Abstract
Objective
To assess current practices in the diagnosis, monitoring, and follow-up of neonatal AKI across Canadian tertiary neonatal units.
Study design
A seven-item electronic survey was distributed to members of the Canadian Neonatal Network between December 2023 and January 2024.
Results
Of the 32 units contacted, 26 (81.3%) responded. On-site nephrology consultation was available at 15/26 sites (57.7%). Only 5 sites (19.2%) actively tracked AKI incidence in infants born < 33 weeks’ gestation. Wide variation was observed in AKI definitions, monitoring practices, and follow-up protocols. Serum creatinine was measured prior to initiating non-steroidal anti-inflammatory drugs at 15 sites (57.7%), and 11 sites (42.3%) monitored creatinine when urine output fell below 0.5 mL/kg/hour.
Conclusions
Substantial variability exists in the identification and monitoring of neonatal AKI among Canadian NICUs. Standardized diagnostic criteria and follow-up strategies are needed to improve early detection and optimize long-term renal outcomes.
Similar content being viewed by others
Introduction
Acute kidney injury (AKI) affects approximately 30% of critically ill neonates in the neonatal intensive care unit (NICU) and is associated with increased mortality, prolonged hospitalization, and a higher risk of chronic kidney disease (CKD) among survivors [1, 2]. Preterm infants are particularly vulnerable to AKI due to ongoing nephrogenesis, which continues until approximately 36 weeks’ gestational age [3]. Additional risk factors in this population include hemodynamic instability, nosocomial infections, and exposure to nephrotoxic medications [4, 5].
International data reveal significant variability in how neonatal AKI is recognized and managed [6, 7]. In 2012, Jetton et al. proposed a modified AKI definition tailored for neonates, which contributed to the development of a neonatal-specific adaptation of the Kidney Disease: Improving Global Outcomes criteria [1]. This definition incorporates both serum creatinine levels and urine output and has been increasingly adopted as the consensus standard for AKI diagnosis and staging in neonatal research and clinical practice [8].
Timely identification of AKI is essential to enable early interventions and reduce modifiable risk exposures, particularly nephrotoxic medications. Systematic surveillance of high-risk infants receiving such agents has been advocated as an effective strategy to lower the incidence and long-term consequences of AKI [7, 9]. Despite growing awareness, there remains limited knowledge regarding how neonatal AKI is diagnosed, monitored, and managed in routine clinical practice, particularly in preterm infants. This study aims to assess current diagnostic and monitoring approaches to neonatal AKI across tertiary-level NICUs in Canada.
Methods
Study population and health care setting
This study was conducted across tertiary-level NICUs in Canada through the Canadian Neonatal Network (CNN). CNN encompasses all 32 tertiary NICUs nationwide, including one institution that operates two separate NICUs, and maintains a standardized national database for neonatal outcomes research, benchmarking, and quality improvement.
Canada’s perinatal care system is highly regionalized, with 17 of the 32 tertiary NICUs located within pediatric hospitals that offer on-site access to pediatric subspecialties. These centers also function as referral hubs for other NICUs requiring specialized pediatric services, including nephrology. Infants needing advanced renal support are transferred to these designated centers, where neonatal dialysis services are available in select institutions.
Survey development and content
The survey instrument was adapted from existing AKI-related questionnaires used in pediatric intensive care and oncology settings across Canada. A seven-item questionnaire (see Table 1) was co-developed by a neonatologist (JT) and a pediatric nephrologist (LD), with input from a multidisciplinary expert panel. The survey targeted diagnostic and management practices for infants born at < 33 weeks’ gestation and focused on three key domains:
-
1.
Criteria and definitions used for AKI diagnosis
-
2.
Monitoring practices, including serum creatinine levels and body weight
-
3.
Availability of on-site pediatric nephrology services and post-discharge follow-up for infants diagnosed with AKI
The survey comprised primarily multiple-choice questions. It was pilot-tested by four neonatologists and two nephrologists to ensure content clarity, clinical relevance, and usability. Revisions were made based on their feedback to improve face validity.
Survey distribution
The final survey was distributed electronically via REDCap to all CNN-affiliated NICUs. The survey was open from December 2023 to January 2024. Site leads (designated neonatologists) were invited to complete one response per site, reflecting practices in place during the 2023 calendar year. Informed consent was integrated into the REDCap platform and obtained prior to survey initiation. Ethical approval was granted by the University of Alberta Health Research Ethics Board (Pro00139191), and administrative approval was obtained from the CNN Executive Committee.
Statistical analysis
Survey responses were analyzed using descriptive statistics. Frequencies and percentages were calculated for each item, with denominators adjusted based on the number of responses received per question.
Results
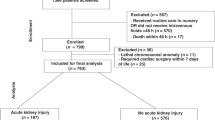
Of the 32 tertiary-level NICUs contacted, 26 (81.3%) completed the survey. On-site pediatric nephrology consultation was available at 15 of 26 sites (57.7%), and 8 sites (30.8%) offered on-site peritoneal dialysis services.
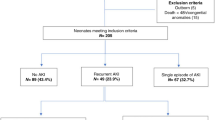
Only 5 sites (19.2%) reported tracking the incidence of AKI in infants born at < 33 weeks’ gestation. Among these, two sites used modified Kidney Disease: Improving Global Outcomes criteria, while two others relied exclusively on urine output thresholds: one using < 0.5 mL/kg/hour and another using < 1.0 mL/kg/hour, both measured over a 24 h period.
All 26 sites reported monitoring serum creatinine levels in preterm infants < 33 weeks’ gestation, though there was considerable variation in diagnostic criteria, monitoring protocols, and follow-up practices. Serum creatinine was assessed prior to the initiation of non-steroidal anti-inflammatory drugs (NSAIDs) at 15 sites (57.7%) and in response to oliguria (<0.5 mL/kg/hour) at 11 sites (42.3%). Additionally, 6 sites (23.1%) measured serum creatinine when aminoglycosides or vancomycin were prescribed for more than 48 h.
Daily body weight monitoring was routinely performed at 23 sites (88.5%), while 19 sites (73.1%) tracked daily fluid intake and output.
Post-discharge follow-up practices varied widely. Most sites arranged outpatient nephrology follow-up for infants with congenital renal anomalies (22/26, 84.6%), nephrocalcinosis (13/26, 50.0%), or hypertension requiring treatment (16/26, 61.5%). However, fewer sites (12/26, 46.2%) referred infants with AKI for nephrology evaluation if renal function had not normalized by the time of discharge.
Discussion
This national survey highlights significant variability in how AKI is identified, monitored, and followed up across Canadian tertiary-level NICUs. Despite the growing recognition of neonatal AKI as a contributor to short- and long-term morbidity, fewer than one-fifth of surveyed sites actively tracked AKI incidence among infants born before 33 weeks’ gestation. Substantial variations were found in serum creatinine monitoring. These findings reflect an important gap in surveillance and may hinder early intervention strategies aimed at mitigating renal injury and its sequelae.
Prevention remains the cornerstone of AKI management, with early recognition essential to avoiding fluid overload, limiting nephrotoxic exposure, and reducing the risk of progression. However, diagnostic challenges persist in the neonatal population. The traditional use of creatinine to estimate glomerular filtration rate (GFR) is unreliable in preterm infants due to its tubular reabsorption by immature kidneys and its dependence on muscle mass as an endogenous marker [10]. Early postnatal serum creatinine levels within first 24–48 h after birth often reflect maternal values, which complicates interpretation, particularly among mothers with pre-eclampsia and renal dysfunction, a common cause of preterm delivery [11]. Creatinine levels often rise markedly during the first 48 h of life, particularly in infants born at less than 30 weeks gestation, and therefore even substantial early increases should not be interpreted in isolation as evidence of renal failure [12]. Urine output is a recognized predictor of mortality in the NICU; however, its measurement is often unreliable in the preterm population, particularly among extremely low gestational age infants [13]. Immature nephrons have a poor capability to concentrate urine leading to free water clearance and the development of polyuria, even in the context of intravascular volume depletion [14]. Modified AKI definitions that include higher urine output thresholds (e.g., ≥1.5–2 mL/kg/hour) have shown improved predictive value for mortality in neonates [15].
Our findings reveal inconsistency in serum creatinine monitoring, especially in the context of nephrotoxic medication exposure. While many sites reported measuring creatinine prior to initiating NSAIDs, routine monitoring during treatment was often lacking. This is concerning given that NSAID-associated AKI has been reported in up to 25% of preterm infants receiving prophylactic or therapeutic treatment for patent ductus arteriosus [5]. Similarly, data from the AWAKEN study identified aminoglycosides, particularly when used in combination with other nephrotoxic agents, as independent risk factors for early AKI [16]. The same study found a direct correlation between the frequency of serum creatinine measurement and the observed incidence of AKI, suggesting that underdiagnosis may result from inconsistent surveillance practices.
Nearly one-quarter of NICUs in our survey did not routinely monitor daily fluid input and output. Given the established association between positive fluid balance in the first 3–10 postnatal days and adverse outcomes including bronchopulmonary dysplasia, prolonged mechanical ventilation, and mortality [17, 18]. This represents another target for quality improvement initiatives. Recent evidence suggests that fluid overload prevention bundles may improve short-term outcomes, such as increased ventilator- and oxygen-free days and reduced NICU length of stay [19].
In the longer term, the renal consequences of preterm birth are increasingly recognized [20, 21]. Epidemiological data, including a national Swedish cohort study, indicate that individuals born before 37 weeks’ gestation face nearly double the lifetime risk of CKD [22]. A meta-analysis demonstrated that preterm birth is associated with reduced glomerular filtration rate, increased albuminuria, reduced renal mass, and elevated blood pressure later in life [23]. Our survey found wide variation in follow-up practices, with fewer than half of NICUs routinely referring infants with unresolved AKI at discharge for nephrology evaluation. A newly published international consensus statement on neonatal kidney health recommends that infants born between 28- and 34-weeks’ gestation who develop AKI should be considered at high risk for CKD and receive structured follow-up, including blood pressure screening and caregiver education by two years of age [24]. Our survey highlights a significant practice gap in the routine assessment of renal function during the long-term follow-up of prematurely born infants.
A key strength of our study is its high response rate, with 81.3% of Canadian tertiary NICUs participating. To our knowledge, this is the first national survey of neonatal AKI practices in Canada, providing valuable insights into current diagnostic, monitoring, and follow-up strategies for infants born before 33 weeks’ gestation. Nonetheless, several limitations must be acknowledged. First, the survey captured site-level responses, which may not fully reflect bedside variability or inter-provider differences within units. We did not specifically inquire about the method used to calculate GFR or the availability of cystatin C across different centers. Second, as the survey targeted only tertiary-level NICUs, our findings may not generalize to level II centers, where a substantial number of preterm infants, particularly those ≥30 weeks’ gestation, receive care. Third, the survey did not capture specific details regarding the provision of hemodialysis or continuous renal replacement therapy at individual centers. Lastly, our study did not assess AKI practices related to infants with congenital heart disease or those undergoing cardiac surgery, who are typically managed outside the NICU environment.
In conclusion, we identified considerable variability in the diagnosis, monitoring, and follow-up of neonatal AKI across tertiary-level NICUs in Canada. These findings underscore an urgent need for the development and implementation of standardized clinical approaches to support early and consistent AKI definition, timely recognition, routine surveillance, and structured long-term follow-up. Establishing such practices is essential to mitigate the risk of adverse renal outcomes in this vulnerable population of preterm infants.
Data availability
The dataset supporting the findings of this study consists of survey responses from individual sites. Aggregate results are presented within the manuscript, and additional anonymized summary data may be made available upon reasonable request to the corresponding author, subject to institutional and network data-sharing policies.
References
Jetton JG, Boohaker LJ, Sethi SK, Wazir S, Rohatgi S, Soranno DE, et al. Incidence and outcomes of neonatal acute kidney injury (AWAKEN): a multicentre, multinational, observational cohort study. Lancet Child Adolesc Health. 2017;1:184–94.
Harer MW, Pope CF, Conaway MR, Charlton JR. Follow-up of Acute kidney injury in Neonates during Childhood Years (FANCY): a prospective cohort study. Pediatr Nephrol. 2017;32:1067–76.
Nada A, Bonachea EM, Askenazi DJ. Acute kidney injury in the fetus and neonate. Semin Fetal Neonatal Med. 2017;22:90–7.
Rhone ET, Carmody JB, Swanson JR, Charlton JR. Nephrotoxic medication exposure in very low birth weight infants. J Matern Fetal Neonatal Med. 2014;27:1485–90.
Ting JY, McDougal K, De Mello A, Kwan E, Mammen C. Acute kidney injury among preterm infants receiving nonsteroidal anti-inflammatory drugs: A pilot study. Pediatrics Neonatol. 2023;64:313–8.
Feeney A, Slagle CL, Harer MW, Charlton JR, Mohamed T, Askenazi DJ, et al. Approaches to neonatal acute kidney injury consultation and follow-up: results of a provider survey. J Perinatol. 2025;45:397–8.
Goldstein SL, Kirkendall E, Nguyen H, Schaffzin JK, Bucuvalas J, Bracke T, et al. Electronic health record identification of nephrotoxin exposure and associated acute kidney injury. Pediatrics. 2013;132:e756–67.
Coleman C, Tambay Perez A, Selewski DT, Steflik HJ. Neonatal Acute Kidney Injury. Front Pediatr. 2022;10:842544.
Goldstein SL, Mottes T, Simpson K, Barclay C, Muething S, Haslam DB, et al. A sustained quality improvement program reduces nephrotoxic medication-associated acute kidney injury. Kidney Int. 2016;90:212–21.
Abitbol CL, DeFreitas MJ, Strauss J. Assessment of kidney function in preterm infants: lifelong implications. Pediatr Nephrol. 2016;31:2213–22.
Martini C, Saeed Z, Simeone P, Palma S, Ricci M, Arata A, et al. Preeclampsia: Insights into pathophysiological mechanisms and preventive strategies. Am J Prev Cardiol. 2025;23:101054.
Miall LS, Henderson MJ, Turner AJ, Brownlee KG, Brocklebank JT, Newell SJ, et al. Plasma creatinine rises dramatically in the first 48 h of life in preterm infants. Pediatrics. 1999;104:e76.
Bezerra CT, Vaz Cunha LC, Libório AB. Defining reduced urine output in neonatal ICU: importance for mortality and acute kidney injury classification. Nephrol Dial Transpl. 2013;28:901–9.
Branagan A, Costigan CS, Stack M, Slagle C, Molloy EJ. Management of Acute Kidney Injury in Extremely Low Birth Weight Infants. Front Pediatr. 2022;10:867715.
De Mul A, Parvex P, Heneau A, Biran V, Poncet A, Baud O, et al. Urine Output Monitoring for the Diagnosis of Early-Onset Acute Kidney Injury in Very Preterm Infants. Clin J Am Soc Nephrology: CJASN. 2022;17:949–56.
Steflik HJ, Charlton JR, Briley M, Selewski DT, Gist KM, Hanna MH, et al. Neonatal nephrotoxic medication exposure and early acute kidney injury: results from the AWAKEN study. J Perinatol. 2023;43:1029–37.
Selewski DT, Gist KM, Nathan AT, Goldstein SL, Boohaker LJ, Akcan-Arikan A, et al. The impact of fluid balance on outcomes in premature neonates: a report from the AWAKEN study group. Pediatr Res. 2020;87:550–7.
Weaver LJ, Travers CP, Ambalavanan N, Askenazi D. Neonatal fluid overload-ignorance is no longer bliss. Pediatr Nephrol. 2023;38:47–60.
Askenazi DJ, Gordon L, Griffin R, Collins M, Black A, Ambalavanan N, et al. Reducing NICU ventilator days by preventing fluid overload with the CAN-U-P-LOTS standardized bundle. Pediatr Res. 2025. https://doi.org/10.1038/s41390-025-04078-x.
Gette F, Aziz Ali S, Ho MSP, Richter LL, Chan ES, Yang CL, et al. Long-term health outcomes of preterm birth: a narrative review. Front Pediatr. 2025;13:1565897.
Richter LL, Shen Y, Lisonkova S, Bone JN, Albert A, Ho MSP, et al. Preterm Birth and Risk of Health Service Use, Morbidity, and Medication Needs at 5 Years. JAMA Pediatr. 2025;179:1172–82.
Crump C, Sundquist J, Winkleby MA, Sundquist K. Preterm birth and risk of chronic kidney disease from childhood into mid-adulthood: national cohort study. Bmj. 2019;365:l1346.
Heo JS, Lee JM. The Long-Term Effect of Preterm Birth on Renal Function: A Meta-Analysis. Int J Environ Res Public Health. 2021;18:2951.
Starr MC, Harer MW, Steflik HJ, Gorga S, Ambalavanan N, Beck TM, et al. Kidney Health Monitoring in Neonatal Intensive Care Unit Graduates: A Modified Delphi Consensus Statement. JAMA Netw Open. 2024;7:e2435043.
Acknowledgements
We are thankful to Ms. Celine Balay and Ms. Mimi Kuan for their support in this study. We are also thankful to all Site Investigators and abstractors of the Canadian Neonatal Network (CNN).
Funding
Dr. Joseph Y. Ting is supported by startup funding from the Women’s and Children’s Health Research Institute, and by endowed funding from Variety, the Children’s Charity, and the Stollery Children’s Hospital Foundation Pediatric Clinical Research Professorship at the University of Alberta.
Author information
Authors and Affiliations
Consortia
Contributions
JYT, KSL, CMa, and CMo conceptualized and designed the study. JYT and LD designed the questionnaire and analyzed the data. All authors interpreted the data. AH and JYT drafted the initial manuscript. All authors critically reviewed and revised the manuscript for important intellectual content.
Corresponding author
Ethics declarations
Competing interests
The authors have no conflicts of interest to declare. The study was conducted in accordance with the Declaration of Helsinki and approved by the University of Alberta Health Research Ethics Board (Pro00139191).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hussein, A., Ding, L., Lee, KS. et al. Neonatal acute kidney injury: a survey of practices in Canadian neonatal intensive care units. J Perinatol (2026). https://doi.org/10.1038/s41372-026-02635-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41372-026-02635-8