Abstract
Background/Objectives
Amid rising global obesity rates and advances in weight-loss therapies, monitoring body composition and ectopic fat could refine trial design. We quantified weight-related changes in body composition and liver steatosis prior to widespread adoption of incretin treatments.
Subjects/Methods
Adults (N = 3070) from the UK Biobank with repeat abdominal MRI scans were included. Percent weight change from baseline was categorised: stable (0 ± 2%), mild change (2–5% weight gain/loss), moderate change (5–10% weight gain/loss), or large change (10–15% weight gain/loss).
Intervention/Methods
MRI data were processed automatically from two visits, spaced 2.7 years apart, to derive volumetric visceral (VAT), subcutaneous adipose tissue (SAT), total skeletal muscle volume (SM, or indexed SM), muscle fat infiltration (MFISM), and psoas muscle cross-sectional area (CSA) in the abdominal region. Liver fat content (LFC) was assessed using LiverMultiScan. Dual-energy x-ray absorptiometry (DXA) measurements were compared.
Results
Weight gain occurred in 28% of all subjects (N = 3070, age 63 years, male 49%, 13% with obesity, 43% with overweight). Moderate or large weight gain increased LFC, VAT, SAT, MFISM, and SM (all p < 0.001). Weight loss also occurred in 28%. Decreases were observed with moderate or large weight loss in LFC, VAT, SAT, SM, SMI, and psoas CSA (all p < 0.001). MFISM was reduced with large weight loss. For VAT and liver fat, prevalent type 2 diabetes exacerbated weight gain-related increases, and blood pressure medication attenuated the impact of weight change. For individuals with overweight or obesity, for every 5% drop in weight, there was 16% reduction in VAT, 11% in SAT, 24% in liver fat, 2.3% in MFISM, 1.5% in SM (or 1.4% in SMI) and 2.1% in psoas CSA. DXA changes in lean mass correlated weakly with changes in SM volume (rho 0.28–0.47).
Conclusions
Using MRI, relative changes in body composition and liver steatosis resulting from weight loss can inform clinical trials, including placebo arm design and power estimations.

Weight changes affect body composition and liver fat and can inform obesity trial design.
Similar content being viewed by others
Introduction
Obesity is a chronic disease currently affecting an estimated 1 billion adults globally and expected to double by 2050 [1, 2]. The precise manifestation of this disease is influenced by genotype, demographics, and environmental factors [3]. The liver is often an early site of pathological fat accumulation in the form of triglycerides; metabolic dysfunction-associated steatotic liver disease (MASLD) is found in 75% of people with obesity [4]. Excess fat accumulation in visceral adipose tissue (VAT), hepatocytes, and skeletal muscle (SM) is associated with increased risk of poor outcomes [5,6,7,8].
In parallel, there has been an explosion in therapeutic advancements for obesity that lead to weight loss and provide additional management options beyond lifestyle and surgical intervention [9]. These drugs can have pleiotropic cardiometabolic benefits and this is associated with a reduction in ectopic fat and/or VAT [10,11,12,13], including in populations with comorbidities like MASLD. There are, however, growing concerns about concurrent loss of skeletal muscle mass and function [14, 15], which correlates with poor outcomes [16, 17] and warrants further investigation.
A clinical workflow for obesity that includes direct measurement of body fat -not BMI- and signs, symptoms or tests for organ dysfunction or substantial limitations in daily activities was recently recommended by the Lancet Diabetes & Endocrinology Commission on Clinical Obesity [2]. The Commission recommended measuring body fat using serum markers such as triglycerides (TGA) or cholesterol, which lack organ specificity, or by dual-energy X-ray absorptiometry (DXA), which estimates three-dimensional mass from coronal two-dimensional images but has limited accuracy for quantifying VAT [18, 19].
MRI is the gold standard for volumetric assessment of VAT, SAT and SM, with single-slice area methods being applied in clinical practice [20, 21]. Use of multiparametric MRI to measure liver fat content is recognized as the most accurate non-invasive method for liver steatosis staging [22, 23].
We sought to provide a comprehensive evaluation of the relative changes in body composition and liver steatosis resulting from general weight loss to inform clinical trial design and clinical management. We compared changes in multiparametric MRI and DXA parameters in individuals with a broad cardiometabolic profile from the UK Biobank, after weight gain and after weight loss prior to widespread adoption of incretin therapies. We evaluate the impact of medication use and pre-existing diabetes and illustrate the application of these data for powering clinical trials of weight-loss treatments.
Materials and methods
Study design and participants
This prospective cohort study involved participants enrolled in the UK Biobank (UKB) imaging sub-study between May 2014 and March 2020, who attended and completed 2 imaging visits within 3 years, by March 2022 [21]. Individuals aged 40–69 years old were invited for MRI examination (heart and abdomen), including quantitative mapping, as previously described [24]. Some participants were invited for a follow-up if the baseline scan was of sufficient quality, and they lived within certain proximity to an imaging centre. UKB has approval from Northwest Multi-Centre Research Ethics Committee as a Research Tissue Bank and obtained written informed consent from all participants prior to the study. UKB complies with the General Data Protection Regulation (GDPR) and the Data Protection Act 2018 and is guided by an independent Ethics and Governance Council to ensure researchers maintain compliance with UKB policies. Data were extracted under access application 9914 and all methods were performed in accordance with the relevant guidelines and regulations. Those with complete imaging data for MRI VAT, SAT, abdominal SM volume, abdominal SM index (SMI, SM volume adjusted for height), abdominal SM fat infiltration (MFISM, the proportion of intramuscular fat in SM, as a %), psoas muscle (PM) and liver fat content were included in the analysis. A single participant who underwent bariatric surgery between repeat imaging visits was excluded from the analysis, to avoid confounding the results.
DXA measurements from the same participants that had previously been processed with automated methods [25] were also included for comparisons without requiring further analysis; the available measurements were trunk lean mass (Field ID 23285) and total lean mass (Field ID 23280). DXA-derived VAT estimates (Field ID 22407) were excluded due to inconsistent quality.
Data collection
Clinical and biochemical data
Clinical data that included demographics (age, sex, ethnicity, weight, BMI, smoking status), and prior history of diabetes, hypertension, recent cancer (within 2 years of imaging visit) and reported medications, were collected at both imaging visits (from ICD-10 codes; Supplementary methods). Blood biomarker data (TGA, total cholesterol) collected at the baseline visit was also accessed.
MRI data acquisition
All participants had been scanned on two occasions at one of the UK Biobank imaging centres, on a Siemens Aera 1.5 T scanner (Siemens Healthineers, Erlangen, Germany), with both a dual-echo Dixon VIBE protocol for body composition and the LiverMultiScan imaging protocol that are encompassed within the UK Biobank imaging protocol [24, 26].
Body composition measurements from MRI
Body composition measurements were derived using fully automated volumetric analyses, as described previously [21] (Supplementary Methods). The region from the centre of the T9 vertebra to the top of the lower of the two femoral heads was selected for volumetric analyses. Volumes for VAT, SAT, abdominal SM volume and MFISM were calculated based on the number of voxels. MFISM was computed as the mean signal fat fraction value of the muscle tissue voxels. Semi-automated single-slice area analyses at the third lumbar vertebra (L3) level were also conducted, as described previously [21] (Supplementary Methods). Axial slices passing through the centre of mass of the L3 vertebra were automatically extracted from the whole-body volumes. The cross-sectional area (CSA) for psoas muscle was calculated based on the number of pixels.
All analyses were performed by trained MR technologists and radiographers blinded to all clinical data.
Liver measurements from MRI
Liver MRI data analysis was performed automatically using LiverMultiScan® software (Perspectum, Oxford, UK) [27], and every case was manually reviewed by trained analysts, blinded to all clinical data.
Statistical analysis
All statistical analyses were performed in R (version 4.3, R Project for Statistical Computing, Vienna, Austria).
Participants were categorized according to percentage weight change from baseline into the following groups: stable weight (0 ± 2%), mild change (2–5% weight gain/loss), moderate change (5–10% weight gain/loss), or large change (10–15% weight gain/loss). These categories were selected to quantify granularity of measurement given the timeframe of this population study [28] and align with categories of treatment responders in clinical trials of obesity treatments (participants with ≥5% weight loss as co-primary endpoints or ≥10%, ≥15%, ≥20% weight loss as secondary endpoints in clinical trials) [29,30,31,32,33] and recommendations in clinical guidelines for management of overweight/obesity [34, 35]. Individuals were excluded from some analysis due to low sample sizes (Supplementary methods).
Initial analyses of variance (ANOVA) were performed to examine overall differences across weight change categories. Subsequently, within the stable weight reference group, paired sample t-tests were used to assess longitudinal changes in body composition from baseline to follow-up to quantify natural temporal variations. Between-group analyses using independent two-sample t-tests were used to compare each weight change category against the stable weight reference group to isolate effects specifically attributable to weight change. All statistical tests were conducted after confirming that parametric test assumptions were met. To control for multiple comparisons in analyses of changes in MRI-derived measurements of body fat, muscle, and liver fat across weight gain or loss groups, p-values were adjusted using the Bonferroni method. Spearman’s rank correlation test was used for correlation analyses (rho). For the correlations, the Benjamini–Hochberg procedure was applied to control the false discovery rate (FDR) due to multiple comparisons. Linear regressions were performed to evaluate whether relationships between weight-change groups and MRI-derived measurements of body fat, muscle, and liver fat differed by 1) age groups (categorised as 50–59, 60–69, and ≥70 years), 2) use of blood pressure medications, and 3) diabetes status, adjusting for sex. Linear regression was also performed to define rate of change in individual parameters after weight change in a subset of individuals in the study population that were living with overweight or obesity. Changes from this regression analysis, occurring after 5% weight loss, were considered clinically meaningful for study powering, in line with precedent on use of proportion of participants with 5% weight loss as co-primary or secondary endpoints in clinical trials [29,30,31,32,33] and recommendations in clinical guidelines for management of overweight/obesity [34, 35]. A power analysis was conducted to determine the sample size required to detect a two-arm (drug vs. placebo) trial, based on detecting a clinically relevant difference in the relative change from baseline in individuals with overweight/obesity. The standard deviation (SD) of percentage change as observed in the subset of individuals with overweight/obesity in our study population (defined as the ‘virtual placebo’) was used to estimate the specific Cohen’s d (effect size) for each measurement calculated as targeted difference (%)/SD of percentage change. Based on the directional hypothesis for each measurement (drug group relative change > placebo group relative change), a one-sided, two-sample t-test was used for the calculation. For co-primary endpoints a Bonferroni correction was applied for the calculation (Supplementary Methods).
All significance tests, except the power calculation, were two-tailed, and p < 0.05 was considered statistically significant. Data are presented as mean ± SD throughout the text. For figures illustrating mean changes in metrics across weight loss/gain groups, standard error of the mean (SEM) is displayed to improve visualization of between-group differences.
The World Health Organisation standards for BMI stratification were applied [36]. Terminology and diagnostic criteria for what was previously “non-alcoholic fatty liver disease” have changed recently to metabolic dysfunction-associated liver disease and metabolic dysfunction-associated steatohepatitis and are adopted herein [37, 38]. MASLD was defined as the presence of 1 cardiometabolic risk factor (diabetes, obesity, hypertension or hyperlipidaemia) and concurrent elevation in liver fat content (>5%), in the absence of high consistent alcohol intake [39].
Results
Study population
3070 individuals enrolled in the UK Biobank between 2014 and 2020 who had repeat MRI imaging over a mean 32 ± 13 months follow up interval were included (Fig. 1). At the first imaging visit, their mean age was 62 ± 8 years, and their BMI was 26 ± 4 kg/m2. 97% were White, 49% were male and 5% were smokers (Table 1). 13% of individuals were living with obesity, 43% with overweight, 15% with metabolic syndrome and 21% had MASLD. Only 5% had type 2 diabetes (T2D) and 1.8% had a recent cancer diagnosis. 14% were on cholesterol–lowering medication and 13% were on blood pressure medications.
CONSORT diagram indicating how selection of the study population was based on demographic information paired with body composition and liver fat measurements following abdominal MRI at 2 imaging visits.
Over the follow-up period, body weight was stable in 43% (1333 of 3070). 864 individuals (28%) gained >2% of baseline weight over the same period. There was mild weight gain in 577 individuals (19%, gaining 2–5% of their baseline weight), with 227 individuals (7.4%) gaining moderate weight (5-10%), and only 60 individuals (2%) gaining 10–15% of their baseline weight.
Additionally, 18% (538 of 3070) lost 2–5% of their baseline weight and 8.3% (255 individuals) lost moderate weight (5–10%), while 80 (3%) lost 10–15% of their original weight. Individuals who lost weight were more likely to have been overweight or obese and have high waist circumference at baseline (p < 0.001). Large weight loss was not associated with prior history of cancer or new cancer diagnoses (both p = 0.2).
Body fat, muscle and liver MRI measurements in individuals maintaining stable weight
During the follow-up period, people who maintained their weight showed relatively small increase in VAT (relative % change of 5 ± 13%, p < 0.001) and SAT (relative % change of 2.3 ± 12%, p < 0.001) compared to baseline (Supplementary Tables 1, 2, Figs. 2 and 3). SM volume and psoas muscle CSA remained relatively stable (−1.2 ± 3% and −0.6 ± 16% relative change, respectively; p < 0.001 for both). Fat infiltration in the abdominal SM (MFISM) was higher at follow-up (3 ± 3% relative change, p < 0.001). In the liver, fat content showed relatively small increases (relative % change of 5.5 ± 29%, p < 0.001).
Box-plots of relative percent changes (mean ± SEM) in: A Fat metrics (LFC, VAT, SAT); B Muscle metrics (SM volume, SMI, MFISM, psoas CSA). Note: The y-axis range varies between sub-figures. *p < 0.05, **p < 0.01, ***p < 0.001, n.s. not significant (all p-values are after Bonferroni correction). Grey lines indicate the SEM. In grey boxes are changes in individuals whose weight remained stable weight over the follow-up period. Red shading indicates changes in individuals who gained weight during the follow-up period, with darker hues indicating larger weight gain.
Box-plots of relative percent changes (mean ± SEM) in: A Fat metrics (LFC, VAT, SAT); B Muscle metrics (SM volume, SMI, MFISM, psoas CSA). Note: The y-axis range differs between sub-figures. *p < 0.05, **p < 0.01, ***p < 0.001, n.s. not significant (all p-values are after Bonferroni correction). Grey lines indicate the SEM. In grey boxes are changes in individuals whose weight remained stable weight over the follow-up period. Green shading indicates changes in individuals who lost weight during the follow-up period, with darker hues indicating larger weight loss.
Body fat, muscle and liver MRI measurements in individuals gaining weight
People who gained weight showed significant changes in body composition compared to those who maintained stable weight. Substantial increases were observed in VAT (ranging from 20 to 58% relative change, p < 0.001), in SAT (12 to 36% relative change, p < 0.001), and in MFISM (4.1 to 8% relative change, p < 0.001) (Supplementary Tables 1, 2, Fig. 2). In contrast, the volume of abdominal SM remained relatively stable (−0.4 to −1.2% relative change, p < 0.001). Mean percent changes in psoas muscle CSA showed statistically significant increases in two larger weight gain groups (4–6% relative change, p < 0.05) but all groups exhibited high inter-individual variability. Liver fat content increased markedly (ranging from 22 to 72% relative change, p < 0.001).
Body fat, muscle and liver MRI measurements in individuals losing weight
Those who lost weight during the follow-up period also had the highest baseline values for VAT, SAT and MFISM across all groups (Table 1). Individuals who lost mild amounts of weight (2–5% of their baseline weight) showed significant but not clinically meaningful changes in all metrics (Supplementary Tables 1, 2, Fig. 3). In individuals with moderate weight loss (5–10% baseline weight lost), more substantial decreases were observed in VAT (−22 ± 13% relative change, p < 0.001) and SAT (−17 ± 10% relative change, p < 0.001). SM volume also reduced by −3 ± 4% relative change (p < 0.001), as did psoas muscle CSA (by −4 ± 14% relative change, p < 0.001).
However, substantial decreases in MFISM (−4 ± 6% relative change, p < 0.001) only occurred with large weight loss (10–15% of baseline weight). In these individuals substantial decreases were also observed in VAT (−38 ± 15% relative change), in SAT (−30% ± 12% relative change), in skeletal (−4.5 ± 4.8% relative change) and psoas muscles (−5 ± 5% relative change), all p < 0.001. In the liver, fat content decreased (−33 ± 29% relative change). Loss of psoas muscle was higher in males (Supplementary Tables 3, 4).
Clinical parameters influencing weight changes
Age did not significantly impact weight changes (p > 0.05). Overall, relative changes in body composition and liver fat occurred across all weight gain or loss categories in the subset of individuals with T2D and in the individuals on blood pressure medications (Supplementary Tables 5, 6). Significant interactions between T2D status and weight-change category were observed for abdominal SM (p = 0.02) but not SMI, with weight loss, and for liver fat (p < 0.001) and VAT (p < 0.001) for both weight gain and weight loss. After adjusting for baseline values, the interactions between T2D status and weight-change category for VAT and liver fat remained significant (p < 0.001), with greater reductions with weight loss and greater increases with weight gain occurring in individuals with T2D.
Individuals on blood pressure medication showed smaller reductions in VAT, liver fat and SM (but not SMI) during weight loss (all p < 0.05). Individuals on these medications also showed larger increases in VAT and liver fat during weight gain (all p < 0.05).
In individuals with obesity or overweight, for every 5% reduction in weight, there was a decrease of 16% in VAT, 11% in SAT, 24% in liver fat, 2.3% in MFISM, 1.5% in SM (or 1.4% in SMI) and 2.1% in psoas muscle (Supplementary Table 7). As the relationship was modelled using linear regression, equivalent relative increases were predicted for every 5% gain in body weight.
Correlations between MRI measures of fat and muscle
At baseline MFISM correlated most strongly with SAT (rho = 0.58), but change in MFISM after the follow-up showed highest correlation with change in VAT (rho = 0.60) and change in SAT (rho = 0.46) (Supplementary Figs. 1, 2). Neither abdominal SM nor psoas muscle CSA demonstrated correlations with any other MRI measure at baseline or with longitudinal changes at follow-up.
DXA assessment of lean mass during weight loss and gain
During follow-up, participants with stable weight showed minimal but statistically significant changes in total lean mass (−0.6 ± 2.1%, p < 0.001) while trunk lean mass remained essentially unchanged (0.1 ± 3.6%, p > 0.05) compared to baseline. Total lean mass decreased with moderate/large weight loss (−2.3 ± 2.5% to −2.6 ± 3.2%, p < 0.05) and increased modestly with weight gain (0.4 ± 2.3% to 2.2 ± 2.8%, all p < 0.001). Trunk lean mass remained statistically unchanged across all weight loss and gain groups (p > 0.05).
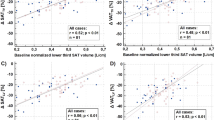
There were strong correlations between baseline DXA-derived lean mass measurements with MRI measurement of abdominal SM volume (rho = 0.95 for trunk lean mass and rho = 0.96 for total lean mass, p < 0.001 (Supplementary Figs. 1, 2). However, the correlations were weak or moderate when longitudinal changes in these measures were considered over the follow-up period (rho = 0.28 for trunk lean mass and rho = 0.47 for total lean mass, p < 0.001) (Fig. 4, Supplementary Fig. 3).
Correlations between relative % change in MRI-derived abdominal skeletal muscle (SM) volume and DXA-derived trunk lean mass (A) and total lean mass (B) over the follow-up period across the entire cohort. Asterisks at each value denote statistical significance after FDR correction (*p < 0.05, **p < 0.01, ***p < 0.001). A black line indicates the line of best fit for the correlation and dotted grey lines indicate where there is 0 change in a measurement.
Case study example for powering weight-loss drug trials
To exemplify use of such body composition datasets for powering studies of weight-loss treatments we calculated the minimum sample sizes required to differentiate a test treatment from a placebo arm. This was based on the results herein for the individuals with overweight or obesity in our dataset (defining a ‘virtual placebo’ group). In a future prospective study of individuals with overweight/obesity, a minimum sample size of 516 (258 per arm) would be required, with 80% power and alpha 0.08, to achieve meaningful differences between placebo and active drug arms for co-primary endpoints on relative change from baseline in body weight, VAT, SAT and liver fat, without meaningful loss of SM (Supplementary Table 8).
Discussion
In this study of 3070 individuals from the general population evaluated over a 3-year follow-up period with multi-parametric MRI, we present quantitative changes in body composition and liver fat according to different degrees of weight gain or loss. There were three key findings. Firstly, substantial increases in adipose tissue (VAT, SAT, MFISM, liver fat) occurred with just moderate weight gain. Secondly, all metrics of fat except muscle fat infiltration (MFISM) decreased with moderate weight loss. Thirdly, both skeletal and psoas muscles decreased with weight loss, with reductions in SM evident even after mild weight loss. For VAT and liver fat, prevalent type 2 diabetes exacerbated weight-gain-related increases, and blood pressure medications attenuated the impact of weight changes. The values reported here provide reference data of real-world weight changes in the pre-incretin era. The changes in body composition may have direct applicability for powering clinical trials and design of placebo arms, as highlighted by the hypothetical example described herein.
In clinical guidelines for obesity and diabetes, large weight loss (>10%) is recommended due to expected improvements in cardiometabolic outcomes [35, 40]. Milder effects are observed even with moderate (5–10%) weight loss: in a study of 15,211 people with recent type 2 diabetes diagnoses, diabetes remission improved 1.7-fold with moderate weight loss compared to 5-fold with large weight loss [41]. Thus, sustained moderate weight loss (5–10%) is advised to improve many conditions associated with overweight/obesity, including MASLD [34, 35]. Incretin-based drugs, in particular, can achieve 15–25% weight loss within 1.5 years, and recent clinical trials frequently have co-primary endpoints on % change from baseline in body weight and % of individuals achieving ≥5% loss of baseline body weight [29,30,31,32,33]. Confirmatory secondary endpoints have included achievement of reduction of ≥10% or 15% of baseline body weight, or in some cases of 20% [29,30,31, 33]. This work preceded the licensing of GLP-1 RA for obesity in the UK (first approval in 2020 [42]), and only 5% of the study cohort had diabetes (GLP-1 RA was first approved for diabetes in 2007 [43]). Nevertheless, the decreases in both VAT and SAT after moderate weight loss herein match the range of changes reported with GLP-1 RA over shorter follow-up periods (−15% for VAT and −20% for SAT with tirzepatide in people with diabetes after 1.5 years [11]; −13% decrease in VAT after 36 weeks of liraglutide in individuals with obesity [44]).
Adverse muscle composition, characterized by elevated muscle fat combined with low muscle volume, has been linked to poor function and is a strong and independent predictor of all-cause mortality [45]. Even small changes in skeletal muscle composition affect muscle function [46] and insulin resistance [47]. The rapid weight loss as a result of incretin treatment has raised concerns about adverse effects on muscle mass and function [14, 15]. Using DXA, trial findings indicate that 10–40% of the weight lost in incretin trials is adjusted fat-free mass [14, 48, 49], estimated to consist of ~50% skeletal muscle [14, 50, 51]. Recent randomized controlled trials employing MRI have examined the effects of incretin mimetics on thigh skeletal muscle. Treatment with liraglutide over 40 weeks was associated with a reduction of thigh muscle MFI (~3%) and 5% weight loss among individuals with obesity [52]. In SURPASS-3 trial, there was a 6–7% reduction in thigh muscle volume and 4–5% reduction in thigh MFI after 52 weeks of tirzepatide in people with type 2 diabetes [53]. Our study evaluated MFISM and SM in the abdomen rather than the thigh because these muscle groups serve as strong markers of whole-body SM mass [54], while also providing operational advantages including shorter scan duration and fewer hardware requirements [21]. Observed reductions in abdominal SM and MFISM with large weight loss herein are comparable. The parabolic relationship that MFISM showed with weight loss in our study may indicate a redistribution of fat from other depots to skeletal muscle following low-level weight loss, consistent with reported changes in fat depots after lifestyle intervention [55].
Clinically meaningful increases in fat occurred only with moderate to large weight gain. In the liver, 30% relative fat changes are considered clinically meaningful in individuals with MASLD, although smaller effects (>1% absolute change) can be monitored and are significant [27, 56]. The gains in liver fat were most pronounced among all fat measurement changes and correlated less with changes in VAT, SAT and MFISM, consistent with previous findings [57]. This may be suggestive of differential temporal patterns of fat mobilization or redistribution after weight gain. More recently, clinical trials with weight-loss treatments in individuals living with obesity or overweight are focused on sustaining weight lost after termination (wash-out) of treatment [30, 32], therefore our quantification of the changes in body composition with increase in body weight may be of relevance.
The high repeatability of measurements using multiparametric MRI [20, 27] is evident in the minimal changes we found in those with stable weight. The availability of DXA has resulted in its use for cross-sectional body composition assessment [58], with recent recommendations endorsing its application for direct measurement of fat [2] in obesity definition. In clinical trials, the lower accuracy of DXA compared to MRI-based volumetric measurements [19] presents a challenge in detecting small changes, particularly in longitudinal trial settings. While less accurate, DXA may be warranted in trials where bone mineral density is a relevant endpoint. However, the weak correlation we found between SM and lean mass changes may reflect its inability to differentiate between SM and MFISM compartments [59, 60]. This prevents retrospective extrapolations of SM from DXA datasets and indicates that lean mass is a poor surrogate for tracking longitudinal changes in SM.
Almost all UK Biobank participants are White, and few in our dataset had type 2 diabetes, highlighting the need for comprehensive research across multi-ethnic or more diseased populations, over multiple follow-up periods. We explored the impact of age differences, prior cancer, diabetes, overweight/obesity and of treatments for hypertension, but other treatments affecting weight changes were not investigated due to missing data and lack of comprehensive medical history in the UK Biobank registry. Efforts were made to remove intentional weight loss due to bariatric surgery, but both intentional and unintentional weight changes remain in this pre-incretin era dataset. While our data suggest measurements are stable and did not change significantly when weight was maintained, it is possible that our results may be confounded by random differences in levels of participant hydration at the time of MRI scanning. Nevertheless, the granularity of changes in body composition provided by MRI in this large-scale population study is a timely and useful reference for exploring the impact of drug candidates that induce weight loss in clinical trials. Of relevance to clinical trial design is that for every 5% relative drop in weight, there was a 16% reduction in VAT, 11% in SAT, 24% in liver fat, 2.3% in MFISM, 1.5% in SM (or 1.4% in SMI) and 2.1% in psoas muscle, in individuals who were obese or overweight. Amid rising global obesity rates, the need to optimize weight-loss therapies and personalize management options is vital, requiring accurate tools to measure adverse effects and stratify response to treatment. Our findings provide data to inform sample size calculations for future clinical trials and support label claims, while also offering a ‘virtual placebo’ arm or population-level estimate [53].
Data availability
Participant data was obtained through UK Biobank Access Application number 9914. UK Biobank has approval from Northwest Multi-Centre Research Ethics Committee and obtained written informed consent from all participants prior to the study. Summary data is included in the manuscript or uploaded as online supplemental information. Anonymized individual patient data and source code for statistical analysis can be shared upon request or as required by law and/or regulation and/or governance by and within the rules of UK Biobank access with qualified external researchers. Approval of such requests is at the discretion of the study sponsors and is dependent on the nature of the request, the merit of the research proposed, the availability of the data, and the intended use of the data. Source code for the processing of body composition measurements from MRI images is not disclosed as it is commercially sensitive.
References
Ng M, Gakidou E, Lo J, Abate YH, Abbafati C, Abbas N, et al. Global, regional, and national prevalence of adult overweight and obesity, 1990–2021, with forecasts to 2050: a forecasting study for the Global Burden of Disease Study 2021. Lancet. 2025;405:813–38.
Rubino F, Cummings DE, Eckel RH, Cohen RV, Wilding JPH, Brown WA, et al. Definition and diagnostic criteria of clinical obesity. Lancet Diab Endocrinol. 2025;13:221–62.
Saeed S, Bonnefond A, Froguel P. Obesity: exploring its connection to brain function through genetic and genomic perspectives. Mol Psychiatry. 2025;30:651–8.
Quek J, Chan KE, Wong ZY, Tan C, Tan B, Lim WH, et al. Global prevalence of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in the overweight and obese population: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2023;8:20–30.
Jung M, Raghu VK, Reisert M, Rieder H, Rospleszcz S, Pischon T, et al. Deep learning-based body composition analysis from whole-body magnetic resonance imaging to predict all-cause mortality in a large western population. EBioMedicine. 2024;110:105467.
Thomas EL, Frost G, Taylor-Robinson SD, Bell JD. Excess body fat in obese and normal-weight subjects. Nutr Res Rev. 2012;25:150–61.
Liu Y, Basty N, Whitcher B, Bell JD, Sorokin EP, van Bruggen N, et al. Genetic architecture of 11 organ traits derived from abdominal MRI using deep learning. eLife. 2022;10:e65554.
Souza ACdoAH, Rosenthal MH, Moura FA, Divakaran S, Osborne MT, Hainer J, et al. Body composition, coronary microvascular dysfunction, and future risk of cardiovascular events including heart failure. JACC Cardiovasc Imaging. 2024;17:179–91.
Liu L, Li Z, Ye W, Peng P, Wang Y, Wan L et al. Safety and effects of anti-obesity medications on weight loss, cardiometabolic, and psychological outcomes in people living with overweight or obesity: a systematic review and meta-analysis. eClinicalMedicine. 2025;79. https://doi.org/10.1016/j.eclinm.2024.103020.
Wang X, Wu N, Sun C, Jin D, Lu H. Effects of SGLT-2 inhibitors on adipose tissue distribution in patients with type 2 diabetes mellitus: a systematic review and meta-analysis of randomized controlled trials. Diabetol Metab Syndr. 2023;15:113.
Gastaldelli A, Cusi K, Fernández Landó L, Bray R, Brouwers B, Rodríguez Á. Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): a substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diab Endocrinol. 2022;10:393–406.
Kramer CM, Borlaug BA, Zile MR, Ruff D, DiMaria JM, Menon V et al. Tirzepatide reduces LV mass and paracardiac adipose tissue in obesity-related heart failure. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2024.11.001.
Loomba R, Hartman ML, Lawitz EJ, Vuppalanchi R, Boursier J, Bugianesi E, et al. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N Engl J Med. 2024;391:299–310.
Conte C, Hall KD, Klein S. Is weight loss-induced muscle mass loss clinically relevant?. JAMA. 2024;332:9–10.
Prado CM, Phillips SM, Gonzalez MC, Heymsfield SB. Muscle matters: the effects of medically induced weight loss on skeletal muscle. Lancet Diab Endocrinol. 2024;12:785–7.
Lopez P, Newton RU, Taaffe DR, Singh F, Buffart LM, Spry N, et al. Associations of fat and muscle mass with overall survival in men with prostate cancer: a systematic review with meta-analysis. Prostate Cancer Prostatic Dis. 2022;25:615–26.
Shachar SS, Williams GR, Muss HB, Nishijima TF. Prognostic value of sarcopenia in adults with solid tumours: a meta-analysis and systematic review. Eur J Cancer. 2016;57:58–67.
Borga M, West J, Bell JD, Harvey NC, Romu T, Heymsfield SB, et al. Advanced body composition assessment: from body mass index to body composition profiling. J invest Med. 2018;66:1–9.
Mouchti S, Orliacq J, Reeves G, Chen Z. Assessment of correlation between conventional anthropometric and imaging-derived measures of body fat composition: a systematic literature review and meta-analysis of observational studies. BMC Med Imaging. 2023;23:127.
Nowak M, Núñez L, Hill CE, Davis T, Cardiel LF, Schooler GR, et al. Repeatability, reproducibility, and observer variability of body composition assessment with single-slice lumbar MRI. Discov Imaging. 2025;2:1.
Nowak M, Nunez L, Hill CE, Pagliaro T, McGonnigle J, Niglas M et al. Automated volumetric MRI quantification of body fat and skeletal muscle in the UK Biobank: technical validation against expert manual segmentations and comparison with single-slice techniques. Abdomimal Radiol. 2025. https://doi.org/10.1007/s00261-025-05170-w.
Beyer C, Hutton C, Andersson A, Imajo K, Nkajima A, Kiker D, et al. Comparison between magnetic resonance and ultrasound-derived indicators of hepatic steatosis in a pooled NAFLD cohort. PLOS ONE. 2021;16:e0249491.
Yokoo T, Serai S, Pirasteh A, Bashir M, Hamilton G, Hernando D, et al. Linearity, bias, and precision of hepatic proton density fat fraction measurements by using MR imaging: a meta-analysis. Radiology. 2018;286:486–98.
Sudlow C, Gallacher J, Allen N, Beral V, Burton P, Danesh J, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12:e1001779–e1001779.
Vasan SK, Osmond C, Canoy D, Christodoulides C, Neville MJ, Di Gravio C, et al. Comparison of regional fat measurements by dual-energy X-ray absorptiometry and conventional anthropometry and their association with markers of diabetes and cardiovascular disease risk. Int J Obes. 2018;42:850–7.
Roca-Fernandez A, Banerjee R, Thomaides-Brears H, Telford A, Sanyal A, Neubauer S, et al. Liver disease is a significant risk factor for cardiovascular outcomes - a UK Biobank study. J Hepatol. 2023;79:1085–95.
Bachtiar V, Kelly MD, Wilman HR, Jacobs J, Newbould R, Kelly CJ, et al. Repeatability and reproducibility of multiparametric magnetic resonance imaging of the liver. PloS one. 2019;14:e0214921.
Paige E, Korda RJ, Banks E, Rodgers B. How weight change is modelled in population studies can affect research findings: empirical results from a large-scale cohort study. BMJ Open. 2014. https://doi.org/10.1136/bmjopen-2014-004860.
Garvey WT, Frias JP, Jastreboff AM, le Roux CW, Sattar N, Aizenberg D, et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2023;402:613–26.
Jastreboff AM, le Roux CW, Stefanski A, Aronne LJ, Halpern B, Wharton S, et al. Tirzepatide for obesity treatment and diabetes prevention. N Engl J Med. 2025;392:958–71.
Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384:989–1002.
Rubino D, Abrahamsson N, Davies M, Hesse D, Greenway FL, Jensen C, et al. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: the STEP 4 randomized clinical trial. JAMA. 2021;325:1414–25.
Davies M, Færch L, Jeppesen OK, Pakseresht A, Pedersen SD, Perreault L, et al. Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet. 2021;397:971–84.
Ryan DH, Yockey SR. Weight loss and improvement in comorbidity: differences at 5%, 10%, 15%, and over. Curr Obes Rep. 2017;6:187–94.
Garvey WT, Mechanick JI, Brett EM, Garber AJ, Hurley DL, Jastreboff AM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pr. 2016;22:1–203.
Cole TJ, Lobstein T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr Obes. 2012;7:284–94.
Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, et al. A multi-society Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78:1966–86.
Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, Abdelmalek MF, Caldwell S, Barb D, et al. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology. 2023;77:1797–835.
Andersson A, Kelly M, Imajo K, Nakajima A, Fallowfield JA, Hirschfield G, et al. Clinical utility of magnetic resonance imaging biomarkers for identifying nonalcoholic steatohepatitis patients at high risk of progression: a multicenter pooled data and meta-analysis. Clin Gastroenterol Hepatol. 2022;20:2451–2461.e3.
American Diabetes Association Professional Practice Committee. 2. Diagnosis and Classification of Diabetes: Standards of Care in Diabetes-2025. Diab Care. 2025;48:S27–S49.
Morieri ML, Rigato M, Frison V, D’Ambrosio M, Sartore G, Avogaro A et al. Early weight loss, diabetes remission and long-term trajectory after diagnosis of type 2 diabetes: a retrospective study. Diabetologia. 2025. https://doi.org/10.1007/s00125-025-06402-w.
Ansari S, Mazaheri T, O’Donnell K, Waite M, Cann A, Abdel-Malek M, et al. Time to unshackle the medical treatment of obesity in the NHS. Clin Med. 2024;24:100206.
Thong KY, Gupta PS, Cull ML, Adamson KA, Dove DS, Rowles SV, et al. GLP-1 receptor agonists in type 2 diabetes - NICE guidelines versus clinical practice. Br J Diab. 2014;14:52–59.
Neeland IJ, Marso SP, Ayers CR, Lewis B, Oslica R, Francis W, et al. Effects of liraglutide on visceral and ectopic fat in adults with overweight and obesity at high cardiovascular risk: a randomised, double-blind, placebo-controlled, clinical trial. Lancet Diab Endocrinol. 2021;9:595–605.
Linge J, Petersson M, Forsgren MF, Sanyal AJ, Dahlqvist Leinhard O. Adverse muscle composition predicts all-cause mortality in the UK Biobank imaging study. J Cachexia Sarcopenia Muscle. 2021;12:1513–26.
Whitcher B, Thanaj M, Cule M, Liu Y, Basty N, Sorokin EP, et al. Precision MRI phenotyping enables detection of small changes in body composition for longitudinal cohorts. Sci Rep. 2022;12:3748.
Maltais A, Alméras N, Lemieux I, Tremblay A, Bergeron J, Poirier P, et al. Trunk muscle quality assessed by computed tomography: association with adiposity indices and glucose tolerance in men. Metab Clin Exp. 2018;85:205–12.
Mechanick JI, Butsch WS, Christensen SM, Hamdy O, Li Z, Prado CM, et al. Strategies for minimizing muscle loss during use of incretin-mimetic drugs for treatment of obesity. Obes Rev. 2025;26:e13841.
Stefanakis K, Kokkorakis M, Mantzoros CS. The impact of weight loss on fat-free mass, muscle, bone and hematopoiesis health: Implications for emerging pharmacotherapies aiming at fat reduction and lean mass preservation. Metabolism. 2024;161:156057.
Tinsley GM, Heymsfield SB. Fundamental body composition principles provide context for fat-free and skeletal muscle loss with GLP-1 RA treatments. J Endocr Soc. 2024;8:bvae164.
Magkos F, Fraterrigo G, Yoshino J, Luecking C, Kirbach K, Kelly SC, et al. Effects of moderate and subsequent progressive weight loss on metabolic function and adipose tissue biology in humans with obesity. Cell Metab. 2016;23:591–601.
Pandey A, Patel KV, Segar MW, Ayers C, Linge J, Leinhard OD, et al. Effect of liraglutide on thigh muscle fat and muscle composition in adults with overweight or obesity: Results from a randomized clinical trial. J Cachexia Sarcopenia Muscle. 2024;15:1072–83.
Sattar N, Neeland IJ, Leinhard OD, Landó LF, Bray R, Linge J et al. Tirzepatide and muscle composition changes in people with type 2 diabetes (SURPASS-3 MRI): a post-hoc analysis of a randomised, open-label, parallel-group, phase 3 trial. Lancet Diabetes Endocrinol. 2025. https://doi.org/10.1016/S2213-8587(25)00027-0.
Lee SJ, Janssen I, Heymsfield SB, Ross R. Relation between whole-body and regional measures of human skeletal muscle. Am J Clin Nutr. 2004;80:1215–21.
Yaskolka Meir A, Shelef I, Schwarzfuchs D, Gepner Y, Tene L, Zelicha H, et al. Intermuscular adipose tissue and thigh muscle area dynamics during an 18-month randomized weight loss trial. J Appl Physiol. 2016;121:518–27.
Alkhouri N, Beyer C, Shumbayawonda E, Andersson A, Yale K, Rolph T, et al. Decreases in cT1 and liver fat content reflect treatment-induced histological improvements in MASH. J Hepatol. 2025;82:438–45.
Mátis D, Hegyi P, Teutsch B, Tornai T, Erőss B, Pár G et al. Improved body composition decreases the fat content in non-alcoholic fatty liver disease, a meta-analysis and systematic review of longitudinal studies. Front Med. 2023;10. https://doi.org/10.3389/fmed.2023.1114836.
Kim D, Lee J, Park R, Oh C-M, Moon S. Association of low muscle mass and obesity with increased all-cause and cardiovascular disease mortality in US adults. J Cachexia Sarcopenia Muscle. 2024;15:240–54.
Basty N, Thanaj M, Whitcher B, Bell JD, Thomas EL. Comparing DXA and MRI body composition measurements in cross-sectional and longitudinal cohorts. medRxiv. 2024, https://www.medrxiv.org/content/10.1101/2024.12.12.24318943v1.full.
Dubin RL, Heymsfield SB, Ravussin E, Greenway FL. Glucagon-like peptide-1 receptor agonist-based agents and weight loss composition: filling the gaps. Diab Obes Metab. 2024;26:5503–18.
Acknowledgements
We wish to acknowledge the UK Biobank participants and Dr Sofia Mouchti for statistical oversight.
Author information
Authors and Affiliations
Contributions
Conceptualization, HTB and MN; methodology, LN and MDR; formal analysis, MN and LN; data curation, MN; writing—original draft preparation, HTB; writing—review and editing, MN, LN, TP, CD, SK, LMK, SH, MDR, HTB, ELT and JDB; supervision, HTB and ELT. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. MN, LN, TP, MDR, and HTB are employees for Perspectum, a company that has developed LiverMultiScan, a UKCA and FDA 510(k)–cleared medical device for liver health measurements that was used in the imaging sub-study of the UK Biobank. HTB and MDR are also shareholders of Perspectum. LN and TP were employees of Perspectum during this work and Carlos Duncker became an employee of Perspectum after this work was completed. ELT and JDB are consultants for Perspectum. LMK is a consultant to Altimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Cytoki, Helicore, Johnson & Johnson, Kallyope, Eli Lilly & Company, MetaVia, Neurogastrx, Novo Nordisk, Oxford Medical Products, Perspectum, Pfizer, Skye Bioscience and Zealand. SBH is on the medical advisory boards of Lilly, Abbott, Tanita Corp., Novo Nordisk, and Regeneron. The remaining authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nowak, M., Núñez, L., Pagliaro, T. et al. Weight-related changes in MRI-derived measures of body composition and liver steatosis: a large-scale analysis for obesity trial design. Int J Obes (2026). https://doi.org/10.1038/s41366-026-02037-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41366-026-02037-y