Abstract
Background
Evidence on cognitive function in relation to per- and polyfluoroalkyl substances (PFAS) exposure in firefighters is scarce.
Objective
We conducted a pilot study aiming to explore the associations of individual PFAS analyte and PFAS mixture with domain-specific cognitive performance in firefighters.
Methods
Firefighters (n = 65), who were actively serving four cities in Central Texas 2003–2025, were assessed for global cognition and processing speed measured by the NIH Toolbox Cognition Battery. Whole blood samples were collected from these participants and analyzed by liquid chromatography-tandem mass spectrometry for 24 PFAS analytes. Linear regression models were used to evaluate associations between cognitive performance and both individual PFAS analytes and the overall 24-PFAS mixture. Additionally, quantile g-computation and Bayesian Kernel Machine Regression (BKMR) were applied to investigate associations between 6-PFAS mixture and cognitive performance, with adjustment for potential covariates.
Results
Six PFAS analytes including potassium perfluoro-1-octanesulfonate (PFOS), perfluoro-n-octanoic acid (PFOA), perfluoro-n-heptanoic acid (PFHpA), perfluoro-n-nonanoic acid (PFNA), perfluoro-n-decanoic acid (PFDA), and potassium perfluoro-1-hexanesulfonate (PFHxS) were detected in the least 70% of participants. PFOS was negatively associated with scores of processing speed (ß = –0.58 p < 0.001). Neither the 6-PFAS mixture nor the 24-PFAS mixture was associated with either cognitive domain.
Significance
These preliminary findings may inform future research seeking to clarify the biological pathways linking PFAS exposure to cognitive outcomes and to explore possible modifiable factors related to cognitive health in firefighters. They might also underscore the potential value of continued effort to minimize workplace PFAS exposure for this occupational group.
Impact
Our study provides the first data on PFAS exposure and cognitive performance for firefighters. Our analysis revealed that PFOS was the primary contributor among the PFAS mixture (consisting of PFOS, PFOA, PFHxS, PFHpA, PFNA, and PFDA) linked to reduced processing speed scores in firefighters. Our findings pave the way for future, larger studies that observe firefighters for longer duration to develop confident estimates of cognitive health in relation to PFAS exposure. These data could inform analyses in support of workplace exposure reduction efforts and revision of preventive health care for first responders.
Similar content being viewed by others
Introduction
Per- and polyfluoroalkyl substances (PFAS) are a class of synthetic fluorinated chemicals. Due to their unique physicochemical properties (e.g., non-stick, heat and acid resistant, waterproof, oil repellent, and stain resistant), PFAS have been widely used in consumer products and industry (e.g., surface coating and protectant formulations) since the 1950s [1]. Owing to their recalcitrant (non-biodegradable) and persistent (i.e., long half-lives) properties, PFAS are now called the “forever chemicals” and have potential for accumulation in human body [1, 2]. PFAS can enter the human body through inhalation, ingestion, and dermal absorption and accumulate in the body for years [3, 4]. Epidemiological studies have documented the adverse effect of legacy or the long-chain PFAS, such as perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA), on human health including cancer, immune suppression, thyroid dysfunction, metabolic effects, increased lipid levels, ulcerative colitis, liver and kidney function in adults [2, 5, 6] and neurodevelopment in children, including executive function deficits [7], cognitive dysfunction [8], and attention deficit and hyperactivity disorder [9]. Additionally, evidence from animal studies has suggested that PFOS and PFOA can reduce function of the nervous system and are potentially neurotoxic and correlate with neurobehavioral dysfunction, such as late-onset Alzheimer’s disease (AD) and learning and memory deficits [1, 10,11,12]. These studies have suggested two major potential pathways of PFAS neurotoxicity: (1) PFAS can disturb calcium homeostasis and induce calcium increase/overload in neurons. Calcium overload can disrupt the normal function of neurons and provoke neuronal excitement, which could lead to neuronal injury and cell death. In particular, PFAS tends to distribute to and accumulate in the hippocampus of the brain, a region implicated in learning and memory, and disturbs hippocampal neurons leading to cognitive impairment [13]; and (2) PFOS and PFOA can cross the blood-brain barrier (BBB) and alter neurotransmitter (e.g., dopamine, glutamate, and acetylcholine) concentrations which can influence the activation of neurons and signal transmission among neurons. Increases or decreases in neurotransmitters could lead to neuron cell death [1, 13].
Recent epidemiological studies have reported mixed findings on the relationship between cognitive function and serum PFAS in middle-aged and older adults in the United States [5, 14,15,16]. Shrestha et al. [14] found that higher serum PFOA and PFOS concentrations were associated with better performance in memory and learning, executive function, and visuospatial function among 126 adults aged 55–74 years old who lived in upper Hudson River communities. A cross-sectional study with 21,024 adults aged 50 and above, who lived in Mid-Ohio Valley, West Virginia, reported the protective impacts of PFOS, PFOA, and PFHxS on self-reported short-term memory [15]. A longitudinal study using data from participants from the National Health and Nutrition Examination Survey (NHANES; 2011–2014), a nationally representative sample of the U.S. population, suggested that a low level of exposure to a PFAS mixture (i.e., PFOA, PFOS, and PFHxS) is positively associated with cognitive performance in Americans aged 60 and above [5]. On the contrary, Park et al. [16], who studied the same NHANES population, revealed that higher levels of serum PFOS was associated with lower scores of global cognition after controlling for chronic kidney disease (CKD), non-monotonic dose-responses, and seafood consumption. Additionally, medical research has found that Alzheimer’s patients had significantly higher PFAS levels in their cerebrospinal fluid than non-AD patients [17], and high concentrations of PFOA have been found impairing dopaminergic neurons in the brain stems of autopsied adults, possibly impairing cognition [18]. While the body of literature on PFAS toxicity has expanded significantly in recent decades, it remains heavily skewed toward legacy PFAS (e.g., PFOS and PFOA) that have been phased out in many regions due to regulatory pressures. In contrast, studies examining the health impacts of PFAS mixtures—particularly those incorporating both legacy compounds and alternatives like shorter-chain analogs or fluorinated ethers [e.g., such as perfluorobutanoic acid (PFBA), perfluorohexanesulfonic acid (PFHxS), perfluorobutane sulfonic acid (PFBS), and perfluoroheptanoic acid (PFHpA)], often designed as less harmful substitutes (lower toxicity profiles and shorter environmental half-lives)—are notably underrepresented, highlighting a critical gap in understanding synergistic or cumulative effects in real-world exposure scenarios.
PFAS exposure may be of particular relevance to firefighting because these compounds are used in turnout gear, training activities in airports and military bases, and are a major ingredient of some firefighting foams, such as aqueous film-forming foams (AFFF) or Class B foam [19, 20]. AFFF are known to contain PFAS surfactants, including PFOS, PFOA, and PFHxS [2, 21]. Firefighters may be exposed to these PFAS compounds through inhalation of aerosolized foam, direct or indirect skin contact with contaminated personal protective equipment (PPE), as well as hand to mouth transfer during the use of AFFF [2, 22,23,24,25,26,27]. A number of biomonitoring studies have reported elevated serum PFAS concentrations in firefighters relative to the general population, likely related at least in part to occupational contact with PFAS-containing materials [20, 27,28,29]. Some studies have reported associations between PFAS exposure and increased risks of certain health issues—like cancer, kidney disease, and thyroid dysfunction—among firefighters, though the evidence is still being evaluated and causality has not been definitively established [30,31,32]. Additionally, firefighters might be under notable stress in their work due to the great workload and psychological stressors during fire and rescue operations. Unlike many other professions, firefighters are engaged in an occupation in which they are exposed to several stressors (e.g., night shift schedules, sleep deprivation, sudden alarm calls, strenuous physical work, exposure to smoke, heat, and other harmful substances during fire suppression) for an unforeseeable amount of time [33]. All the above factors can contribute to short-term and long-term impairment of mental health in firefighters [34]. Despite mounting concerns about firefighter health, few studies have assessed how exposure to PFAS may influence cognitive function in firefighters and cognitive health of firefighters is largely understudied worldwide. Such data are urgently needed given rapid population aging and the increase in the number of people having Alzheimer’s disease and related dementias (AD/ADRD). To address these notable gaps, objectives of this pilot study were to (1) identify and quantify concentrations of individual PFAS analyte and their mixture in blood, and (2) assess the association between PFAS and domain-specific cognitive performance in firefighters.
Materials/subjects and methods
Participants and recruitment
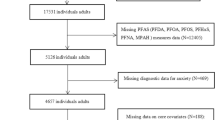
Participant recruitment, cognitive assessments, and general surveys capturing sociodemographic, basic health, and occupational characteristics were conducted between June 2023 and March 2025. Blood samples were collected between January and March 2025. Fire departments in Kyle, San Marcos, New Braunfels, and San Antonio in Texas assisted with the recruitment of firefighter participants by sending announcements through their listservs and newsletters. To recruit firefighter participants from San Antonio, a recruitment booth was established at the annual health fair in February 2024 and 2025 supported by the San Antonio Fire Department. Interested firefighters were invited to complete a screening survey to determine their eligibility for the study. Study inclusion criteria included career firefighters who were age of 18 and above, having at least 5 years of service at the city level, without a history of blood disease, central neurological disease, major psychiatric disorder, alcohol or substance abuse, serious medical illness (renal, hepatic, cardiac, or pulmonary insufficiency, cancer), or psychoactive drug use. In addition, career firefighters needed to be currently on “activity duty” at the time of recruitment. Following eligibility screening, 68 participants were scheduled for cognitive assessment and requested to complete a general survey. Of these, 65 were scheduled for blood collection, while 3 participants declined to provide their blood samples. Therefore, our final sample consisted of 65 participants with complete data. Participants who completed their cognitive assessment and provided their blood samples received a $60 gift card. All participants were consented into the study following protocols approved by the Institutional Review Board of Texas State University (#8639).
Cognitive function assessment
Trained research staff conducted a 30–45-min cognitive assessment per standard protocols with each participant using an iPad in a reserved quiet room. We selected the NIH Toolbox Cognition Battery (NIHTB-CB) to assess the cognitive performance of our participants due to its validated sensitivity to domain-specific cognitive variations in a younger, healthier study population, including middle-aged individuals (ages 20–66 years), as demonstrated in recent validation studies [35, 36]. The Cognitive Battery is a computerized (iPad-based administration) neurobehavioral assessment consisting of multiple tests to measure cognitive domains including executive function, attention, episodic memory, language, processing speed, and working memory for a wide range of ages (7–85) [37, 38]. The Cognitive Battery has good scores on test-retest reliability, age effects on performance, and convergent and discriminant construct validity [37]. Following suggestions from neurologists and psychologists in our research team, we selected 7 tests to build our own battery to align with our research objectives and participant characteristics. The selected tests included Dimensional Change Card Sort (DCCS) Test for executive function, Flanker Inhibitory Control and Attention Test for attention, Picture Sequence Memory Test (PSMFA) for episodic memory, Pattern Comparison Processing Speed Test (PC) and Oral Symbol Digit (OSD) for processing speed, Face Name Associative Memory Exam (FNAMED) for associative memory, and List Sorting Working Memory Test (LSWM) for working memory. The new battery was piloted with our trained research staff to ensure clarity and functionality prior administration to study participants.
For each participant, an age-adjusted scale score for each individual test was produced by the Cognitive Battery after the administration. We calculated two summary scores to measure global cognition and processing speed. Calculation for the composite score of global cognition used all 7 tests, while computation for the composite score of processing speed used the PC and OSD tests. The composite scores were calculated by the average of the standardized z-scores of each test. Global cognition captures the overall cognitive function across multiple domains, reflecting the integrity of general cognitive abilities. Processing speed is a core component of cognitive function that allows first responders to handle information-rich, rapidly evolving situations, make critical decisions quickly, and perform their duties effectively under pressure [39]. Higher scores on these tests indicate better cognitive performance.
PFAS exposure assessment
Capillary whole blood (via finger prick) as the sample matrix was selected to prioritize logistical feasibility. This method eliminated the requirement for on-site centrifugation, thereby minimizing the time burden on the firefighter participants and simplifying field collection. Moreover, several human studies have identified specific PFAS (e.g., FOSA, PFOSA, and PFHxA) that partition into red blood cells are found only in whole blood [40,41,42]. Blood samples were collected by trained research staff at the health fairs and fire stations where participants were assigned. The collection was done through finger prick using lancets (21 gauge and 2.2 depth) and vials. Each participant’s fingertip was disinfected with a 70% isopropanol alcohol wipe before sample collection. The first drop of blood was discarded to avoid PFAS contamination from disinfection products or finger prick needles. Three drops of blood (~150 µL) were collected into a heparin-containing 2 mL Eppendorf polypropylene vials (approximately 30 USP units per mL of whole blood). The vials were stored in a cooler with ice for transportation within 3 h from the fingerstick site (e.g., health fairs or fire stations) to Texas State University and stored at –20 °C in a biological lab until shipped on dry ice to the laboratory at the Yale School of Public Health for analysis. Once received by the laboratory, samples will be stored at -20 °C until analysis per standard protocols. The Data Use Agreement (DUA) on data sharing were signed by Texas State University and Yale University.
Whole blood (50 µL) was aliquot into a 2-mL Eppendorf microcentrifuge tubes and spiked with a mixture of mass-labeled PFAS internal standards (20 µL). Samples were vortexed and sonicated for 15 min. Methanol (130 µL) was added and then samples were vigorously shaken on a for 30 min. Samples were transferred to a polypropylene cellulose acetate filter tube and then centrifuged for 15 min to remove protein precipitation and cell debris. The supernatant was collected for each sample and transferred to a polypropylene autosampler vial for analysis of 24 PFAS analytes on an Agilent 1290 Infinity liquid chromatograph (LC) coupled to an Agilent 6545 quadrupole-time-of-flight mass spectrometer (LC-QTOF-MS) in negative electrospray ionization mode. Measured analytes included perfluorohexane sulfonic acid (PFHxS), perfluoroheptanoic acid (PFHpA), perfluorooctanoic acid (PFOA), perfluorooctane sulfonic acid (PFOS), perfluorononanoic acid (PFNA), perfluorodecanoic acid (PFDA), perfluorobutane sulfonic acid (PFBS), perfluoroundecanoic acid (PFUnDA), sodium perfluoro-1-heptanesulfonate (PFHpS), perfluorohexanoic acid (PFHxA), perfluoropentane sulfonic acid (PFPeS), perfluorooctane sulfonamide (PFOSA), perfluorododecanoic acid (PFDoA), perfluorobutanoic acid (PFBA), perfluoro-n-pentanoic acid (PFPeA), perfluoro-n-tridecanoic acid (PFTrDA), perfluoro-n-tetradecanoic acid (PFTeDA), sodium perfluoro-1-nonanesulfonate (PFNS), sodium perfluoro-1-decanesulfonate (PFDS), sodium 1H,1H,2H,2H-perfluoro-1-hexanesulfonate (4:2 FTS), sodium 1H,1H,2H,2H-perfluoro-1-octanesulfonate (6:2 FTS), sodium 1H,1H,2H,2H-perfluoro-1-decanesulfonate (8:2 FTS), N-methylperfluoro-1-octanesulfonamidoacetic acid [linear and branched] (MeFOSAA), and N-ethylperfluoro-1-octanesulfonamidoacetic acid [linear and branched] (EtFOSAA).
Samples were injected (10 µL) onto a InfinityLab Poroshell 120 EC C18 analytical column (2.1 mm × 100 mm × 2.7 µm; Agilent) preceded by a SecurityGuard C18 Guard Cartridge (4 mm × 2 mm I.D.; Phenomenex) and two Zorbax DIOL guard columns (4.6 mm × 12.5 mm × 6 µm; Agilent). LC binary pump flow rate was set at 0.4 mL/min and the starting mobile phase composition was 75% A (2.5 mM ammonium acetate in water) and 25% B (2.5 mM ammonium acetate in methanol) between 0 to 1 min. From 1 min to 3 min, the organic mobile phase B percentage increased from 25 to 75%. From 3 min to 8 min, the percentage of mobile phase B further increased from 75% to 100% and was held at 100% until 12 min, when the end of the run was reached. Data-dependent acquisition mode was used with iterative exclusion over mass-to-charge ratio (m/z) ratios between 118 and 1500 Dalton (Da) with fragmentation spectra collected at a fixed collision energy at 40 electron votes (eV) from 50 to 1500 Da. Dynamic exclusion was enabled so that a feature would be excluded after one spectrum collected and released after 0.5 min. The samples were analyzed along with a six-point calibration curve with native PFAS concentrations at 0.01, 0.05, 0.1, 0.5, 1, 2 parts per billion (ppb) limits of detection (LOD).
Of the 24 PFAS analytes, PFHxS, PFHpA, PFOA, PFOS, PFNA, and PFDA were selected for this study because the detection frequency (DF) of these PFAS were > 70% in our participants and they were also monitored in the U.S. general population in NHANES (Center for Disease Control, NHANES 2013–2014 Laboratory Data. https://www.n.cdc.gov/nchs/nhanes/search/datapage.aspx?Component=Laboratory&CycleBeginYear=2013). Levels of these PFAS analytes in this pilot study were measured in whole blood, whereas the NHANES reported concentrations in serum. Despite different biological matrices, levels of these PFAS analytes in this study could be compared to those measured in NHANES, but only after applying well-established conversion factors (e.g., a 2:1 serum/whole-blood ratio ± 0.2 depending on hematocrit and specific PFAS). PFDoDA, PFTrDA, PFTeDA, PFNS, PFDS, 4:2 FTS, 6:2FTS, MeFOSAA, and EtFOSAA were not detected, while 6 PFAS analytes including PFBA, PFPeA, PFBS, FOSA, 8:2 FTS, and PFHxA were measured in less than 20% of participants. We log-transformed concentrations of PFHxS, PFHpA, PFOA, PFOS, PFNA, and PFDA. To capture the combined exposure profile of 24 PFAS analytes, we calculated a composite score (a weighted sum of 24 PFAS analytes, Bayesian Weighted Sums [BWS)] for each participant representing the overall mixture exposure to PFAS, while accounting for uncertainty, non-detected values/left censoring, and correlations among the analytes. Given 9 undetected PFAS analytes, this calculation was performed using Bayesian hierarchical model with censoring through the PROC MCMC procedure in SAS. Bayesian hierarchical model with censoring is designed for treating undetected values as censored observations rather than missing or zero. The censoring mechanism in the hierarchical model directly handles this by modeling the latent (true) concentrations for censored values, ensuring their contributions (often small weights for undetected analytes) are included in the weighted sum. PFAS analytes exhibited a linear association with each cognitive outcome (see Figs. S3 and S4 in the Supplementary Material).
Covariates
Sociodemographic and behavioral factors, basic health information, and occupational characteristics were obtained by self-reported questionnaires. A group of covariates was selected a priori based on experience in previous studies and a literature search: age (continuous, years), race (Non-Hispanic White vs. others), education (some college or less vs. Bachelor’s degree or above), annual individual income (≤$60,000 vs. >$60,000), smoking history (ever smoked cigarettes or electronic cigarettes), past alcohol consumption (average weekly standard drinks over the past year) and current consumption (average weekly standard drinks in the past 30 days), and depressive symptoms (Center for Epidemiologic Studies Depression Scale score ≥16 and/or use of antidepressant medication). We additionally considered seafood consumption (frequency of fish or shellfish consumption during the past 30 days: never, 1 to 3 times, ≥4 times) [16]. We also controlled for the potential impact of occupational characteristics including years of service at the city level, use of self-contained breathing apparatus (SCBA) last year, frequency of shower after a fire event, and frequency of use of firefighting foam for a fire event last year. We did not include gender as covariate due to the predominance of male firefighters in our sample.
Statistical analysis
Power analysis was conducted to estimate statistical power with the sample of 65 using PROC POWER and MULTIREG statements in SAS. When the R-squared (R2) for the full regression model and the R2 difference for predictors of interest was set to 0.30 and 0.105, respectively; α error was set to 0.05 and the statistical power (1–β error) to 95%, the actual statistical power was moderate, 0.865.
Descriptive analyses were conducted to examine the sociodemographic characteristics of the participants, distributions of each PFAS analytes, and cognitive performance in the sample. Independent t tests were performed for continuous variables and Chi-squared tests for categorical variables. We calculated summary statistics including DF, arithmetic mean, standard deviation, and percentiles for each PFAS analytes.
To assess the associations of individual PFAS analyte (PFHxS, PFHpA, PFOA, PFOS, PFNA, and PFDA) and the 24-PFAS mixture with cognitive performance, we conducted separate sequential linear regression models to check if the relationship can be explained by different potential covariates. The initial model (Model 1) was the unadjusted model. Model 2 was based on Model 1 adjusting for age, race, education, income, cigarette smoking status, alcohol consumption, seafood consumption, and depressive symptoms. Model 3 controlled for variables in Model 2 as well as occupational characteristics: years of service, use of SCBA, frequency of shower after a fire event, and frequency of use of firefighting foam. We utilized PROC GENMOD procedure in SAS for model analyses.
We used quantile g-computation and Bayesian kernel machine regression (BKMR) to assess the association of the 6-PFAS mixture (PFHxS, PFHpA, PFOA, PFOS, PFNA, and PFDA) with cognitive performance. These methods can handle non-linearities, non-additive interactions among PFAS analytes, and variable selection in mixture, and estimate the joint association of a simultaneous one-quartile increase in the mixture with performance in each cognitive domain. We utilized bkmr package in R for model analyses.
In our sensitivity analyses, we applied generalized additive models (GAMs) with penalized splines to examine the potential non-linear associations between PFAS concentrations and cognitive domains. The PROC GAMPL procedure with Restricted Maximum Likelihood (REML) smoothing in SAS was used due to its more robust estimation for small samples.
Results
As shown in Table 1, The mean age of our participants was 43 years (SD = 9.0), 92.3% were men, 56.9% were non-Hispanic White, and 49.2% with a Bachelor’s degree or above education. The average annual income was >$60,000. The average number of standard weekly alcohol or alcoholic beverage consumption was about 8 (SD = 1.2) over the past year and 1–7 (SD = 0.6) at present. Participants self-reported smoking cigarettes or electronic cigarettes (29.3%). In the past 30 days, 13.0% of participants reported not consuming any seafood, 32% indicated eating seafood 1 to 3 times, and 20% reported consuming it 4 or more times. For occupational characteristics, the average years of service was 11 years (SD = 6.8), 92.7% of participants reported having used SCBA last year, and 77.9% reported having one shower after a fire event. Additionally, 49.2% of participants reported that they occasionally used fluorine-free or Class A foam for a fire event last year, 33.8% indicated using most or frequently, and 7.7% reported using always. The average concentration of 24-PFAS mixture was 1.01 pg/μL (SD = 0.42). The composite z-score for global cognition had the mean of –0.002 (SD = 0.7) and ranged from –1.6 to 1.4, while the composite z-score for processing speed had the mean of 0.12 (SD = 0.78) and ranged from –1.86 to 2.45. Correlations between PFAS exposure and each covariate were demonstrated in Table S3 in Supplementary Material. Higher monthly seafood consumption was significantly associated with elevated PFOS concentration (r = 0.38, p < 0.01).
Table 2 shows that among the 24 PFAS analytes, PFHpA, PFOA, PFOS, and PFHxS were detected in 100% of study participants, while PFNA and PFDA were detected in 93.75% and 77.78%, respectively. The arithmetic means of blood concentrations were 0.40 pg/μL (SD = 0.31) for PFHpA; 0.56 pg/μL (SD = 0.27) for PFOA; 2.4 pg/μL (SD = 1.24) for PFOS; 0.90 pg/μL (SD = 0.57) for PFHxS; 0.16 pg/μL (SD = 1.00) for PFNA; and 0.07 pg/μL (SD = 0.05) for PFDA. Distributional comparisons of these 6 PFAS analytes between this study and the NHANES were demonstrated in Figure S1 (Supplementary Materials). Details of the distributions of 24 PFAS in this study and their comparison with NHANES are presented in Table S1. Table S2 showed that 5(7.7%) firefighters who reported always using firefighting foam in the past year had higher levels of all 6 PFAS analytes than those who reported not using firefighting foams in the past year. In particular, PFHxS and PFOS levels were 2 times higher in these firefighters compared to firefighters who did not use firefighting foam in the past year before our study.
Results from linear regression models assessing the relationship between PFAS concentrations and cognitive performance in both unadjusted and adjusted models are presented in Table 3. For PFOS, the absolute value of the correlation coefficient increased progressively from the unadjusted Model 1 to the fully adjusted Model 3 (β =–0.13 to –0.58 for processing speed; β = –0.13 to –0.36 for global cognition), indicating a stronger and more statistically significant association between exposure to these PFAS analytes and cognitive performance after adjustment for covariates. After adjusting for the potential covariates, PFOS was negatively associated with processing speed (ß = –0.58, p < 0.001). For a 10-fold increase in levels of PFOS (e.g., 0.1 pg/μL to 1 pg/μL), there was 0.58 SD (8.7 points) decrease in processing speed, with the test having a SD of 15 points. PFOS was not associated with global cognition. PFOA, PFNA, PFDA, PFHpA, or PFHxS was not individually associated with either cognitive domain. Additionally, 24-PFAS mixture was not associated with global cognition or processing speed.
Results from the quantile g-computation and BKMR investigating the associations of 6-PFAS mixture and each cognitive domain were presented in Table 4. The Markov Chain Monte Carlo (MCMC) diagnostics indicated satisfactory acceptance rates for the kernel parameters in the model examining associations between the 6-PFAS mixture and cognitive domains (r = 0.34 for processing speed; 0.37 for global cognition). The chains exhibited good mixing (λ = 0.34) for processing speed, indicating efficient sampling and supporting reliable posterior inferences. In contrast, mixing was suboptimal for global cognition (λ = 0.21), suggesting less efficient exploration of the posterior and weaker support for reliable inference. Moreover, the overall mixture exhibited no significant association with processing speed as the credible interval for the primary comparison (75th vs. 25th) included 0. Posterior mean estimates were small and displayed opposing directions in opposite directions (slightly positive at lower exposure levels and slightly negative at higher levels), while the associated uncertainty (posterior standard deviations, SDs) exceeded the magnitude of the estimates themselves. However, this null overall mixture effect appeared to be driven primarily by heterogeneity across individual PFAS analytes: PFOS exhibited a strong association with processing speed (posterior inclusion probability [PIP] = 0.80), whereas the PIPs for the other PFAS analytes were substantially weaker (PIPs ≈ 0.3–0.5), suggesting that the estimated joint effect of the mixture was dominated by PFOS. These findings were further corroborated by visualizations of the exposure-response function h(z), which depict the estimated relationship between the overall PFAS mixture and cognitive outcomes (see Figs. 1 and 2).
“est” column is the posterior mean estimate to quantify how the exposure to PFAS mixture influences processing speed.
“est” column is the posterior mean estimate to quantify how the exposure to PFAS mixture influences global cognition.
In sensitivity analyses, we evaluated potential non-linearity using smoothing plots shown in Fig. S2 (see the Supplementary Materials). The smoothing component panel visualized the spline transformations for PFOS, PFOA, PFNA, PFDA, PFHxS, PFHpA, and 24-PFAS mixture in addition to 95% Bayesian curvewise confidence bands. There were clear linear negative associations of PFOS and 24-PFAS mixture with z-scored global cognition and processing speed. The results were consistent with those of linear regression models.
Discussion
This pilot study is one of the first epidemiological studies to assess the association between PFAS exposure and cognitive performance in firefighters. Of the 24 PFAS we measured, detection frequencies ranged from 0 to 100% and all participants had at least 6 PFAS analytes (PFHxS, PFHpA, PFOA, PFOS, PFNA, and PFDA) detected in their whole blood samples. Our findings suggest that levels of PFOS had a moderate-to-large effect, potentially impacting occupational performance in tasks requiring rapid processing abilities, which are critical for firefighting. The association is statistically significant, but the low overall PFAS levels (<1 pg/μL) suggest high sensitivity to small exposure changes. For individual firefighters, this decline may affect job efficacy but is unlikely to indicate clinical impairment unless cumulative. For the firefighter population, it supports exposure reduction and cognitive monitoring. Additionally, the magnitude of the correlation coefficients increased progressively from the unadjusted Model 1 to the fully adjusted Model 3 (which included occupational characteristics). This strengthening of associations after covariate adjustment highlights the potential for confounding bias in studies of PFAS exposure and cognitive outcomes when key occupational factors are not considered.
These findings align with evidence suggested by prior epidemiological studies, [15,16,17]which report that neurotoxicity of PFAS varies by carbon chain lengths and the functional group: larger adverse effects were exhibited in legacy or long-chain PFAS (e.g., PFOS and PFOA, compared to alternative shorter-chain PFAS) and the sulfonate functional group [16, 43]. In cell-cultured rat models, Berntsen et al. (2017) [44] also confirmed that the cytotoxicity of neurons increases with increasing carbon chain lengths and the sulfonate functional group and leads to a greater toxicity than the carboxylate group if the chain length is the same. Additionally, PFOS exhibits a lower elimination rate from the body compared to other PFAS analytes due to its longer half-lives or biological persistence [17, 45]. In our study, the average concentration of PFOS was the highest among the 6 PFAS analytes (x̄=2.40 pg/μL), and its concentrations at each quartile (25th, 50th, and 75th percentiles) were also consistently the highest (1.53 pg/μL for the 25th, 2.13 pg/μL for the 50th, and 2.95 pg/μL for the 75th) as shown in Table 2. Even after the 2002 voluntary phase-out of PFOS production, legacy foams remain in use or contaminate sites, equipment, and turnout gear. PFOA was present in much lower amounts (often <10%) in these foams, either as an impurity or byproduct [2, 45]. Additional sources like PFAS-treated turnout gear may contribute more to PFOS migration through layers during use or laundering. These could be the potential mechanism to explain the adverse impact of PFOS on processing speed over PFOA in our study as well as the NHANES study for the general population [16].
Our findings add to the current literature in providing evidence that PFOS exerted the predominant influence within the PFAS mixture on processing speed among firefighters. These results are consistent with the findings of similar prior research studied PFAS concentrations on neurohealth in early and late life [17, 46]. In an exploratory study of older French cohort at a hospital setting, branched PFOS was found significantly higher among 8 older patients with both clinical and biological signs of neurological impairment than those without [17]. A literature review on the impact of PFAS exposure on neurodevelopment in early life suggests that the legacy PFAS concentrations, such as PFOS, PFHxS, and PFDA, are persistently associated with lower trajectories of cognitive function including grow motor and problem solving in children between 3 and 36 months [46]. The potential mechanism for these results is that legacy PFAS analytes can freely cross the BBB and have significant accumulation in brain, which disrupts calcium homeostasis and dopamine and glutamate signaling, and consequently affects cognitive functions and contributes to unfavorable cognitive performance [46].
We did not find significant associations of cognitive domains with either the 6-PFAS mixture or the 24-PFAS mixture in this firefighter population, after adjusting for the occupational characteristics. This finding is not unexpected, as it can be attributed to two primary factors. First, epidemiological evidence on childhood PFAS exposure and neurodevelopmental scores suggests that despite the adverse cognitive outcomes in relation to PFAS mixture, legacy PFAS were major contributors that drove the mixture associations, with little contribution from alternative PFAS [46, 47]. Second, the detection rates of alternative PFAS were low in this population resulting in their minimal impact on cognitive domains.
Widespread use for PFAS in consumer products, contamination of food and water sources, and their environmental persistence may contribute to the high levels of exposure in firefighters. Turnout gear may be a major source of PFAS exposure in firefighters in our study. Recent studies have shown that PFAS are present in all three layers of firefighter turnout gear: the outer shell, moisture barrier, and thermal liner [25, 48]. These studies highlight the health risks associated with the materials and finishes used in turnout gear even before it is exposed to its first fire. In our study, firefighters all wear turnout gear during their firefighting, overhaul, and working in smoke. Although clean procedures, including basic spot cleaning, full wash, and specialized for heavy chemicals, that generally have been performed to reduce the PFAS exposure before and after fire or rescue events, PFAS residues may persist on turnout gear and can penetrate the skin through dermal absorption, entering the bloodstream and potentially impacting cognitive function. In particular, insufficient processing speed could slow their responses, increase the risk of errors, and jeopardize both rescue missions and personal safety. Firefighting foams may be another important source of PFAS exposure. In our study, 61 firefighters reported that they have only used fluorine-free or Class A foam after their training in the past year before our study, while 4 firefighters reported that they used both Class A and AFFF. A study in Finland found that firefighters using AFFF in training activities had increased levels of PFHxS in their blood after the training activities [19], however we lacked access to valid, individual-specific data on the types or chemical compositions of firefighting foams to which participants were exposed. As a result, non-differential exposure misclassification likely attenuated the observed associations meaning our reported effect estimates were probably conservative.
Following adjustment using a 2:1 serum/whole-blood conversion factor, comparison of seven PFAS analytes (PFOA, PFNA, PFDA, PFUnDA, PFHxS, PFHpS, and PFOS) revealed that firefighters in the present study had elevated PFHxS and PFOS concentrations compared with the NHANES 2013–2014 cohort. PFDA and PFUnDA concentrations were nearly identical to those reported in NHANES, whereas PFOA, PFNA, and PFHpS levels were slightly lower. The NHANES data were collected 2013–2014 and our samples were collected 2023-2025. This may reflect the temporal trends associated with the phase-out of these compounds in consumer products and firefighting equipment [2]. Additionally, firefighter participants in our study were without CKD, while HNANES participants included those with CKD. CKD may result in higher serum concentration for specific PFAS analytes [16, 49].
Our study has several limitations. First, the absence of a significant association between PFAS exposure and global cognition may be attributable, at least in part, to the healthy worker effect. Because our cohort consisted of active-duty career firefighters, a population subject to rigorous pre-employment screening and ongoing medical surveillance, individuals with poorer baseline cognitive function or neurological health are less likely to enter or remain in this occupation, potentially rendering them less susceptible to or less likely to exhibit PFAS-global cognition deficits within the observed exposure range. Second, the overall small sample size (N = 65) may diminish statistical power to assess the relationship between the 24-PFAS mixture and cognitive performance. Given the complexity of mixtures, larger sample sizes are needed to produce reliable estimates. Third, we could not investigate differences in the PFAS-cognition relationship by specific racial/ethnic group and disparities among them. Gender heterogeneity in the PFAS-cognition relationship was also not able to be examined due to the predominance of male firefighters in our sample. Generalizability of our findings should be tested with a large and diverse sample. Fourth, cross-sectional study design cannot draw causal inference for the impact of PFAS exposure on cognitive performance. Epidemiological studies of longitudinal trajectory of PFAS exposure and cognitive function are urgently needed to replicate our findings. Moreover, control groups, such as office workers who were not first responders, need to be included to differentiate occupational risks in relation to PFAS exposure. Fifth, we adjusted the data for most identified and measured potential confounders based on the limited literature, but we cannot rule out biases due to unmeasured confounders. For example, it was difficult to assess the association between PFAS exposure and fire events. Although our firefighters had responded to a fire event in the 24 h prior to the sample collection. We also did not have information on the intensity of the fire, the amount of firefighting foam used at a fire event, the formulations of firefighting foam, or the participants assigned role at the fire event (e.g., whether the firefighter was involved in direct fire suppression activities or providing back up and support). Additionally, all occupational characteristics for firefighters were based on self-reported data, which is often influenced by recall bias.
Despite these limitations, our study is the first epidemiological study to explore the association between PFAS exposure and cognitive performance in firefighters. This study fills the data gaps and contributes to the existing environmental health literature in enriching and replenishing the examination of environment–cognition relationship in firefighters and providing the basis for future research to elucidate mechanisms of PFAS affecting domain-specific cognitive function and subsequently characterize potential modifiable risks for cognitive impairment and AD/ADRD. Moreover, findings of the study highlight opportunities to develop effective intervention and prevention strategies to reduce workplace exposure for firefighters. These findings will also educate health care professionals about the relationship between workplace exposure and cognitive function, and promote neurocognitive screening tests and assessment as a part of routine exams for firefighters to reduce their risk for developing dementia. Finally, our findings inform the risk assessment process used by government officials to derive health-based guidelines and standards that regulate PFAS in environment and protect the public from potential unsafe level of exposure.
In conclusion, our study provides the first data on PFAS exposure and cognitive performance for firefighters. We found that PFOS was the primary contributor among the PFAS mixture (consisting of PFOS, PFOA, PFHxS, PFHpA, PFNA, and PFDA) linked to reduced processing speed scores in firefighters. While confirmative studies are required, our findings recommend interventions to reduce PFAS exposure and highlight opportunities to assess the effectiveness of workplace exposure reduction efforts. Our findings also suggest that screening tests for cognitive decline should be recommended by health care providers to firefighters, who are first responders.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Foguth RM, Flynn RW, de Perre C, Iacchetta M, Lee LS, Sepulveda MS, et al. Developmental exposure to perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) selectively decreases brain dopamine levels in Northern leopard frogs. Toxicol Appl Pharmacol. 2019;377:114623.
Trowbridge J, Gerona RR, Lin T, Rudel RA, Bessonneau V, Buren H, et al. Exposure to Perfluoroalkyl Substances in a Cohort of Women Firefighters and Office Workers in San Francisco. Environ Sci Technol. 2020;54:3363–74.
Zhou YLQ, Wang P, et al. Associations of prenatal PFAS exposure and early childhood neurodevelopment: evidence from the Shanghai Maternal-Child Pairs Cohort. Environ Int. 2023;173:107850.
Shi L, Deng X, Liu X, Yan S, Wang Y, Li Y, et al. The effect of chronic exposure to a low concentration of perfluorooctanoic acid on cognitive function and intestinal health of obese mice induced by a high-fat diet. Food Chem Toxicol. 2022;168:113395.
Weng X, Liang H, Tan Y, Chen J, Fei Q, Liu S, et al. Mixed effects of perfluoroalkyl and polyfluoroalkyl substances exposure on cognitive function among people over 60 years old from NHANES. Environ Sci Pollut Res Int. 2022;29:32093–104.
Bassler JDA, Elliott M, et al. Environmental perfluoroalkyl acid exposures are associated with liver disease characterized by apoptosis and altered serum adipocytokines. Environ Pollut. 2019;247:1055–63.
Vuong AM, Yolton K, Webster GM, Sjodin A, Calafat AM, Braun JM, et al. Prenatal polybrominated diphenyl ether and perfluoroalkyl substance exposures and executive function in school-age children. Environ Res. 2016;147:556–64.
Skogheim TS, Villanger GD, Weyde KVF, Engel SM, Surén P, Øie MG, et al. Prenatal exposure to perfluoroalkyl substances and associations with symptoms of attention-deficit/hyperactivity disorder and cognitive functions in preschool children. Int J Hyg Environ Health. 2020;223:80–92.
Forns J, Verner MA, Iszatt N, Nowack N, Bach CC, Vrijheid M, et al. Early Life Exposure to Perfluoroalkyl Substances (PFAS) and ADHD: A Meta-Analysis of Nine European Population-Based Studies. Environ Health Perspect. 2020;128:57002.
Grønnestad RJS, Müller MHB, Müller MHB, Schlenk D, Tanabe P, Krøkje Å, et al. Effects of an environmentally relevant PFAS mixture on dopamine and steroid hormone levels in exposed mice. Toxicol Appl Pharmacol. 2021;428:115670.
Sammi SR, Foguth RM, Nieves CS, De Perre C, Wipf P, McMurray CT, et al. Perfluorooctane Sulfonate (PFOS) Produces Dopaminergic Neuropathology in Caenorhabditis elegans. Toxicol Sci. 2019;172:417–34.
Pierozan P, Karlsson O. Differential susceptibility of rat primary neurons and neural stem cells to PFOS and PFOA toxicity. Toxicol Lett. 2021;349:61–8.
Cao Y, Ng C. Absorption, distribution, and toxicity of per- and polyfluoroalkyl substances (PFAS) in the brain: a review. Environ Sci: Process Impacts. 2021;23:1623–40.
Shrestha S, Bloom MS, Yucel R, Seegal RF, Rej R, McCaffrey RJ, et al. Perfluoroalkyl substances, thyroid hormones, and neuropsychological status in older adults. Int J Hyg Environ Health. 2017;220:679–85.
Gallo V, Leonardi G, Brayne C, Armstrong B, Fletcher T. Serum perfluoroalkyl acids concentrations and memory impairment in a large cross-sectional study. BMJ Open. 2013;3:e002414.
Park SK, Ding N, Han D. Perfluoroalkyl substances and cognitive function in older adults: Should we consider non-monotonic dose-responses and chronic kidney disease?. Environ Res. 2021;192:110346.
Delcourt N, Pouget AM, Grivaud A, Nogueira L, Larvor F, Marchand P, et al. First observations of a potential association between accumulation of Per- and polyfluoroalkyl substances in the central nervous system and markers of Alzheimer’s disease. J Gerontol A Biol Sci Med Sci. 2024;79.
Di Nisio A, Pannella M, Vogiatzis S, Sut S, Dall’Acqua S, Rocca MS, et al. Impairment of human dopaminergic neurons at different developmental stages by perfluoro-octanoic acid (PFOA) and differential human brain areas accumulation of perfluoroalkyl chemicals. Environ Int. 2022;158:106982.
Laitinen JAKJ, Koikkalainen J, Kiviranta H. Firefighters’ exposure to perfluoroalkyl acids and 2-butoxyethanol present in firefighting foams. Toxicol Lett. 2014;231:227–32.
Jin C, Sun Y, Islam A, Qian Y, Ducatman A. Perfluoroalkyl Acids Including Perfluorooctane Sulfonate and Perfluorohexane Sulfonate in Firefighters. J Occup Environ Med. 2011;53:324–8.
Rotander A, Toms LM, Aylward L, Kay M, Mueller JF. Elevated levels of PFOS and PFHxS in firefighters exposed to aqueous film forming foam (AFFF). Environ Int. 2015;82:28–34.
Gainey SJ, Horn GP, Towers AE, Oelschlager ML, Tir VL, Drnevich J, et al. Exposure to a firefighting overhaul environment without respiratory protection increases immune dysregulation and lung disease risk. PloS One. 2018;13:e0201830.
Navarro KM, Cisneros R, Noth EM, Balmes JR, Hammond SK. Occupational exposure to polycyclic aromatic hydrocarbon of wildland firefighters at prescribed and wildland fires. Environ Sci Technol. 2017;51:6461–9.
Nilsson S, Smurthwaite K, Aylward LL, Kay M, Toms LM, King L, et al. Serum concentration trends and apparent half-lives of per- and polyfluoroalkyl substances (PFAS) in Australian firefighters. Int J Hyg Environ Health. 2022;246:114040.
Peaslee GFW, Wilkinson JT, McGuinness SR, Tighe M, Caterisano N, Lee S, et al. Another pathway for firefighter exposure to per- and polyfluoroalkyl substances: firefighter textiles. Environ Sci Technol Lett. 2020;7:594–9.
Seow J. Fire fighting foams with perfluorochemicals-Environmental Review. Department of Environment and Conservation Western Australia. 2013. https://cswab.org/wp-content/uploads/2018/03/Fire-Fighting-Foams-with-PFAS-Env-Review-June-2013-Australia-.pdf
Shaw SD, Berger ML, Harris JH, Yun SH, Wu Q, Liao C, et al. Persistent organic pollutants including polychlorinated and polybrominated dibenzo-p-dioxins and dibenzofurans in firefighters from Northern California. Chemosphere. 2013;91:1386–94.
Dobraca D, Israel L, McNeel S, Voss R, Wang M, Gajek R. Biomonitoring in california firefighters: metals and perfluorinated chemicals. Occup Environ Med. 2015;57:88–97.
Mesfin Tefera Y, Gaskin, S, Mitchell, K, Springer, D, Mills, S. Temporal decline in serum PFAS concentrations among Metropolitan Firefighters: Longitudinal Study on post-exposure changes following PFAS foam cessation. Environ Int. 2023;179.
Furlong MA, Liu T, Jung A, Beitel S, Hughes J, Krause R, et al. Per- and polyfluoroalkyl substances (PFAS) and microRNA: An epigenome-wide association study in firefighters. Environ Res. 2025;279:121766.
Pedersen JE, Petersen KKU, Andersen MHG, Saber AT, Vogel U, Ebbehøj N, et al. Non-malignant kidney diseases in Danish firefighters. Occup Environ Med. 2025;82:423–8.
Gould Rothberg BE, Caban-Martinez AJ, Barr DB, Jara MA, Rodriguez V, Louzado Feliciano P, et al. Polybrominated diphenyl ethers, occupational exposures, and thyroid function among US and Canadian firefighters: a cross-sectional study. J Occup Environ Med. 2025;67:935–42.
Hemmatjo R, Hajaghazadeh M, Allahyari T, Zare S, Kazemi R. The effects of live-fire drills on visual and auditory cognitive performance among firefighters. Ann Glob Health. 2020;86:144.
Orysiak J, Mlynarczyk M, Piec R, Jakubiak A. Lifestyle and environmental factors may induce airway and systemic inflammation in firefighters. Environ Sci Pollut Res Int. 2022;29:73741–68.
Cole KR, Yen CL, Dudley-Javoroski S, Shields RK. NIH toolbox cognition battery in young and older adults: reliability and relationship to adiposity and physical activity. J Geriatr Phys Ther. 2021;44:51–9.
Chaytor NS, Trapani VR, Braffett BH, Fonseca LM, Lorenzi GM, Gubitosi-Klug RA, et al. Utility of the NIH toolbox cognition battery in middle to older aged adults with longstanding type 1 diabetes: the DCCT/EDIC study. Clin Neuropsychol. 2024;38:1007–27.
Weintraub S, Dikmen SS, Heaton RK, Tulsky DS, Zelazo PD, Bauer PJ, et al. Cognition assessment using the NIH Toolbox. Neurology. 2013;80:S54–S64.
Tulsky DS, Carlozzi NE, Holdnack J, Heaton RK, Wong A, Goldsmith A, et al. Using the NIH Toolbox Cognition Battery (NIHTB-CB) in individuals with traumatic brain injury. Rehabil Psychol. 2017;62:413–24.
Perroni F, Mol E, Walker A, Alaimo C, Guidetti L, Cignitti L, et al. Reaction time to visual stimulus in firefighters and healthy trained subjects: a preliminary comparative study. Open Sports Sci J. 2018;11:69–77.
Carignan CC, Bauer RA, Patterson A, Phomsopha T, Redman E, Stapleton HM, et al. Self-collection blood test for PFASs: comparing volumetric microsamplers with a traditional serum approach. Environ Sci Technol. 2023;57:7950–7.
Poothong S, Thomsen C, Padilla-Sanchez JA, Papadopoulou E, Haug LS. Distribution of novel and well-known poly- and perfluoroalkyl substances (PFASs) in human serum, plasma, and whole blood. Environ Sci Technol. 2017;51:13388–96.
Hanssen L, Dudarev AA, Huber S, Odland JØ, Nieboer E, Sandanger TM. Partition of perfluoroalkyl substances (PFASs) in whole blood and plasma, assessed in maternal and umbilical cord samples from inhabitants of arctic Russia and Uzbekistan. Sci total Environ. 2013;447:430–7.
He Q, Yang Q, Wu L, He Y, Zeng N, Wang Z. Neurotoxic effects of per- and polyfluoroalkyl substances (PFAS) mixture exposure in mice: Accumulations in brain and associated changes of behaviors, metabolome, and transcriptome. J Hazard Mater. 2025;489:137699.
Berntsen HF, Berg V, Thomsen C, Ropstad E, Zimmer KE. The design of an environmentally relevant mixture of persistent organic pollutants for use in in vivo and in vitro studies. J Toxic Environ Health. 2017;A80:1002–16.
Mesfin Tefera Y, Gaskin S, Mitchell K, Springer D, Mills S. Temporal decline in serum PFAS concentrations among metropolitan firefighters: Longitudinal study on post-exposure changes following PFAS foam cessation. Environ Int. 2023;179:108167.
Ames JL, Sharma V, Lyall K. Effects of early‑life PFAS exposure on child neurodevelopment: a review of the evidence and research gaps. Curr Environ Health Rep. 2025;12:9.
Oh J, Shin HM, Kannan K, Busgang SA, Schmidt RJ, Schweitzer JB, et al. Childhood exposure to per- and polyfluoroalkyl substances and neurodevelopment in the CHARGE case-control study. Environ Res. 2022;215:114322.
Muensterman DJTI, Peaslee GF, Minc LD, Cahuas L, Rodowa AE, Horiuchi Y, et al. Disposition of fluorine on new firefighter turnout gear. Environ Sci Technol. 2022;56:974–83.
Dhingra RWA, Darrow LA, Klein M, Steenland K. A study of reverse causation: examining the associations of perfluorooctanoic acid serum levels with two outcomes. Environ Health Perspect. 2017;125:416–21.
Acknowledgements
We express our sincere gratitude to all firefighters who participated in our study. We are grateful to staff and leadership of the fire departments in Kyle, San Marcos, New Braunfels, and San Antonio in Texas for their dedicated efforts in facilitating participant recruitment. Additionally, we thank the faculty and staff at Texas State University and Yale University, who supported sample storage, meticulously managed sample delivery, maintained the integrity of samples critical to our research. Finally, we express our gratitude to Dr. Timothy M. Hughes (Gerontology and Geriatric Medicine, School of Medicine, Wake Forest University) for his guidance on the NIH Toolbox Cognition Battery and to Dr. Carmen Westerberg (Department of Psychology, Texas State University) for her advice on selecting the cognitive tests for this study.
Funding
This project was supported by the Research Accelerated Grant of Texas State University 2023–2025.
Author information
Authors and Affiliations
Contributions
XP planned the study, conducted participant recruitment, collected data, conducted literature review, performed all statistical analyses, led data interpretation, and wrote the manuscript; KJGP provided instructions and essential tools for blood sample collection, advised blood sample analysis, and provided critical revisions to the manuscript for important intellectual content; SL provided instruction and essential tools for blood sample collection, conducted blood sample analyses, and interpreted the results; FA collected blood samples and conducted literature review; BA conducted cognitive data collection. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the Institutional Review Boards (IRB) at Texas State University under the protocol number IRB#8639. Informed consent was obtained from all participants prior to data collection. All data were de-identifiable. All study procedures adhered to the ethical principles outlined in the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pan, X., Pollitt, K.J.G., Liu, S. et al. Association of per- and polyfluoroalkyl substances (PFAS) exposure with cognitive function in firefighters. J Expo Sci Environ Epidemiol (2026). https://doi.org/10.1038/s41370-026-00861-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41370-026-00861-y