Abstract
This study used compositional data techniques that address the interdependence of 24-h movement behaviors (sleep, sedentary behavior [SB], light-intensity physical activity [LPA], moderate-to-vigorous intensity physical activity [MVPA]) to examine: (1) how patients undergoing metabolic bariatric surgery (MBS) allocate time among these behaviors before MBS, and (2) whether overall time-use composition and modeled reallocation patterns relate to early weight loss after MBS. Participants wore an accelerometer 24 h/day for 10 days before MBS to measure time in sleep, SB, LPA, and MVPA. Isotemporal substitution models estimated differences in 6-month post-MBS percentage total weight loss (%TWL) associated with reallocations of these pre-surgery movement behaviors. Forty-five participants provided valid data. Pre-MBS time-use composition was associated with %TWL (23.8 ± 5.1%; F = 2.66, p = 0.047). Reallocating 15–60 SB or LPA minutes/day to MVPA was estimated to relate to 0.9–3.5% greater %TWL. Reallocating 15–30 MVPA minutes/day to SB or LPA was estimated to relate to 1.4–5.0% less %TWL (all comparisons p < 0.05). Other reallocations were non-significant. In conclusion, modeled shifts in time from SB or LPA to MVPA and vice versa were associated with estimated increases or decreases in early post-surgical weight loss, respectively. Experimental research is needed to clarify causal relationships and inform interventions to improve MBS outcomes.
Similar content being viewed by others
Introduction
Higher levels of moderate-to-vigorous intensity physical activity (MVPA) before and following metabolic bariatric surgery (MBS) are associated with more favorable postoperative weight outcomes [1,2,3]. However, MVPA is a movement behavior that accounts for the smallest proportion of a 24-h day [1, 4]. Recent work suggests that other daily movement behaviors, including sedentary behavior (SB) and sleep, may also influence weight outcomes [2, 4, 5]. Additionally, for some individuals undergoing MBS, increasing light-intensity physical activity (LPA) may be more practical than MVPA given physical or other limitations [6]. These findings support the idea of shifting from an MVPA-focused view to a more holistic and inclusive 24-h movement perspective [7].
Several recent studies have examined multiple 24-h movement behaviors in relation to postoperative weight outcomes [2, 4]. However, they have used analytic approaches that treat these behaviors separately, rather than reflecting their codependence [7,8,9]. Overlooking this codependence hinders understanding of how each 24-h movement behavior relates to weight outcomes in the context of the others [8,9,10]. Such approaches also do not address how reallocating time spent in one behavior (e.g., SB) to others (e.g., LPA) may influence weight outcomes [8,9,10].
To address these gaps, this study used compositional data analysis (CoDA) to 1) describe how patients allocate their time among sleep, SB, light-intensity PA (LPA), and MVPA before MBS from a 24-h perspective; 2) examine whether observed pre-MBS time-use composition correlates with weight loss at 6-months post-MBS; and 3) estimate differences in 6-month weight loss based on modeled compositional time reallocations among these movement behaviors pre-MBS. Although these time reallocations (Aim 3) were statistically modeled rather than experimentally tested, they can offer insights into how shifts in 24-h time use might influence weight loss and inform further experimental investigation. We hypothesized that overall time-use composition and reallocating time away from SB and LPA to MVPA would be associated with greater weight loss, while the reciprocal reallocation pattern would be associated with less weight loss.
Methods
Data were collected as part of a larger study investigating psychosocial and behavioral predictors of post-MBS weight loss (R01DK108579) [11]. All protocol elements relevant to the current study are reported below. All procedures were approved by The Miriam Hospital Institutional Review Board (#2112).
Participants
Participants were recruited from two academic MBS clinics in the Northeastern United States between May 2016 and April 2018. Participants were ≥21 years old, had a body mass index (BMI) ≥ 35.0 kg/m2, and were scheduled to undergo Roux-en-Y gastric bypass (RYGB) or sleeve gastrectomy (SG)—the two most common MBS procedures [12]. Additional details on inclusion and exclusion criteria, as well as recruitment and screening procedures, have been previously reported [11].
Procedures
After eligibility was confirmed and informed consent was obtained, participants completed an assessment visit 3–8 weeks before their scheduled MBS procedure. During this visit, they received an Actigraph GT9X accelerometer to wear on their non-dominant wrist 24 h/day for 10 days, completed a sociodemographic questionnaire, and had their height and weight measured. Participants’ height and weight were re-measured by clinic staff at 6 months post-MBS. Participants received USD75 compensation for each assessment.
Measures
Actigraph data were processed using GGIR, a commonly used R package for processing accelerometry data [13]. Participants who wore the device for ≥16 h/day on ≥4 days were included in the analysis [13]. Time/day spent in sleep was determined using the vanHees2025 algorithm [14] for sustained inactivity bouts detection, and the Heuristic algorithm looking at Distribution of Change in Z-Angle (HDCZA) algorithm [15] as the guider. For awake hours, vector magnitude counts per minute thresholds were used to identify time/day spent in SB, LPA, and MVPA as follows: <2000 counts/min = SB, 2000–7499 counts/min = LPA and ≥7500 counts/min = MVPA [16, 17].
Participants self-reported their age, gender, race, ethnicity, and MBS procedure type. Percent total weight loss (%TWL) was calculated using pre- and 6-month post-MBS weights.
Statistical analysis
Aim 1 was evaluated using descriptive statistics (mean and standard deviation). For Aim 2, Compositional Data Analysis (CoDA) was used to perform linear regression models on the 6-month %TWL while allowing the 24-h movement behaviors composition (i.e., using the relative values of time/day between behaviors which sum up to 1 representing 1440 min) before MBS to be included as one of the model predictors. In the same linear regression model, age, sex, race, pre-MBS BMI, and surgery type were each evaluated as potential covariates and adjusted for if significant. The four movement behaviors (sleep, SB, LPA, MVPA) in the composition were transformed to four sets of three isometric log ratio (ILRs) coordinates, using the compositions package [18]. Of note, the relative time spent in each of the four 24-h movement behaviors as a composition is represented by a 4-part simplex, which is not compatible with operations suitable for real space (e.g., multiplication). By using ILR coordinates, the 4-part simplex can be structured as a 3-dimensional real space, while the relative positions of the data points are preserved from the simplex to the real space (i.e., isometric). Data represented by the new 3-dimensional real space is then compatible with conventional statistical models such as multiple linear regression [19]. If the overall composition (ILRs’ coordinates) was significantly associated with %TWL (p < 0.05), isometric time reallocation analyses were conducted (Aim 3), using the codaredistlm package [20, 21], to estimate the average difference in 6-month %TWL when time (−60 to +60 min at 15 min increments) in one behavior at pre-MBS was replaced in the model by time in another behavior at pre-MBS, while keeping all remaining behaviors constant. All analyses were performed in R version 4.4.1. Statistical significance was set at α = 0.05.
Results
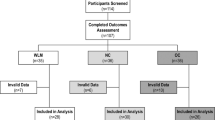
Forty-five (90%) participants had 6-month weight loss data and sufficient baseline accelerometer wear time. On average, participants were middle-aged (45.2 ± 11.3 years old) and had class III obesity before MBS (46.4 ± 7.7 kg/m2). Most identified as women (88.9%), White (59.1%), and non-Hispanic (82.2%). Nine (20.0%) patients underwent RYGB and 36 (80.0%) underwent SG. Surgery type was controlled for in the regression models. Other variables were not significant and were not included.
Aims 1 and 2. On average, participants had 447.3 ± 70.7 sleep, 583.7 ± 100.9 SB, 366.0 ± 75.3 LPA, and 42.6 ± 23.0 MVPA minutes/day at pre-MBS over 9.4 ± 2.4 days. Mean 6-month TWL was 23.8 ± 5.1%. Pre-MBS time-use composition was associated with 6-month %TWL (F = 2.66, p = 0.047).
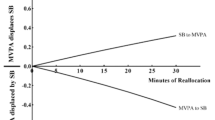
Aim 3. As shown in Table 1 and Fig. 1, per the estimated models, shifting 15–60 min of SB or LPA to MVPA was associated with greater 6-month %TWL (all comparisons were statistically significant at p < 0.05). Conversely, shifting 15 or 30 min of MVPA to SB or LPA was associated with less %TWL (all comparisons p < 0.05). All other reallocations between movement behaviors and %TWL were statistically non-significant.
Each panel within the figure depicts a separate time-use reallocation model. The labels along the top show the 24-h time use behavior that was increased (reallocated to) for each model, and the labels along the right side show the time use behavior that was decreased (reallocated from) for each model. The x-axis at the bottom shows the number of minutes for each modeled reallocation. The y-axis on the left side shows estimated 6-month post-MBS percentage total weight loss. The plotted values within each panel show the estimated change in percentage total weight loss for each modeled reallocation with 95% confidence intervals. SB sedentary behavior, LPA light-intensity physical activity, MVPA moderate-to-vigorous intensity physical activity, TWL total weight loss.
Discussion
This study is the first to use compositional data analysis to evaluate whether time spent in movement behaviors across a 24-h day before MBS relates to early postoperative weight loss. Unlike earlier studies that focused on individual 24-h movement behaviors and associations with weight loss without considering the proportional time spent in other behaviors [1,2,3,4,5], we found that the overall 24-h composition of time in sleep, SB, LPA, and MVPA before MBS was associated with weight loss afterward. Additionally, our findings indicated that certain modeled time reallocations among these behaviors were associated with estimated differences in weight loss at 6-month post-MBS.
Reallocating 15–60 min per day from SB or LPA to MVPA before MBS was estimated (modeled) to relate to greater 6-month postoperative weight loss (up to 3.5% greater %TWL), while reallocating time away from MVPA was estimated to relate to less 6-month postoperative weight loss (up to 5.0% less %TWL). Although these results do not reflect experimental manipulation of these movement behaviors, they suggest the need for further research to determine whether even modest shifts in time-use patterns—such as replacing 15 min of SB or LPA with MVPA, which may be more feasible in this population—could lead to clinically meaningful changes in postoperative weight loss.
Our findings align with recent research in nonsurgical samples, which shows meaningful changes in obesity and cardiometabolic health indicators with small actual or predicted time reallocations (e.g., 10 min), with more pronounced benefits with larger shifts [21, 22, 23]. It is noteworthy that replacing MVPA with lower-intensity behaviors was estimated to have a stronger, negative association with weight change; for example, replacing 30 min of MVPA with LPA was associated with an estimated 5% less %TWL. These findings suggest that reallocating time to and especially away from MVPA before MBS may influence weight loss during the early postoperative period, when MBS has its greatest impact on body weight, although experimental verification is needed.
Results also showed that participants’ observed (measured), pre-surgical time-use patterns related to 6-month postoperative weight loss. The reasons why preoperative movement patterns relate to early postoperative weight loss are not yet fully understood. However, individuals with more favorable movement patterns may have higher total energy expenditure, be healthier overall, face lower risks of postoperative complications, and be more inclined to follow a healthier lifestyle that promotes more weight loss after MBS. Additional research is needed to examine these possibilities while accounting for potential confounders, as well as to explore how shifts in time-use composition and movement behaviors relate to other clinical- and patient-centered outcomes.
Study strengths include device-based measurement of 24-h movement behaviors, calculation of 24-h time-use patterns from multiple days of data, rigorous statistical methods that account for the interdependence of these behaviors, measured weight, and a diverse sample. However, certain limitations must be considered: our allocation analyses relied on estimated (statistically modeled) rather than observed effects; without experimental testing, it remains uncertain whether, for example, reallocating time from SB or LPA to MVPA before MBS indeed increases %TWL after MBS. The modest sample size also limited the investigation of changes in movement behaviors or their associated with %TWL beyond six months post-surgery. Moreover, this study did not assess causal pathways, potential mechanisms driving the observed relationships, eating behaviors or other factors that could influence weight loss after MBS, the role of 24-h movement behaviors in other clinical and patient-centered outcomes, or the context and other aspects of these behaviors (e.g., sleep quality or prolonged versus dispersed SB) that might affect weight loss and general health. Future work that addresses these gaps, including larger samples with more men, is needed to confirm these findings and improve generalizability.
In conclusion, this study is the first to examine how patients undergoing MBS allocate their time to different movement behaviors over the 24-h day before MBS using a compositional approach and to model how time-use changes may relate to postoperative weight loss. Both the overall, observed 24-h composition and the modeled reallocation of time to and from MVPA—including smaller increments (e.g., 15 min), which may be more achievable for this population—were associated with early weight loss after MBS. These findings highlight the importance of expanding beyond an exercise-focused perspective and adopting a holistic view of movement in the context of MBS to better understand how movement behaviors intersect and evolve over time, potentially enhancing and sustaining clinical and health outcomes.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
King WC, Hinerman AS, White GE, Courcoulas AP, Saad MAB, Belle SH. Associations between physical activity and changes in weight across 7 years after Roux-en-Y gastric bypass surgery: a multicenter prospective cohort study. Ann Surg. 2022;275:718–26.
Herman KM, Carver TE, Christou NV, Andersen RE. Keeping the weight off: physical activity, sitting time, and weight loss maintenance in bariatric surgery patients 2 to 16 years postsurgery. Obes Surg. 2014;24:1064–72.
Browning MG, Baugh NG, Wolfe LG, Kellum JK, Maher JW, Evans RK. Evaluation of pre-and postoperative physical activity participation in laparoscopic gastric banding patients. Obes Surg. 2014;24:1981–6.
Nielsen MS, Alsaoodi H, Hjorth MF, Sjödin A. Physical activity, sedentary behavior, and sleep before and after bariatric surgery and associations with weight loss outcome. Obes Surg. 2021;31:250–9.
Santos C, Carvalho M, Oliveira L, Palmeira A, Rodrigues LM, Gregório J. The long-term association between physical activity and weight regain, metabolic risk factors, quality of life and sleep after bariatric surgery. Int J Environ Res Public Health. 2022;19:8328.
King WC, Chen JY, Belle SH, Courcoulas AP, Dakin GF, Elder KA, et al. Change in pain and physical function following bariatric surgery for severe obesity. JAMA. 2016;315:1362–71. 5.
Ross R, Chaput J-P, Giangregorio LM, Janssen I, Saunders TJ, Kho ME, et al. Canadian 24-hour movement guidelines for adults aged 18–64 years and adults aged 65 years or older: an integration of physical activity, sedentary behaviour, and sleep. Appl Physiol Nutr Metab. 2020;45:S57–S102.
Chastin SF, Palarea-Albaladejo J, Dontje ML, Skelton DA. Combined effects of time spent in physical activity, sedentary behaviors and sleep on obesity and cardio-metabolic health markers: a novel compositional data analysis approach. PloS One. 2015;10:e0139984.
Rollo S, Antsygina O, Tremblay MS. The whole day matters: understanding 24-hour movement guideline adherence and relationships with health indicators across the lifespan. J Sport Health Sci. 2020;9:493–510.
Dumuid D, Pedišić Ž, Palarea-Albaladejo J, Martín-Fernández JA, Hron K, Olds T. Compositional data analysis in time-use epidemiology: what, why, how. Int J Environ Res Public Health. 2020;17:2220.
Goldstein SP, Thomas JG, Vithiananthan S, Blackburn GA, Jones DB, Webster J, et al. Multi-sensor ecological momentary assessment of behavioral and psychosocial predictors of weight loss following bariatric surgery: study protocol for a multicenter prospective longitudinal evaluation. BMC Obes. 2018;5:1–12.
Clapp B, Ponce J, Corbett J, Ghanem OM, Kurian M, Rogers AM, et al. American Society for Metabolic and Bariatric Surgery 2022 estimate of metabolic and bariatric procedures performed in the United States. Surg Obes Relat Dis. 2024;20:425–31.
van Hees V, Migueles J, Sabia S, Patterson MR, Fang Z, Heywood J, et al. GGIR: raw accelerometer data analysis. R package version 3.2-1. 2025. https://doi.org/10.5281/zenodo.1051064, https://CRAN.R-project.org/package=GGIR.
Van Hees VT, Sabia S, Anderson KN, Denton SJ, Oliver J, Catt M, et al. A novel, open access method to assess sleep duration using a wrist-worn accelerometer. PloS One. 2015;10:e0142533.
van Hees V, Sabia S, Jones S, Wood A, Anderson K, Kivimaki M. Estimating sleep parameters using an accelerometer without sleep diary. Sci Rep. 2018;8:12975.
Kamada M, Shiroma EJ, Harris TB, Lee I-M. Comparison of physical activity assessed using hip-and wrist-worn accelerometers. Gait Posture. 2016;44:23–28.
Schumacher LM, Thomas JG, Vithiananthan S, Webster J, Jones DB, Bond DS. Prolonged sedentary time adversely relates to physical activity and obesity among preoperative bariatric surgery patients. Surg Obes Relat Dis. 2020;16:562–7.
Van den Boogaart KG, Tolosana-Delgado R. Compositions”: a unified R package to analyze compositional data. Comput Geosci. 2008;34:320–38.
Dumuid D, Pedišić Ž, Stanford TE, Martín-Fernández J-A, Hron K, Maher CA, et al. The compositional isotemporal substitution model: a method for estimating changes in a health outcome for reallocation of time between sleep, physical activity and sedentary behaviour. Stat Methods Med Res. 2019;28:846–57.
Stanford T, Lund Rasmussen C, Dumuid D. codaredistlm: compositional data linear models with composition redistribution. R package version 0.1.0. 2022. https://CRAN.R-project.org/package=codaredistlm.
Gába A, Hartwig TB, Jašková P, Sanders T, Dygrýn J, Vencálek O, et al. Reallocating time between 24-h movement behaviors for obesity management across the lifespan: a pooled data meta-analysis of more than 9800 participants from seven countries. Sports Med. 2025;55:641–54.
Talarico R, Janssen I. Compositional associations of time spent in sleep, sedentary behavior and physical activity with obesity measures in children. Int J Obes. 2018;42:1508–14.
Suorsa K, Gupta N, Leskinen T, Andersen LL, Pasanen J, Hettiarachchi P, et al. Modifications of 24-h movement behaviors to prevent obesity in retirement: a natural experiment using compositional data analysis. Int J Obes. 2023;47:922–30.
Acknowledgements
This work was supported by funding from the National Institute of Diabetes and Digestive and Kidney Diseases (R01DK108579) and the National Heart, Lung, and Blood Institute (L30 HL154167). We thank all study participants.
Author information
Authors and Affiliations
Contributions
LMS helped to conceptualize the study, prepared the data for analysis, and wrote the initial draft of the manuscript. YW performed data analysis, prepared the figure and table, and helped to draft and revise the manuscript. JGT helped to secure funding for and oversee the parent trial, interpret results, and revise the manuscript. AB and PKP helped to interpret results and revise the manuscript. SV and DBJ helped to execute the parent trial, interpret results, and revise the manuscript. JW assisted with execution of the parent trial, including participant recruitment and data collection. DSB helped to conceptualize the study, secure funding for and oversee the parent trial, interpret results, and draft and revise the manuscript.
Corresponding author
Ethics declarations
Competing interests
LMS reports funding from the National Institutes of Health and editorial board membership for Surgery for Obesity and Related Diseases. JGT reports grant funding from the National Institutes of Health, consulting fees from Medifast Inc. and Lummé Health, Inc., and stock options for Lummé Health, Inc. AB reports Salary Awards from the Fonds du recherche en santé du Québec. PP reports consulting fees from Medtronic and Boehringer and leadership or fiduciary roles in the American Society for Metabolic and Bariatric Surgery (Clinical Issues Committee, Research Committee) and The Obesity Society (Bariatric Surgery and Devices Section). SV reports five planned, issued or pending patents related to intestinal flow or obesity treatment. DBJ reports editorial board membership for Surgery for Obesity and Related Diseases and stock in Allurion. DB reports grand funding from the National Institutes of Health and being an associate editor for Surgery for Obesity and Related Diseases and Obesity Surgery. YW and JW report no potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schumacher, L.M., Wu, Y., Thomas, J.G. et al. Preoperative 24-hour movement behaviors and early weight loss after metabolic bariatric surgery: a compositional analysis. Int J Obes 50, 679–683 (2026). https://doi.org/10.1038/s41366-025-01983-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-025-01983-3