Abstract
Background
Understanding how chemical exposure varies within and between people over time is a critical component of characterizing the exposome—the totality of lifetime exposures. However, variability remains an understudied aspect of exposomic research.
Objective
Our objective was to investigate trends in variability for chemical exposure data between and within people that appear across differing study designs.
Methods
Thirty-five people in Eugene, Oregon, and 46 people in St. Helens, Oregon, wore silicone wristbands over multiple seasons, including a span of heavy wildfire smoke in Eugene. Each participant wore between four and 14 wristbands. We analyzed 586 wristbands for 94 (Eugene) and 58 (St. Helens) chemicals. While analytic tests differed between the studies, the same 43 polycyclic aromatic hydrocarbons (PAHs) were measured in both studies. We also evaluated three environmental variables for their impact on chemical concentrations. We fit generalized mixed effects models to each chemical, and used variance partitioning to understand and quantify sources of variability across environmental factors and inter- and intra-individual variables.
Results
We observed PAHs that were consistent within people across different days. For a subset of these PAHs, results did not agree well between studies, indicating the importance of measuring chemical data at different time points across studies. Environmental variables were not sufficient for explaining data variability for most chemicals. Only 21% and 30% of the modeled chemicals for Eugene and St. Helens, respectively, had a combined environmental variable R2 at or above 0.1. Yet, environmental factors still revealed valuable information; we observed higher combined R2 values for styrene, o-xylene, ethylbenzene, and phenanthrene in the Eugene detection model, which came from a combination of fine particulate matter and smoke density information.
Impact Statement
-
Our manuscript is the largest investigation of intra- and inter- variability in silicone wristband concentrations, containing over 23,000 chemical data points across two different personal chemical exposure studies.
-
Certain chemicals were consistent within people across different days.
-
For a subset of chemicals, results did not agree well between the two wristband studies.
-
Our findings highlight the importance of measuring chemical data at different time points across studies to better understand the exposome.
-
Environmental variables included in this study were not sufficient for explaining the data variability for most chemicals.
Similar content being viewed by others
Introduction
The human exposome is defined as the combined impact of all environmental factors, including chemical exposures, on health throughout all life stages [1, 2]. The U.S. National Institutes of Environmental Health Sciences (NIEHS) highlights “exposomics” as one of six research areas of emphasis for the agency in their 2025–2029 Strategic Plan [3]. In addition, one of the goals of the 2025 Exposome Moonshot Forum is to shape the future direction of the Human Exposome Project by creating a “clear and actionable plan for the comprehensive characterization and utilization of the Human Exposome” [4]. Exposome-related research is a growing and integral focus area within environmental health.
Exposome research cannot rely solely on air quality data from regional outdoor stationary monitors or smoke density data from satellites to accurately estimate personal chemical exposure. Often, these types of datasets are used as proxies for personal chemical exposure because long-term data is publicly available for large geographic areas and populations [5, 6]. However, our recent work compared personalized chemical exposure data from silicone wristbands to fine particulate matter (PM2.5) data and Hazard Mapping System (HMS) smoke density data and found that using PM2.5 AQI data or HMS data alone is not enough to characterize the complexities of personal chemical exposure [5].
There have been significant advancements over the past decade making it easier to assess personal chemical exposure to a wide variety of chemicals. For example, silicone wristbands were first introduced as an easy-to-wear, novel personal exposure assessment tool in 2014 [7] and, since then, wristbands and other silicone configurations (e.g., some samplers worn on the wrist or attached to the lapel also include sorbent-bar samplers) have been used in many exposure studies [8,9,10,11,12]. Hereafter, we use the term “wristbands” to refer to silicone wristbands [7]. Researchers have analyzed wristband extracts for over 1500 chemicals, including polycyclic aromatic hydrocarbons (PAHs), phthalates, and flame retardants [13]. Researchers have also found that wristbands provide similar exposure information as metabolites in biological samples [14,15,16]. Wristbands capture a combination of inhalation exposure, dermal exposure, and dermal excretion [8] that correspond strongly with internal biomarkers [14].
Wristbands in research studies are worn for a set time period, ranging from a few hours to one month [8]. A hurdle for exposome research moving forward is understanding how findings from personal chemical exposure assessment studies spanning a short period of time (few hours to a month) can be used to estimate a person’s chemical exposure across their lifetime. Researchers need to learn more about the factors that influence chemical exposure within a person (intra-individual) and between people (inter-individual) over time to address this challenge and inform the future direction of exposomics.
Two existing wristband studies have investigated intra- and inter-individual variability in personal chemical exposure [17, 18]. In Donald et al., 35 participants actively engaged in farming in Senegal wore one wristband at a time in two sequential periods [17]. The researchers used signed-rank and Spearman’s correlation analyses to compare pesticide concentrations between the two wristbands worn by each participant and found that the wristband pairs yielded similar results [17].
In Bonner et al., 56 firefighters from two fire departments in Missouri, U.S. wore two different silicone dog tags while on- and off-duty [18]. Researchers used Bray–Curtis dissimilarity scores to investigate inter- and intra-variability for all pairwise comparisons (on- vs. off-duty dog tag from the same firefighter); this statistical method was used to calculate the similarity of observed chemical ordinal quantiles across all chemicals for the pairs of dog tags. Researchers concluded that intra-pair dissimilarity scores were often smaller than inter-pair dissimilarity scores, even within the same department and duty status, highlighting similarities in personal chemical exposure within the same individual [18].
To expand on this work, we investigated the inter- and intra- variability of personal chemical exposure using data from two large wristband studies. Thirty-five people in Eugene, Oregon wore 426 wristbands and 46 people in St. Helens, Oregon wore 160 wristbands, with participants each wearing between 4 and 14 wristbands. Our research is the first to look at intra- and inter- variability between studies in wristband concentrations with more than two repeated measures per person. The larger sample sizes and additional repeated measures in this manuscript enabled us to examine sources of variability for individual chemicals in a more detailed manner than previous studies.
We also evaluated additional environmental data (PM2.5, outdoor temperature, and HMS) from the U.S. Environmental Protection Agency (EPA) and National Oceanic and Atmospheric Administration (NOAA) for their impact on chemical concentrations. Our statistical methods presented here move beyond correlative summaries used in previous studies and allow for a more comprehensive evaluation of the sources of variability contributing to personal chemical exposure.
Our objective was to understand sources of variability in chemical exposure. We report how chemical detections and concentrations change over time within and between people. We establish statistical best practices for future evaluations on (a) inter- and intra- chemical variability and (b) how additional environmental factors affect chemical exposure variability. These results will inform and guide future exposome research.
Materials and methods
Site descriptions and study cohorts
Our investigation included data from two different studies: one in Eugene, Oregon, U.S., and one in St. Helens, Oregon, U.S.; hereafter, referred to as the “Eugene study” and the “St. Helens study”. For the Eugene study, participants were required to live within a 32-kilometer (km) radius of Eugene, Oregon, be over the age of 18, and have a current asthma diagnosis. Other enrollment criteria are described in Bramer et al. [5]. The wristband dataset collected in the Eugene study was collected as part of a larger research project; other studies utilized paired lung function data [19, 20] and global positioning system data [5]. Eugene wristbands were worn between August 2017 and April 2018, and participants wore one wristband per day for 7 days twice in a 9-month period.
As of the 2020 U.S. census, Eugene had a population of about 170,000 people and was the second most populous city in Oregon [21]. There are several site-specific features in Eugene that could influence chemical exposure. For example, there are large highways that run through Eugene as well as an industrial corridor that includes sites like the J.H. Baxter & Co. Wood Treatment Facility in West Eugene [22]. In July 2025, the EPA added the J.H. Baxter site to the Superfund National Priorities List after proposing to do so in 2024 [23]. Prior to J.H. Baxter halting their wood treatment operations in 2022, this facility was the source of many community odor complaints [22]. Eugene has industrial zones directly next to residential zones [22]. Our Eugene study also captured two different periods of heavy wildfire smoke and included days where the PM2.5 air quality index (AQI) was in the unhealthy and very unhealthy range [5].
To participate in the St. Helens study, participants were required to live within a five km radius from the center of town. Other enrollment criteria included being over the age of five and residing or working within St. Helens, all with no more than three participants per household. There were eight households with more than one study participant. The wristband component of the St. Helens study is part of a larger study combining personal exposures with environmental conditions that is still ongoing. Participants in this study wore wristbands for 7 days once every other month for 8 months from November 2022 to June 2023 (4 total sampling periods per participant). A visual comparison of the Eugene and St. Helens study designs can be seen in Fig. 1.
Visualization of the locations, wristband wear times, and datasets collected in the Eugene and St. Helens studies. Eugene participants wore one wristband each day for seven consecutive days and repeated this at two different time periods to wear a maximum of 14 wristbands. St. Helens participants wore one wristband for a seven- to eight-day period and repeated this four times over a one-year period, wearing a maximum of four wristbands. Created by Pacific Northwest National Laboratories.
St. Helens is approximately 224 km from Eugene. St. Helens is a port town at the confluence of the Multnomah channel and the Columbia River near Portland, Oregon. Industrial activity over the last century, including transporting logging products and producing coal tar creosote products, may contribute to chemical exposures in the area. Like Eugene, several large transit routes may also influence chemical exposure. Unlike Eugene, St. Helens had a population of near 14,000 at the 2020 census [21] and has no dedicated industrial center. However, within city limits is the former Pope and Talbot wood treatment site, which produced and transported coal tar creosote in the early twentieth century. Community members have expressed concerns about chemical exposures from this site, and previous research has investigated environmental contamination in this community [24, 25]. No major wildfires or unhealthy PM2.5 AQI days took place during this study’s sampling period.
In both studies, the majority of participants identified as White/Caucasian (86% and 94%, respectively). A higher percentage of participants identified as female in Eugene compared to St. Helens (80% and 52%, respectively). Table S1 in the Supplementary Information (SI) includes participant demographics for both studies.
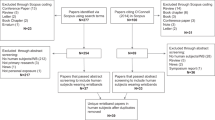
We included data from 426 wristbands and 35 people in the Eugene study and from 160 wristbands and 46 people in the St. Helens study (Fig. 1). Both studies were approved by Oregon State University’s Institutional Review Board (Eugene #IRB-2020-0899, St. Helens #IRB-2020-0529). In addition to wristband data, researchers also collected outdoor daily maximum temperature, smoke, and PM2.5 data from the EPA and NOAA; all datasets are described in detail below.
Wristband methodology
Preparation and deployment
We purchased silicone wristbands from 24hourwristbands.com (Houston, TX, U.S.) and prepared the wristbands for sampling as described in Anderson et al. [26]. We stored prepared wristbands in airtight polytetrafluoroethylene (PTFE) bags and distributed them to participants either in-person (Eugene) or by mail (St. Helens) at ambient temperature. The sampling period began when participants took the wristband out of the PTFE bag. Upon return to the laboratory, wristbands were stored at −20 °C before extraction.
Inclusion and exclusion criteria
For the Eugene study, we only analyzed wristbands that (a) were worn within eight hours of the 24-h period, (b) had date and time information for when the wristband was put on and taken off, (c) were in airtight PTFE bags upon receipt, (d) were put on between 12:01 am and 11:59 am, and (e) were worn within the seven-day sampling period for the participant [5]. Overall, we excluded 22 wristbands (5% of all wristbands collected) in the Eugene study.
For the St. Helens study, we only analyzed wristbands that (a) were worn for at least five of the seven (\(\pm\)1) day sampling period, (b) had date and time information for when the wristband was put on and taken off, and (c) was intact and within a sealed PTFE bag. Overall, we excluded two wristbands (1.2% of all wristbands collected) in the St. Helens study.
Cleaning, extraction, and chemical analysis
We cleaned all wristbands that met our inclusion criteria. We removed particles on the wristband surface with 18 MΩ*cm water and isopropanol [14] and then extracted chemicals from the wristbands using ethyl acetate as described in Bramer et al. [5]. Extracts were stored at −20 °C prior to chemical analysis. The St. Helens samples were stored at 4 °C in between solid phase extraction and analysis.
The Eugene and St. Helens studies were originally set up to learn more about different environmental health issues (Section “Site descriptions and study cohorts”) which determined the chosen analytical method for each study. We quantitatively analyzed all Eugene wristband extracts for 94 organic chemicals using an Agilent (Santa Clara, CA, U.S.) 7890 A gas chromatograph (GC) interfaced with a 5975 C mass spectrometer (MS) detector as well as an Agilent 6890 N GC interfaced with a 5975B MS. The method includes 94 VOCs and SVOCs including PAHs, OPAHs, tri-r-phosphates, and alkanes. St. Helens wristband extracts were analyzed for 58 parent and alkylated PAHs using an Agilent 7890 A GC and triple quad MS (7000 MS/MS). A list of the target analytes and molecular weights for both studies are in Table S2. Additional information on chemical analysis, limits of detection (LOD), laboratory materials, and quality control samples is described in Bramer et al. for the Eugene study and in Paulik et al. for the St. Helens study [5, 27].
We utilized solid phase extraction (SPE) in the St. Helens study as an additional cleaning process for removing lipid residues from the wristband extracts, which could be from sources such as personal care products or sweat [13, 28]. This process reduces matrix interference during chemical analysis. SPE was not utilized in the Eugene study and, as described in Bramer et al., we were not able to detect certain deuterated chemical surrogates in a few wristband extracts due to matrix interference and unable to quantify the related target chemical in those instances [5]. Table S2 in Bramer et al. includes the number of wristbands for each target chemical that did not have matrix interference in Eugene [5]. No wristbands from the St. Helens study had matrix interference.
PM2.5
We downloaded daily mean PM2.5 data from the EPA for days where wristbands were worn in the Eugene and St. Helens studies [29]. PM2.5 data was available from multiple stationary air monitors in Eugene. We prioritized PM2.5 data from the air monitor at the Eugene Airport (EPA site name = Eugene – Hwy 99; E99), which had PM2.5 data available for 97.7% of Eugene study days and is the same location where the climate data was collected (see Section “Outdoor temperature”). For the two days where PM2.5 data was not available from the Eugene Airport air monitor, we used PM2.5 data from a monitor at a Eugene park approximately 12 miles southeast of the airport (EPA site name = Eugene – Amazon Park; EAP). The PM2.5 data collected at Amazon Park and Eugene Airport monitors were not significantly different during our Eugene study period. We looked at how close the PM2.5 data was between the two stations on the 87 days where we had data from both monitoring locations. On average, PM2.5 differed by 2.1 µg/m3 between the sites on the same day. The PM2.5 value was exactly the same at both sites for 74% of the days we had values at both locations.
PM2.5 data was only available from one stationary air monitor in Columbia County, the county where St. Helens is located. The air monitor is on Sauvie Island (EPA site name = Sauvie Island – SIS), which is a large island along the Columbia River and about 10 miles straight-line distance from St. Helens.
HMS
We downloaded daily coordinates of smoke polygons and smoke plume density from the NOAA HMS database [30]. NOAA satellite imagery identifies smoke during sunlight over North America, and NOAA categorizes observed smoke into either none, light, medium, or heavy smoke density groups based on opacity information [31]. HMS data may not identify dilute smoke and does not provide information about smoke plume height [31, 32]. We used the highest observed smoke category at the location of the respective PM2.5 monitor as the daily observed HMS value.
Outdoor temperature
We downloaded NOAA hourly dry bulb globe temperatures from two climatological data stations: one in Eugene and one near St. Helens [33]. For Eugene, we downloaded temperature data from the station at the Eugene Airport (NOAA station ID = GHCND:USW00024221). We selected the Eugene Airport station due to proximity to study participants and availability of PM2.5 data, which we used for the majority of Eugene PM2.5 data. For St. Helens, we downloaded temperature data from the closest station, which was at the Scappoose Industrial Airport (NOAA station ID = WBAN:04201) and about 10 miles from downtown St. Helens. We then calculated the maximum daily dry bulb globe temperature at each location.
Data analysis
All statistical analyses were conducted using R version 4.4.3 [34]. For each study, we constructed two datasets, a chemical concentration dataset using only concentration values which were above the LOD and a detection dataset with a binary value indicating if each concentration observation was above the LOD or not. For the chemical concentration dataset, we analyzed all chemicals that were detected in at least 5% of all wristbands. For the detection datasets, we excluded chemicals with less than 5% or more than 95% of observations above the LOD. For both models, chemicals were required to have at least two individuals each with a minimum of two observations that were above LOD to ensure that downstream statistical models were estimable. Chemicals not meeting these criteria were filtered and not used in analysis. We computed the coefficient of variation (CV) value, equation given in the SI, for each chemical (Fig. 2).
Plot of the log2 coefficients of variation for each chemical with color denoting study: Eugene (red) and St. Helens (blue). Horizontal lines correspond to 95% confidence intervals.
For the concentration dataset, we fit a mixed effects linear model with a conditional normal distribution to the log2 transformed concentration values for each chemical in each study. For the detection dataset, we fit a generalized linear mixed effects model with a conditional binomial distribution to the binary indicator values for each chemical in each study. Hereafter, we refer to these models as the “concentration model” and “detection model”, respectively. Both models included PM2.5, HMS, and maximum temperature as fixed effects and participant as a random effect. Models were estimated using the lme4 [35] package v1.1-35.4. For each fitted model, we computed the proportion of variability in the response variable that could be explained by the combination of fixed effect variables, the proportion of variability that could be explained by each environmental variable uniquely, and the proportion of variability that could be attributed to observations being from the same participant (intra-subject) or not (inter-subject), after accounting for the fixed effect variables. We used the partR2 [36] package v0.9.2 to partition the sources of variability and estimate the amount of variability explained by each partition, for each model. Full model specifications are given in the SI, and details on variance partition estimators are given [36].
Results
Characterization of wristband and other environmental data sets
Wristband
Of the 94 chemicals measured in the 426 Eugene study wristbands, 59 chemicals were detected in at least one wristband. Of the 58 chemicals measured in the 160 St. Helens study wristbands, 50 chemicals were detected in at least one wristband. Twenty-six of the detected chemicals overlap between studies (Table S2). In the Eugene study, the median chemical concentrations were highest for n-nonane, xylenes (m and p), and n-heptadecane (12.7, 9.8, and 8.9 log2 ng/g wristband, respectively). In the St. Helens study, the median chemical concentrations were highest for phenanthrene, 2-methylphenanthrene, and 1,6- and 1,3-dimethylnaphthalene (4.8, 3.6, and 3.6 log2 ng/g wristband, respectively). Detection frequency and median concentration for each chemical in both studies is provided in Table S2.
The concentration model included 42 chemicals for the Eugene study and 37 chemicals for the St. Helens study. The detection model included 38 chemicals for the Eugene study and 27 chemicals for the St. Helens study. Chemicals that did not meet requirements to be included in detection modeling were either consistently observed above LOD or below LOD in nearly all wristbands regardless of the individual. For example, 2-methylnaphthalene was not included in detection modeling because it was detected in 96% and 99% of Eugene and St. Helens wristbands, respectively. In addition, dibenzothiophene was not included in detection modeling because it was detected in only 1.6% and 100% of Eugene and St. Helens wristbands, respectively. A full list of these chemicals for each study is provided in Table S2.
PM2.5
In the Eugene study, the minimum, median, and maximum daily mean PM2.5 values were 1.73 µg/m3, 6.6 µg/m3, and 179.0 µg/m3, respectively. In the St. Helens study, the minimum, median, and maximum daily mean PM2.5 values were 2.9 µg/m3, 6.2 µg/m3, and 13.1 µg/m3, respectively. A figure showing the distribution of daily mean PM2.5 values between the Eugene and St. Helens studies is in the SI (Fig. S1).
HMS
All HMS smoke density categories were observed in the Eugene study; “none” was the most frequently detected category and “heavy” was the second most frequently detected category (Fig. S2). Only two HMS smoke density categories were observed in the St. Helens study (“none” and “light” categories) and “none” was the most frequently observed (Fig. S2).
Outdoor temperature
In the Eugene study, the minimum, median, and maximum outdoor daily temperatures were 36 °F, 64 °F, and 97 °F, respectively. In the St. Helens study, the minimum, median, and maximum outdoor daily temperatures were 45 °F, 52 °F, and 87 °F, respectively. A figure showing the distribution of daily outdoor temperatures between the Eugene and St. Helens studies is in the SI (Fig. S3).
Variance attribution
We looked at the consistency of concentration values and the probability of detecting a chemical within an individual after accounting for environmental factors in the Eugene (Fig. 3) and St. Helens studies (Fig. 4).
The percentage of variability in the response variable attributed to the variance in response values between subjects against the variance in response values within subjects for the (A) log2 chemical concentration and B probability of chemical detection in the Eugene study. The size of a point corresponds to the percentage of variability in the response that is explained by the observed environmental variables: maximum daily temperature, smoke category, and PM2.5.
The percentage of variability in the response variable attributed to the variance in response values between subjects against the variance in response values within subjects for the (A) log2 chemical concentration and B probability of chemical detection in the St. Helens study. The size of a point corresponds to the percentage of variability in the response that is explained by the observed environmental variables: maximum daily temperature, smoke category, and PM2.5.
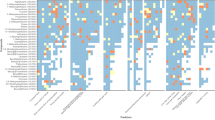
The sources of variability for many chemicals differ between Eugene (Fig. 3) compared to St. Helens (Fig. 4). For example, in Fig. 3A, 1,2-dimethylnaphthalene is in the lower right-hand corner for Eugene, which indicates there is high consistency in measured concentration within each individual after accounting for environmental factors. In Fig. 4A, B, 1,2-dimethylnaphthalene is located more toward the upper left corner in St. Helens, indicating this chemical varies considerably within each individual.
In Fig. 3A, pyrene and TCEP are located in the upper left corner, indicating that these chemical concentrations vary quite a bit within each individual after accounting for environmental factors in Eugene. Variability between people accounts for 87.2% and 78.5% of the total variation in concentration for pyrene and TCEP, respectively. In Fig. 4A, there is only 1 chemical (coronene) in the upper left corner for St. Helens. Differences in chemical concentrations between people accounted for more than half of the total variability in 18 chemicals in Eugene and 5 chemicals in St. Helens.
There are also differences in the lower right-hand corner of the plots between the studies. In Fig. 3B, there are no chemicals in the lower right-hand corner for Eugene; indicating there are no chemicals with high consistency in detection probability within each individual after accounting for environmental factors. However, there are several chemicals in St. Helens in the lower right-hand corner (Fig. 4A, B). For Fig. 4B, there is a group of PAHs detected in St. Helens (with relatively higher molecular weights) with high consistency within each individual.
In Fig. S4, the proportion of variability in each observation type, for each chemical, explained by measurements coming from the same individual is shown. Chemicals near the line are consistent in terms of the amount of variability that can be attributed to observations coming from the same individual. In Eugene’s concentration and detection values for retene, the intrasubject measurements accounted for approximately 50% of the total variability in measurements (Fig. S4A). This means that chemical concentration values and detection values (i.e., a chemical is consistently above or consistently below LOD for an individual) are very consistent within an individual. Additionally, for styrene in Eugene, the variability explained by intrasubject measurements is low (bottom left of the plot) and inconsistent in both concentration and detection values (Fig. S4A). In contrast, for phenanthrene in Eugene, the variability explained by intrasubject concentration measurements is higher (i.e., the concentration of phenanthrene within a person is more consistent) than the variability explained by intrasubject detection measurements (i.e., whether phenanthrene is detected within a person is not consistent; Fig. S4A).
Figures 3 and 4 highlight which chemicals have more variability explained by environmental factors. For example, in Fig. 3B, the circles are larger for o-xylene, styrene, and ethylbenzene compared to the majority of other chemicals detected in Eugene, indicating that these chemical detection probabilities are explained more by environmental factors.
Figure 5 shows the proportion of the variability in the response variable, for each model, that could be explained by the combination of environmental factors. Environmental variables were not helpful in explaining observed variability in the data for a majority of chemicals.
The Eugene study is in (A), and St. Helens is in B. Chemical names are displayed when the combined environmental variable R2 was at or above 0.1 for at least one of the models. Red circles indicate chemicals where the detection model was not run and R2 values for the detection model were assumed to be zero for visualization purposes.
Chemicals where the environmental variables had explanatory value tended to be in the detection model only (Fig. 5). In the detection model for the Eugene study, the R2 values attributed to the combined set of environmental variables ranged from 0.51 to 0.76 for styrene, o-xylene, ethylbenzene, and phenanthrene (Fig. 5A). In Eugene, a combination of information from PM2.5 and HMS is driving the higher combined R2 values for styrene, o-xylene, ethylbenzene, and phenanthrene in the detection model (Table S3). For phenanthrene in the detection model, the individual R2 for PM2.5 and HMS is 0.20 and 0.75, respectively (Table S3). In the detection model for the St. Helens study, the R2 values attributed to the combined set of environmental variables ranged from 0.46 to 0.93 for naphthalene, 2-ethylnaphthalene, and 1,4-dimethylnaphthalene (Fig. 5B). In St. Helens, a combination of information from HMS and maximum daily temperature information is driving the higher combined R2 values in the detection model (Table S4). For 2-ethylnaphthalene, the individual R2 for HMS and maximum daily temperature is 0.66 and 0.63, respectively (Table S4).
Comparison between studies
The chemicals that were highly consistent within an individual did not agree well between the studies (see lower right- and upper left-hand corners; Fig. 6). These chemicals include 1,2-dimethylnaphthalene, phenanthrene and fluoranthene. For example, in Eugene, 1,2-dimethylnaphthalene is more consistent within subjects compared to between subjects (opposite true for St. Helens).
The ratio of variability in chemical concentration values for the 12 chemicals observed in both studies explained by within subject measurements over between subject measurements for Eugene (x-axis) and St. Helens (y-axis) studies. The dashed line represents where the two ratios are equal between studies.
The chemicals in the lower left-hand corner (1-methylnaphthalene, 2-methylnaphthalene, retene, naphthalene, and 1,4-dimethylnaphthalene) had similar proportions of total variance attributed to within and between subject variability across the two studies (Fig. 6). The detection probability results only had three chemicals so those are not included in Fig. 6 (Table S5).
Discussion
Repeated measures
A 2025 commentary from a lead exposome researcher, Dr. Wild, states that the “capture of the time dimension of the exposome remains crucial” and recommends that future research collect exposure data at more than one time point [37]. A strength of our study is that we enhance understanding of this time dimension data gap and our results demonstrate the importance of measuring personal chemical exposure at different time points for certain chemicals, and that this varies between sites.
Figure 6 shows that chemicals close to the dotted line and with a ratio of 1 or more (i.e., 1-methylnaphthalene, 2-methylnaphthalene, 1,4-dimethylnaphthalene, retene, and naphthalene) were consistent within an individual across different days and agreed well between the studies. While more research is needed to confirm this trend, chemical data for these five chemicals is potentially similar day-to-day for a person. If these are the primary chemicals of interest in a study, then repeated measures may not be as important as capturing a larger swath of the population when determining how to prioritize sampling.
In contrast, the results from our two studies in Fig. 6 also reveal a group of six chemicals that are highly consistent within an individual across different days but that do not agree well between the studies (i.e., 1,2-dimethylnaphthalene, fluoranthene, pyrene, fluorene, 1-methylphenanthrene, and 2-methylphenanthrene). Guo et al. also showed a high consistency within an individual for 1,2-dimethylnaphthalene [11], in agreement with St. Helens but not Eugene. For this subset of chemicals, our results indicate that measuring chemical data at different time points across study groups in future studies is important.
More studies are needed to evaluate and determine consistency of variability patterns observed in exposome research. Often, studies are limited in the number of samples that can be taken or analyzed and prioritizing sample types is an important consideration in the study design. As additional studies become available, the data analysis approach we demonstrate here can help identify which chemicals would benefit most from consistently generating repeated measurement data, rather than measurements across more individuals, when determining study design. Our approach can also help evaluate and quantify sources of variability such as behavioral or environmental factors in future studies. These approaches may help identify chemicals that are less affected by certain behavioral or environmental factors. Figure 6 shows that all 11 chemicals have higher within-subject variability than between-subject variability, which may suggest that environmental factors impacted chemical exposures more than behavioral factors.
Concentration and detection data reveal different, but complementary, information about exposure variability between and within people. Our analysis demonstrates the importance of looking at both these types of data together rather than just one type (i.e., concentration or detection results) alone. For example, Fig. 3A indicates that pyrene concentrations in Eugene vary quite a bit within each individual after accounting for environmental factors. We would not have reached the same conclusion had we only looked at the detection results (Fig. 3B). Further, Fig. S4 highlights differences and similarities between the concentration and detection model accounted for by within subject observations; for example, in the Eugene study, when detected, the concentration of phenanthrene within a person is consistent, but whether phenanthrene is detected for the same person is not consistent (Fig. S4A).
Environmental factors
Characteristics of the Eugene and St. Helens studies are fundamentally different, so it is not surprising that we observed different R2 values for different environmental factors (Tables S3 and S4; R2 values represent the proportion of variability in the chemical data explained by each environmental factor). For example, R2 patterns look different for the Eugene study compared to the St. Helens study, with a combination of information from PM2.5 and HMS driving the higher combined R2 values in the detection model for Eugene (Table S3) and a combination of information from HMS and maximum daily temperature information driving the higher combined R2 values in the detection model for St. Helens (Table S4). The Eugene and St. Helens studies contrast for many reasons, including different sets of chemicals (Table S2); different observed PM2.5 values (Fig. S1), HMS categories (Fig. S2), and temperatures (Fig. S3); and different participant demographics (Table S1). Differing PM2.5, temperature, and smoke values between the two cities may be the result of localized wildfires near Eugene within the sampling window. In addition, each study was in a different geographic location with different features that could influence chemical exposure. For example, Eugene is a larger city with an industrial corridor while St. Helens is a smaller town (Section “ Site descriptions and study cohorts”).
Overall, for most chemicals, environmental variables were not sufficient for explaining the variability in the data (Fig. 5). Only 21% of the modeled chemicals (9 out of 42) for Eugene and 30% of the modeled chemicals (11 out of 37) for St. Helens, had a combined environmental variable R2 at or above 0.1 for at least one of the models. This finding is in agreement with conclusions in Bramer et al. which found that using fine particulate matter data from stationary air monitors and smoke density data from satellites is not enough to understand personal chemical exposure [5]. We also observed that most cases where the combined environmental variable R2 was above 0.1 was in the detection model rather than the concentration model (Tables S3 and S4), which could indicate that environmental factors play a bigger role in getting to a minimum threshold of detection but are less important to concentration gradients in exposures.
There is still value to including these high-level environmental factors in an analysis of chemical exposure variability data. Trends emerged from our analysis, such as observed higher combined R2 values for styrene, o-xylene, ethylbenzene, and phenanthrene in the Eugene detection model (Fig. 5A) which came from a combination of PM2.5 and HMS information (Table S3). Some of the explanatory power here could be from the heavy wildfire smoke that was present during portions of the Eugene study.
Future studies could benefit from including additional environmental factors and participant behavior information to better understand chemical exposure variability trends. Some of the environmental factors in our investigation may partially be a proxy for behavior; for example, people may spend more time indoors when it is hot outside or when there is wildfire smoke. Future research could focus on understanding which environmental factors are linked with behaviors and how certain behaviors like spending time indoors, turning on the AC, or traveling influences personal chemical exposure variability.
Modeling considerations
Our statistical methods provide a distinct advantage over previous correlative metrics used to characterize variability in wristband studies with repeated measures. In addition to the quantification of inter- and intra-subject variability, variability measures can be obtained while accounting for known and unknown sources (such as environmental conditions or measured subject behavior) of variability in measured chemical exposure. Unlike previous methods, the models presented here can be used to simultaneously evaluate the influence of any potential explanatory variables on the exposome while accounting for inter- and intra-subject variability. Finally, we demonstrated that models of chemical concentration provide different information to models of chemical detection for the same chemical. Thus, the capability of the modeling approach in this work provide the advantage of evaluating both chemical outcome variables.
Additional considerations
Different sets of chemicals were measured in the Eugene and St. Helens studies (Table S2). A strength of this work is that a broad range of chemicals were measured in each study. Forty-three chemicals were measured in both studies, but only 12 chemicals had enough data to be modeled in both studies, which reflects that the chemical measurement patterns were fundamentally different between the two studies. Our analysis demonstrates the importance of measuring the same chemicals across different studies to fully understand variability between and within people on a per-chemical basis.
A general limitation of community-driven research is that people who choose to volunteer for a research study are likely not representative of the entire community. The demographics of the 81 participants in our study are not representative of the overall Oregon or U.S. population. Here, participants in both St. Helens and Eugene were primarily white and had some college education (Table S1). Further, all participants in Eugene were asthmatics. Participants with asthma potentially may have different behaviors (and exposures) compared to people without asthma; for example, asthmatics may choose to stay indoors all day with air filters on during periods of heavy wildfire smoke.
We were able to include some potential location and source-specific influences in this work such as heavy wildfire smoke in Eugene that may have contributed to chemical exposures in the Eugene cohort. However, chemicals detected from wildfire smoke can also be prevalent in car exhaust, indoor sources such as stovetops, and smoking products. For example, phenanthrene is detected in both the Eugene and St. Helens cohorts at relatively high frequencies (Table S2). Phenanthrene is commonly found in wildfire smoke, but also in tobacco smoke and coal/oil combustion.
Conclusions
This manuscript is the largest investigation of intra- and inter- variability in wristband concentrations, containing 23,812 chemical data points from 586 wristbands across two different personal exposure assessment studies. This is also the first wristband study to investigate intra- and inter- variability between studies with more than two repeated measures per person. Repeated measures from two different wristband studies enabled us to investigate personal chemical exposure variability patterns throughout different months and years (Section “ Site descriptions and study cohorts”) and during varying outdoor environmental conditions (e.g., the Eugene study captured much higher levels of PM2.5 and HMS than the St. Helens study; Figs. S1 and S2). Overall, the large sample size, additional repeated measures, inclusion of environmental factors, and advanced statistical methods in this manuscript enabled us to reach several conclusions and recommendations for future exposomic research.
Data availability
Non-personally identifiable wristband data for participants that provided consent for their data to be shared are available in the PNNL DataHub repository, [https://data.pnnl.gov/group/nodes/dataset/34274].
References
Wild CP. The exposome: from concept to utility. Int J Epidemiol. 2012;41:24–32.
Exposome Moonshot Forum Infographic: Exposome Moonshot Forum; 2025 [Available from: https://exposomemoonshot.org/two-pager/].
NIEHS. NIEHS 2025-2029 Strategic Plan: Health at the Intersection of People and Their Environments Research Triangle Park, NC: U.S. National Institute of Environmental Health Sciences (NIEHS), U.S. Department of Health and Human Services; 2025 [Available from: https://www.niehs.nih.gov/sites/default/files/strategic-plan-2025_long_print_508.pdf].
About the Exposome Moonshot Forum: Exposome Moonshot Forum; 2025 [Available from: https://exposomemoonshot.org/about/].
Bramer LM, Dixon HM, Rohlman D, Scott RP, Miller RL, Kincl L, et al. PM2.5 is insufficient to explain personal PAH exposure. GeoHealth. 2024;8:e2023GH000937.
Liu JC, Pereira G, Uhl SA, Bravo MA, Bell ML. A systematic review of the physical health impacts from non-occupational exposure to wildfire smoke. Environ Res. 2015;136:120–32.
O’Connell SG, Kincl LD, Anderson KA. Silicone wristbands as personal passive samplers. Environ Sci Technol. 2014;48:3327–35.
Samon SM, Hammel SC, Stapleton HM, Anderson KA. Silicone wristbands as personal passive sampling devices: current knowledge, recommendations for use, and future directions. Environ Int. 2022:107339.
Murcia-Morales M, Díaz-Galiano FJ, Gómez-Ramos MJ, Fernández-Alba AR. Human exposure to PAHs through silicone-based passive samplers: Methodological aspects and main findings. TrAC, Trends Anal Chem. 2024;173:117643.
Hamzai L, Lopez Galvez N, Hoh E, Dodder NG, Matt GE, Quintana PJ. A systematic review of the use of silicone wristbands for environmental exposure assessment, with a focus on polycyclic aromatic hydrocarbons (PAHs). J Expo Sci Environ Epidemiol. 2022;32:244–58.
Guo P, Lin EZ, Koelmel JP, Ding E, Gao Y, Deng F, et al. Exploring personal chemical exposures in China with wearable air pollutant monitors: A repeated-measure study in healthy older adults in Jinan, China. Environ Int. 2021;156:106709.
Huang Z, Peng C, Rong Z, Jiang L, Li Y, Feng Y, et al. Longitudinal mapping of personal biotic and abiotic Exposomes and transcriptome in underwater confined space using wearable passive samplers. Environ Sci Technol. 2024;58:5229–43.
Dixon HM, Armstrong G, Barton M, Bergmann AJ, Bondy M, Halbleib ML, et al. Discovery of common chemical exposures across three continents using silicone wristbands. R Soc Open Sci. 2019;6:181836.
Dixon HM, Bramer LM, Scott RP, Calero L, Holmes D, Gibson EA, et al. Evaluating predictive relationships between wristbands and urine for assessment of personal PAH exposure. Environ Int. 2022;163:107226.
Hammel SC, Phillips AL, Hoffman K, Stapleton HM. Evaluating the Use of Silicone Wristbands To Measure Personal Exposure to Brominated Flame Retardants. Environ Sci Technol. 2018;52:11875–85.
Hoffman K, Levasseur JL, Zhang S, Hay D, Herkert NJ, Stapleton HM. Monitoring human exposure to organophosphate esters: comparing silicone wristbands with spot urine samples as predictors of internal dose. Environ Sci Technol Lett. 2021;8:805–10.
Donald CE, Scott RP, Blaustein KL, Halbleib ML, Sarr M, Jepson PC, et al. Silicone wristbands detect individuals’ pesticide exposures in West Africa. R Soc Open Sci. 2016;3:160433.
Bonner EM, Poutasse CM, Haddock CK, Poston WSC, Jahnke SA, Tidwell LG, et al. Addressing the need for individual-level exposure monitoring for firefighters using silicone samplers. J Expo Sci Environ Epidemiol. 2025;35:180–95.
Evoy R, Kincl L, Rohlman D, Bramer LM, Dixon HM, Hystad P, et al. Impact of acute temperature and air pollution exposures on adult lung function: a panel study of asthmatics. PLoS One. 2022;17:e0270412.
Rohlman D, Dixon HM, Kincl L, Larkin A, Evoy R, Barton M, et al. Development of an environmental health tool linking chemical exposures, physical location and lung function. BMC Public Health. 2019;19:854.
City and Town Population Totals: 2020-2023 [Internet]. United States Census Bureau. 2024 [cited 8-20-2024]. Available from: https://www.census.gov/data/tables/time-series/demo/popest/2020s-total-cities-and-towns.html.
OSU. ArcGIS StoryMap: The J.H. Baxter & Co. Wood Treatment Facility in West Eugene. Pacific Northwest Center for Translational Environmental Health Sciences Research at Oregon State University (OSU), with assistance from the Oregon Department of Environmental Quality, Oregon Health Authority, Lane Regional Air Protection Agency, and the City of Eugene; 2022.
EPA proposes adding JH Baxter site in West Eugene to Superfund cleanup list [press release]. U.S. Environmental Protection Agency (EPA) 2024.
Bonner EM, Clark AE, Bramer LM, Rohlman D, Tidwell LG, Waters KM, et al. Integrating personal behaviors, demographics, and housing characteristics with PAH exposure through silicone wristbands. In Review, Environmental Pollution. 2025.
Ghetu CC, Moran IL, Scott RP, Tidwell LG, Hoffman PD, Anderson KA. Concurrent assessment of diffusive and advective PAH movement strongly affected by temporal and spatial changes. Sci Total Environ. 2024;912:168765.
Anderson, Points III KA, GL, Donald CE, Dixon HM, Scott RP, et al. Preparation and performance features of wristband samplers and considerations for chemical exposure assessment. J Expo Sci Environ Epidemiol. 2017;27:551.
Paulik LB, Hobbie KA, Rohlman D, Smith BW, Scott RP, Kincl L, et al. Environmental and individual PAH exposures near rural natural gas extraction. Environ Pollut. 2018;241:397–405.
Kile ML, Scott RP, O’Connell SG, Lipscomb S, MacDonald M, McClelland M, et al. Using silicone wristbands to evaluate preschool children’s exposure to flame retardants. Environ Res. 2016;147:365–72.
Outdoor Air Quality Data: Download Daily Data Tool [Internet]. U.S. Environmental Protection Agency (EPA). 2024 [cited 5-24-2024]. Available from: https://www.epa.gov/outdoor-air-quality-data/download-daily-data.
Hazard Mapping System (HMS) [Internet]. National Oceanic and Atmospheric Administration (NOAA) Office of Satellite and Product Operations. 2024 [cited 5-24-2024]. Available from: https://www.ospo.noaa.gov/Products/land/hms.html#data.
Ruminski M, Simko J, Kibler J, Kondragunta S, Draxler R, Davidson P, et al., editors. Use of multiple satellite sensors in NOAA’s operational near real-time fire and smoke detection and characterization program. Remote Sensing of Fire: Science and Application; 2008: SPIE.
O’Dell K, Ford B, Burkhardt J, Magzamen S, Anenberg SC, Bayham J, et al. Outside in: the relationship between indoor and outdoor particulate air quality during wildfire smoke events in western US cities. Environ Res: Health. 2022;1:015003.
Climate Data Online: Dataset Discovery [Internet]. National Oceanic and Atmospheric Administration (NOAA). National Centers for Environmental Information. 2024 [cited 5-24-2024]. Available from: https://www.ncei.noaa.gov/cdo-web/datatools/lcd.
R Development Core Team R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing;; 2024.
Bates D, Mächler M, Bolker B, Walker S. Fitting linear mixed-effects models using lme4. J Stat Softw. 2015;67:1–48.
Stoffel MA, Nakagawa S, Schielzeth H. partR2: partitioning R2 in generalized linear mixed models. PeerJ. 2021;9:e11414.
Wild CP. The exposome at twenty: a personal account. Exposome. 2025;5:osaf003.
Acknowledgements
We thank the participants in each study. Additionally, at Oregon State University, we thank Laurel Kincl, Richard Scott, Michael Barton, Peter Hoffman, Carolyn Poutasse, Clarisa Caballero-Ignacio, Lane Tidwell, Kaley Adams, Caoilinn Haggerty, Jessica Scotten, and other members of the Food Safety and Environmental Stewardship Laboratory. Particular thanks to Emily Bonner for her work on the St. Helens project. We also thank the OSU Superfund Research Center (specifically Core D for chemistry support) and DMAC at PNNL for modeling support. PNNL is a multi-program laboratory operated by Battelle for the U.S. Department of Energy under contract DEAC05-76RL01830.
Funding
Research reported in this publication was supported by the National Institute of Environmental Health Sciences (NIEHS) under award numbers 1R21ES024718, 4R33ES024718, P30ES030287, and P42ES016465. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or NIEHS. Holly M. Dixon was supported in part by the ARCS Foundation® and both Holly M. Dixon and Alison E. Clark were supported in part by the NIEHS Fellowship T32ES007060.
Author information
Authors and Affiliations
Contributions
LMB: Methodology, Investigation, Formal analysis, Data Curation, Visualization, Software, Validation, Writing—Original Draft, Writing—Review & Editing. HMD: Methodology, Investigation, Visualization, Project administration, Writing—Original Draft, Writing—Review & Editing. AEC: Visualization, Writing—Original Draft, Writing—Review & Editing. DR: Investigation, Writing—Review & Editing. KMW: Conceptualization, Methodology, Visualization, Supervision, Funding acquisition, Writing—Review & Editing. KAA: Conceptualization, Methodology, Visualization, Supervision, Project administration, Funding acquisition, Writing—Review & Editing.
Corresponding author
Ethics declarations
Competing interests
KAA and DR, authors of this research, disclose a financial interest in MyExposome, Inc., which is marketing products related to the research being reported. The terms of this arrangement have been reviewed and approved by Oregon State University in accordance with its policy on research conflicts of interest. The authors have no other relevant financial or non-financial interests to disclose.
Ethical approval
We obtained informed consent from all study participants in accordance with the Oregon State University Institutional Review Board (#IRB-2020-0899, # IRB-2020-0529). All participants included in this study provided informed written consent to participate and for publication of the aggregate data. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bramer, L.M., Dixon, H.M., Clark, A.E. et al. Characterizing variability in personal chemical exposure to improve exposomics. J Expo Sci Environ Epidemiol 36, 221–230 (2026). https://doi.org/10.1038/s41370-025-00822-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41370-025-00822-x