Abstract
Objective
To analyze the sclerotherapy of giant (≥8 mm) lymphatic malformations in neonates and discuss effective treatment.
Methods
This was a single-center retrospective study. Twelve patients received bleomycin (BLM) (six patients) or BLM combined with polidocanol (POL) (eight patients). The safety and effects of BLM and BLM + POL were analyzed using a t-test for categorical data and likelihood ratios or Fisher’s exact test for continuous data. Factors resulting in an increased number of treatment sessions were studied.
Results
Treatment began 2–5 days after birth. The patients in the BLM group underwent more treatment sessions. Gestational age, lesion size and type, total response, and complications showed no significant differences between groups. Larger and mixed-ype lesions required more treatment sessions.
Conclusions
Both BLM group and BLM + POL group appear relatively safe and effective in neonates with giant lymphatic malformations. The combination of the two agents reduced the number of procedures and general anesthesia required.
Similar content being viewed by others
Introduction
Lymphatic malformations (LMs) are vascular malformations that result from the abnormal development of the lymphatic system during the embryonic period [1]. LMs are divided into three morphological types: macrocystic, microcystic, and mixed [2]. LMs can occur in any region; however, the neck is the most common location. Patients with giant LMs often face several risks during the newborn stage. The most severe complication is airway compression, including that of the larynx and trachea, especially in patients with spontaneous hemorrhage [3, 4]. In addition, infection and subsequent abscesses may occur in LM cysts [5].
Although spontaneous regression has been observed in some LMs, giant LMs require treatment even in neonates because of their high risk of compression. Therapies for LMs include sclerotherapy, surgery, and systemic drugs [6,7,8]. Considering its minimal injury, effectiveness, and low complication rates, sclerotherapy has been regarded as the optimal choice in recent years, particularly for macrocystic lesions. However, neonatal treatment remains challenging. Neonates, especially preterm babies and early neonates, face a higher risk of anesthesia and other possible complications during the perinatal period. Giant and multi-cystic lesions often require higher drug doses; however, the low body weight and immature liver and renal function of neonates restrict the drug type and dosage. The optimal choice of sclerosants for neonates remains unclear.
This study aimed to analyze sclerotherapy for giant LMs in neonates, identify the possible factors that may affect the outcome, and discuss the treatment strategy for this disease in neonates.
Methods
Patients and clinical data
Fourteen patients with LMs >8 cm at the West China Second University Hospital of Sichuan University were retrospectively enrolled from September 2017 to October 2022. The LMs were found either during the fetal period or at birth. The following data were collected: sex, gestational age, body weight at birth, site of LM, type of lesion, lesion diameter, tissue/organ compression, treatment, and complications. The patients’ parents provided informed consent, including permission to use their clinical data and publishing patient photos. This study was approved by the ethics committee of West China Second University Hospital of Sichuan University. This study followed the ethical guidelines of the Declaration of Helsinki.
Treatment
The patients underwent color Doppler ultrasonography and/or magnetic resonance image (MRI)/computed tomography (CT) for diagnosis. All patients were treated with bleomycin and/or a 3% polidocanol injection under ultrasonographic and radiographic guidance. If an obvious airway compression was present, tracheal intubation was performed at birth or before treatment. The neonates received general anesthesia, and cysts within the lesions were punctured with 23-gauge needles under ultrasound guidance. The cystic fluid was extracted before drug injection. In total, 15 mg of bleomycin was dissolved in 10 ml iopamidol, and the solution was injected into different cysts under X-ray guidance. No more than 1 mg/kg of bleomycin was administered to each patient. In patients receiving combination therapy, 3% polidocanol (POL) was used as an additional treatment after bleomycin injection. The polidocanol foam was prepared using the Tessari method. Two injection syringes containing 2 ml 3% polidocanol and 8 ml air, respectively, were connected using a three-way valve. The agent and air were injected 20 times to form polidocanol foam, which was injected into the lesions. The polidocanol dose was no greater than 2 ml. The lesions were compressed using a bandage for 5–7 d. Six patients were treated with bleomycin (BLM group) and eight patients were treated with bleomycin + polidocanol (BLM + POL group). Most of the neonates had airway compression in this group, they received trachea intubation with ventilation and all received antibiotics treatment before or after treatment.
The effect of treatment was evaluated one month after drug injection. A second or third drug injection session was performed if the lesions were not completely involuted. Outcomes were classified using the following criteria [9]: 1. Excellent (≥90% reduction in lesions), 2. Moderate (≥50% reduction in lesions), 3. Mild (<50% reduction in lesions), 4. None (no response to treatment). The patients were followed up for 6–28 months.
To study the possible risk factors that might increase the number of treatment sessions, we compared gestational age, body weight, lesion size and type, and sclerosants used for treatment between patients undergoing ≤2 sessions and those undergoing >2 sessions.
Statistical analysis
SPSS (version 23.0) software was used for statistical analysis. Normally distributed continuous data between the two groups were analyzed using a t-test. Categorical data were analyzed using the likelihood ratio or Fisher’s exact test. Statistical significance was set at p < 0.05.
Results
Fourteen patients were included in this study, of whom 12 were diagnosed during the fetal period and two were diagnosed after birth. There were nine male and five female patients. Four patients were born preterm, and the lowest gestational age was 34+6 weeks. The size of the LMs ranged from 8 to 18 cm and included 11 macrocystic and three mixed lesions. Twelve patients experienced airway compression. One patient experienced compression of the thoracic wall. Only one patient had no compression, but the lesion size was 10 cm. Considering the possible risk of intralesional hemorrhage, the patient was treated at birth. Six patients were in the BLM group and eight patients were in the BLM + POL group. Table 1 shows the patient characteristics.
Treatment was initiated 2–5 days after birth. The average age at first treatment in the two groups was 3.6 days in the BLM group and 3.0 days in the BLM + POL group (p = 0.325). The mean lesion size in the BLM group was 9.5 cm while that in the BLM + POL group was 11.5 cm (p = 0.223). There were five macrocystic lesions and one mixed lesion in the BLM group, and six macrocystic lesions and two mixed lesions in the BLM + POL group (p = 0.615). Six patients in the BLM group and eight patients in the BLM + POL group underwent 16 and 15 treatments, respectively. Patients in the BLM group seemed to receive more treatment sessions (2.7 sessions) than those in the BLM + POL group (1.9 sessions), although the difference was not statistically significant (p = 0.064). All patients in the BLM + POL group showed an excellent response after 1–3 treatment sessions. However, five patients in the BLM group showed an excellent response after 2–4 treatment sessions. One patient in the BLM group showed a moderate response after three sessions and finally underwent surgical treatment. After the first session, three patients (37.5%) in the BLM + POL group showed an excellent response; however, no patient in the BLM group showed an excellent response (p = 0. 122). After the second treatment session, seven patients (87.5%) in the BLM + POL group and three (50%) in the BLM group showed an excellent response (p = 0.127) (Table 2).
Complications included fever (25.8%), vomiting (12.9%), and intralesional hemorrhage (16.1%) during the 31 procedures. The fever and vomiting lasted for no more than 48 h. No additional antibiotics were administered to treat fever after sclerotherapy. The ratios of fever and vomiting were not significantly different between the two groups (p = 0.685 and p = 0.325, respectively). Two patients in the BLM group and one in the BLM + POL group had intralesional hemorrhage after the procedures (p = 0.532); however, blood transfusion was not needed. Liver function was tested before and after the treatment and showed no abnormalities. The patient’s urine output was normal before and after treatment. None of the patients in either group exhibited pulmonary fibrosis at follow-up (Table 3).
Four patients received >2 treatment sessions and 10 patients received ≤2 sessions. The sclerosants used in the two groups were not significantly different (p = 0.594). The size (p = 0.03) and type (p = 0.011) of the lesions were significantly different between the two groups, suggesting that larger LMs and more complex lesions may require increased treatment sessions. However, age, preterm status, and body weight did not affect the number of treatment sessions (Table 4).
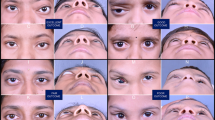
Typical images of patients in the two groups are shown in Figs. 1 and 2.
After the treatment, the lesion decreased significantly and the compression at the neck was relieved.
After the treatment, the size of the lesion reduced and chest radiography showed relief of the chest wall compression.
Discussion
Sclerotherapy has been used to treat LMs in neonates for decades. To date, there have been limited studies focusing on neonates [3], and many studies have been case reports [10, 11]. Besides, few studies have focused on early neonates (≤7 days). A large cohort study included 47 patients diagnosed at birth; however, treatment in the neonatal period was not specifically analyzed [12]. A recent study found sclerotherapy effective for LMs in the head and neck in neonates and infants ≤12 months of age [13]. In this study, we report a series of treatments for giant LMs in neonates that were successfully treated with sclerotherapy with slight complications. The first-time treatment was initiated at ≤5 days in all patients.
Neonates, especially preterm babies, usually have immature organ functions, resulting in an increased risk when undergoing anesthesia and drug therapy. Safety is always the first consideration when treating newborns. Sclerotherapy is a safe and effective method for treating LMs [14]. However, the safest sclerosant for neonates remains unclear. Bleomycin is a sclerosant that is widely used to treat LMs. In a pharmacokinetic study, bleomycin was systemically absorbed as a sclerosant in venous malformations and LMs [15], suggesting possible systemic adverse events in patients. There have been no pharmacokinetic studies on bleomycin in neonates. Complications of bleomycin include fever, vomiting, intralesional hemorrhage, acute lung injury, and pulmonary fibrosis [16,17,18]. Acute lung injury is rare and its underlying mechanism is unclear. It can also occur in patients treated with high or low doses [17]. Pulmonary fibrosis induced by bleomycin is dose-dependent. The cumulative dose of LMs is always too low to cause pulmonary fibrosis [19]. Pulmonary fibrosis has not been documented in bleomycin-treated LMs. Polidocanol, a detergent sclerosant, is often used to treat venous malformation and has recently been used in LMs [20]. Polidocanol-related complications are low. Intralesional hemorrhage has been reported in 13% of the patients treated with polidocanol [9]. In fact, bleomycin can also induce intralesional hemorrhage [12]. In this study, the incidence of short-term fever (25.8%) after the procedures was higher than in the previously reported cases [12, 16], suggesting that neonates and young infants might be more sensitive to sclerosants, especially bleomycin. Intralesional hemorrhage is another common complication. Bandage compression is believed to reduce the frequency of hemorrhage; however, further case-control studies are needed to confirm this.
Several sclerosants have been proven to be effective in the treatment of LMs. Both bleomycin and OK-432 are widely used agents [6, 21]. At our center, bleomycin is preferred because of its availability, effectiveness, and safety at an appropriate dosage. In neonates, a single dose of bleomycin cannot cover giant and complex cysts because of safety-related dose limitation. Previously reported combined therapies included bleomycin/pingyangmycin (bleomycin A5) + polidocanol, bleomycin + ethanol, polidocanol + ethanol, and pingyangmycin + triamcinolone acetonide [12, 22, 23]. The combined use of pingyangmycin and polidocanol results in a better response than polidocanol alone [23]. However, large-scale prospective studies are needed to establish the optimal treatment strategies for neonates.
Other alternative therapies for LMs include surgical excision, oral administration of sirolimus [24, 25]. However, to minimize injury and complications, sclerotherapy has been regarded as the optimal treatment for large cyst lesions. Surgery is regarded as the first-line treatment for circumscribed superficial microcystic LMs [26]. Sirolimus is used for long-term treatment of LMs, especially complex lesions [27]. Considering its immunosuppressive effects, careful consideration is required when using sirolimus in early newborns.
General anesthesia is needed for safety in neonates and young children undergoing sclerotherapy, especially in those undergoing tracheal intubation. The effect of general anesthesia on the nervous systems of neonates and young infants is not fully understood. A study of children aged <3 years suggested that multiple, but not single, exposures to procedures requiring general anesthesia are related to a pattern of neuropsychological changes associated with behavioral and learning difficulties [28]. Based on this study, a reduced number of procedures under general anesthesia may be helpful in preventing nervous system disorders in children. In our study, 37.5% of the patients in the BLM + POL group underwent one procedure and showed an excellent response. None of the BLM groups showed excellent response after the first session. Although some patients in other studies could be cured with one session of bleomycin treatment, in our study the patients were first treated at age ≤5 days with relatively large lesions. Low body weight restricted the dosage of bleomycin, whereas large lesions responded relatively poorly to the inadequate agent. In this study, large mixed-type lesions were associated with an increased number of procedures. Therefore, combined sclerosant therapy may be a better choice for neonates with giant LMs to reduce the number of procedures and the associated general anesthesia.
Conclusions
Both bleomycin and bleomycin+polidocanol appear to be relatively safe and effective for the treatment of neonates with giant LMs. The combination of the two agents seemed to reduce the number of procedures and general anesthesia required.
Limitations
The number of patients in this study was small because giant LMs in neonates are not very common in a single center. Large-scale multicenter studies may further verify the findings of the present study.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Sadick M, Müller-Wille R, Wildgruber M, Wohlgemuth WA. Vascular Anomalies (Part I): classification and diagnostics of vascular anomalies. Gefäßanomalien (Teil I): Klassifikation und Diagnostik von Gefäßanomalien. Rofo. 2018;190:825–35.
Mäkinen T, Boon LM, Vikkula M, Alitalo K. Lymphatic malformations: genetics, mechanisms and therapeutic strategies. Circ Res. 2021;129:136–54.
Bouwman FCM, Klein WM, de Blaauw I, Woiski MD, Verhoeven BH, Botden SMBI. Lymphatic malformations adjacent to the airway in neonates: Risk factors for outcome. J Pediatr Surg. 2021;56:1764–70.
Usui H, Shinkai M, Kitagawa N, Mochizuki K, Kawakita I, Tsuzuki Y, et al. Lymphatic malformations compromising the upper airway in children: ultrasound-guided intralesional focal sclerotherapy with bleomycin targeting culprit lesions. Pediatr Surg Int. 2020;36:1047–54.
Khosla S, Caton N, Zhang TT, Davies-Husband CR. Parapharyngeal abscess secondary to lymphovenous malformation. J Laryngol Otol. 2019;133:256–9.
Bhatnagar A, Neyaz Z, Singh B, Mishra A, Yusuf, Upadyayaya VD, et al. Bleomycin sclerotherapy in massive macrocystic lymphatic malformation: minimal complications with maximum results. J Cutan Aesthet Surg. 2020;13:319–25.
Kulungowski AM, Patel M. Lymphatic malformations. Semin Pediatr Surg. 2020;29:150971.
Ricci KW, Hammill AM, Mobberley-Schuman P, Nelson SC, Blatt J, Bender JLG, et al. Efficacy of systemic sirolimus in the treatment of generalized lymphatic anomaly and Gorham-Stout disease. Pediatr Blood Cancer. 2019;66:e27614.
Yamaki T, Sasaki Y, Hasegawa Y, Osada A, Konoeda H, Hamahata A, et al. Percutaneous ultrasound-guided sclerotherapy with polidocanol microfoam for lymphatic malformations. J Vasc Surg Venous Lymphat Disord. 2017;5:707–14.
Kenton A, Duncan N, Bhakta K, Fernandes CJ. Laryngeal lymphatic malformation in a newborn. J Perinatol. 2003;23:567–71.
Tang CY, Wijnen M, Sambeeck van SJ, Halbertsma FJ. Acute neonatal presentation of a lymphatic malformation. BMJ Case Rep. 2013;2013:bcr2012006784.
Bouwman FCM, Kooijman SS, Verhoeven BH, Schultze Kool LJ, van der Vleuten CJM, Botden SMBI, et al. Lymphatic malformations in children: treatment outcomes of sclerotherapy in a large cohort. Eur J Pediatr. 2021;180:959–66.
Caton MT, Duvvuri M, Baker A, Smith ER, Narsinh KH, Amans MR, et al. Percutaneous sclerotherapy for head and neck lymphatic malformations in neonates and infants ≤12 months of age. J Neurointerv Surg. 2023;15:1242–6.
De Maria L, De Sanctis P, Balakrishnan K, Tollefson M, Brinjikji W. Sclerotherapy for lymphatic malformations of head and neck: Systematic review and meta-analysis. J Vasc Surg Venous Lymphat Disord. 2020;8:154–64.
Mack JM, Peterson EC, Crary SE, Moran JH, Neville K, Pierce CD, et al. Pharmacokinetics of bleomycin sclerotherapy in patients with vascular malformations. Pediatr Blood Cancer. 2022;69:e29733.
Bawazir OA, Bawazir R, Bawazir A, Kausar N, Said H. Efficacy and clinical outcomes of bleomycin in the treatment of lymphangiomas: a multicenter experience. Dermatol Surg. 2021;47:948–52.
Méndez-Echevarría A, Fernandez-Prieto A, de la Serna O, Lopez-Gutierrez JC, Parron M, Marin-Aguilera B, et al. Acute lung toxicity after intralesional bleomycin sclerotherapy. Pediatrics. 2018;141:e20161787.
Tetikkurt C, Ongel D, Tetikkurt S. A case of bleomycin-induced lung toxicity. Monaldi Arch Chest Dis. 2018;88:981.
Niramis R, Watanatittan S, Rattanasuwan T. Treatment of cystic hygroma by intralesional bleomycin injection: experience in 70 patients. Eur J Pediatr Surg. 2010;20:178–82.
De Corso E, Cina A, Salonna G, Di Cintio G, Gaudino S, Panfili M, et al. Sclerotherapy with polidocanol microfoam in head and neck venous and lymphatic malformations. Acta Otorhinolaryngol Ital. 2022;42:116–25.
Aluffi Valletti P, Brucoli M, Boffano P, Benech A, Toso A, Dell’Era V, et al. A single-center experience in the management of head and neck lymphangiomas. Oral Maxillofac Surg. 2020;24:109–15.
Luo QF, Gan YH. Pingyangmycin with triamcinolone acetonide effective for treatment of lymphatic malformations in the oral and maxillofacial region. J Craniomaxillofac Surg. 2013;41:345–9.
Wang Q, Zhou Q, Zhao Z, Liu C, Zheng J. Successful sclerotherapy for cervicofacial macrocystic lymphatic malformations using polidocanol and pingyangmycin combined foam sclerosants. Lymphat Res Biol. 2022. https://doi.org/10.1089/lrb.2021.0053.
Poget M, Fresa M, El Ezzi O, Saliou G, Doan MT, de Buys Roessingh A. Lymphatic malformations in children: retrospective review of surgical and interventional management. Pediatr Surg Int. 2022;39:36.
Wiegand S, Dietz A, Wichmann G. Efficacy of sirolimus in children with lymphatic malformations of the head and neck. Eur Arch Otorhinolaryngol. 2022;279:3801–10.
Greene AK, Perlyn CA, Alomari AI. Management of lymphatic malformations. Clin Plast Surg. 2011;38:75–82.
Wiegand S, Wichmann G, Dietz A. Treatment of lymphatic malformations with the mTOR inhibitor sirolimus: a systematic review. Lymphat Res Biol. 2018;16:330–9.
Warner DO, Zaccariello MJ, Katusic SK, Schroeder DR, Hanson AC, Schulte PJ, et al. Neuropsychological and behavioral outcomes after exposure of young children to procedures requiring general anesthesia: the Mayo Anesthesia Safety in Kids (MASK) Study. Anesthesiology. 2018;129:89–105.
Funding
National Natural Science Foundation of China, Grant Number: 82102897.
Author information
Authors and Affiliations
Contributions
FH designed the study and drafted the manuscript; XL collected the data; FM participated in the writing of the manuscript; JY helped statistical analysis, revised the manuscript and provided the funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The patients’ parents provided informed consent, including permission to use their clinical data. The authors have obtained written informed consent for publication of the images of the patients. This study was approved by the ethics committee of West China Second University Hospital of Sichuan University. This study followed the ethical guidelines of the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hu, F., Ma, F., Liu, X. et al. Sclerothrapy of giant lymphatic malformation in neonates. J Perinatol 45, 213–217 (2025). https://doi.org/10.1038/s41372-024-02113-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41372-024-02113-z