Abstract
Perinatal transition is uniquely complex in newborns with critical congenital heart disease (CCHD), whose cardiopulmonary physiology often diverges from the standard neonatal resuscitation framework. We developed lesion-specific clinical practice guidelines (CPGs) for delivery room (DR) management of six high-risk CCHD diagnoses – hypoplastic left heart syndrome, dextro- transposition of the great arteries, Ebstein anomaly, congenital complete heart block, total anomalous pulmonary venous return, and tetralogy of Fallot with absent pulmonary valve. Developed collaboratively between neonatologists, cardiologists, and cardiac intensivists, these guidelines standardize DR stabilization for high-risk, low frequency events, promote a shared mental model among multidisciplinary teams, and provide structured escalation thresholds for oxygen, respiratory support, intubation, and medication use. Beyond clinical standardization, these CPGs enhance provider education, team preparedness, and family counseling. They offer a scalable framework adaptable to centers with or without on-site cardiac care, bridging physiologic principles with practical implementation.
Similar content being viewed by others
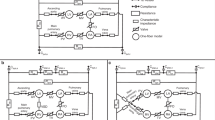
Perinatal transition from fetal to neonatal physiology poses distinct challenges for infants with critical congenital heart disease (CCHD), defined as life-threatening heart disease likely to require emergent cardiac intervention (catheter- or surgical-based) and/or highly specialized care within hours of birth [1]. While the Neonatal Resuscitation Program (NRP®) provides a foundational algorithm for newborn resuscitation, infants with CCHD require tailored adaptations to standard protocols to concomitantly address the high risk of decompensation shortly after delivery and alterations to usual cardiopulmonary physiology. Depending on the lesion, these adaptations include modified oxygen saturation goals, planned intubation with individualized ventilation strategies, early intravenous access, prostaglandin-E1 infusion, inotropic support, inhaled nitric oxide, and sedation with or without neuromuscular blockade [2,3,4,5,6]. Effective management of these complex cases, with severe but often predictable physiologic derangements, benefits from clinical guidance grounded in expected physiology and informed by expert consensus and experience at high-volume centers. Although similar frameworks have been described previously, institutional perspectives remain valuable to illustrate how these principles are applied across different care settings [4, 5, 7]. Sharing these experiences highlights variation, identifies shared challenges and supports broader efforts to standardize delivery room (DR) management for newborns with CCHD. This perspective article presents our institutional clinical practice guidelines (CPGs), aimed at translating physiologic principles into practical delivery room education and management (Fig. 1).
HLHS hypoplastic left heart syndrome, d-TGA d- transposition of the great arteries, TOF-APV tetralogy of Fallot with absent pulmonary valve, TAPVR total anomalous pulmonary venous return.
Prenatal diagnosis, multidisciplinary collaboration, and family counseling are essential components of optimal care for newborns with CCHD [5]. Prenatal diagnosis of CCHD has been associated with improved preoperative survival, likely due to better preparation for delivery and early postnatal management [8]. Integrating lesion-specific physiology into delivery room planning is an important part of reducing adverse perinatal events. Perinatal instability, prolonged severe hypoxemia, and asphyxia in infants with CCHD have been linked to short-term morbidity, including prolonged mechanical ventilation and hypoxic ischemic brain injury, and higher preoperative mortality [9, 10].
We identified six high-risk CCHD diagnoses that would benefit from targeted DR strategies to support rapid stabilization and optimize systemic oxygen delivery and cardiac output [11]. These included (1) hypoplastic left heart syndrome (HLHS), particularly with an intact or restrictive atrial septum (2); dextro-transposition of the great arteries (d-TGA), particularly with a restrictive atrial septum (3); severe Ebstein anomaly, particularly with concern for circular shunt physiology (4); congenital complete heart block, particularly with inadequate ventricular escape (5); total anomalous pulmonary venous return (TAPVR), particularly obstructed TAVPR; and (6) Tetralogy of Fallot with absent pulmonary valve (TOF-APV) (Supplementary Figs. 1–6). Neonatologists at Brigham and Women’s Hospital (BWH), a high-volume delivery center (~7000 births annually) with a high-risk delivery service, collaborated with pediatric cardiologists and cardiac intensivists at Boston Children’s Hospital (BCH) to develop lesion-specific DR CPGs for these conditions. Each participant contributed expertise in DR stabilization or postnatal management of CCHD, and consensus was reached for each guideline.
At our institutions, deliveries occur at BWH, where the NICU team provides initial resuscitation and rapid stabilization in the mother’s labor room or operating room, followed by a short transport across an indoor bridge to BCH for CICU care. All deliveries are attended by the NICU team, which includes a neonatologist, neonatology fellow or advanced practice provider, pediatric resident, respiratory therapist, and two neonatal nurses. A cardiologist or CICU attending is present in the DR for an average of five deliveries per year and otherwise is available by phone for support as needed. For example, a cardiologist or CICU attending was present at the deliveries of infants with severe Ebstein anomaly requiring inhaled nitric oxide, complete heart block with fetal hydrops and potential need for transcutaneous pacing, and severe cardiomyopathy with possible need for expedited extracorporeal membrane oxygenation support. Since 2021 (1/1/21-9/4/25), BWH has delivered 947 newborns with prenatally diagnosed structural CHD or congenital arrhythmia (median: 186 per year), including newborns with one of the six high-risk diagnoses: 85 HLHS, 110 d-TGA, 20 Ebstein anomaly, 17 complete heart block, 14 TAPVR, and 4 TOF-APV. There were additional patients with more complex anatomy for whom the CPG most similar to expected physiology was used to guide DR management.
Our CPGs were designed to standardize care for high-risk, low frequency events and foster a shared mental model among maternal-fetal medicine, NICU, and CICU teams [12]. In line with previously published institutional guidelines and the recently introduced NRP® Cardiac modules [3, 5, 6, 13, 14], our CPGs offer lesion-specific recommendations for stabilization. They add further value by providing a detailed, practical framework reflecting the sequence of events encountered in high-risk deliveries, starting at birth and incorporating typical clinical practice through transport to the CICU. Clinical variability is addressed through well-defined escalation thresholds for oxygen administration, non-invasive respiratory support, intubation, sedation, and initiation of inotropic support. For example, the d-TGA CPG (Supplementary Fig. 2) specifies that patients meeting criteria for intubation (pre-ductal oxygen saturation <70% at 5–10 min after birth despite non-invasive respiratory support) should receive sedation and neuromuscular blockade to reduce metabolic demand in the setting of severe hypoxemia, a practice not included in standard neonatal resuscitation protocols and therefore less commonly performed prior to guideline implementation.
The CPGs have also become a valuable peer-to-peer educational tool in preparing for high-risk deliveries. Each CPG provides a concise synthesis of the expected CCHD physiology and potential postnatal complications, along with the rationale for specific DR resuscitation recommendations. This format enables fellows or attendings to review, teach and orient the entire multidisciplinary team, and serves as a shared reference during the pre-delivery huddle. During the huddle, the multidisciplinary DR team - including neonatologists, cardiology specialists (if present), advanced practice providers, fellows or residents, respiratory therapists, and bedside and charge nurses - use a structured checklist (Supplementary Fig. 7) to review the case. The huddle empowers the DR team to align around goals for oxygenation and hemodynamics, anticipate degree of illness, and clarify potential medical and procedural escalation of care pathways. This process has enhanced confidence and preparedness, particularly among less experienced team members, in carrying out specialized resuscitation strategies for newborns with CCHD [15].
Developing and implementing these guidelines has also strengthened partnerships among maternal-fetal medicine, NICU, CICU, fetal cardiology and cardiovascular surgical teams. This work has fostered a collaborative and respectful culture of communication that supports coordinated planning across the full spectrum of cases, from straightforward deliveries guided by established CPGs to those requiring individualized strategies for complex anatomy or physiology, including cases complicated by prematurity, fetal hydrops, or additional anomalies. Monthly multidisciplinary meetings are used to review upcoming high-risk deliveries and serve as a platform for continuous quality improvement, allowing teams to reflect on recent cases, evaluate key processes, and identify opportunities to refine DR planning and communication. The CPGs additionally inform the anticipatory guidance provided by the NICU and fetal cardiologists during prenatal counseling, ensuring consistent expectations for DR care.
An important evolution of this collaborative framework has been its extension beyond the specific lesions covered by formal CPGs. The core principles of reviewing high-risk cases, understanding how CCHD physiology may present at the time of delivery, and establishing a documented, detailed management plan that is reviewed by the multidisciplinary team prior to delivery is a process that we have applied to atypical or rare cases where no predefined protocol exists. For example, an infant with a large pericardial teratoma and effusion, at risk for tamponade, was safely delivered at BWH, with the CICU in attendance and interventional cardiology and cardiovascular surgery on standby in the BCH CICU. The plan incorporated the standard CPG framework of outlining the patient’s physiology, what would be expected in the post-delivery transition, and a discussion of potential resuscitative interventions and necessary resources to prepare. This example illustrates how the overall framework of this program has facilitated consistent, collaborative decision-making and safe perinatal management, even for cases that fall outside existing CPGs.
These experiences have also laid the groundwork for systematic evaluation of their clinical impact. To advance from expert-driven consensus toward evidence-based practice, future work will report on our ongoing quality improvement initiatives and quantitative analyses comparing clinical practice before and after CPG implementation. CPG implementation may influence delivery location (labor room versus operating room delivery), rates of delivery room procedures, sedation practices, time to transport, and time to critical interventions such as balloon atrial septostomy. Subsequent studies will also examine how standardized DR interventions affect opportunities for delayed cord clamping, early bonding, preoperative oral feeding, and clinical outcomes such as severity of illness on CICU admission, length of stay and survival to hospital discharge. Balancing timely intervention with avoidance of unnecessary procedures remains an ongoing challenge, underscoring the importance of evaluating physiologic outcomes alongside developmental and family-centered perspectives.
In summary, our institution-specific CPGs have enabled our multidisciplinary teams to standardize DR care for newborns with high-risk CCHD, aligning DR management with the unique pathophysiology of each lesion. These guidelines can be adapted for use in fetal care delivery units within a free-standing children’s hospital, or for separate delivery hospitals requiring neonatal transport to specialized centers. We hope our CPGs serve as a model for other institutions seeking to improve DR management for this vulnerable population. Future work should aim to transition these expert-driven practices toward evidence- and quality-based protocols supported by multicenter data.
References
Słodki M, Respondek-Liberska M, Pruetz JD, Donofrio MT. Fetal cardiology: changing the definition of critical heart disease in the newborn. J Perinatol. 2016;36:575–80.
Thomas AR, Ma AL, Weinberg DD, Huber M, Ades A, Rychik J, et al. Delivery room oxygen physiology and respiratory interventions for newborns with cyanotic congenital heart disease. J Perinatol. 2021;41:2309–16.
Patel SR, Michelfelder E. Prenatal diagnosis of congenital heart disease: the crucial role of perinatal and delivery planning. J Cardiovasc Dev Dis. 2024;11:108.
Donofrio M. Predicting the future: delivery room planning of congenital heart disease diagnosed by fetal echocardiography. Am J Perinatol. 2018;35:549–52. http://www.ncbi.nlm.nih.gov/pubmed/29694994%0A.
Sethi N, Klugman D, Said M, Hom L, Bowers S, Berger JT, et al. Standardized delivery room management for neonates with a prenatal diagnosis of congenital heart disease: A model for improving interdisciplinary delivery room care. J Neonatal Perinatal Med. 2021;14:317–29.
Ali N, Donofrio MT. Delivery room and early postnatal management of neonates with congenital heart disease. Prenat Diagn. 2024;44:915–24.
Donofrio MT, Levy RJ, Schuette JJ, Skurow-Todd K, Sten MB, Stallings C, et al. Specialized delivery room planning for fetuses with critical congenital heart disease. Am J Cardiol. 2013;111:737–47.
Holland BJ, Myers JA, Woods CR. Prenatal diagnosis of critical congenital heart disease reduces risk of death from cardiovascular compromise prior to planned neonatal cardiac surgery: a meta-analysis. Ultrasound Obstet Gynecol. 2015;45:631–8.
Boos V, Kocjancic L, Berger F, Bührer C. Delivery room asphyxia in neonates with ductal-dependent congenital heart disease: a retrospective cohort study. J Perinatol. 2019;39:1627–34.
Petit CJ, Rome JJ, Wernovsky G, Mason SE, Shera DM, Nicolson SC, et al. Preoperative brain injury in transposition of the great arteries is associated with oxygenation and time to surgery, not balloon atrial septostomy. Circulation. 2009;119:709–16.
Kwiatkowski DM, Ball MK, Savorgnan FJ, Allan CK, Dearani JA, Roth MD, et al. Neonatal congenital heart disease surgical readiness and timing. Pediatrics. 2022;150:e2022056415D.
Thomas AR, Levy PT, Donofrio MT, Law B, Joynt C, Gupta R, et al. Call to action: prioritizing delivery room care for neonates with critical congenital heart disease. J Perinatol. 2024;44:321–4.
Donofrio MT, Moon-Grady AJ, Hornberger LK, Copel JA, Sklansky MS, Abuhamad A, et al. Diagnosis and treatment of fetal cardiac disease. Circulation. 2014;129:2183–242.
NRP® Cardiac. 2025. Available from: https://www.aap.org/en/pedialink/neonatal-resuscitation-program/nrp-cardiac/?srsltid=AfmBOoqVeWOui5rcxUPlU0i2e09mHB_qe_QOce-M194zNcytcsxh7AKt
Thomas AR, Bowen C, Abdulhayoglu E, Brennick E, Woo K, Everett MF, et al. Structured pre-delivery huddles enhance confidence in managing newborns with critical congenital heart disease in the delivery room. J Perinatol. 2025;45:1009–16.
Acknowledgements
We thank Kai-ou Tang, M.A. for her illustration of Fig. 1.
Author information
Authors and Affiliations
Contributions
AT and DG contributed equally to the project and conceptualized and wrote the first draft of the CPGs and manuscript. EK, CA, EA, EF, AD, JK, EV, SM, and SG critically reviewed and revised the CPGs and the manuscript. SM and SG contributed equally by providing senior clinical guidance in neonatology and cardiac intensive care to support the content of the CPGs and manuscript. All authors agree to be held accountable for the content of the work and its publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41372_2025_2552_MOESM6_ESM.pdf (download PDF )
Supplemental Figure 6: Tetralogy of Fallot with Absent Pulmonary Valve Delivery Room Management Clinical Practice Guideline
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thomas, A.R., Geisser, D., Kaza, E. et al. Importance of clinical practice guidelines for specialized delivery room resuscitation of newborns with prenatally diagnosed critical congenital heart disease. J Perinatol 46, 491–494 (2026). https://doi.org/10.1038/s41372-025-02552-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41372-025-02552-2