Abstract
Objective
Extrauterine growth restriction (EUGR) affects 30–97% of preterm infants and is associated with poor outcomes. We used machine learning (ML) to assess how clinical and nutritional factors, particularly during the transition from parenteral to enteral nutrition, influence EUGR.
Study design
This retrospective observational study included 1165 patients (46% with EUGR) born below 33 weeks’ gestation or 1500 g. We developed 10 models to predict EUGR combining two sets of features (all and nutritional features only) across five subgroups of patients (all, extremely preterm, very preterm, moderately preterm, small for gestational age).
Results
Model accuracy was 0.71 (F1-score = Recall = AUROC = 0.71, Precision = 0.72) with nutritional features and 0.79 (F1-score = AUROC = 0.79, Precision = 0.80, Recall = 0.79) with all features. Lower EUGR risk was linked to female sex, higher growth velocity, and lipid intake in week one. Influential factors differed by subgroup.
Conclusion
ML models accurately predicted EUGR across preterm subgroups, highlighting the role of early nutritional and clinical variables.
Similar content being viewed by others
Introduction
Extrauterine growth restriction (EUGR) refers to inadequate growth during hospitalization [1]. At discharge, 30% to 97% of preterm infants, particularly those born very preterm or with very low birth weight (VLBW), do not achieve the expected growth based on intrauterine growth charts [1,2,3,4,5].
Inadequate growth in the neonatal intensive care unit (NICU) is associated with adverse short-term outcomes, such as sepsis and prolonged dependence on mechanical ventilation. It can also significantly impact neurodevelopment at 18–22 months corrected age in extremely low birth weight infants [3]. Therefore, providing adequate nutrition and ensuring appropriate growth in the NICU is crucial for this high-risk population [6,7,8,9].
The nutritional care of preterm infants in the NICU consists of three phases. The first is the parenteral nutrition (PN) phase, during which the infant relies entirely on intravenous nutrients, with only minimal enteral feeding, to prevent the consequences of fasting and support intestinal health and development. The second phase, known as transitional nutrition (TN), involves weaning from PN to enteral nutrition (EN) with a gradual increase in enteral intake. The third phase is the EN, where the infant is fully established on milk feeds without parenteral support.
The TN phase is the most critical [10,11], as numerous factors can interfere with adequate nutrition. Transitioning from full parenteral support to EN requires crucial decisions regarding modifiable nutritional practices, including vascular access, choice of milk, fortification, formula selection, and the amount of tolerated enteral intake. The EN is frequently interrupted due to feeding intolerance (e.g., abdominal distension, regurgitation) or medical conditions affecting feeding tolerance, such as severe sepsis, necrotizing enterocolitis (NEC), or cardiorespiratory instability.
While interruption of EN is necessary in certain circumstances, promoting early EN is beneficial to prevent gut atrophy, stimulate gastrointestinal maturation, enhance feeding tolerance, and reduce the incidence of NEC [12]. It is noteworthy that EN requirements significantly differ from parenteral requirements as intestinal absorption reduces the bioavailability of some nutrients. Unfortunately, international recommendations on nutrient intake refer solely to PN or EN [13,14,15,16,17], with no guidelines on weaning PN while minimizing nutrient delivery disruption.
Recently, the availability of big data in healthcare has enabled advanced statistical and machine learning (ML) methods to conduct large retrospective studies to evaluate optimal diagnostic and therapeutic strategies, predict outcomes, and support clinical decisions [18]. Specifically, ML methods are valuable for analyzing the complex impact of various clinical and nutritional factors affecting postnatal growth in preterm infants [18,19]. Specifically, the NICU is a highly data-rich environment, which is ideal for the application of ML and artificial intelligence in general. These technologies have the potential to revolutionize image interpretation, leverage electronic health record data to predict acute events (e.g., sepsis, NEC, acute kidney injury), prognosticate critical outcomes (e.g., bronchopulmonary dysplasia, retinopathy of prematurity), and uncover patterns in monitored parameters to enhance clinical decision-making [20,21,22,23].
ML combined with electronic clinical records and computerized programs for PN prescription has the potential to improve the analysis of the complex impact of various clinical and nutritional factors affecting postnatal growth in preterm infants. Previous studies employing ML to predict postnatal growth failure have focused on longitudinal microbiome [20] or metabolome data [21]. These studies evaluated the effect of timing, amount, and type of milk [20,22] or total parenteral intake [22]. However, the impact of specific nutrient intakes and TN strategy on postnatal growth has not been investigated.
The present study aims to investigate the effect of clinical and nutritional factors, including specific nutrient intakes during TN, on EUGR, leveraging ML tools applied to the electronic health records of preterm infants below 33 weeks of gestation or 1500 g of birth weight admitted to a neonatal intensive care unit from January 2005 to November 2021.
Methods
Study design and population
This retrospective observational study includes preterm infants admitted to the Fondazione IRCCS San Gerardo dei Tintori Neonatal Intensive Care Unit between January 2005 and November 2021. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by Comitato Etico Territoriale Lombardia 3 (ID 4281). Written informed consent was waived because the data were anonymized, and no re-identification codes linking the data to clinical record IDs were retained.
We included infants with a gestational age (GA) below 33 weeks and/or with a birth weight below 1500 g, with complete nutritional data. We excluded infants with medical conditions potentially affecting growth, including chromosomal abnormalities, congenital diseases, and major abdominal surgery. To ensure a diverse dataset for ML algorithms, we did not restrict the sample based on power analysis.
Clinical data collection
Data were extracted from the electronic medical records to derive 171 features, including patient demographics, anthropometric measurements, antenatal and perinatal factors, clinical complications, respiratory support needs, antibiotic use, nutritional strategies, and average growth velocity during the first week of life [23] (see eTable 1). Figure 1 provides an overview of the computed nutritional features based on macronutrient (glucose, proteins, and lipids) raw data.
MeanIncrementEN: Mean daily pro kg increment during TN; DaysToMaxPN: Days to the maximum pro kg amount for a given intake; DaysPNmacronutrients: Days with different parenteral macronutrients; Avg: average amount in g/kg/day, kcal/kg/day or ml/kg/day; Min/Max: maximum/minimum amount in g/kg/day, kcal/kg/day or ml/kg/day; *: computed for PN, EN, and cumulative pro kg intakes of proteins, glucose, lipids, fluids, calories, and non-protein calories.
Weight z-scores were calculated using the Intergrowth-21 standards [24], which have demonstrated good concordance with the Fenton reference standard [25]. EUGR was binary coded, considering a loss of at least one weight z-score between birth and the 36th week of postmenstrual age (PMA) or at discharge, whichever occurred first [26].
Machine learning models development
To assess the impact of the nutritional strategy on postnatal growth, we developed several ML models to predict EUGR using clinical data and parameters describing the nutritional strategy.
Specifically, we built 10 datasets combining 5 groups of patients: (a) all patients, (b) below 28 + 0 weeks’ gestation (extremely preterm, EP), (c) between 28 + 0 and 31 + 6 weeks’ gestation (very preterm, VP), (d) above 32 + 0 weeks (moderate-to-late preterm, MP), and (e) small for gestational age (SGA); and two subsets of features: (1) all features, (2) nutritional features only.
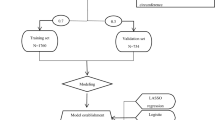
Figure 2 summarizes the ML models development. First, we performed majority-class undersampling—stratifying by GA for group (a)—to balance the number of EUGR and non-EUGR patients. Next, we split the dataset into training (80%) and validation (20%) sets to assess the internal and external validity of the prediction.
The steps marked with double lines are performed on the all-patients all-features dataset only, and applied to the others. Circular arrow: iterations for undersampling (5 seeds).
Then, we performed the following steps on the EUGR-balanced training set:
-
Correlation-based feature selection [27] on the all-features all-patients dataset (1a), considering different thresholds (ρ = 0.7, 0.8, 0.85, 0.9) for feature elimination.
-
Standardization and missing values imputation for each of the 5 subsets of patients. Missing weight values were linearly interpolated from the patient’s available data; continuous variables were imputed with 5-Nearest Neighbor; categorical data were imputed with the mode.
-
Training of several algorithms (Naïve Bayes, Classification Tree, Random Forest, Support Vector Classifier, Logistic Regression, and CatBoost) on the all-features all-patients dataset (1a) in k-fold cross-validation, considering k a tunable parameter (5 or 10).
The optimal correlation threshold for feature elimination (dataset 1a only—all patients, all features), the best performing model (dataset 1a only), and the number of folds for cross-validation (all datasets) were determined through hyperparameter tuning during cross-validation, optimizing for accuracy.
The best-performing model and configuration were refitted on each of the 10 feature-subgroup combinations.
The following steps were performed on the EUGR-balanced test set:
-
Standardization and missing values imputation using parameters computed on the training set to mitigate the risk of bias.
-
Fitting of the best-performing model
-
Model evaluation and interpretation
The significance of features in predicting EUGR was assessed using Shapley values and expressed as a percentage of importance. The model performance was evaluated in terms of accuracy, F1-score, precision, recall, and area under the receiving operating characteristic curve (AUROC). For each of the 10 feature-subgroup combinations, undersampling, model fitting, and validation were repeated with five random seeds, and performance was averaged. This improvement in robustness included a diverse subset of patients for each repetition. Data were analyzed using Python 3.10.12.
Statistical analysis
The main descriptors of infants’ condition were expressed as medians and interquartile ranges or numbers and percentages. The significance of differences was evaluated using the Wilcoxon rank sum test for continuous variables and Pearson’s Chi-squared test for dichotomous variables. Alpha values < 0.05 were considered statistically significant. Data were analyzed in R 4.0.1.
Results
Study population
One thousand five hundred and fifty-six newborns <33 weeks’ gestation or below 1500 g of birth weight were born at our center between January 2005 and November 2021.
Ninety patients were excluded due to congenital chromosomal abnormalities, congenital diseases, or major abdominal surgery; 301 were excluded because the nutritional data did not include the TN phase until full enteral feeding (FEF) was achieved (Supplementary Fig. 1). Among patients without complete nutritional data, 87 (29%) received only EN, 4 (2%) were still hospitalized when data were extracted, 118 (39%) died before achieving FEF, 76 (25%) were transferred before achieving FEF, and 16 (5%) had data relative to less than 72 h of FEF. 1165 were included in the analysis, 320 were moderate-to-late preterm, 641 very preterm, 204 extremely preterm, and 309 SGA. Of these 1165 patients, 531 (45.6%) developed EUGR (Table 1). The EUGR subgroup showed significant differences from the non-EUGR group, including more males, lower GA and BW, fewer SGA cases, worse Apgar scores, higher early-onset sepsis rates, poorer respiratory outcomes, and longer hospital stays. Regarding nutritional factors, they started EN later after birth, but at a higher PMA, they had a longer TN phase. They also began FEF at a later postnatal age. Although the difference in PMA at FEF initiation was statistically significant, the difference—less than 1 week—was clinically negligible.
Surgical NEC and death occurred in three and two cases, respectively, but such proportions do not reflect the prevalence in the overall population. Indeed, of the 301 infants excluded due to lack of TN data and/or because they did not reach FEF, 118 died before 36 weeks of PMA, and 76 were transferred to other hospitals, some for abdominal surgery.
Best-performing model and configuration
The optimal number of folds for cross-validation was 10 for datasets comprising all patients, MP, and VP; 5 for datasets comprising EP and SGA. We identified an optimal correlation threshold of 0.85 for feature elimination, leading to the selection of 54 features, including 31 nutritional parameters and the average growth velocity over the first week of life growth (see eTable 1). CatBoost was selected as the best-performing model with an accuracy of 0.76 ± 0.05 (mean and standard deviation across validation folds during internal validation) (see eTable 2 for the complete comparison).
Features importance
On the whole population, the model including only nutritional features had an accuracy of 0.71 ± 0.02 (mean and standard deviation across the five EUGR-balanced datasets) in external validation; the model’s performance in terms of F1-score, precision, recall, and AUROC was 0.71 ± 0.01, 0.72 ± 0.04, 0.71 ± 0.03, and 0.71 ± 0.02, respectively. The model with all features had an accuracy of 0.79 ± 0.03, F1-score = 0.79 ± 0.02, Precision = 0.80 ± 0.05, Recall = 0.78 ± 0.02, and AUROC 0.79 ± 0.03. Details on internal and external validation are reported in eTable 3 for one EUGR-balanced dataset and in Figs. 3 and 4 for all the seeds. Figures 3 and 4 show the most important features for EUGR prediction, considering all features and only nutritional features, respectively. Lower growth velocity in the first week of life was among the most important factors associated with EUGR across groups. Lipid intake was associated with better growth, especially in SGA patients. Being male was among the most important predictors of EUGR in the whole population, but it was relatively less important in extremely preterm infants. Regarding TN variables, in the entire population, the most important factor was the average parenteral protein intake (importance = 12.5%), whilst, in the SGA sub-group, it was the mean increment of enteral proteins (importance = 18.4%), with high values associated with increased risk of EUGR. TN features were generally more important in EP and SGA infants than in other subgroups.
Positive values are associated with better outcomes (decreased risk of EUGR). External performance metrics across the five EUGR-balanced datasets are reported in the boxes. Acc accuracy, F1 F1-score, Prec precision, Rec recall, AUROC area under the receiving operating characteristic curve.
Positive values are associated with better outcomes (decreased risk of EUGR). External performance metrics across the five EUGR-balanced datasets are reported in the boxes. Acc accuracy, F1 F1-score, Prec precision, Rec recall, AUROC area under the receiving operating characteristic curve.
Discussion
We investigated the effect of clinical and nutritional factors on EUGR, focusing on the transition from parenteral to enteral feeding. This study significantly advances understanding of the complex factors contributing to EUGR in preterm infants. Specifically, it provides an innovative and comprehensive analysis of the interplay between clinical and nutritional variables by leveraging ML tools and a robust dataset from over 15 years of electronic health records. Furthermore, the use of explainable artificial intelligence techniques (e.g., Shapley values) offers a transparent interpretation of feature importance, enhancing the clinical relevance of the findings across different patient subgroups.
Models accuracy
The prediction accuracy was comparable to previous reports [28] and, more importantly, consistent between internal and external validation, demonstrating good generalization capability and robustness of the models. Even if the models which include both clinical and nutritional features achieved the best results—confirming a multifactorial influence on preterm growth—nutritional features alone achieved accuracies that were only slightly below the state of the art, confirming the key role of an adequate nutrition strategy to meet growth goals. The finding that the predictive accuracy improved when integrating nutritional features underscores the importance of early macronutrient management, such as lipid and protein intake, in preventing EUGR.
Opportunities for an early intervention
Growth velocity during the first week of life consistently affected EUGR regardless of the patient subgroup, indicating an association between slow early growth rates and nutritional deficits [29,30]. Lipid intake during the first week of life was also important across subgroups, highlighting the critical role of early nutritional interventions. Indeed, starting adequate PN within the first hours of life is essential to promote optimal growth and prevent energy deficits in the early postnatal period [31,32]. Interestingly, even if extremely preterm and SGA infants are characterized by long hospital stays (median and quartiles: 79 (63, 100) days and 42 (29, 61) days, respectively), variables related to the first day and the first week of life remain important, as in the other groups.
Effect of nutrition during the transition phase
An innovative aspect of the present study is the focus on TN. Interestingly, we identified specific factors relative to the TN phase that significantly predicted EUGR. In particular, we observed a positive impact of protein content in PN on growth. In contrast, increased fluid intake, both in PN and EN, negatively impacted growth. These findings suggest that adequate provision of amino acids is more critical for promoting lean mass accretion and supporting growth during the TN phase than fluid provision, as supported by the scientific literature [33]. Such an interpretation is consistent with previous reports. Meio et al. [33] recently analyzed the total energy-to-protein ratio, finding that during the second week of life, typically corresponding to the TN phase, a median ratio below 25 kcal/g of protein was significantly associated with weight-based EUGR [34]. A Cochrane Review comparing higher versus lower parenteral amino acid intake revealed that increased amino acid intake in PN reduced the incidence of postnatal growth failure without affecting mortality [35].
Variables related to the TN phase are among the most important predictors of EUGR in extremely preterm and SGA infants. This finding, together with the longer duration of the TN phase (extremely preterm: 13 (9, 20) days; SGA: 8 (6, 12) days), suggests a higher risk of feeding difficulties and complications during the TN phase in these subgroups of patients, likely due to their smaller size and immature digestive systems [36].
Effect of specific macronutrients
Another specificity of our study is that we included individual macronutrients in the prediction models, allowing us to speculate on their relative importance in determining adequate postnatal growth. Interestingly, we found that high fluid and glucose intakes are associated with an increased risk of EUGR. The discrepancy between the beneficial effect of lipid intake through PN on growth and the negative impact of high fluid and glucose intake may be explained by the fact that lipid administration can be considered a proxy for complete PN. Our findings are consistent with a meta-analysis that demonstrated a correlation between the early introduction of high lipid intake and growth in preterm infants [26].
Differential effect of nutritional factors on specific subgroups of patients
The stratification of the study population into homogeneous subgroups had technical and clinical implications. From a technical perspective, increased data homogeneity could benefit ML models, potentially compensating for the reduction in sample size. Such a dual effect is evident from the accuracy results, which remained nearly stable. From a clinical perspective, stratification could help identify differences in the determinants of growth among subgroups of patients and guide tailored corrective actions.
Regarding the EP group, our findings indicate that ensuring adequate parenteral protein intake is essential, especially immediately following birth. During the TN phase, it is crucial to maintain sufficient caloric intake through both parenteral and enteral routes. Additionally, a higher enteral protein intake has a positive impact on growth.
Although a rapid transition might benefit the general preterm population, it may not be appropriate for SGA infants. In these cases, a more cautious advancement of enteral feeding and a gradual reduction of PN is recommended, as a swift increase in enteral protein intake could hinder growth. This suggests that their ability to absorb and utilize amino acids may be impaired. Indeed, a faster increase in enteral caloric intake was associated with a higher risk of EUGR in these infants. Additionally, we observed a positive impact of increased parenteral protein intake on growth in SGA infants. Such a finding aligns with other studies that link poor growth outcomes at discharge to protein deficits during weaning from PN [5,10]. An adequate protein intake is also crucial for brain growth and development, as proteins serve as the primary structural framework of the brain, as suggested by the positive correlation between higher protein intake, increased head circumference growth, and enhanced neurodevelopmental outcomes [37].
Differential effect of clinical factors on postnatal growth in specific subgroups of patients
Surprisingly, gestational age and birth weight were not among the most important variables for predicting growth outcomes, despite being well-established risk factors for EUGR [38]. Similarly, we observed an unexpected behavior in the role of sex: while males are generally considered at higher risk for EUGR [38], sex becomes less significant in more critical situations, such as in extremely preterm or SGA infants. This finding suggests that, in a multidimensional approach, commonly recognized risk factors are less critical compared to other variables.
Study limitations
A limitation of this study is that excluding patients who did not complete the TN phase led to the loss of a significant number of infants who were transferred for abdominal surgery and who died (particularly in the extremely preterm subgroup), introducing survivorship bias. However, since our analysis focused specifically on the effect of TN on postnatal growth, we found it reasonable to exclude patients with incomplete data on the intervention of interest. Another limitation is the single-centre study design, which may limit the generalizability of results. In our unit, the electronic clinical report has been customized to receive detailed data from the software used for the computerized prescription of PN and integrate them with the prescription of EN. Such customization allows us to analyze the association of individual nutrients across time on relevant outcomes, but makes it difficult to aggregate our data with other centres. Our results hold value as they may serve as preliminary data for designing multicentric, prospective studies investigating the effect of specific TN strategies on postnatal growth.
Conclusions
This study is the first to analyze such a comprehensive volume of data, encompassing both nutritional and non-nutritional factors, to assess the risk factors for EUGR using an innovative machine-learning model. The approach not only represents a significant advancement in understanding EUGR but also sets a new standard for predictive modeling in neonatal care.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to privacy or ethical restrictions, but are available from the corresponding author on reasonable request.
Code availability
The code for machine learning models is available from the corresponding author on reasonable request.
References
Clark RH, Thomas P, Peabody J. Extrauterine growth restriction remains a serious problem in prematurely born neonates. Pediatrics. 2003;111:986–90.
Griffin IJ, Tancredi DJ, Bertino E, Lee HC, Profit J. Postnatal growth failure in very low birthweight infants born between 2005 and 2012. Arch Dis Child Fetal Neonatal Ed. 2016;101:50–5.
Ehrenkranz RA, Dusick AM, Vohr BR, Wright LL, Wrage LA, Poole WK. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics. 2006;117:1253–61.
Stevens TP, Shields E, Campbell D, Combs A, Horgan M, La Gamma EF, et al. Variation in enteral feeding practices and growth outcomes among very premature infants: a report from the New York State Perinatal Quality Collaborative. Am J Perinatol. 2015;33:9–19.
Miller M, Vaidya R, Rastogi D, Bhutada A, Rastogi S. From parenteral to enteral nutrition. J Parenter Enter Nutr. 2014;38:489–97.
Lucas A, Morley R, Cole TJ. Randomised trial of early diet in preterm babies and later intelligence quotient. Br Med J. 1998;317:1481–7.
Hayakawa M, Okumura A, Hayakawa F, Kato Y, Ohshiro M, Tauchi N, et al. Nutritional state and growth and functional maturation of the brain in extremely low birth weight infants. Pediatrics. 2003;111:991–5.
Tan MJ, Cooke RW. Improving head growth in very preterm infants—a randomised controlled trial I: neonatal outcomes. Arch Dis Child Fetal Neonatal Ed. 2008;93:F337–F341.
Ziegler EE, Carlson SJ. Early nutrition of very low birth weight infants. J Matern Fetal Neonatal Med. 2009;22:191–7.
Brennan AM, Fenton S, Murphy BP, Kiely ME. Transition phase nutrition recommendations: a missing link in the nutrition management of preterm infants. J Parenter Enter Nutr. 2018;42:343–51.
Miller M, Donda K, Bhutada A, Rastogi D, Rastogi S. Transitioning preterm infants from parenteral nutrition: a comparison of 2 protocols. J Parenter Enter Nutr. 2017;41:1371–9.
Pietz J, Achanti B, Lilien L, Stepka EC, Mehta SK. Prevention of necrotizing enterocolitis in preterm infants: a 20-year experience. Pediatrics. 2007;119:e164–e170.
Mesotten D, Joosten K, van Kempen A, Verbruggen S, Braegger C, Bronsky J, et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: carbohydrates. Clin Nutr. 2018;37:2337–43.
Lapillonne A, Fidler Mis N, Goulet O, van den Akker CHP, Wu J, Koletzko B, et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: lipids. Clin Nutr. 2018;37:2324–36.
van Goudoever JB, Carnielli V, Darmaun D, Sainz de Pipaon M, Braegger C, Bronsky J, et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: amino acids. Clin Nutr. 2018;37:2315–23.
Joosten K, Embleton N, Yan W, Senterre T, Braegger C, Bronsky J, et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: energy. Clin Nutr. 2018;37:2309–14.
Embleton ND, Jennifer Moltu S, Lapillonne A, Van Den Akker CHP, Carnielli V, Fusch C, et al. Enteral nutrition in preterm infants (2022). J Pediatr Gastroenterol Nutr. 2023;76:248–68.
Nair S, Hsu D, Celi LA. Challenges and opportunities in secondary analyses of electronic health record data. Second analysis of electronic health records. Springer, Cham. 2016; 17–26.
Alanazi A. Using machine learning for healthcare challenges and opportunities. Inform Med Unlocked. 2022;30:100924.
Lugo-Martinez J, Xu S, Levesque J, Gallagher D, Parker LA, Neu J, et al. Integrating longitudinal clinical and microbiome data to predict growth faltering in preterm infants. J Biomed Inf. 2022;128:104031.
Younge NE, Newgard CB, Cotten CM, Goldberg RN, Muehlbauer MJ, Bain JR, et al. Disrupted maturation of the microbiota and metabolome among extremely preterm infants with postnatal growth failure. Sci Rep. 2019;9:1–12.
Greenbury SF, Ougham K, Wu J, Battersby C, Gale C, Modi N, et al. Identification of variation in nutritional practice in neonatal units in England and association with clinical outcomes using agnostic machine learning. Sci Rep. 2021;11:1–15.
Patel AL, Engstrom JL, Meier PP, Jegier BJ, Kimura RE. Calculating postnatal growth velocity in very low birth weight (VLBW) premature infants. J Perinatol. 2009;29:618.
Villar J, Ismail LC, Victora CG, Ohuma EO, Bertino E, Altman DG, et al. International standards for newborn weight, length, and head circumference by gestational age and sex: the Newborn Cross-Sectional Study of the INTERGROWTH-21st Project. Lancet. 2014;384:857–68.
González-García L, García-López E, Fernández-Colomer B, Mantecón-Fernández L, Lareu-Vidal S, Suárez-Rodríguez M. et al. Extrauterine growth restriction in very low birth weight infants: concordance between Fenton 2013 and INTERGROWTH-21st growth charts. Front Pediatr. 2021;9. https://doi.org/10.3389/FPED.2021.690788.
Peila C, Spada E, Giuliani F, Maiocco G, Raia M, Cresi F, et al. Extrauterine growth restriction: definitions and predictability of outcomes in a cohort of very low birth weight infants or preterm neonates. Nutrients. 2020;12. https://doi.org/10.3390/nu12051224.
Hall MA, Smith LA. Practical feature subset selection for machine learning. Comput Sci. 1998;98:181–91.
Han JH, Yoon SJ, Lee HS, Park G, Lim J, Shin JE, et al. Application of machine learning approaches to predict postnatal growth failure in very low birth weight infants. Yonsei Med J. 2022;63:640–7.
Gao C, Ehsan L, Jones M, Khan M, Middleton J, Vergales B, et al. Time to regain birth weight predicts neonatal growth velocity: a single-center experience. Clin Nutr ESPEN. 2020;38:165–71.
Steward DK, Pridham KF. Growth patterns of extremely low-birth-weight hospitalized preterm infants. J Obstet Gynecol Neonatal Nurs. 2002;31:57–65.
Sharma D. Golden hour of neonatal life: need of the hour. Matern Heal Neonatol Perinatol. 2017;3:1–21.
Wang N, Cui L, Liu Z, Wang Y, Zhang Y, Shi C, et al. Optimizing parenteral nutrition to achieve an adequate weight gain according to the current guidelines in preterm infants with birth weight less than 1500 g: a prospective observational study. BMC Pediatr. 2021;21:1–9.
Méio MDBB, de Miranda Salgado GG, Villela LD, da Costa ACC, Lima PAT, Milanesi BG, et al. Influence of morbidity, early nutritional intake, and total energy: protein ratio on longitudinal extrauterine growth restriction of very preterm newborns at term-equivalent age: an observational study. Eur J Pediatr. 2024;183:3327–36.
Embleton ND, Cleminson J, Zalewski S. What growth should we aim for in preterm neonates?. Paediatr Child Health. 2017;27:18–22.
Osborn DA, Schindler T, Jones LJ, Sinn JKH, Bolisetty S. Higher versus lower amino acid intake in parenteral nutrition for newborn infants. Cochrane Database Syst Rev. 2018. https://doi.org/10.1002/14651858.CD005949.
Behrman RE, Butler AS. Preterm birth: causes, on sequences, and prevention. National Academies Press. 2006. p. 1–772.
Morgan C, McGowan P, Herwitker S, Hart AE, Turner MA. Postnatal head growth in preterm infants: a randomized controlled parenteral nutrition study. Pediatrics. 2014;133:e120–e128.
Shan HM, Cai W, Cao Y, Fang BH, Feng Y. Extrauterine growth retardation in premature infants in Shanghai: a multicenter retrospective review. Eur J Pediatr. 2009;168:1055–9.
Funding
Open access funding provided by Politecnico di Milano within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Linda Greta Dui performed formal data analysis and drafted the first version of the manuscript; Valentina Bozzetti contributed to the study design, interpreted the data and drafted the first version of the manuscript; Emanuela Zannin interpreted the data, reviewed and revised the manuscript; Silvia Riccò analyzed the data, drafted the figures and tables; Paola Coglianese designed the database and supervised data collection; Valeria Cavalleri and Lucia Iozzi contributed to data collection and results interpretation; Maria Luisa Ventura and Simona Ferrante led study design and supervised results interpretation and formal analysis, respectively. All authors approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical statement
The study protocol was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by Comitato Etico Territoriale Lombardia 3 (ID 4281).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bozzetti, V., Dui, L.G., Zannin, E. et al. AI to predict extrauterine growth restriction during transitional nutrition of preterm infants: a retrospective study. J Perinatol 46, 416–424 (2026). https://doi.org/10.1038/s41372-025-02445-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41372-025-02445-4