Abstract
In this position statement the British and Irish Hypertension Society (BIHS) present a review of the current evidence for blood pressure (BP) treatment thresholds and targets. The BIHS recommend initiating pharmacological antihypertensive therapy, irrespective of cardiovascular disease risk, following a confirmed diagnosis of hypertension (sustained out-of-office BP ≥ 135/85 mmHg despite diet and lifestyle advice). The BIHS recommend an on-treatment BP target < 130/80 mmHg or as low as reasonably achievable without causing unacceptable side-effects, within 6-months of initiating treatment, for all adults. Possible subgroups to whom this may not apply are those who are frail and/or have limited life expectancy where higher targets may be appropriate based on clinical judgement and the individuals’ tolerance to treatment. The BIHS believe that this simple 2-step approach will facilitate practitioners deliver evidence-based best practice, discourage therapeutic inertia around BP lowering and improve heath outcomes for all adults living with high BP.
Similar content being viewed by others
Statement
High blood pressure (BP) is the leading modifiable risk factor for premature morbidity and mortality from cardiovascular disease (CVD) worldwide, and significantly contributes to chronic kidney disease, heart failure and dementia [1, 2].
Epidemiological studies demonstrate a continuous positive association between BP and major adverse cardiovascular events (MACE) [3,4,5]. Observational data are supported by randomized clinical trials, demonstrating the benefits of antihypertensive drug treatment in reducing MACE [6, 7]. The Blood Pressure Lowering Treatment Trialists’ Collaboration reported that the reduction in MACE was proportional to the magnitude of the fall in SBP down to at least 120 mmHg in those aged < 75 years, with and without prior cardiovascular disease [6, 7]. Overall, MACE was reduced by 10% per 5 mmHg reduction in Systolic BP (SBP), with a greater effect in younger (age <55 years: HR 0·82 [95% CI: 0·76–0·88]) compared with older individuals (age 65–74 years: HR 0·91 [95%CI: 0·87–0·96]) [6, 7].
Over the last 15 years, several large outcome trials have been conducted to specifically compare aiming for an intensive BP treatment target (e.g. SBP < 120 mmHg) with a less intensive target (e.g. SBP < 140 mmHg) among men and women, with and without pre-existing CVD and diabetes [8,9,10,11,12,13,14]. These trials consistently demonstrate that aiming for an SBP treatment target <120 mmHg resulted in a greater reduction in MACE compared with an SBP target < 140 mmHg, with no increase in all-cause mortality [Table 1] [8,9,10,11,12,13,14]. There was no overall significant difference in reported serious adverse events between treatment groups [Table 1] [8,9,10,11,12,13,14].
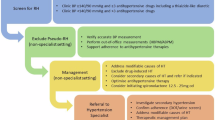
Results were consistent across these trials whether BP was measured by attended or unattended office readings or home BP measurements (HBPM) [8,9,10,11,12,13,14]. This aligns with a UK study by McManus and colleagues which demonstrated that at lower levels of BP the difference between office and HBPM was small [15]. Based on this evidence, the British and Irish Hypertension Society (BIHS) recommend that BP targets remain the same whether BP is measured in the office, by 7-day average HBPM [Fig. 1] or day-time average ambulatory BP measurement (ABPM).
BIHS Guide to Home Blood Pressure Measurement.
The current National Institute of Clinical Excellence (NICE) Adult Hypertension Guideline NG136, recommended antihypertensive treatment to be initiated based on a combination of BP-level and 10-year CVD risk [16]. This approach targets treatment to those at highest short-term absolute risk. In light of the continuing excess of premature morbidity and mortality from BP-related diseases [1, 2], the BIHS questions the ethics of waiting until sufficient irreversible damage has been done to meet an arbitrary risk threshold, before cheap, safe and effective antihypertensive treatment (both lifestyle and pharmacological) is initiated.
Current NICE BP treatment targets for people aged under 80 years are <140/90 mmHg, with <130/80 mmHg only recommended in specific clinical situations (e.g. some patients with diabetes and chronic kidney disease) [16]. The BIHS believes there is now sufficient new evidence to recommend one consistent BP target of <130/80 mmHg or as low as reasonably achievable (ALARA) without causing unacceptable side-effects within 6-months of initiating treatment, among all adults with a confirmed diagnosis of hypertension. Possible subgroups to whom this may not apply are those who are frail and/or have limited life expectancy where higher targets may be appropriate based on clinical judgement and the individuals’ tolerance to treatment.
Thus, based on current evidence, the BIHS calls for NICE to update its guidance in NG136 “Hypertension in adults: diagnosis and management” [16] in line with current recommendations from Europe and the United States of America [17,18,19,20] as follows:
BP threshold for treatment
Following a confirmed diagnosis of hypertension (sustained BP ≥ 135/85 mmHg based on 7-day average HBPM or day-time average ABPM despite diet and lifestyle advice) starting BP-lowering treatment with pharmacological therapy on top of diet and lifestyle interventions is recommended irrespective of CVD risk. We recommend the BIHS algorithm in “Adult Hypertension Referral Pathway and Therapeutic Management: British and Irish Hypertension Society Position Statement” for a titration protocol and exemplar therapeutic options within the NICE treatment pathway [21].
On-treatment BP target
On treatment BP target < 130/80 mmHg (measured by 7-day average HBPM or day-time average ABPM or office BP*) or ALARA without causing unacceptable side-effects, and within 6-months of initiating treatment, for all adults. Possible subgroups to whom this may not apply are those who are frail and/or have limited life expectancy where higher targets may be appropriate based on clinical judgement and the individuals’ tolerance to treatment.
*Among people with diagnosed white-coat hypertension out-of-office measurements should always be used to evaluate treatment.
The BIHS believe that this simple 2-step approach will facilitate practitioners deliver evidence-based best practice, discourage therapeutic inertia around BP lowering and improve health outcomes for all adults living with high BP.
Data availability
All data generated and analysed for the development of this statement are included in this published article.
Change history
28 July 2025
References have been corrected on page 1.
References
British Heart Foundation (2025) UK Factsheet January 2025. Available from: https://www.bhf.org.uk/-/media/files/for-professionals/research/heart-statistics/bhf-cvd-statistics-uk-factsheet.pdf [accessed 19/5/2025]
Lewington S, Clarke R, Qizilbash N, Peto R, Collins R, Prospective Studies Collaboration. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360:1903–13. Erratum in: Lancet. 2003 Mar 22;361(9362):1060
Rapsomaniki E, Timmis A, George J, Pujades-Rodriguez M, Shah AD, Denaxas S, et al. Blood pressure and incidence of twelve cardiovascular diseases: lifetime risks, healthy life-years lost, and age-specific associations in 1·25 million people. Lancet. 2014;383:1899–911.
Arvanitis M, Qi G, Bhatt DL, Post WS, Chatterjee N, Battle A, et al. Linear and nonlinear Mendelian randomization analyses of the association between diastolic blood pressure and cardiovascular events: the J-curve revisited. Circulation. 2021;143:895–906.
Whelton SP, McEvoy JW, Shaw L, Psaty BM, Lima JAC, Budoff M, et al. Association of normal systolic blood pressure level with cardiovascular disease in the absence of risk factors. JAMA Cardiol. 2020;5:1011–8.
The Blood Pressure Lowering Treatment Trialists’ Collaboration. Pharmacological blood pressure lowering for primary and secondary prevention of cardiovascular disease across different levels of blood pressure: an individual participant-level data meta-analysis. The Lancet. 2021;397:1625–36.
The Blood Pressure Lowering Treatment Trialists’ Collaboration. Age-stratified and blood-pressure-stratified effects of blood-pressure-lowering pharmacotherapy for the prevention of cardiovascular disease and death: an individual participant-level data meta-analysis. Lancet. 2021;398:1053–64.
The ACCORD Study Group. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2010;362:1575–85.
SPS3 Study Group. Blood-pressure targets in patients with recent lacunar stroke: the SPS3 randomised trial. Lancet. 2013;382:507–15.
SPRINT Research Group. Final report of a trial of intensive versus standard blood-pressure control. N Engl J Med. 2021;384:1921–30.
RESPECT Study Group. Effect of standard vs intensive blood pressure control on the risk of recurrent stroke: a randomized clinical trial and meta-analysis. JAMA Neurol. 2019;76:1309–18.
STEP Study Group. Trial of intensive blood-pressure control in older patients with hypertension. N Engl J Med. 2021;385:1268–79.
ESPRIT Collaborative Group. Lowering systolic blood pressure to less than 120 mm Hg versus less than 140 mm Hg in patients with high cardiovascular risk with and without diabetes or previous stroke: an open-label, blinded-outcome, randomised trial. Lancet. 2024;404:245–55.
BPROAD Research Group. Intensive blood-pressure control in patients with type 2 diabetes. N Engl J Med. 2025;392:1155–67.
McManus RJ, Little P, Stuart B, Morton K, Raftery J, Kelly J, et al. Home and Online Management and Evaluation of Blood Pressure (HOME BP) using a digital intervention in poorly controlled hypertension: randomised controlled trial. BMJ. 2021;372:m4858.
National Institute for Health and Care Excellence. Hypertension in adults: diagnosis and management. NICE guideline [NG136]. Published 28 August 2019, Last updated: 21 November 2023. https://www.nice.org.uk/guidance/ng136 [accessed 20/5/2023].
McEvoy JW, McCarthy CP, Bruno RM, Brouwers S, Canavan MD, Ceconi C, et al. 2024 ESC Guidelines for the management of elevated blood pressure and hypertension. Eur Heart J. 2024;45:3912–4018.
Mancia G, Kreutz R, Brunstrom M, Burnier M, Grassi G, Januszewicz A, et al. 2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA). J Hypertens. 2023;41:1874–2071.
Unger T, Borghi C, Charchar F, Khan NA, Poulter NR, Prabhakaran D, et al. 2020 International society of hypertension global hypertension practice guidelines. Hypertension. 2020;75:1334–57.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr., Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/ PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the american college of cardiology/ american heart association task force on clinical practice guidelines. Hypertension. 2018;71:e13–115.
Lewis P, George J, Kapil V, Poulter NR, Partridge S, Goodman J, et al. Adult hypertension referral pathway and therapeutic management: British and Irish Hypertension Society position statement. J Hum Hypertens. 2024;38:3–7.
Acknowledgements
We would like to thank the following members of the BIHS Guideline Standing Committee for their expert review of this manuscript: Professor Adrian Brady, Dr Andrew Jordan, Dr Spoorthy Kulkarni, Dr Philip Lewis, Professor Terry McCormack, Ms Michaela Nuttall, Professor Peter Sever and Dr Wayne Sunman. We also thank the BIHS Executive Committee for their expert review: Professor Philip Chowienczyk, Dr Pankaj Gupta, Dr Ryan McNally, Mr Sam Olden, Dr Manish Saxena, Dr James Sheppard, Dr Helen Warren.
Funding
No financial assistance was received to develop this statement.
Author information
Authors and Affiliations
Contributions
IBW and NP proposed the statement. NT conducted the literature review. LF, NT, NP, JG, VK, AG, PS, TH, ES, SP, IBW provided expert input on the formulation of the position statements. SP provided medical writing and editorial support. All authors reviewed and approved the final draft.
Corresponding author
Ethics declarations
Ethical approval
Ethical approval was not required for this work as it did not involve any human or animal studies.
Competing interests
Professor Adrian J.B. Brady has received honoraria from Daiichi-Sankyo, Amgen, Sanofi-Aventis, Bayer, MSD, and Novartis. Professor Phil Chowienczyk has an interest in Centron Diagnostics, a company that has produced technology for blood pressure measurement. Professor Jacob George has received grants and travel funding from Astra Zeneca, Novartis and Daiichi Sankyo, is on Advisory Boards for AstraZeneca, Menarini and Novartis, consulting fees from Roche and PwC and is a Principal Investigator for studies funded by AstraZeneca, Alnylam, Novartis, Esperion. Dr Pankaj Gupta has received research grants, lecture honoraria and funding for conference attendance from Sanofi-Aventis and Amgen, and consulting fees from Ionis Pharmaceuticals. Dr Spoorthy Kulkarni is a PhD student at the University of Cambridge funded by AstraZeneca and has attended scientific advisory boards for Viatris. Professor Terry McCormack has received lecture honoraria and/or consultation fees from AstraZeneca, Daichi-Sankyo, and Medtronic. He is a co-investigator in the Alnylam study Kardia 3. Professor Neil R Poulter has received lecture honoraria and/or consultation fees from several pharmaceutical companies that manufacture blood pressure lowering agents including AstraZeneca, Eva Pharma, Lri Therapharma, Napi, Pfizer, Servier and Sanofi-Aventis. Dr Manish Saxena has received grant support from Recor Medical, Ablative Solutions, Applied Therapeutics, Vascular Dynamics, and MSD; and has received consulting fees from Recor Medical, Esperion Inc., Daiichi Sankyo Inc., Novartis, and Vifor Pharma. Professor Peter Sever has received renumeration for scientific advisory boards and lectures from Viatris. Professor Ian B Wilkinson has received research grants from AstraZeneca, GSK and scientific advisory board consultation fees for Viatris, Astra Zeneca and Roche. Dr Luca Faconti, Dr Ajay Gupta, Professor Anthony Heagerty, Dr Andrew Jordan, Dr Vikas Kapil, Dr Philip Lewis, Dr Ryan McNally, Ms Michaela Nuttall, Mr Sam Olden, Dr Sarah Partridge, Dr Eduard Shantsila, Professor James Sheppard, Dr Wayne Sunman, Dr Pauline Swift, Dr Nayanatara Tantirige, and Dr Helen Warren have no competing interests to declare for this manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Faconti, L., Tantirige, N., Poulter, N.R. et al. Call to action: British and Irish hypertension society position statement on blood pressure treatment thresholds and targets. J Hum Hypertens 39, 537–540 (2025). https://doi.org/10.1038/s41371-025-01055-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41371-025-01055-z