control group waited to receive the intervention for 6 months and then received the intervention for 6 months. Further analyses are planned for both 48 and 96 weeks. The study protocol has been approved by the ethics committee of the Berlin Medical Association (vote number: Eth-57/23) and was registered in the German Clinical Trials Register (Registration number: DRKS00033045). All participants provided written informed consent before participation, and all procedures were performed in accordance with the relevant guidelines and regulations.
Study population
Participants were recruited via multiple social media platforms (e.g., Facebook, Instagram) and directly via the Oviva website between 27th November 2023 and 7th March 2024. Participants who met the following eligibility criteria were included: adults (females, males, non-binary), age between 18 and 75 years, BMI 30–45 kg/m², with a diagnosis of obesity confirmed by a physician, ownership of a smartphone compatible with the Oviva Direkt app, no severe conditions contraindicating lifestyle changes, and no recent or planned use of weight loss medications. Inclusion criteria were checked through a pre-screening survey and video consultations with a medical investigator and the study team. Informed consent was provided via a digital signing procedure. Included participants were randomized via stratified block randomization into the intervention group or control group in a ratio of 1:1. Stratification aimed to ensure that the influencing variables of sex, age, and BMI were equally distributed in both study groups. Pre-planned recruitment quotas to ensure even distribution of participants across the strata were in place. All participants attended five digital, remote study visits (baseline, week 6, week 12, week 18, week 24) and filled in digital questionnaires at these timepoints. Participants received compensation for completing the study visits (electronic vouchers worth 20 EUR at 3 months and 60 EUR at 6 months).
Digital health application
The digital health application Oviva Direkt for Obesity (Oviva AG, Potsdam, Germany) is an app-based, multimodal weight management intervention (targeting diet, physical activity, and disease-related psychological states and behaviors) in accordance with the German guidelines for the prevention and treatment of obesity [7]. The Oviva Direkt for Obesity app is certified as a medical device class IIa and available for iOS and Android. To ensure participants’ safety and the appropriate use of the medical device, a certified dietitian checked in with the participants shortly after they began the therapy and again after 3 months. During the check-ins, the dietitians addressed potential safety issues and explained the functioning of Oviva Direkt for Obesity to empower participants to self-administer the intervention independently. In addition, the dietitians were available via a reactive private chat.
Oviva Direkt for Obesity is a self-administered, app-based therapy designed to support participants to achieve clinically meaningful weight loss. Key intervention elements are self-monitoring, self-management, education, and feedback (details are provided in Table 1, Fig. 1, and in the app manual [8]).
Intervention elements in Oviva Direkt for Obesity.
The mechanism of action incorporates behavior change techniques (BCT) that enable behavioral modifications, which in turn drive weight loss [9]. For example, participants use the photo-based meal logging feature that documents their meals in a nutrition diary (Fig. 1). The act of meal logging corresponds to the BCT self-monitoring, which leads to heightened self-awareness of the types and quantities of food consumed, which supports participants to make conscious, alternative food choices [10]. The generated nutrition diary (based on multiple meal logs) digitally embodies the BCT feedback on behavior, again designed to alter undesirable dietary intake. Importantly, Oviva Direkt for Obesity has been continuously improved based on feedback from patients and health care professionals since the first study on its efficacy [6]. The educational content was improved for readability and actionability. Calorie-reduced, lower-carb meal plans including recipes were integrated, and the goal-setting functionality was upgraded, allowing participants to set and track therapy-related goals (Fig. 1).
Participants in the control group were eligible for care as usual at their discretion, which typically consisted of counseling by a general practitioner, and received access to Oviva Direkt for Obesity at 6 months.
Data collection
Due to the decentralized setup of the study, data were collected remotely during video calls as well as via electronic questionnaires (provided via e-mail and personal links). Sociodemographic data, comorbidities, and concomitant medication were collected at baseline via questionnaires. Study assistants collected weight data via provided, calibrated digital scales in supervised study calls via video calls at all measurement time points (baseline, 6, 12, 18, and 24 weeks). Quality of life and food literacy data were collected via questionnaires at baseline, 12, and 24 weeks. Quality of life was assessed with the WHOQOL-BREF [11], consisting of a score for the four components: physical health, psychological component, social relationships, and the environmental component. Component scores had a range of 0 to 100, while the individual questions were scored on a scale of 1 to 5, with higher values representing a higher quality of life. Also, question 1 (“How do you rate your quality of life?”) and question 11 (“Can you accept your appearance?”) of the WHOQOL-BREF were used to assess the perceived overall quality of life and the acceptance of the physical appearance of the respondent, respectively. Food literacy was assessed with the Short Food Literacy Questionnaire (SFLQ; example question: “If I have questions about healthy eating, I know where I can find information.”) [12]. A higher SFLQ sum score (range of 0–52) indicated higher food literacy. All outcome analyses for the data above were prespecified.
Statistical analyses
The primary endpoint was weight loss expressed as a percent change in body weight at 6 months. The primary analysis population was the intention-to-treat (ITT) population with missing values imputed using multiple imputation by chained equations (MICE) to allow for flexible modeling of missing data with multiple imputations across variable types while preserving relationships [13]. For the primary endpoint weight change, a multiple linear regression analysis adjusted for sex, age, BMI category, baseline weight, comorbidities, and use of concomitant medication (yes/no) was conducted for comparison between the intervention and control group after 6 months. The Bonferroni–Holm procedure was used on each p value for multiple comparisons. Subgroup analyses (unpowered) were conducted across the three strata (sex, age, BMI category; using the ITT population) by fitting the regression model to each stratum of the subgroups. For the secondary endpoint quality of life, the score for each of the four domains of the WHOQOL-BREF (physical health, psychological well-being, social relationships, environment) was calculated, which resulted in scores between 0 (lowest health state) to 100 (perfect state of health) for each domain. For each domain, the difference in scores between baseline and 6 months was calculated, and a multiple linear regression model with the adjustments specified above was conducted, allowing for comparisons between treatment arms. For the exploratory endpoints (relative and absolute weight change over time, change in quality of life over time, change in food literacy over time) mixed effects models for multiple measurements adjusted for sex, age, BMI category, baseline weight were used. Moreover, a logistic regression model was employed to identify the proportion of responders—defined as a decrease in body weight from baseline to 6 months of 5%, 7.5%, or 10%. Sensitivity analyses were conducted to assess the effect of missing data and the imputation mechanism on the calculated group effect using the full analysis set (FAS) population, the complete cases (CC) population, as well as last-observation-carried-forward imputation (LOCF) and copy-increments-in-reference imputation (CIR). Sample size was calculated using a significance level of 0.05, a statistical power of 90%, and an effect size of d = 0.78 (Cohen’s d; estimated from [6]). An assumed 25% drop-out rate, as well as augmentation of the sample to allow for sufficient representation within the strata, yielded a sample size of 160. All statistical analyses were performed using R Version 4.4.2 and RStudio Version 2024.12.0 + 467.
Results
Study profile
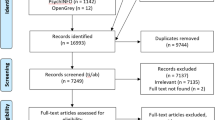
Figure 2 depicts the study flow. A total of 164 patients were included into the study between January 18, 2024 (first patient first visit) and March 7, 2024 (last patient first visit). The last patient completed the 6-month assessment corresponding to the primary endpoint on September 4, 2024 (last patient 6-month visit). The overall 6-month retention rate was 76%, with 69% retention in the intervention group and 84% in the control group.
T1 = baseline, T2 = 6 weeks, T3 = 12 weeks, T4 = 18 weeks, T5 = 24 weeks (primary endpoint), ePRO electronic patient-reported outcome.
Baseline data
Baseline characteristics of total study participants (n = 164) are shown in Table 2. Across both groups, 69 (42%) participants were female, and 95 (58%) were male. The mean BMI was 37.82 ± 4.25 kg/m², and mean age was 45.92 ± 10.66 years. Due to recruitment challenges, the pre-planned quota for the age stratum “61+ years” could not be observed, resulting in a small sample size in this stratum.
Weight change
At 6 months, the intervention group achieved a mean weight loss of 5.29% ([95% CI: −6.73% to −3.86%], p < 0.001) compared to 1.76% ([95% CI: −3.10% to −0.42%], p = 0.010) in the control group (estimated marginal mean difference: −3.53% [95% CI: −5.16% to −1.91%]; p < 0.001) with a large effect size (d = 0.80, [95% CI: 0.43–1.19]) (Fig. 3, see Table S1 for the observed, unadjusted values).
Weight change (%) at 6 months (i.e., 24 weeks) (primary endpoint).
Relative weight loss over time differed between the intervention and the control group at all measurement timepoints (all p < 0.05; see Table S2). The early divergence between the two groups indicates that the intervention group achieved a faster and more substantial weight loss compared to the control group from the beginning of the study. The intervention’s effect appears progressive, as weight loss continues to increase over time (Fig. 4). Absolute weight loss over time showed a corresponding pattern with statistically significant group differences at all timepoints (all p < 0.05) and an estimated marginal mean weight loss of −6.98 kg ([95% CI: −8.71 kg to −5.24 kg], p < 0.001) in the intervention group compared to −1.41 kg ([95% CI: −3.11 kg to 0.28 kg], p = 0.010) in the control group at 6 months.
Estimated marginal means (EMMs) for percent change in body weight over time in the intention-to-treat (ITT) population (exploratory endpoint; data in Table S2).
Regarding response, a logistic regression model identified an association between treatment arm (intervention vs. control) and the chance of a clinically relevant weight reduction of at least 5% at 6 months (OR: 0.04 [95% CI: 0.01–1.14]; p < 0.001). Descriptively, almost half (49%) of the participants in the intervention group achieved a weight loss of ≥5%, whereas in the control group, only 4.8% achieved this category. Moreover, in the intervention group 46% achieved ≥7.5% weight loss and 17% achieved ≥10% weight loss (Table 3).
Subgroup analyses for sex, age, BMI category revealed stable effects which indicated a uniformly higher weight loss in the intervention group compared to the control group, including the group of participants with a BMI of ≥40–45 kg/m² (estimated marginal mean difference: −3.86%, d = 0.67; p = 0.027) (see Table S3 in the appendix for details).
Quality of life
Descriptive analyses of the four components of the WHOQOL-BREF indicated a positive effect in the intervention group, testified to by positive estimated marginal mean differences, but no statistical significance was observed (physical health: 3.77 [95% CI: −0.37 to 8.34], p = 0.148; psychological component: 6.30 [95% CI: 0.68–12.73], p = 0.061; social relationships: 4.54 [95% CI: 1.30–11.01], p = 0.133, environmental component: 2.30 [95% CI: −1.83 to 4.00], p = 0.191). Analyzing the group differences in reported quality of life over time revealed a statistically significant effect for the psychological component regarding estimated marginal mean differences at 3 months (6.86, d = 0.41, p = 0.043) and 6 months (6.56, d = 0.39, p = 0.014) and for social relationships at 3 months (9.5, d = 0.44, p < 0.001). No other statistically significant effects were found for the components. However at 6 months, participants’ perceived overall quality of life (question 1 of the WHOQOL-BREF; estimated marginal mean difference = 0.33, d = 0.40, p = 0.010) and their acceptance of their physical appearance (question 11; 0.47, d = 0.51, p = 0.001) was higher in the intervention group than in the control group.
Food literacy
Between-group contrasts for food literacy based on the respective intra-group change from baseline indicated statistically significant estimated marginal mean differences at 3 months (3.30%, d = 0.59, p < 0.001) and 6 months (2.77%, d = 0.49, p < 0.001), indicating improved food literacy in the intervention group. A higher baseline score was significantly associated with a smaller change in food literacy over time (p < 0.001).
Sensitivity analyses
Sensitivity analyses for the primary endpoint indicated that the estimated marginal mean difference between groups was robust regarding missing data (FAS: −4.27%, d = 1.28, p < 0.001; CC: −4.24%, d = 1.29, p < 0.001) and imputation mechanisms (LOCF: −2.76%, d = 0.83, p < 0.001; CIR: −2.91%, d = 0.66, p < 0.001).
Discussion
The digital health application Oviva Direkt for Obesity demonstrated efficacy in achieving clinically relevant weight loss (>5%) in people with obesity at 6 months, as well as improvements in food literacy and nuanced improvements in quality of life. The control group, receiving only care as usual, showed only a small, clinically irrelevant decline in weight. The early and sustained divergence in weight loss between the intervention and control groups indicates early and sustained effects of the intervention. The observed weight loss was clinically meaningful as stipulated in the German guidelines for the prevention and treatment of obesity that define a weight loss of at least 5–10% at 6 to 12 months as a goal—with higher recommended weight loss for patients with a BMI ≥ 35 kg/m² [7]. Clinically meaningful weight loss is associated with a variety of health benefits, such as a reduction of cardiometabolic risk [14, 15]. Regarding the magnitude of the weight loss, the guidelines state that 2–3 kg for patients with a baseline BMI of 27–35 kg/m² can be achieved when treated by general practitioners, including personalized recommendations and information brochures [16]. In this study, the average weight loss was −6.98 kg (95% CI: −8.71 to −5.24 kg) for patients with a baseline BMI of 30–45 kg/m². Comparing the magnitude of the achieved weight loss with other smartphone interventions is challenging due to their heterogeneity and variations in research methods employed [4]. However, Antoun et al. [17] state in a systematic review that the average weight loss for smartphone app-based interventions at 6 months was –2.80 kg (95% CI –3.03 to –2.56 kg), indicating that participants in the present study lost around −5 kg more weight. Moreover, the observed large effect size in this study (d = 0.81 [95% CI: 0.43–1.19]) and the response rate of 49% of participants achieving ≥5% weight loss testify to the practical significance of the weight loss outcome. Importantly, more direct comparisons are possible: In a trial on another digital health application for weight management, which used patient-reported data as weight outcomes, participants in the intervention group lost on average −7.75% (95% CI: −9.66% to −5.84%) after 12 months [18]. Furthermore, a previous RCT with an earlier version of Oviva Direkt for Obesity yielded 3.2 ± 3.0% weight loss at 3 months for people with obesity with a BMI of 30–40 kg/m² [6]. In the present study, weight loss at 3 months was 4.07% (see Fig. 4 and Table S2) for people with obesity with a BMI of 30–45 kg/m², which may be a result of improvements made to the intervention in the time between the trials.
The robustness of the outcomes is bolstered by the consistent results of the subgroup analyses (unpowered) across the sample strata. Notably, the group of participants with a BMI of ≥40–45 kg/m² also achieved relevant weight loss, pointing to the efficacy of the intervention across all BMI categories researched in this study. Moreover, results from the responder analyses indicated that the treatment effect unfolded over time. For example, the number of patients responding with 7.5% weight loss increased from 9.8% at 3 months to 46% at 6 months. While there were early responders, a majority of the patients profited from sustained treatment, buttressing the beneficial effect of extended treatment from 3 to 6 months.
Additionally, improvements in perceived quality of life and acceptance of physical appearance were observed. Descriptive quality of life outcomes for the four components of the WHOQOL-BREF at 6 months uniformly indicated improvements in the intervention compared to the control group, but did not reach statistical significance. However, when analyzing changes over time improvements in the psychological component were apparent. The obtained mixed results could indicate a lack of sensitivity of the used questionnaire to the intervention under scrutiny. For example, the environmental component of the WHOQOL-BREF contains questions on available means of transportation and living conditions, which are unlikely to be impacted by a weight management intervention. Food literacy, a factor more indicative of the intervention at hand, improved over time, suggesting holistic treatment benefits beyond weight loss [19]. This improvement may lead to healthier eating behaviors and more effective self-management, which over time may contribute to reduced risk of obesity-related complications and chronic conditions, potentially lowering healthcare utilization [20]. The interrelation of weight loss, quality of life, and food literacy warrants further investigation to explore their temporal sequence, positive association, or even potential causation, as their interplay remains unclear.
A strength of this study was the decentralized setup, enabling Germany-wide recruitment of participants, and thus the representativeness of the sample. The remotely supervised data collection ensured data integrity and quality while limiting the burden on participants by making the need for study center visits redundant. Moreover, 58% of the participants were male, a peculiarity in weight management studies. A limitation of the study was the sole inclusion of participants in Germany and the small number of participants aged 61+, limiting generalizability. Moreover, effective blinding regarding the treatment assignment of participants in the intervention and control groups was not possible due to the nature of the intervention.
Conclusion
This study adds to the growing body of research on the efficacy of digital health applications for weight management. Due to their scalability and comparatively low cost, such interventions can be a meaningful therapy to support the rising number of people with obesity. As digital health applications are established and reimbursed in Germany, their increased utilization can address the unmet therapeutic needs of patients with obesity. Further research is needed to examine the longer-term efficacy of digital health applications for weight management, their effectiveness in real-world care, potential effects of adherence, as well as the patient-treatment fit of such interventions.
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available for data privacy reasons but are available from the corresponding author on reasonable request.
References
Busetto L, Dicker D, Frühbeck G, Halford JCG, Sbraccia P, Yumuk V, et al. A new framework for the diagnosis, staging and management of obesity in adults. Nat Med. 2024;30:2395–9.
Mensink GBM, Schienkiewitz A, Haftenberger M, Lampert T, Ziese T, Scheidt-Nave C. Übergewicht und Adipositas in Deutschland. Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz. 2013;56:786–94.
Blüher M. Versorgungssituation für Menschen mit Adipositas in Deutschland. Dtsch Med Wochenschr. 2023;148:957–63.
Metzendorf MI, Wieland LS, Richter B. Mobile health (m-health) smartphone interventions for adolescents and adults with overweight or obesity. Cochrane Database Syst Rev. 2024;2:CD013591.
Federal Institute for Drugs and Medical Devices. Digital Health Applications (DiGA). Accessed April 2025. https://diga.bfarm.de/de/.
Gemesi K, Winkler S, Schmidt-Tesch S, Schederecker F, Hauner H, Holzapfel C. Efficacy of an app-based multimodal lifestyle intervention on body weight in persons with obesity: results from a randomized controlled trial. Int J Obes. 2024;48:118–26.
Deutsche Adipositas-Gesellschaft (DAG) e.V. S3-Leitlinie Adipositas - Prävention und Therapie. 2024. https://register.awmf.org/de/leitlinien/detail/050-001.
Oviva AG. Oviva Direkt App Manual. Accessed April 2025. https://ovivadiga.com/manuals/Oviva_Direkt_App_User_Manual_v1.15.0_DE.pdf.
Tate DF, Lytle LA, Sherwood NE, Haire-Joshu D, Matheson D, Moore SM, et al. Deconstructing interventions: approaches to studying behavior change techniques across obesity interventions. Transl Behav Med. 2016;6:236–43.
Patel ML, Wakayama LN, Bennett GG. Self-Monitoring via digital health in weight loss interventions: a systematic review among adults with overweight or obesity. Obesity. 2021;29:478–99.
World Health Organisation. WHOQOL-BREF: Measuring Quality of Life. German version. Accessed April 2025. https://www.who.int/tools/whoqol/whoqol-bref/docs/default-source/publishing-policies/whoqol-bref/german-whoqol-bref.
Gréa Krause C, Beer-Borst S, Sommerhalder K, Hayoz S, Abel T. A short food literacy questionnaire (SFLQ) for adults: findings from a Swiss validation study. Appetite. 2018;120:275–80.
Buuren Svan, Groothuis-Oudshoorn K. mice: multivariate imputation by chained equations in R. J Stat Softw. 2011;45:1–67.
Dhar D, Packer J, Michalopoulou S, Cruz J, Stansfield C, Viner RM, et al. Assessing the evidence for health benefits of low-level weight loss: a systematic review. Int J Obes. 2025;49:254–68.
Blüher M. An overview of obesity-related complications: the epidemiological evidence linking body weight and other markers of obesity to adverse health outcomes. Diabetes Obes Metab. 2025. https://dom-pubs.pericles-prod.literatumonline.com/; https://doi.org/10.1111/dom.16263.
Jebb SA, Ahern AL, Olson AD, Aston LM, Holzapfel C, Stoll J, et al. Primary care referral to a commercial provider for weight loss treatment versus standard care: a randomised controlled trial. Lancet. 2011;378:1485–92.
Antoun J, Itani H, Alarab N, Elsehmawy A. The effectiveness of combining nonmobile interventions with the use of smartphone apps with various features for weight loss: systematic review and meta-analysis. JMIR MHealth UHealth. 2022;10:e35479.
Roth L, Ordnung M, Forkmann K, Mehl N, Horstmann A. A randomized-controlled trial to evaluate the app-based multimodal weight loss program zanadio for patients with obesity. Obesity. 2023;31:1300–10.
Zheng S, Edney SM, Goh CH, Tai BC, Mair JL, Castro O, et al. Effectiveness of holistic mobile health interventions on diet, and physical, and mental health outcomes: a systematic review and meta-analysis. eClinicalMedicine. 2023;66:102309.
Herman PM, Nguyen P, Sturm R. Diet quality improvement and 30-year population health and economic outcomes: a microsimulation study. Public Health Nutr. 2021;25:1265–73.
Acknowledgements
The authors would like to thank all study participants for participating in this study and Dr. Jan Multmeier for conducting the statistical analyses, as well as Sarah Meyer and Isabelle Springer for the management of the trial.
Author information
Authors and Affiliations
Contributions
AL and FS wrote the manuscript. MB, AM, and LJ provided feedback and refined the manuscript. All authors approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The research was fully funded by Oviva AG. AM, FS, and LJ are employed at Oviva AG. MB received honoraria as a consultant and speaker from Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Daiichi-Sankyo, Lilly, Novo Nordisk, Novartis, Pfizer, and Sanofi. AL received honoraria as a consultant and speaker from AstraZeneca, Lilly, Novo Nordisk, Boehringer Ingelheim, and Oviva.
Ethics approval and consent to participate
The study protocol has been approved by the ethics committee of the Berlin Medical Association (vote number: Eth-57/23) and was registered in the German Clinical Trials Register (Registration number: DRKS00033045). All participants provided written informed consent before participation, and all procedures were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lautenbach, A., Blüher, M., Mijuskovic, A. et al. Efficacy of a digital health application for weight management in people with obesity: 6-months results from a randomized controlled trial. Int J Obes 50, 466–473 (2026). https://doi.org/10.1038/s41366-025-01967-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-025-01967-3