Abstract
Background
In Australia, the rising prevalence of metabolic syndrome (MetS) presents a significant public health challenge. However, research on geographic and ethnic disparities remains limited. This study aimed to investigate the prevalence and temporal trends of MetS by geographic remoteness and between Indigenous and non-Indigenous Australians.
Methods
We analysed data from 44,760 adults (aged ≥18 years) derived from the National Health Survey (2014–2015 and 2017–2018) and the National Aboriginal and Torres Strait Islander Health Survey (2012–2013 and 2018–2019). Weighted prevalence estimates of MetS were calculated overall and stratified by remoteness. The Average Annual Rate of Change (AARC) in MetS prevalence was computed to assess temporal trends.
Results
MetS prevalence varied notably by remoteness and ethnicity. In the most recent surveys, 7.1% (95% CI: 6.19–8.19) of Indigenous adults (2018–2019) and 4.6% (95% CI: 4.23–4.99) of non-Indigenous adults (2017–2018) had MetS. Prevalence was higher in remote areas for both groups. Among non-Indigenous adults, MetS declined across most regions but increased in remote areas from 4.5% to 7.1% (AARC: +15.77%), while among Indigenous adults it remained stable in remote areas but rose in major cities and regional settings. Central obesity and type 2 diabetes (T2D) were the most prominent contributors to MetS among Indigenous adults, whereas hypertension and high cholesterol were more prevalent among non-Indigenous adults in regional areas. Central obesity was the most common MetS risk factor, affecting 57.4% (95% CI: 55.14–59.63) of Indigenous and 40.9% (95% CI: 39.90–41.85) of non-Indigenous adults. High cholesterol was the least common risk factor among Indigenous adults (7.6% [95% CI: 6.58–8.67]), whereas elevated blood sugar was the least common among non-Indigenous adults (4.8% [95% CI: 4.44–5.21]).
Conclusions
Substantial disparities in MetS exist across Australia, disproportionately affecting Indigenous Australians and residents of remote areas. Culturally tailored, region-specific interventions targeting obesity are urgently needed through Local Health Districts and Aboriginal Community Controlled Health Organisations.
Similar content being viewed by others
Introduction
Non-communicable diseases (NCDs) are the leading cause of death and disability worldwide, responsible for approximately 43 million deaths annually and accounting for 75% of global mortality [1]. Their rising prevalence places substantial pressure on health care systems, economic productivity, and overall societal well-being [2,3,4,5]. Metabolic syndrome (MetS), a clustering of interrelated metabolic abnormalities, is increasingly recognised as a global public health threat [6,7,8,9]. It is characterised by the presence of at least three out of five cardiometabolic risk factors: abdominal (central) obesity, elevated blood pressure, hyperglycaemia, hypertriglyceridemia, and reduced high-density lipoprotein (HDL) cholesterol levels [6, 10, 11]. Collectively, these factors markedly accelerate the risk of major NCDs, including cardiovascular disease (CVD), type 2 diabetes (T2D), stroke, macular degeneration, and chronic kidney disease (CKD) [12,13,14,15]. Globally, MetS affects approximately one-quarter of adults, and its prevalence is rising in many regions, including Australia [8, 9].
In Australia, the increasing prevalence of MetS is influenced by a complex interplay of socioeconomic, demographic, and lifestyle factors [11, 16, 17]. Recent evidence highlights significant rural–urban disparities in MetS prevalence, with rural and remote areas often disproportionately affected [18,19,20]. These disparities in MetS prevalence are compounded by broader social determinants of health, including limited access to health care services, economic disadvantage, and lifestyle factors associated with rural living (e.g., lower physical activity and higher obesity prevalence) [18]. Australians living in rural and remote areas experience a higher burden of chronic conditions, including obesity, hypertension, and T2D than their urban counterparts, as documented by the Australian Institute of Health and Welfare (AIHW) [18]. Aboriginal and Torres Strait Islander peoples (hereafter respectfully referred to as Indigenous) face an even greater burden as they are 3.3 times more likely to have diabetes than non-Indigenous Australians [18,19,20,21], and tend to develop metabolic risk factors at a younger age (teenage years) [22]. The intersection of ethnicity and remoteness further amplifies health disparities, particularly in remote Indigenous communities. Despite the clinical and public health importance of MetS as a clustering of risk factors, research on its prevalence and geographic distribution in Australia remains insufficient.
Similar patterns have been reported among Indigenous populations in other nations with colonial histories. In Canada, studies among Cree, First Nations, and Métis adults indicate MetS prevalence frequently exceeding 40%, substantially higher than non-Indigenous Canadians [23,24,25]. In New Zealand, population-based surveys and regional cohort studies show Māori and Pacific adults have nearly double the prevalence of MetS compared with people of European descent, with central obesity and dyslipidaemia being key contributors [26,27,28]. In the United States, American Indian and Alaska Native populations, including participants in the Pima and Strong Heart cohorts, record some of the highest adult prevalence worldwide, approaching 60% in certain groups and well above rates observed the general population [29, 30].
The National Health Survey (NHS) and the National Aboriginal and Torres Strait Islander Health Survey (NATSIHS), both conducted by the Australian Bureau of Statistics (ABS), provide nationally representative data that enable the assessment of MetS prevalence across diverse population groups and geographic regions in Australia [31, 32]. Although MetS is increasingly recognised as a major public health concern, critical knowledge gaps remain regarding its temporal trends and geographic variation, particularly in rural and remote settings. Most existing studies have focused on urban populations or specific regions, often overlooking the unique health challenges faced by people living outside major cities [33,34,35,36]. To our knowledge, this is the first comprehensive study to examine MetS prevalence and trends across all levels of geographic remoteness in Australia, using nationally representative samples of both Indigenous and non-Indigenous adults. The study aimed to estimate the prevalence of MetS among Indigenous and non-Indigenous Australians across urban, regional and remote areas, and to analyse temporal trends between 2012 and 2019. These findings are intended to provide evidence-based insights to inform public health policies, guide equitable resource allocation, and ultimately reduce the burden of MetS in underserved rural and Indigenous communities across Australia.
Methods
Data sources
We conducted a secondary analysis of data drawn from two nationally representative population health surveys administered by the ABS: the NHS and the NATSIHS. The NHS is a periodic cross-sectional survey of the general Australian population’s health status and health-related behaviours, while the NATSIHS specifically surveys the health of Aboriginal and Torres Strait Islander population. For this study, we included two waves of each survey that contained relevant metabolic health data: the 2014–2015 and 2017–2018 NHS, and the 2012–2013 and 2018–2019 NATSIHS. Although the ABS released the 2022–2023 NHS and NATSIHS datasets during our study period, confidential unit record files were not accessible through the ABS DataLab at the time of analysis. Given this restriction and project timelines, we proceeded with the most recent available datasets. Together, these surveys provide a robust, population-wide profile of MetS prevalence and associated risk factors among both Indigenous and non-Indigenous Australian adults.
Sampling design
Both the NHS and NATSIHS employed stratified multistage random sampling methods to ensure national and regional representativeness. In the NHS, private dwellings were randomly selected, and within each household one adult ( ≥ 18 years) and one child (0–17 years) were randomly invited to participate. Proxy interviews were permitted for adults who were unable to respond due to illness, disability, or language barriers. The NATSIHS used a comparable multistage sampling strategy, specifically designed to reflect the diversity of the Aboriginal and Torres Strait Islander communities. The sample comprised two components: a community sample (from discrete Indigenous communities) and a non-community sample (from private dwellings). Operational exclusions were applied to some areas (e.g., Tasmania, and certain non-remote parts of Western Australia and the Northern Territory) to maximise feasibility and sample quality. To ensure geographic representativeness, targeted sampling was conducted in the Torres Strait region. In non-remote households, up to two adults and two children were randomly selected, while in remote households, up to one adult and one child were included per dwelling.
Data collection
Data were collected through structured face-to-face interviews by trained ABS interviewers. The NHS and NATSIHS recorded demographic and socioeconomic characteristics; smoking and alcohol consumption; diet and physical activity; health care access; and self-reported long-term conditions. Where available, standardised physical measurements (height, weight, waist circumference, and blood pressure) were obtained. The NATSIHS additionally included culturally relevant questions to reflect the unique social and environmental contexts of Indigenous communities. Health conditions in both surveys were classified according to the International Classification of Diseases, 10th Revision (ICD-10) to identify the individual components of MetS [37]. Missing physical measurements (height, weight, waist circumference, and blood pressure) were imputed by the ABS using hot-deck donor methods based on respondent characteristics. We undertook no additional imputation, and no laboratory biomarkers were imputed [32, 38].
Selection of the analytic sample
We restricted the analysis to adults aged 18 years and older. Across both waves of the NHS, a total of 40,572 individuals were surveyed. After excluding children (n = 9642) and Indigenous participants (n = 750) to avoid duplication with the NATSIHS dataset, 30,180 non-Indigenous adults were retained for analysis. Similarly, the NATSIHS datasets included 23,526 individuals, and following the exclusion of children (n = 8946), 14,580 Indigenous adults remained eligible for analysis. The final analytic sample comprised 44,760 adults, including 30,180 non-Indigenous and 14,580 Indigenous individuals.
Outcome measurements
The primary outcome of this study was the presence of MetS, defined using criteria adapted from internationally recognised guidelines, including the National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III) and the International Diabetes Federation (IDF) [7, 10, 39]. However, due to limitations in lipid biomarker (HDL Cholesterol and triglycerides) availability across survey cycles, in this study MetS was defined using only four of the five interrelated cardiometabolic risk factors: (1) central obesity, defined as a waist circumference of ≥94 cm for males and ≥80 cm for females; (2) T2D, defined as meeting any of the following criteria: a self-reported diagnosis of diabetes (excluding gestational diabetes), self-reported elevated blood glucose levels previously diagnosed by a health professional, or current use of glucose-lowering medication, consistent with ICD-10 code E11 [40]; (3) hypertension, defined as a self-reported diagnosis of hypertension, measured systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg, or current use of antihypertensive medication; and (4) high cholesterol, defined as a self-reported diagnosis of hyperlipidaemia (ICD-10 code E78) [40] or current use of lipid-lowering medication.
Exposure of interest
The primary exposure variable in this study was geographic remoteness, defined according to the Australian Statistical Geography Standard (ASGS) Remoteness Structure developed by the ABS [41]. The ASGS categorises areas into five standard levels of remoteness: Major Cities, Inner Regional, Outer Regional, Remote, and Very Remote. To ensure sufficient statistical power across strata, and in line with ABS reporting practices, the Remote and Very Remote categories were combined into a single group. Accordingly, the final exposure variable included four categories: Major Cities, Inner Regional, Outer Regional, and Remote. This variable was used to examine spatial variation in the prevalence of MetS across different levels of service accessibility and population distribution.
Statistical analysis
All statistical analyses were conducted using Stata version 18.0 [42]. Survey weights provided by the ABS were applied to account for the complex survey design, differential selection probabilities, and non-response. Weighted analyses were conducted separately for the NHS and NATSIHS to generate nationally representative estimates of MetS prevalence among non-Indigenous and Indigenous populations, respectively. Both the NHS and NATSIHS employed multistage, stratified sampling designs, and provided corresponding survey weights. These weights were incorporated to adjust for unequal sampling probabilities and participant non-response, ensuring that prevalence estimates accurately reflected the national population distributions [38]. Population-weighted estimates of MetS prevalence were calculated for each survey year, stratified by Indigenous status and geographic remoteness. To assess temporal trends, the AARC in MetS prevalence was estimated between the earliest and most recent survey cycles for each subgroup. The AARC was calculated using a method adapted from the United Nations Children’s Fund (UNICEF) approach [43], which assumes a constant annual percentage change over time. According to this method, the prevalence is denoted as Yt and the AARC remains constant at b%, the prevalence of nth year (Yt + n) can thus be calculated:
With the estimated slope β, we computed average annual rate of reduction (AARR) = 1 − exp (β), and average annual rate of increment (AARI) as AARI = exp (β) − 1. Therefore, an AARC with a negative value was reported as a reduction, while a positive value was regarded as an increment.
Ethics
This study was approved by the Charles Sturt University Human Research Ethics Committee (Protocol number #2023-118). The NHS and NATSIHS surveys were conducted with informed consent and under ethical oversight. De-identified data were accessed through the secure ABS DataLab. All analyses adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline for cross-sectional studies [44].
Results
Characteristics of study participants
A total of 44,760 adults were included in the study, comprising 30,180 non-Indigenous and 14,580 Indigenous participants. Overall, non-Indigenous adults represented the majority (60%) of the combined study population. Both groups showed comparable age and sex distributions, with older adults ( ≥ 45 years) comprising the largest proportion in each group and younger adults (18–29 years) the smallest. However, younger adults were more prominently represented among the Indigenous participants (27.1%) compared to the non-Indigenous participants (14.6%). In both populations, females outnumbered males (58.6% of Indigenous and 53.8% of non-Indigenous participants) (Table 1). Geographic distribution of residence varied markedly by Indigenous status: most non-Indigenous participants resided in Major Cities (64.3%), with only 2.6% living in Remote areas. In contrast, Indigenous participants were far more likely to live in Remote areas (41.8%) than in Major Cities (21.1%) (Table 1).
MetS prevalence and risk factors in the latest survey period
In the latest survey cycle for each population, the overall prevalence of MetS was higher among Indigenous adults than non-Indigenous adults. MetS affected 7.1% (95% CI: 6.19–8.20) of Indigenous Australians in 2018–2019, compared to 4.6% (95% CI: 4.23–4.99) of non-Indigenous Australians in 2017–2018 (Table 2). Among the individual MetS risk factors, central obesity was the most prevalent component in both groups. In 2018–2019, 57.4% (95% CI: 55.14–59.63) of Indigenous adults had central obesity, compared to 40.9% (95% CI: 39.90–41.85) of non-Indigenous adults in 2017–2018. In contrast, high cholesterol was the least common risk factor among Indigenous adults (7.6%, 95% CI: 6.58–8.67), while elevated blood sugar (T2D) was the least common among non-Indigenous adults (4.8%, 95% CI: 4.44–5.21) (Table 2). Notably, the prevalence of diagnosed elevated blood sugar was more than twice as high in Indigenous adults (9.9%, 95% CI: 8.85–11.06) than in non-Indigenous adults (4.8%, 95% CI: 4.44–5.21). The prevalence of hypertension was similar between groups (13.9% in Indigenous vs. 13.7% in non-Indigenous) (Table 2).
Geographic variations in MetS prevalence and risk factors
MetS prevalence varied significantly by geographic remoteness in both populations. Overall, rural and remote areas had higher MetS prevalence than urban areas for both Indigenous and non-Indigenous Australians (Table 3). Among non-Indigenous adults in 2017–2018, the MetS prevalence ranged from 4.3% (95% CI: 3.9–4.8) in Major Cities to 7.1% (95% CI: 3.9–12.6) in Remote areas. The Indigenous population showed a somewhat different pattern: in 2018–2019 the highest MetS prevalence was observed in Inner Regional areas (8.1%, 95% CI: 6.2–10.4), slightly exceeding the Remote area prevalence (7.8%, 6.5–9.4), while the lowest was in Major Cities (5.9%, 4.5–7.8). Despite minor differences in the rank order of prevalence, both Indigenous and non-Indigenous groups exhibited their lowest MetS prevalence in Major Cities and higher rates in all non-urban regions (Table 3).
Central obesity exhibited a geographic distribution that was similar to overall MetS, with a higher prevalence observed in rural and remote areas. Among non-Indigenous adults, central obesity prevalence in 2017–2018 was highest in Remote areas at 48.5% (95% CI: 41.3–55.7) and lowest in Major Cities at 38.4% (37.3–39.6). Indigenous adults in 2018–2019 had the highest central obesity prevalence in Inner Regional areas (61.1%, 95% CI: 56.4–65.6), with the lowest in Major Cities (55.3%, 95% CI: 51.0–59.6) (Table 3). Notably, even in urban settings, more than half of Indigenous adults were affected by central obesity. Other MetS risk factors showed a similar urban–rural gradient, with higher prevalences reported outside of Major Cities for both populations. For example, T2D and hypertension were more common in Regional and Remote areas than in Major Cities (Table 3). An exception to the overall pattern was observed for hypercholesterolaemia, which showed lower prevalence in Remote areas than in Major Cities. In Remote regions, high cholesterol affected 6.5% (95% CI: 5.5–7.8) of Indigenous and 6.4% (95% CI: 3.5–11.4) of non-Indigenous adults, compared to 7.6% of those in Major Cities (Indigenous: 95% CI: 5.97–9.67; non-Indigenous: 95% CI: 7.02–8.19) (Table 3).
Geographic trends in MetS and its risk factors by Indigenous status
The prevalence of MetS demonstrated contrasting temporal trends by Indigenous status and geographic distribution. Among non-Indigenous adults, the prevalence of MetS declined from 5.5% in 2014–2015 to 4.6% in 2017–2018 (AARC: –6.07%), despite a slight increase in central obesity during the same period. This overall decline was primarily driven by modest reductions in T2D, hypertension, and particularly high cholesterol (Table 2). In contrast, MetS prevalence increased among Indigenous adults, rising from 4.1% in 2012–2013 to 7.1% in 2018–2019 (AARC: +9.59%) (Table 2). Central obesity remained the most prevalent MetS risk factor in both populations, increasing slightly from 39.9% to 40.9% among non-Indigenous adults (AARC: +0.78%) and from 55% to 57.4% among Indigenous adults (AARC: +0.71%) (Table 2). The prevalence of T2D remained relatively stable over the study period among both Indigenous adults (9.8% to 9.9%) and non-Indigenous adults (5.0% to 4.8%) (Table 2). While there was a slight numerical decrease among non-Indigenous adults, the magnitude of change in both groups was minimal and unlikely to be clinically significant. Hypertension increased notably in Indigenous adults (9.5% to 13.9%; AARC: +6.5%) but declined modestly in non-Indigenous adults (14.6% to 13.7%; AARC: –2.06%). High cholesterol declined among non-Indigenous adults (9.1% to 7.8%; AARC: –4.98%) but rose markedly among Indigenous adults (4.7% to 7.6%; AARC: +8.17%) (Table 2).
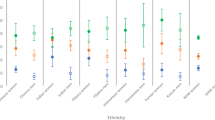
MetS prevalence increased with remoteness and showed widening disparities across population groups. In 2018–2019, prevalence was higher in Remote areas than in Major Cities for both Indigenous (7.8% vs. 5.9%) and non-Indigenous (7.1% vs. 4.3%) adults (Table 3). The most contrasting trends were observed in Inner Regional areas: Indigenous prevalence rose from 3.3% to 8.1% (AARC: +16.33%), while non-Indigenous prevalence declined from 7.1% to 5.3% (AARC: –9.69%) (Table 3). In Major Cities, Indigenous prevalence more than doubled (2.5% to 5.9%), while non-Indigenous prevalence declined modestly (5.1% to 4.3%). In Remote areas, MetS prevalence rose substantially among non-Indigenous adults (4.5% to 7.1%; AARC: +15.77%) but remained stable in Indigenous adults (7.9% to 7.8%; AARC: –0.34%) (Table 3). In the most recent surveys, Indigenous adults exhibited higher MetS prevalence than non-Indigenous adults in all geographic regions, with the largest disparities observed in regional areas (Fig. 1).
Prevalence estimates of metabolic syndrome, central obesity, type 2 diabetes, hypertension, and high cholesterol among Indigenous and non-Indigenous Australian adults across Major Cities, Inner Regional, Outer Regional, and Remote areas, based on data from the National Health Survey (2014–2015 and 2017–2018) and the National Aboriginal and Torres Strait Islander Health Survey (2012–2013 and 2018–2019).
Major MetS risk factors varied across regions and populations. Central obesity increased slightly in most areas, with Indigenous adults consistently bearing the highest burden. Among non-Indigenous adults, the largest increase occurred in Remote areas (41.3% to 48.5%; AARC: +5.44%), while the most notable rise among Indigenous adults was observed in Major Cities (52.0% to 55.3%; AARC: +1.04%) (Table 3). The prevalence of T2D declined in non-Indigenous adults across most regions, especially in Inner Regional areas (6.0% to 5.2%; AARC: –4.41%) but increased in Remote areas (6.4% to 7.5%; AARC: +5.72%). Among Indigenous adults, T2D prevalence rose in Inner Regional (9.5% to 10.6%; AARC: +1.98%) and Remote areas (11.9% to 13.0%; AARC: +1.48%), while declining slightly in Major Cities (Table 3). Hypertension declined across all regions among non-Indigenous adults, with the largest reduction observed in Inner Regional areas (19.7% to 17.1%; AARC: –4.51%). In contrast, hypertension prevalence increased considerably among Indigenous adults, particularly in Inner Regional areas (6.8% to 15.0%; AARC: +14.0%). A slight decrease was observed in Remote areas among Indigenous adults, where hypertension fell from 17% to 16% (AARC: –1.02%) (Table 3). High cholesterol also declined in all regions among non-Indigenous adults, with the most pronounced drop in Remote areas (8.1% to 6.4%; AARC: –7.8%). Conversely, Indigenous adults experienced increases in high cholesterol across all areas except Remote areas, with the sharpest rise in Outer Regional areas (3.9% to 8.5%; AARC: +14.08%). Notably, remote areas had the lowest cholesterol prevalence for both populations by the latest survey years, contrasting with the distribution of other MetS risk factors (Fig. 1).
Discussion
This study presents the first nationally representative analysis of MetS prevalence, major risk factors, trends and geographic variation among Indigenous and non-Indigenous Australians. By integrating data from the NHS and NATSIHS across multiple time points, we found significant disparities in MetS prevalence and its associated risk factors, particularly by ethnicity and remoteness. Indigenous Australians consistently exhibited a higher burden of MetS and its components, especially central obesity and self-reported T2D, compared to non-Indigenous Australians. Between 2012–2013 and 2018–2019, MetS prevalence increased among Indigenous adults, whereas non-Indigenous adults experienced a modest decline, a divergence of particular concern given the established association between MetS and elevated risk of CVD, T2D, macular degeneration, and CKD [6, 9, 45]. These findings are consistent with national reports documenting persistently higher rates of chronic disease among Indigenous Australians and in rural and remote communities [46,47,48]. Central obesity emerged as the most prevalent MetS component in both populations, affecting more than half of Indigenous adults and exceeding the global average of 41% [49, 50]. This disproportionate burden represents a significant public health threat for Indigenous communities.
Central abdominal obesity is a critical driver of insulin resistance and cardiometabolic disease [51]. Waist circumference, as used in this study, is a stronger indicator of visceral adiposity and metabolic risk than body mass index (BMI) [52]. The disproportionately high prevalence among Indigenous adults reflects longstanding inequalities in nutrition, physical activity, and access to preventive healthcare [47, 53]. Beyond individual health behaviours, historical trauma, intergenerational disadvantage, and colonisation-related disruption of cultural practices have also contributed to this burden. A shift from traditional food systems to energy-dense, nutrient-poor products, reinforced by high food prices and constrained access to affordable fresh produce in many remote and regional communities, has further driven central adiposity [48, 54,55,56]. Overcrowded and insecure housing, inadequate access to safe spaces for physical activity, and chronic psychosocial stress reduce opportunities for healthy living and are associated with greater waist circumference and elevated cardiometabolic risk [57, 58]. Given the central role of abdominal obesity in the pathophysiology of MetS, these findings emphasise the urgent need for targeted, culturally safe interventions in Indigenous communities to reduce this burden.
T2D prevalence was substantially higher among Indigenous adults and remained relatively stable over time, in contrast to the slight decline observed among non-Indigenous adults. This persistent disparity reflects the well-documented, disproportionate burden of diabetes among Indigenous Australians [18,19,20,21]. The highest prevalence was observed in remote Indigenous communities, reaching 13%, which is nearly double the prevalence reported in major cities. This geographic gradient reflects broader patterns in the distribution of chronic disease and is shaped by structural inequities such as limited health care access, food insecurity, and substandard living conditions [47, 59]. Addressing these entrenched challenges requires community-led, locally tailored interventions that target both behavioural risk factors and the social determinants of health.
Geographic disparities in MetS prevalence were most pronounced in remote communities. While non-Indigenous adults experienced improvements across most remoteness categories, prevalence increased notably in remote areas. In contrast, Indigenous adults exhibited a substantial rise in inner regional and urban areas, while prevalence remained relatively stable in remote regions. These patterns differ from global trends, where urban populations typically show higher MetS prevalence due to greater exposure to sedentary lifestyles and processed foods [60]. In the Australian context, this inverse gradient likely reflects persistent socio-structural inequalities, including limited access to health care, food insecurity, and suboptimal dietary practices in remote communities [18]. These challenges are particularly pronounced among Indigenous populations [47, 59, 61]. The steeper increase among Indigenous adults in inner regional areas may reflect rapid nutrition and lifestyle transitions, the influence of obesogenic retail food environments, and less consistent access to culturally safe preventive care compared with that available in major cities [54, 62, 63]. Greater uptake of Indigenous-specific Medicare-funded health checks in regional settings relative to very remote areas, may also have contributed to higher detection of metabolic abnormalities [64]. In very remote communities, where obesity and T2D are already highly prevalent, there may be limited scope for further measurable increases [47].
Culturally appropriate, community-led nutritional interventions are urgently needed to address the growing burden of MetS among Indigenous Australians and those living outside major cities, particularly in rural and remote areas. Programmes promoting healthy diets, physical activity, and routine health screening should be co-designed and implemented with Aboriginal Community Controlled Health Organisations (ACCHOs) and Local Health Districts (LHDs), which are well-positioned to deliver culturally safe and effective care [65, 66]. Evidence shows that interventions grounded in traditional knowledge such as incorporating bush foods, community gardens, and Indigenous games, can enhance engagement and promote sustained behaviour change [54]. In regional and remote non-Indigenous populations, where MetS and its components have risen, tailored prevention is equally important. These communities may be underserved by urban-centric prevention strategies. Expanding rural health services, scaling up telehealth, and investing in region-specific health promotion could help reverse these trends. Addressing broader social determinants, such as poverty, housing insecurity, and limited educational opportunities, is fundamental. A multisectoral approach that integrates health, education, agriculture, and housing sectors is required to overcome structural barriers to healthy living for both Indigenous and non-Indigenous Australians [48, 67].
Geographic variation in MetS prevalence calls for regionally tailored policy response. In major cities, where prevalence is lower but rising among Indigenous adults, priorities include weight management, culturally appropriate nutritional support, and improved preventive screening through ACCHOs. In inner regional areas, where the sharpest increases were observed, strengthening chronic disease programmes and expanding culturally safe primary care are essential. In outer regional areas, where service availability is constrained, workforce retention, telehealth expansion, and school and community-based health promotion should be emphasised. In remote areas, poor outcomes are compounded by workforce instability, high food costs, and inadequate infrastructure [68, 69]. Policy strategies should therefore extend beyond food supply and housing to ensure local access to core primary health care, even in very small communities. Evidence indicates that chronic and metabolic disease management is most effective when delivered by resident primary health care providers, even in smaller communities, and guided by principles of equity, cultural safety, and accessibility of care [48, 68, 69].
Between 2012 and 2019, MetS prevalence increased among Indigenous Australians but declined modestly among non-Indigenous adults, reflecting the combined influence of structural determinants, behavioural risk factors, and health system changes. Among Indigenous populations, overweight and obesity rose from 66% in 2012–2013 to 74% in 2018–2019 among adults, suggesting a growing burden of metabolic risk during this period, although causality cannot be inferred due to the cross-sectional design [70, 71]. This trend occurred within a context of entrenched socioeconomic disadvantage, food insecurity, poor availability and affordability of nutritious foods, and systemic inequities recognised as key determinants of obesity and T2D [48]. During the same period, uptake of Medicare funded annual health assessments for Indigenous Australians expanded, improving detection of hypertension, diabetes, and dyslipidaemia and likely contributing to the higher recorded prevalence of MetS [64]. In contrast, the modest decline observed among non-Indigenous adults aligns with improved risk factor management, including reductions in self-reported high cholesterol from 7.1% in 2014–2015 to 6.1% in 2017–2018, supported by sustained dispensing of statins and other lipid-lowering medicines between 2013 and 2019 [72, 73]. Overall, these findings suggest that rising obesity, persistent social disadvantage, and enhanced case detection are intensifying metabolic risk among Indigenous Australians, while improved clinical management has stabilised prevalence among non-Indigenous adults, underscoring the need to reduce inequities while sustaining progress in the broader population.
Strengths and limitations
This study leveraged large, nationally representative datasets (NHS and NATSIHS) and conducted stratified analyses by remoteness and ethnicity, providing valuable insights into geographic and population-based disparities in MetS. The use of consistent data collection methods across survey years enhances comparability, and the application of population weights and the calculation of average annual rate of change (AARC) add rigour to trend analysis.
However, several limitations should be acknowledged when interpreting these findings. First, the reliance on self-reported data for conditions such as diabetes (elevated blood glucose), high cholesterol, and partly for blood pressure may have introduced misclassification. These conditions are prone to both underestimation and overestimation due to recall error and social desirability bias, with self-reported blood pressure particularly vulnerable. These reporting and measurement limitations may affect estimates for individual conditions and, in turn, contribute to misclassification of MetS status, because classification requires multiple risk factors. National estimates suggest that up to 30% of people living with diabetes in Australia remain undiagnosed, underscoring that self-reported survey data may not fully capture the true burden of disease, particularly in underserved populations [74]. Second, although survey weights accounted for the complex sampling design and unequal selection probabilities by remoteness, the survey selected up to two adults per household in non-remote areas and one adult in remote areas. This differential within-household sampling may introduce systematic bias, particularly if multiple individuals in remote households share similar cardiometabolic risk profiles. Such design features could result in underestimation of MetS prevalence in remote populations, especially where risk factors are known to cluster. Third, the cross-sectional design of the NHS and NATSIHS precludes causal inference and limits the ability to observe individual level changes over time. Fourth, the exclusion of very remote communities from the NHS sample may have led to an underrepresentation of populations experiencing the most significant health disparities. Finally, our operational definition excluded HDL cholesterol and triglycerides because these biomarkers were not consistently available across survey waves. Standard definitions (ATP III, IDF, and the Joint Interim Statement) define dyslipidaemia as low HDL cholesterol and elevated triglycerides [6, 7, 10, 39], whereas our reliance on a general ‘high cholesterol’ indicator did not fully capture this profile. As a result, some individuals who would qualify under standard criteria were likely missed, making our prevalence estimates conservative and limiting comparability with studies using full lipid panels [10, 39]. Despite these limitations, the use of large, nationally representative datasets, together with a consistent methodology across survey waves, strengthens the validity of the findings and supports their relevance for guiding public health policy and targeted intervention strategies.
Conclusion
This study highlights significant disparities in MetS prevalence across Australia, with Indigenous Australians and residents of regional and remote areas bearing the greatest burden. Among non-Indigenous adults, prevalence declined overall but rose in remote areas. Among Indigenous adults, prevalence increased in Major Cities and in inner and outer regional areas, whereas it remained stable in remote communities. Central obesity and T2D were the principal contributors to MetS among Indigenous adults, whereas hypertension and high cholesterol were more common among non-Indigenous adults in regional areas. These findings highlight the urgent need for culturally responsive and geographically targeted public health strategies. Priorities include strengthening partnerships with LHD and ACCHOs, improving access to affordable nutritious food, promoting physical activity, and expanding preventive care with consistent follow-up after Medicare-funded Indigenous health checks, alongside stronger continuity of care between urban, regional, and remote services. Future surveillance should incorporate lipid biomarkers and include very remote communities, guided by Indigenous data governance, to enable accurate monitoring. Reducing the burden of MetS in Australia requires coordinated public health action that addresses central obesity, T2D, and hypertension, and delivers region-specific prevention and culturally safe primary care across urban, regional, and remote settings for both Indigenous and non-Indigenous populations.
Data availability
This study used data from two surveys conducted by the Australian Bureau of Statistics (ABS): the National Health Survey (NHS) and the National Aboriginal and Torres Strait Islander Health Survey (NATSIHS). Weighted estimates from both surveys are available on the ABS website. Access to the de-identified unit record microdata is provided through the secure ABS DataLab to approved users, and all outputs are reviewed by ABS prior to release.
References
WHO. Noncommunicable diseases. 2023. https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases.
Bloom DE, Cafiero E, Jané-Llopis E, Abrahams-Gessel S, Bloom LR, Fathima S, et al. The global economic burden of noncommunicable diseases. Program on the Global Demography of Aging; (Geneva, Switzerland, 2012).
Chaker L, Falla A, van der Lee SJ, Muka T, Imo D, Jaspers L, et al. The global impact of non-communicable diseases on macro-economic productivity: a systematic review. Eur J Epidemiol. 2015;30:357–95.
Jan S, Laba T-L, Essue BM, Gheorghe A, Muhunthan J, Engelgau M, et al. Action to address the household economic burden of non-communicable diseases. Lancet. 2018;391:2047–58.
World Health Organization. Noncommunicable diseases 2021. 2024. https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases.
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5.
Alberti KGM, Zimmet P, Shaw J. The metabolic syndrome—a new worldwide definition. Lancet. 2005;366:1059–62.
Noubiap JJ, Nansseu JR, Lontchi-Yimagou E, Nkeck JR, Nyaga UF, Ngouo AT, et al. Geographic distribution of metabolic syndrome and its components in the general adult population: a meta-analysis of global data from 28 million individuals. Diab Res Clin Pract. 2022;188:109924.
Saklayen MG. The global epidemic of the metabolic syndrome. Curr Hypertens Rep. 2018;20:1–8.
IDF. The IDF consensus worldwide definition of the metabolic syndrome. International Diabetes Federation. (Brussels, Belgium, 2006).
Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Circulation. 2005;112:2735–52.
Cameron AJ, Shaw JE, Zimmet PZ. The metabolic syndrome: prevalence in worldwide populations. Endocrinol Metab Clin. 2004;33:351–75.
Mottillo S, Filion KB, Genest J, Joseph L, Pilote L, Poirier P, et al. The metabolic syndrome and cardiovascular risk: a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56:1113–32.
Wilson PW, D’Agostino RB, Parise H, Sullivan L, Meigs JB. Metabolic syndrome as a precursor of cardiovascular disease and type 2 diabetes mellitus. Circulation. 2005;112:3066–72.
Cameron AJ, Magliano DJ, Zimmet PZ, Welborn T, Shaw JE. The metabolic syndrome in Australia: prevalence using four definitions. Diab Res Clin Pract. 2007;77:471–8.
Harris MF. The metabolic syndrome. Aust Fam Physician. 2013;42:524–7.
Bankoski A, Harris TB, McClain JJ, Brychta RJ, Caserotti P, Chen KY, et al. Sedentary activity associated with metabolic syndrome independent of physical activity. Diab Care. 2011;34:497–503.
Australian Institute of Health and Welfare. Rural and remote health: AIHW. 2024. https://www.aihw.gov.au/reports/rural-remote-australians/rural-and-remote-health.
Australian Institute of Health and Welfare. Diabetes: Australian facts: AIHW. 2024. https://www.aihw.gov.au/reports/diabetes/diabetes/contents/about.
Australian Institute of Health and Welfare. Diabetes: Australian facts: overweight and obesity: AIHW. 2024. https://www.aihw.gov.au/reports/diabetes/diabetes/contents/diabetes-risk-factors/overweight-and-obesity.
Gubhaju L, McNamara BJ, Banks E, Joshy G, Raphael B, Williamson A, et al. The overall health and risk factor profile of Australian Aboriginal and Torres Strait Islander participants from the 45 and up study. BMC Public Health. 2013;13:1–14.
Welfare AIoHa. Determinants of health: 2.22 Overweight and obesity National Indigenous Australian Agency; 2025 [cited 2025 8 April]. Available from: https://www.indigenoushpf.gov.au/measures/2-22-overweight-and-obesity
Liu J, Hanley AJ, Young TK, Harris S, Zinman B. Characteristics and prevalence of the metabolic syndrome among three ethnic groups in Canada. Int J Obes. 2006;30:669–76.
Kaler SN, Ralph-Campbell K, Pohar S, King M, Laboucan CR, Toth E. High rates of the metabolic syndrome in a First Nations Community in western Canada: prevalence and determinants in adults and children. Int J Circumpolar Health. 2006;65:389–402.
Lavigne-Robichaud M, Moubarac J-C, Lantagne-Lopez S, Johnson-Down L, Batal M, Sidi EAL, et al. Diet quality indices in relation to metabolic syndrome in an Indigenous Cree (Eeyouch) population in northern Québec, Canada. Public Health Nutr. 2018;21:172–80.
Simmons D, Thompson CF. Prevalence of the metabolic syndrome among adult New Zealanders of Polynesian and European descent. Diab Care. 2004;27:3002–4.
Gentles D, Metcalf P, Dyall L, Sundborn G, Schaaf D, Black P, et al. Metabolic syndrome prevalence in a multicultural population in Auckland, New Zealand. N Z Med J. 2007;120.
Yu D, Cai Y, Osuagwu UL, Pickering K, Baker J, Cutfield R, et al. Metabolic profiles of Maori, Pacific, and European New Zealanders with type 2 diabetes over 25 years. Diab Care. 2021;44:e183–5.
Godfrey TM, Cordova-Marks FM, Jones D, Melton F, Breathett K. Metabolic syndrome among American Indian and Alaska Native populations: implications for cardiovascular health. Curr Hypertens Rep. 2022;24:107–14.
Schumacher C, Ferucci ED, Lanier AP, Slattery ML, Schraer CD, Raymer TW, et al. Metabolic syndrome: prevalence among American Indian and Alaska native people living in the southwestern United States and in Alaska. Metab Syndr Relat Disord. 2008;6:267–73.
Statistics ABo. National Health Survey 2022. 2022. https://www.abs.gov.au/statistics/health/health-conditions-and-risks/national-health-survey/latest-release.
Australian Bureau of Statistics. National Aboriginal and Torres Strait Islander Health Survey methodology: ABS. 2024. https://www.abs.gov.au/methodologies/national-aboriginal-and-torres-strait-islander-health-survey-methodology/2022-23.
Esgin T, Hersh D, Rowley KG, Macniven R, Glenister K, Crouch A, et al. Physical activity and self-reported metabolic syndrome risk factors in the Aboriginal population in Perth, Australia, measured using an adaptation of the global physical activity questionnaire (gpaq). Int J Environ Res Public Health. 2021;18:5969.
Dunstan DW, Zimmet PZ, Welborn TA, De Courten MP, Cameron AJ, Sicree RA, et al. The rising prevalence of diabetes and impaired glucose tolerance: the Australian Diabetes, Obesity and Lifestyle Study. Diab Care. 2002;25:829–34.
Zhang H, Rogers K, Sukkar L, Jun M, Kang A, Young T, et al. Prevalence, incidence and risk factors of diabetes in Australian adults aged≥ 45 years: a cohort study using linked routinely-collected data. J Clin Transl Endocrinol. 2020;22:100240.
Mitchell P, Smith W, Wang JJ, Cumming RG, Leeder SR, Burnett L. Diabetes in an older Australian population. Diab Res Clin Pract. 1998;41:177–84.
Brämer GR. International statistical classification of diseases and related health problems. Tenth revision. World Health Stat Q. 1988;41:32–6.
Australian Bureau of Statistics. 4363.0—National Health Survey: Users’ Guide, 2017-18. 2019. https://www.abs.gov.au/ausstats/abs@.nsf/mf/4363.0.
Detection NEPO. Executive summary of the third report of the National Cholesterol Education Program (NCEP)(Adult treatment Panel III). JAMA. 2001;285:2486–97.
WHO. International Statistical classification of diseases and related health problems 10th Revision. 2016.
Australian Bureau of Statistics. Australian Statistical Geography Standard (ASGS): volume 5—remoteness structure Canberra: ABS. 2016. https://www.abs.gov.au/ausstats/abs@.nsf/Latestproducts/1270.0.55.005Main%20Features5July%202016.
StataCorp LLC. Stata statistical software: release 18. College Station, Texas, USA: StataCorp LLC; 2023. https://www.stata.com/company/.
UNICEF. Technical Note: How to calculate average annual rate of reduction (AARR) of underweight prevalence. 2007. https://data.unicef.org/resources/technical-note-calculate-average-annual-rate-reduction-aarr-underweight-prevalence/.
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370:1453–7.
Kassi E, Pervanidou P, Kaltsas G, Chrousos G. Metabolic syndrome: definitions and controversies. BMC Med. 2011;9:1–13.
AIHW. Health and wellbeing of First Nations people. Australian Institute of Health and Welfare; 2024. https://www.aihw.gov.au/reports/australias-health/indigenous-health-and-wellbeing.
AIHW. Aboriginal and Torres Strait Islander: health performance framework summary report in. Australian Institute of Health and Welfare, editor; 2024.
Australian Institute of Health and Welfare. Australia’s health 2020 in brief. Canberra: AIHW; 2020.
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384:766–81.
Wong MC, Huang J, Wang J, Chan PS, Lok V, Chen X, et al. Global, regional and time-trend prevalence of central obesity: a systematic review and meta-analysis of 13.2 million subjects. Eur J Epidemiol. 2020;35:673–83.
Després J-P. Body fat distribution and risk of cardiovascular disease: an update. Circulation. 2012;126:1301–13.
Ross R, Neeland IJ, Yamashita S, Shai I, Seidell J, Magni P, et al. Waist circumference as a vital sign in clinical practice: a Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat Rev Endocrinol. 2020;16:177–89.
Blüher M. Adipose tissue dysfunction in obesity. Exp Clin Endocrinol Diab. 2009;117:241–50.
Lee A, Ride K. Review of nutrition among Aboriginal and Torres Strait Islander people. 2018.
Thurber KA, Banwell C, Neeman T, Dobbins T, Pescud M, Lovett R, et al. Understanding barriers to fruit and vegetable intake in the Australian Longitudinal Study of Indigenous Children: a mixed-methods approach. Public Health Nutr. 2017;20:832–47.
Gracey M, King M. Indigenous health part 1: determinants and disease patterns. Lancet. 2009;374:65–75.
Markwick A, Ansari Z, Sullivan M, Parsons L, McNeil J. Inequalities in the social determinants of health of Aboriginal and Torres Strait Islander People: a cross-sectional population-based study in the Australian state of Victoria. Int J Equity Health. 2014;13:91.
Browne J, de Leeuw E, Gleeson D, Adams K, Atkinson P, Hayes R. A network approach to policy framing: a case study of the National Aboriginal and Torres Strait Islander Health Plan. Soc Sci Med. 2017;172:10–8.
AIHW. Determinants of health for First Nations people. Australian Institute of Health and welfare; 2024. https://www.aihw.gov.au/reports/australias-health/social-determinants-and-indigenous-health.
Kaur J. [Retracted] A comprehensive review on metabolic syndrome. Cardiol Res Pract. 2014;2014:943162.
Browne J, Hayes R, Gleeson D. Aboriginal health policy: is nutrition the ‘gap’in ‘Closing the Gap’? Aust NZ J Public Health. 2014;38:362–9.
Brimblecombe J, Ferguson M, Chatfield MD, Liberato SC, Gunther A, Ball K, et al. Effect of a price discount and consumer education strategy on food and beverage purchases in remote Indigenous Australia: a stepped-wedge randomised controlled trial. Lancet Public Health. 2017;2:e82–95.
Cameron AJ, Charlton E, Ngan WW, Sacks G. A systematic review of the effectiveness of supermarket-based interventions involving product, promotion, or place on the healthiness of consumer purchases. Curr Nutr Rep. 2016;5:129–38.
AIHW. Health checks and follow-ups for Aboriginal and Torres Strait Islander people. Australian Institute of Health and Welfare; 2025. https://www.aihw.gov.au/getmedia/be51e5e1-ffb0-463f-b637-a38008b95393/health-checks-and-follow-ups-for-aboriginal-and-torres-strait-islander-people.pdf?v=20250227103516&inline=true.
Campbell MA, Hunt J, Scrimgeour DJ, Davey M, Jones V. Contribution of Aboriginal Community-controlled health services to improving Aboriginal health: an evidence review. Aust Health Rev. 2017;42:218–26.
Couzos S, Murray R. Aboriginal primary health care: an evidence-based approach. Oxford University Press; 2008.
Australian Government. National Aboriginal and Torres Strait Islander Health Plan 2021–2031. Health Do, editor. Canberra: Commonwealth of Australia as represented by the Department of Health; (South Melbourne, Victoria, Australia, 2021).
Thomas SL, Wakerman J, Humphreys JS. Ensuring equity of access to primary health care in rural and remote Australia-what core services should be locally available? Int J Equity Health. 2015;14:111.
Wakerman J, Humphreys J, Russell D, Guthridge S, Bourke L, Dunbar T, et al. Remote health workforce turnover and retention: what are the policy and practice priorities? Hum Resour Health. 2019;17:99.
Australian Institute of Health and Welfare. Determinants of health: overweight and obesity. AIHW: Australian Government; 2019. https://www.indigenoushpf.gov.au/measures/2-22-overweight-and-obesity.
Statistics ABo. National Aboriginal and Torres Strait Islander Health Survey 2018-19. Australian Bureau of Statistics; 2019. https://www.abs.gov.au/statistics/people/aboriginal-and-torres-strait-islander-peoples/national-aboriginal-and-torres-strait-islander-health-survey/latest-release.
ABS. High cholesterol. Australian Bureau of Statistics; 2018. https://www.abs.gov.au/statistics/health/health-conditions-and-risks/high-cholesterol/2017-18.
Talic S, Hernandez CM, Ofori-Asenso R, Liew D, Owen A, Petrova M, et al. Trends in the utilization of lipid-lowering medications in Australia: an analysis of national pharmacy claims data. Curr Probl Cardiol. 2022;47:100880.
Sainsbury E, Shi Y, Flack J, Colagiuri S. Burden of diabetes in Australia: it’s time for more action. Preliminary Report. (Novo Nordisk, Sydney, Australia, 2018).
Acknowledgements
The authors acknowledge the traditional owners of the lands from which they contributed to this research and pay respects to the Elders, past, present, and emerging. They also thank the Australian Bureau of Statistics (ABS) and its Microdata Access team for facilitating data access through DataLab.
Funding
This study was funded by a grant from the Commonwealth of Australia, represented by the Department of Health and Aged Care (Grant Activity 4-DGEJZ1O/4-CW7UT14). Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
UKM, AGR, and MMH conceptualised the study and led the design, with MMH responsible for data acquisition, conducting analysis, and interpretation. UKM drafted the initial manuscript. MMH, AEA, SCP, BK, FHA, ST, KYA, SM, SBA, FR, MJAS, and AGR critically reviewed and revised the manuscript for important intellectual content. UKM, together with AGR, refined the research objectives and ensured accurate interpretation of the findings. MMH had full access to all raw data and verified its integrity, with additional verification by UKM. All authors meet the ICMJE authorship criteria, contributed significantly to the work, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mondal, U.K., Huda, M.M., Anyasodor, A.E. et al. Metabolic syndrome in Australia: nationwide survey results by remoteness and Indigenous status, 2012–2019. Int J Obes (2026). https://doi.org/10.1038/s41366-025-02013-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41366-025-02013-y