Abstract
Objective
Substance use during pregnancy may affect fetal development and have implications for newborn, childhood, and life-long health. While standardized maternal screening protocols may reduce testing disparities, there are limited data describing the effects of standardized drug screening protocols on neonatal outcomes.
Study design
We conducted a retrospective cohort study of deliveries at a tertiary care center during pre-(7/1/2020–6/9/2021) and post-intervention(6/10/2021–5/31/2022) periods around implementation of a standardized obstetric substance use screening/testing protocol. Associations between neonatal testing and maternal race, ethnicity, and insurance status were assessed using generalized estimating equations (SAS 9.4,α = 0.05).
Result
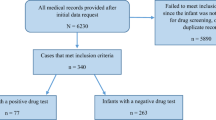
Among 3163 pre-intervention and 3389 post-intervention neonates, testing decreased post-intervention (4.5% vs. 6.2%, p = 0.0035), though a higher proportion lacked corresponding maternal tests (70.8% vs. 32.3%, p < 0.001). Black neonates had higher testing odds, which attenuated after adjusting for insurance status.
Conclusion
Use of a maternal screening protocol was associated with a reduction in neonatal testing, but disparities persist, warranting further study.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
SAMHSA. Substance Abuse and Mental Health Services Administration, Center for Behavioral Health Statistics and Quality, National Survey on Drug Use and Health, 2021 and 2022. (2022).
Behnke M, Smith VC. Prenatal substance abuse: short- and long-term effects on the exposed fetus. Pediatrics. 2013;131:e1009–24.
NIDA. Substance Use While Pregnant and Breastfeeding. National Institute on Drug Abuse. https://nida.nih.gov/publications/research-reports/substance-use-in-women/substance-use-while-pregnant-breastfeeding
Simpson WJ. A preliminary report on cigarette smoking and the incidence of prematurity. Am J Obstet Gynecol. 1957;73:807–15.
Persson PH, Grennert L, Gennser G, Kullander S. A study of smoking and pregnancy with special references to fetal growth. Acta Obstet Gynecol Scand Suppl. 1978;78:33–9.
Tiesler CM, Heinrich J. Prenatal nicotine exposure and child behavioural problems. Eur Child Adolesc Psychiatry. 2014;23:913–29.
Ernst M, Moolchan ET, Robinson ML. Behavioral and neural consequences of prenatal exposure to nicotine. J Am Acad Child Adolesc Psychiatry. 2001;40:630–41.
Patrick SW, Schumacher RE, Benneyworth BD, Krans EE, McAllister JM, Davis MM. Neonatal abstinence syndrome and associated health care expenditures: United States, 2000-2009. Jama. 2012;307:1934–40.
Ecker J, Abuhamad A, Hill W, Bailit J, Bateman BT, Berghella V, et al. Substance use disorders in pregnancy: clinical, ethical, and research imperatives of the opioid epidemic: a report of a joint workshop of the Society for Maternal-Fetal Medicine, American College of Obstetricians and Gynecologists, and American Society of Addiction Medicine. Am J Obstet Gynecol. 2019;221:B5–b28.
Jarlenski M, Shroff J, Terplan M, Roberts SCM, Brown-Podgorski B, Krans EE. Association of Race With Urine Toxicology Testing Among Pregnant Patients During Labor and Delivery. JAMA Health Forum. 2023;4:e230441.
Perlman NC, Cantonwine DE, Smith NA. Racial differences in indications for obstetrical toxicology testing and relationship of indications to test results. Am J Obstet Gynecol MFM. 2022;4:100453.
Winchester ML, Shahiri P, Boevers-Solverson E, Hartmann A, Ross M, Fitzgerald S, et al. Racial and Ethnic Differences in Urine Drug Screening on Labor and Delivery. Matern Child Health J. 2022;26:124–30.
Patel E, Bandara S, Saloner B, Stuart EA, Goodman D, Terplan M, et al. Heterogeneity in prenatal substance use screening despite universal screening recommendations: findings from the Pregnancy Risk Assessment Monitoring System, 2016-2018. Am J Obstet Gynecol MFM. 2021;3:100419.
Schoneich S, Plegue M, Waidley V, McCabe K, Wu J, Chandanabhumma PP, et al. Incidence of newborn drug testing and variations by birthing parent race and ethnicity before and after recreational cannabis legalization. JAMA Netw Open. 2023;6:e232058.
Peterson JA, Koelper NC, Curley C, Sonalkar SR, James AT. Reduction of racial disparities in urine drug testing after implementation of a standardized testing policy for pregnant patients. Am J Obstet Gynecol MFM. 2023;5:100913.
Gateway CWI. Plans of Safe Care: Infants With Prenatal Substance Exposure and Their Families – North Carolina. U.S. Department of Health and Human Services, Administration for Children and Families. Updated November 2024. 2025. https://www.childwelfare.gov/resources/plans-safe-care-infants-prenatal-substance-exposure-and-their-families-north-carolina/
Soos A, Plegue M, Darwiche A, Oshman L, Frank CJ. Racial disparities in newborn drug testing after implementation of question-based screening for prenatal substance use. Obstet Gynecol. 2024;144:233–40.
Drake B, Jones D, Kim H, Gyourko J, Garcia A, Barth RP, et al. Racial/ethnic differences in child protective services reporting, substantiation and placement, with comparison to non-CPS risks and outcomes: 2005–2019. Child Maltreat. 2023;28:683–99.
Roberts SC, Nuru-Jeter A. Universal screening for alcohol and drug use and racial disparities in child protective services reporting. J Behav Health Serv Res. 2012;39:3–16.
Acknowledgements
The Duke Biostatistics, Epidemiology, and Research Design (BERD) Methods Core’s support of this project was made possible in part by CTSA Grant (UL1TR002553) from the National Center for Advancing Translational Sciences (NCATS) of the National Institutes of Health (NIH), and the NIH Roadmap for Medical Research. The content is solely the responsibility of the authors and does not represent the official views of the National Institutes of Health.
Funding
This work was supported by the Charles B. Hammond Research Fund, Duke University School of Medicine, Durham, NC.
Author information
Authors and Affiliations
Contributions
ABB conceptualized the study, performed data collection and chart review, drafted the initial manuscript, and led manuscript revision and editing. SB, JC, and LF contributed to data collection and chart review. LS and TT conducted the statistical analysis and contributed to data interpretation. YM and LK provided critical review and revision of the manuscript. SMW and JJMC provided study supervision and contributed to manuscript editing. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations and the principles of the Declaration of Helsinki. This study was reviewed and approved by the Duke University Institutional Review Board (protocol number: Pro00109350). The requirement for informed consent was waived by the ethics committee due to the retrospective nature of the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baffoe-Bonnie, A.A., Bethell, S., Cato, J. et al. The impact of an obstetric substance use screening and biologic testing protocol on neonatal drug testing. J Perinatol (2026). https://doi.org/10.1038/s41372-026-02598-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41372-026-02598-w