Abstract
Luminescent metal halides are attracting growing attention as scintillators for X-ray imaging in safety inspection, medical diagnosis, etc. Here we present brand-new hybrid Eu(II)-bromide scintillators, 1D type [Et4N]EuBr3·MeOH and 0D type [Me4N]6Eu5Br16·MeOH, with spin-allowed 5d-4f bandgap transition emission toward simplified carrier transport during scintillation process. The 1D/0D structures with edge/face -sharing [EuBr6]4− octahedra further contribute to lowing bandgaps and enhancing quantum confinement effect, enabling efficient scintillation performance (light yield ~73100 ± 800 Ph MeV−1, detect limit ~18.6 nGy s−1, X-ray afterglow ~ 1% @ 9.6 μs). We demonstrate the X-ray imaging with 27.3 lp mm−1 resolution by embedding Eu(II)-based scintillators into AAO film. Our results create the new family of low-dimensional rare-earth-based halides for scintillation and related optoelectronic applications.
Similar content being viewed by others
Introduction
Scintillators, producing visible photons by transforming high-energy X-rays, have been widely applied in scientific research, safety inspection, medical diagnosis, etc1,2. In the last decades, solution-processed scintillating materials, e.g., metal-organic frameworks3,4, organic molecules5,6,7, metal halide perovskites2,8,9, and hybrid metal halides (HMHs)10,11,12, have emerged as promising alternatives to conventional inorganic scintillators due to the characteristics of low-cost and easy-processing. In particular, HMHs with metal-halide polyhedrons isolated by organic cations possess inherent merits in terms of tunable structure determination and luminescence, effective radioluminescence, and heavy-atom (metals and halides) for X-ray absorption13. However, most of the present HMH scintillators are difficult to meet requirements for the state-of-the-art X-ray imaging, such as Mn(II)/Cu(I)-based HMHs (long decay time) and Pb(II)-based compounds (environmental toxicity)14,15,16,17. Thus, the exploration of novel HMH scintillators that are highly sensitive to X-rays is of high importance.
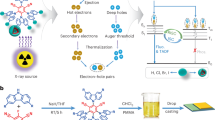
Radioluminescence of scintillators generally undergo three stages18,19,20. First, the large amounts of electron-hole pairs are generated under X-ray radiation interacting with the heavy atoms in scintillators. Second, the charge carriers transport to defects or the bottom of the conduction band (CB) and the top of the valence band (VB), and captured at the luminescence centers (LCs) of scintillators, such as rare-earth Eu2+ 4f-5d (Scheme 1a). Last, radiative recombination of captured carriers at the LCs generates visible radioluminescence. Accordingly, it can be extrapolated that three elements induce efficient radioluminescence, i.e., heavy atoms, few defects and low energy level D-value between CB/VB and LCs, and high luminescent efficiency. These factors are mainly dominated by regulating appropriate B-site ions, e.g., rare-earth ions (Ce3+ and Eu2+). However, the Eu-based halide scintillators generally possess higher light yield for Ce-based counterparts, such as Cs4EuBr6: 78000 Ph MeV−1, Cs3CeBr6: 28000 Ph MeV−1 21,22. Moreover, the energy bandgaps (EEu2+ free = 4.216 eV) of free ions (Eu2+ 5d-4f) is much smaller than that (ECe3+ free = 6.118 eV) of Ce3+ 5d-4f23, which enhances the feasibility of synthesizing Eu2+-based small bandgap hybrid materials and obtaining higher light yield, as shown in Scheme 1b. Therefore, we reason the Eu2+ ions that can be considered as B-site ones to design brand-new HMH scintillators based on their heavy atom effectivity and spin-allowed 5d-4f bandgap emission for efficient carrier transport (Scheme 1c). Additionally, reduced molecular dimensionality in HMHs could be employed to enhance luminescent efficiency due to the localized charge carriers (Scheme 1c)24,25. From optical imaging perspective, in addition, various large-area HMH scintillators were explored, i.e., nanocrystal thin films, thick films, hybrid film (mixture of HMH and polydimethylsiloxane), and transparent ceramics/glasses10,11,26. However, the abovementioned forms remain challenging for durability or poor optical transparency/output. Combining with solution processability of HMHs, their AAO films are thought to generate optical waveguide effect and thus enhance X-ray imaging quality. Despite several Eu2+-doped/-based scintillators, such as Cs4EuBr6, SrI2:Eu2+, LiI: Eu2+ and BaCl2:Eu2+, have been reported22,27,28,29, the synthesis conditions are challenging, including high-cost, time-consuming, high-temperature, and vacuum/high-pressure sintering processes. Thus, the solution processability of rare-earth-based scintillators combining highly sensitive to X-rays is an alternative way.
a The scintillation process of traditional rare-earth doped scintillators. b The energy bandgaps free ions Eu2+ and Ce3+. c Mechanism illustrations and rational design of RE-based scintillators with efficient 5d-4f bandgap emission
Here we demonstrate an example of Eu(II)-based scintillators from adjusting molecular dimensionality and coordination environment in 3D CsEuBr3 (corner-shared octahedra) and discovered two low-dimensional Eu(II)-based scintillators (one is 1D type [Et4N]EuBr3·MeOH (EtEu) with edge-shared octahedra; the other is 0D type [Me4N]6Eu5Br16·MeOH (MeEu) with face-shared octahedra). The scintillation of MeEu shows great light yield of ~73100 ± 800 Ph MeV−1, low detection limit of 18.6 nGy s−1, weak X-ray afterglow of ~1% @ 9.6 μs. The efficient scintillation performance is originated from 4f-5d bandgap emission, lowing bandgaps and enhancing quantum confinement effect. Furthermore, X-ray scintillation imaging by embedding Eu(II)-based scintillators into AAO film exhibits ultrahigh spatial resolution of ca. 27.3 lp mm−1. These findings provide valuable guidelines for designing novel HMH scintillators with high sensitivity and low cost.
Results
Design and structural characteristics
As a conceptual design, we selected tetramethylammonium bromide (Me4NBr) and tetraethylammonium bromide (Et4NBr) as organic cations due to good processability. The anhydrous MeOH with high polarity of ca. 6.6 is able to dissolve Me4NBr/Et4NBr and EuBr2 to form a clear solution and safeguard B-site Eu(II) from oxidation30, as shown in Fig. S1. The colorless and viridescent single crystals can be easily obtained with dissolvent volatilization and emit bright blue and cyan emission under ultraviolet (UV) irradiation, respectively (Fig. S2). The details of the synthesis process are depicted in the Methods section. Scanning-electron-microscope (SEM) images of as-prepared crystals reveal high crystallization quality, and energy-dispersive-spectrometer (EDS) images with homogenous distribution of N, Eu and Br elements confirm that we synthetize new hybrid Eu(II) halides (Fig. S3). Considering the solution processability of these materials, the large-area scintillation screens can be facilely fabricated enabling X-ray imaging.
We measured single crystal X-ray diffraction (SCXRD) to gain crystal complementary data. Detailed crystal cell parameters summarized in Tables S1–S3. Compared with 3D structure of CsEuBr3 (Fig. 1a), crystal of [Et4N]EuBr3·MeOH (EtEu) posseses one-dimensional (1D) chain structure isolated by Et4N+ cations (Fig. 1b), whereas [Me4N]6Eu5Br16·MeOH (MeEu) only contains zero-dimensional (0D) [EuBr6]4− polyhedron surrounded by Me4N+ cations (Fig. 2c). Notably, the strong quantum confinement from reduced structural dimensionality is able to enhance luminescence efficiency24. The high phase-purity of EtEu and MeEu can be further approved by Powder X-ray diffraction (PXRD) patterns (Fig. S4). The XPS spectrum is employed to demonstrate the testify divalent valence state of Eu ions in EtEu and MeEu hybrids (Fig. S5)31,32. In particular, coordination environment of Eu(II)-Br transfer from corner-shared to edge-shared octahedra, and even face-shared octahedra with structural dimensionality decreasing from 3D to 0D (Fig. 1d, e). The differences in local coordination could affect bandgap structure33,34. The Eu–Eu metallic bonding in face-shared motifs (3.9379–3.9768 Å) is obviously shorter than ones in edge/corner-shared clusters (5.892 Å and 4.4867 Å). The face/edge-shared structures, with stronger couple of [EuBr6]4− polyhedrons, exhibits good structural stability and low trap states34,35,36. The distortion levels of [EuBr6]4– octahedra are calculated and summarized in Fig. 1g. The face/edge-shared [EuBr6]4– octahedra reveals higher distortion level from the bond length distortion and bond angle variance, which is similar to the tendency between distortion level and connectivity modes in the 2D lead bromide perovskites33. Meanwhile, we also observe that the MeOH molecules coordinate to Eu in [EuBr5·MeOH]3– octahedra, resulting in locally collective hydrogen bonding. By hydrogen bonding comparison, EtEu process stronger hydrogen bonding effect along the direction perpendicular to the 1D chains, preserving 1D structure (Fig. 1h). However, the chain structure is destroyed by the steric effects of organic cation in MeEu without muscular hydrogen bonding, forming 0D clusters (Fig. 1i)37. The forementioned results indicate that EtEu and MeEu hybrids owns quantum confinement, structural stability and decreasing trap states, contributing to enhanced luminescent properties.
Structural characteristics of Eu(II)-based hybrids. Crystal structures of a 3D CsEuBr3, b 1D EtEu and c 0D MeEu hybrids. Local connection pattern of [EuBr6]4− octahedrons with d corner-sharing for CsEuBr3, e edge-sharing for 1D EtEu, and f face-sharing for 0D MeEu. g Calculated bond length distortion of bond angle variance of the compounds. Highlights of the hydrogen bonds marked in red dashed line between organic cations and MeOH in the h 1D EtEu and i 0D MeEu hybrids
Photoluminescence/radioluminescence characterizations of Eu(II)-based halides. a Normalized PL and PLE spectra at room temperature of CsEuBr3, EtEu and MeEu halides. b PL decay of CsEuBr3, EtEu and MeEu halides as a function of time recorded at the PL maximum peaks under 340 nm pulsed excitation. c ΦIQE and ΦEQE of CsEuBr3,EtEu and MeEu halides, and the ΦEQE is equal to the ΦIQE multiplied by the absorption coefficient. d X-ray absorption spectra of CsEuBr3, EtEu and MeEu halides and commercial LuAG:Ce3+ scintillator. e Calculated X-ray attenuation efficiencies of CsEuBr3, EtEu and MeEu halides and LuAG:Ce3+ versus thickness at photon energy of 17.5 keV. f RL intensity spectra CsEuBr3, EtEu and MeEu halides and LuAG:Ce3+ with normalized to the X-ray attenuation efficiencies at the thickness of 500 μm (X-ray tube voltage, 50 kV; dose rate, 1.503 mGy s−1). g Detection limits under low-dose-rate X-ray excitation at SNR of 3. h The afterglow of commercial CsI:Tl, EtEu and MeEu halides under X-ray off. i Normalized RL intensities of 3D CsEuBr3, 1D EtEu and 0D MeEu halides a total dose of 8.4 Gy during X-ray irradiation
Photoluminescence/radioluminescence properties
Figure 2a displays the steady-state photoluminescence (PL) and excitation (PLE) spectra of 3D CsEuBr3, 1D EtEu and 0D MeEu hybrids, respectively, with detailed PL parameters summarized in Table S4. Remarkably, under UV excitation from 300 to 400 nm (Fig. S6a), 3D CsEuBr3 shows narrow-blue emission peaking at 445 nm with full width at half maximum (FWHM) approximately 26 nm. Similarly, 1D EtEu also produces narrow-blue emission (FWHM ~ 24 nm) peak at 455 nm when excited by UV from 270 to 400 nm (Fig. S6b), while 0D MeEu hybrids exhibit narrow-cyan photoluminescence (FWHM ~ 40 nm) centered at 500 nm with broad PLE band ranging from 300 to 445 nm (Fig. S6c). The emission redshift of MeEu hybrids with higher Stokes shift (ΔS(A)) is contributed by higher Huang−Rhys (HR) factor ~6.58 (Fig. S7)38. Following Dorenbos’ semiempirical model39,40, the Eabs and Eem of CsEuBr3, EtEu and MeEu hybrids were calculated and almost consistent with experimental results (Table S5). Apparently, the emissions of the designed HMHs are originated from the 4f–5d transitions of Eu(II). The luminescence decays at the emission maximum as bi/mono-exponential functions for 3D, 1D EtEu and 0D MeEu with short lifetime of 953.2 ns, 387.1 ns, and 368.2 ns (Fig. 2b), respectively. The decreasing luminescence lifetime is mainly attributed to shorter Eu-Eu distances (dEu-Eu (CsEuBr3) ~ 5.892 Å, dEu-Eu (EtEu) = 4.4867 Å, dEu-Eu (MeEu) ~ 3.9379–3.9768 Å)41,42. The typical PLE, PL and PL lifetime spectra indicate their emission is indeed originated from the spin-allowed 4f–5d transitions of Eu(II)43. It should be pointed out that the short decaying lifetime in 1D EtEu and 0D MeEu hybrids is great significance for fast X-ray scintillation imaging. Impressively, photoluminescent internal/external quantum efficiency (ΦIQE and ΦEQE) gradually increase from 78%/43% to 97%/61% accompanied with reduced molecular dimensionality (Fig. 2c and Table S6). Moreover, the energy barriers of thermal quenching ΔE of CsEuBr3, EtEu and MeEu hybrids are at the same level as most Eu2+ doped inorganic phosphors (Fig. S8), meaning a good thermal stability44. As the thermal stability of MeEu remained above 80% at 100 °C (Fig. S9), thanks to its higher ΔE value and higher structural stability (more rigid face-sharing local structure and organic cations isolated 0D structure). Strikingly, the enhanced quantum confinement in low-dimensional hybrid gives rise to the higher ΦIQE and ΦEQE, which are also in favor of efficient scintillation performance.
It is noteworthy that the X-ray absorption coefficients of [EuBr6]4− octahedra (Z = 35 and Kα = 13.475 keV for bromine; Z = 63 and Kα = 48.515 keV for europium) is far outweigh than those of organic cations (Z = 1–8, Kα = 0.0136–0.531 keV for C, H, N and O). As a result, 3D, 1D and 0D Eu(II) bromides show identical resonant absorption edges, even commercial scintillator LuAG:Ce (Fig. 2d). To investigate the radioluminescence (RL) properties, we compare X-ray attenuation efficiency vs sample thicknesses (Fig. 2e). The difference of X-ray attenuation efficiency is related to the densities of 3D CsEuBr3, 1D EtEu, and 0D MeEu hybrids with 4.31 g cm−3, 2.19 g cm−3, and 2.51 g cm−3, respectively. Moreover, we evaluate light yields (LY) of X-ray to visible photons (Figs. 2f), and 1D EtEu and 0D MeEu exhibit high LY of ~ 73100 ± 800 and ~ 32200 ± 700 ph MeV−1, all of which evidently outperform those of 3D CsEuBr3 (21900 ± 300 ph MeV−1) and LuAG:Ce (25000 ph MeV−1), related to their high luminescent efficiencies and low optical bandgaps. The low detection limit of 18.6 nGy s−1 in 0D MeEu (Fig. 2g), <0.5% of X-ray diagnostic dosage of 5.5 μGy s−1 45, is inferred by linear dependence of RL intensity on X-ray dose rate at signal-to-noise ratio (SNR) ~ 3 (Fig. S10). As-prepared Eu(II) scintillators reveal superiority in X-ray afterglow respect. As shown in Fig. 2h, the afterglow of 1D EtEu and 0D MeEu is 1% @12.5, 9.6 μs, respectively, which is superior to the commercial scintillator CsI:Tl (1.5% @ 3.1 ms)46,47. In addition, the X-ray fatigue stability is another important limiting factor of metal halide scintillators. Figure 2i illustrates the RL intensity of Eu(II)-based halides exposure at a total dose of 8.4 Gy, demonstrating the superb stability. Consequently, it can be concluded that the 0D MeEu process transcendent X-ray scintillation performance, propelling the favorable for X-ray imaging devices by solution processed scintillation screens.
Modified radioluminescence mechanism
Generally, the scintillation performance is determined by bandgap (Eg) of the scintillator, and quantum efficiencies in the transfer (S) and luminescence stages (Q) as follows48:
where β is a phenomenological parameter (2 ~ 3). With the goal of revealing origination of efficient luminescence, we first turned to studying the exciton binding energy Eb by Arrhenius fitting from temperature-dependent PL (Fig. S11)49.
where I0 is the intensity at 0 K and kB is the Boltzmann constant. It was found that MeEu halide displayed a larger value (216 meV) of the Eb than those (132 meV and 53 meV) of other two halides (Fig. 3a), manifesting the effect of quantum confinement in effectively enhancing luminescence. This trend matches well with their corresponding scintillation performance. Typically, the defect states in metal halides originate from the vacancies of halide. Thus, we used electron paramagnetic resonance (EPR) measurement (Fig. 3b) to estimate the concentration of Br vacancy (VBr)50,51. The three samples show lower a characteristic signal for g-factor of 2.003 than that of (PA)4AgBiBr8 and CsPbBr3, reflecting lesser defect states for as-prepared Eu-based halides. The lower defect states (VBr) could be further corroborated by shifting higher binding energy of Br 3d5/2 and 3d3/2 in Eu-based halides (Fig. 3c)52,53, and no spectral signal at thermoluminescence measurement (Fig. S12)54. The lower The defect states contribute to short X-ray afterglow performance55. This observation could be attributed to the more rigid local structure (face-sharing > edge-sharing > corner-sharing).
Mechanism of scintillation in Eu-based halides. a Fitting results of the integrated PL intensity–temperature dependence of CsEuBr3, EtEu and MeEu halides. b EPR spectra, and c XPS spectra Br-3d core level of CsEuBr3, EtEu and MeEu halides. d The plots of (αhv)2 and PL versus the light energy of CsEuBr3, EtEu and MeEu hybrids. e The density of states (DOS) plots of MeEu. f Partial density contours in the VB and CB for MeEu. g Proposed mechanism of radioluminescence for Eu-based halides
The optical bandgaps were calculated from UV-vis diffuse reflectance spectra (Fig. 3d) with the values of 2.77 eV for CsEuBr3, 2.66 eV for EtEu, 2.54 eV for MeEu, indicating improved probability of captured electron-hole pairs carriers under X-ray radiation19. Additionally, we also observed good matching at energy between absorption edge and PL emission at room temperature (Fig. 3d), which describes band-to-band (5d-4f) recombination in these Eu(II)-based halides56. To gain further insight into luminescent mechanism of 3D CsEuBr3, 1D EtEu and 0D MeEu, we performed density functional theory (DFT) calculations on the electronic configurations. The direct bandgap can be observed in all systems with ca. 3.39 for 3D CsEuBr3, 3.10 for 1D EtEu and 2.95 eV for 0D MeEu (Fig. S13), which is consistent with results of absorption data. The dispersive bands and smaller band gap in 1D edge/0D face-shared structures in comparation with 3D corner-ones clearly illustrates that the molecular dimensionality and connectivity of the octahedra is impacted on the electronic structure33,34. The projected density of states (DOS) shows that valence band (VB) is mainly contributed by the 4 f orbitals of Eu(II), whereas the conductive band (CB) is derived from 5d orbitals of Eu(II) (Fig. 3e and Fig. S14). There is no contribution from the Br 4p or organic cations to the band edges. Similar results can be observed in the electronic states (Fig. 3f and Fig. S15). We also found that the electronic states were localized on the Eu(II) units isolated by organic cations in low-dimensional Eu-based halides (Fig. 3f), indicating reinforced quantum confinement effect. The emission, therefore, was attributed to 5d-4f electronic transitions of Eu(II). These results, heavy atoms (Eu), low defect states, and smaller band gap with efficient 5d-4f bandgap luminescence, contribute to vibrant radioluminescence. Thus, radioluminescence of Eu-based scintillators contains the following three stages in a modified process (Fig. 3g): (1) conversion stage, heavy atoms (Eu) under X-ray radiation generate a large of electron-hole pairs; (2) transport stage, the charge carriers mainly migrate to the conduction band (CB) Eu2+-5d and valence band (VB) Eu2+-4f without being captured at detect states (traps). (3) luminescence stage, excitonic radiative recombination at band-to-band (5d-4f) generates efficient radioluminescence.
X-ray imaging with solution processability
Inspired by the promising scintillation properties of 0D MeEu halide, we further investigated the utility for X-ray imaging applications. To obtain large-area scintillation film, we take advantage of characteristics of solution processed for MeEu scintillator to incorporate into two-pass AAO matrix templates (Fig. 4a). The AAO@MeEu scintillation film possess higher transparent at top view than that at tilting 45° view (Fig. 4b). The formation of the optical waveguide effect reduces optical scattering at lateral surface to enhance X-ray imaging resolution. And the waveguide structure could be further corroborated by the SEM image of AAO@MeEu cross-section (Fig. 4d) compared to ones of AAO matrix templates (Fig. 4c). Besides, the EDS images with uniformly distributed Eu, Br, and N elements (Fig. 4e) form red box in Fig. 4d reveal that the MeEu is homogeneously embedded into AAO matrix templates.
X-ray imaging of AAO@MeEu scintillation film. a Schematic diagram of AAO@MeEu scintillation film by the suction crystallization method. b Photographs of AAO@MeEu scintillation film at top view and tilting 45° view. c The cross-sectional SEM image of AAO film. d The cross-sectional SEM image of AAO@MeEu scintillation film. e EDS images of Eu, Br, and N elements from the element Mapping scanning in the red dashed box of d. Bright-field and X-ray imaging of f longicorn specimen, g chip drive, and h jaffeite bottle iron knife aluminum pot hiding into plastic box. i Multi-color imaging through MATLAB simulation. j X-ray images of a copper mesh (diameter: 100 μm) using AAO@MeEu and PDMS@MeEu scintillation films. k X-ray imaging of partial region of the standard X-ray test pattern based on AAO@MeEu. l Modulation transfer function curves of the AAO@MeEu and PDMS@MeEu scintillation films measured by the slanted-edge method
A home-built X-ray imaging system integrating with X-ray source, imaging object, reflector, and COMS camera, is employed to perform X-ray imaging (Fig. S16). We demonstrate X-ray imaging with inserting a metal needle (diameter: 100 μm) into the body and clear joint structures of longicorn specimen under rate dose of 0.78 μGy s−1 (Fig. 4f), 60 times lower than X-ray diagnostic dosage of 5.5 μGy s−1 45. The imaging easily distinguished interior circuit of a chip drive, as shown in Fig. 4g, emphasizing capacity of AAO@MeEu film in industrial inspection. Moreover, we prepared three objects, i.e., jaffeite bottle iron knife aluminum pot, to hide into plastic box (Fig. 4h), to evaluate potential of AAO@MeEu film for application in security. Clear imaging accompanied by different grayscale values can be observed for objects of different materials. Furthermore, the types of materials can be further defined by color depth through MATLAB simulation (Fig. 4i). Thanks to the low X-ray afterglow performance, the AAO@MeEu film exhibit no imaging ghost in dynamic X-ray imaging compared to CsI:Tl scintillators, as shown in Fig. S17 and Video S1. In order to appraise superiority of AAO@MeEu film in imaging resolution, the mixed film (PDMS@MeEu) by incorporating the MeEu into a polydimethylsiloxane (PDMS) was prepared as a comparison. As expected, AAO@MeEu film manifest more distinct imaging of a tiny copper hexagonal mesh (diameter: 100 μm, Fig S18) than that using PDMS@ MeEu (Fig. 4j). The X-ray imaging spatial resolutions of two films are defined by a standard line-pair card (TYPE 39 b) (Fig. S19). X-ray imaging employed AAO@MeEu demonstrates spatial resolution up to ~ 28 lp mm−1 (Fig. 4k), outclassing ~ 16.5 lp mm−1 of PDMS@ MeEu (Fig. S20). Additionally, we measured X-ray imaging using coupling MeEu in AAO thin films with high transparency glass substrates (thickness ~ 1 mm) and a silica template with capillary microholes (thickness ~ 1 mm)57. As shown in Fig. S21, X-ray imaging employed AAO@MeEu with glass substrates demonstrates spatial resolution up to ~ 27 lp mm−1, which is almost consistent with a single AAO@MeEu film, superior to ~20 lp mm−1 of MeEu with a silica template. As shown in Fig. 4l, the spatial resolution results can be further quantified by calculating the modulation transfer function (MTF) using slanted-edge method58,59. AAO@MeEu owns higher spatial resolution of ~ 27.3 lp mm−1 at MTF = 0.2, which is superior to the commercial CsI:Tl ~ 10 lp mm−1 and competitive to those of reported most metal halide scintillators (Table S7).
Discussion
In conclusion, we combined the B-site screening and molecular dimensional engineering for developing rare-earth-based halide scintillators for sensitive X-ray detection. As expected, low dimensional Eu(II)-based halides demonstrated edge-sharing for 1D and face-sharing for 0D structures, respectively. The efficient spin-allowed 5d-4f bandgap emission, lowing bandgaps and enhancing quantum confinement effect lead to the highly enhanced scintillation performance, i.e., light yield ~ 73100 ± 800 Ph MeV−1, detection limit 18.6 nGy s−1, X-ray afterglow ~1% @ 9.6 μs. In addition, the AAO@MeEu scintillation film with optical waveguide effect exhibits high X-ray imaging resolution to >27 lp mm−1, superior to those of most commercial and metal halide scintillators, potential of applications in medical diagnosis, industrial inspection and security. Our findings offer insights for developing low-dimensional rare-earth-based halides as next-generation high-performance scintillation materials.
Methods
Synthesis of Eu(II)-based hybrid single crystals
EtEu: 1 mmol EuBr2 (99.5%, Grirem Advanced Materials Co. Ld.) and 1 mmol Tetraethylammonium bromide (99.7%, Alfa Aesar) were added to a 10 ml Pyrex-bottle with 3 mL anhydrous MeOH (99.7%, Alfa Aesar). The solution was stirred for 1 h at 60 °C to obtain a well-soluble solution. The viridescent single crystals were grown by slow evaporation at room temperature. After 1 week, the single crystals could be recovered and stored in the glovebox for further characterization. MeEu: the similar method was used to obtain MeEu single crystals. The only difference is that 0.6 mmol EuBr2 and 0.5 mmol Tetramethylammonium bromide (99.7%, Alfa Aesar) were dissolved into 8 mL anhydrous MeOH. All manipulations were performed in a glovebox filled with argon where H2O and O2 levels < 0.1 ppm.
Fabrication of MeEu@AAO scintillation screens
The MeEu hybrids were dissolved into MeOH. The solution was uniformly dropped onto the two-pass AAO matrix templates with pore size (interval of 60 nanometers, diameter of 30 nanometers). The solvent MeOH slowly evaporated and MeEu scintillators were embedded into pores of AAO matrix templates at room temperature. Repeat the above steps until MeEu scintillators fully filled into AAO film. All manipulations were performed in glovebox filled with argon where H2O and O2 levels <0.1 ppm.
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Roques-Carmes, C. et al. A framework for scintillation in nanophotonics. Science 375, eabm9293 (2022).
Chen, Q. S. et al. All-inorganic perovskite nanocrystal scintillators. Nature 561, 88–93 (2018).
Orfano, M. et al. Efficient radioactive gas detection by scintillating porous metal–organic frameworks. Nat. Photonics 17, 672–678 (2023).
Perego, J. et al. Composite fast scintillators based on high-Z fluorescent metal–organic framework nanocrystals. Nat. Photonics 15, 393–400 (2021).
Wang, X. et al. Organic phosphors with bright triplet excitons for efficient X-ray-excited luminescence. Nat. Photonics 15, 187–192 (2021).
Wang, J. X. et al. Heavy-atom engineering of thermally activated delayed fluorophores for high-performance X-ray imaging scintillators. Nat. Photonics 16, 869–875 (2022).
Ma, W. B. et al. Thermally activated delayed fluorescence (TADF) organic molecules for efficient X-ray scintillation and imaging. Nat. Mater. 21, 210–216 (2022).
Zhang, H. et al. Reproducible X-ray imaging with a perovskite nanocrystal scintillator embedded in a transparent amorphous network structure. Adv. Mater. 33, 2102529 (2021).
Morad, V. et al. Disphenoidal zero-dimensional lead, tin, and germanium halides: highly emissive singlet and triplet self-trapped excitons and X-ray scintillation. J. Am. Chem. Soc. 141, 9764–9768 (2019).
Han, K. et al. Seed-crystal-induced cold sintering toward metal halide transparent ceramic scintillators. Adv. Mater. 34, 2110420 (2022).
Xu, L. J. et al. Highly efficient eco-friendly X-ray scintillators based on an organic manganese halide. Nat. Commun. 11, 4329 (2020).
Zhao, X. et al. Solution-processed hybrid europium (II) iodide scintillator for sensitive X-ray detection. Research 6, 0125 (2023).
Akkerman, Q. A. & Manna, L. What defines a halide perovskite? ACS Energy Lett. 5, 604–610 (2020).
Zheng, J. X. et al. Hydrogen-rich 2D halide perovskite scintillators for fast neutron radiography. J. Am. Chem. Soc. 143, 21302–21311 (2021).
He, T. Y. et al. Copper iodide inks for high-resolution X-ray imaging screens. ACS Energy Lett. 8, 1362–1370 (2023).
Jin, T. et al. Self-wavelength shifting in two-dimensional perovskite for sensitive and fast gamma-ray detection. Nat. Commun. 14, 2808 (2023).
Yang, B. et al. Lead-free halide Rb2CuBr3 as sensitive X-ray scintillator. Adv. Mater. 31, 1904711 (2019).
Han, K. et al. Band alignment engineering in ns2 electrons doped metal halide perovskites. Laser Photonics Rev. 17, 2200458 (2023).
Gektin, A. & Vasil’ev, A. Scintillation, phonon and defect channel balance; the sources for fundamental yield increase. Funct. Mater. 23, 183–190 (2016).
Dujardin, C. et al. Needs, trends, and advances in inorganic scintillators. IEEE Trans. Nucl. Sci. 65, 1977–1997 (2018).
Zhuravleva, M. et al. Crystal growth and scintillation properties of Cs3CeX6 and CsCe2X7 (X = Cl, Br). Proceedings of the IEEE Nuclear Science Symposium & Medical Imaging Conference. Knoxville 1296–1299 (IEEE, 2010).
Wu, Y. T. et al. Zero-dimensional Cs4EuX6 (X = Br, I) all-inorganic perovskite single crystals for gamma-ray spectroscopy. J. Mater. Chem. C 6, 6647–6655 (2018).
Dorenbos, P. Relation between Eu2+ and Ce3+ f ↔ d-transition energies in inorganic compounds. J. Phys. Condens. Matter 15, 4797–4807 (2003).
Gong, X. W. et al. Electron–phonon interaction in efficient perovskite blue emitters. Nat. Mater. 17, 550–556 (2018).
Li, M. Z. & Xia, Z. G. Recent progress of zero-dimensional luminescent metal halides. Chem. Soc. Rev. 50, 2626–2662 (2021).
Zhang, M. Y. et al. Oriented-structured CsCu2I3 film by close-space sublimation and nanoscale seed screening for high-resolution X-ray imaging. Nano Lett. 21, 1392–1399 (2021).
Cherepy, N. J. et al. Strontium and barium iodide high light yield scintillators. Appl. Phys. Lett. 92, 083508 (2008).
Murray, R. B. Use of Li6I(Eu) as a scintillation detector and spectrometer for fast neutrons. Nucl. Instrum. 2, 237–248 (1958).
Selling, J. et al. Europium-doped barium halide X-ray scintillators. Phys. Status Solidi C. 4, 976–979 (2007).
Choi, D. S. et al. Diamine functionalization of a metal–organic framework by exploiting solvent polarity for enhanced CO2 adsorption. ACS Appl. Mater. Interfaces 13, 38358–38364 (2021).
Wang, Z. Y. et al. Color tunable Ba0.79Al10.9O17.14: xEu phosphor prepared in air via valence state control. J. Adv. Ceram. 6, 81–89 (2017).
Nagpure, I. M. Nanoarchitectonics and properties of KMgSO4Cl: Eu phosphor: oxidation state of Eu ion, TL kinetic parameters and fading response. Appl. Phys. A 128, 431 (2022).
Mao, L. L. et al. Structural diversity in white-light-emitting hybrid lead bromide perovskites. J. Am. Chem. Soc. 140, 13078–13088 (2018).
Ke, F. et al. Quasi-one-dimensional metallicity in compressed CsSnI3. J. Am. Chem. Soc. 144, 23595–23602 (2022).
Guan, D. Q. et al. Exceptionally robust face-sharing motifs enable efficient and durable water oxidation. Adv. Mater. 33, 2103392 (2021).
Ye, M. et al. Covalency-driven collapse of strong spin-orbit coupling in face-sharing iridium octahedra. Phys. Rev. B 98, 201105 (2018).
Cui, B. B. et al. Locally collective hydrogen bonding isolates lead octahedra for white emission improvement. Nat. Commun. 10, 5190 (2019).
De Jong, M. et al. Resolving the ambiguity in the relation between Stokes shift and Huang–Rhys parameter. Phys. Chem. Chem. Phys. 17, 16959–16969 (2015).
Dorenbos, P. 5d-level energies of Ce3+ and the crystalline environment. I. Fluoride compounds. Phys. Rev. B 62, 15640–15649 (2000).
Dorenbos, P. Energy of the first 4f7→4f6 5d transition of Eu2+ in inorganic compounds. J. Lumin. 104, 239–260 (2003).
Mao, L. L. et al. Design principles for enhancing photoluminescence quantum yield in hybrid manganese bromides. J. Am. Chem. Soc. 142, 13582–13589 (2020).
Meijerink, A., Nuyten, J. & Blasse, G. Luminescence and energy migration in (Sr, Eu)B4O7, a system with a 4f7-4f6 5d crossover in the excited state. J. Lumin. 44, 19–31 (1989).
Han, K. et al. Narrow-band green-emitting hybrid organic–inorganic Eu (II)-iodides for next-generation micro-LED displays. Adv. Mater. 36, 2313247 (2024).
Dorenbos, P. Thermal quenching of Eu2+ 5d–4f luminescence in inorganic compounds. J. Phys. Condens. Matter 17, 8103–8111 (2005).
Wei, H. T. et al. Sensitive X-ray detectors made of methylammonium lead tribromide perovskite single crystals. Nat. Photonics 10, 333–339 (2016).
Zhu, W. J. et al. Low-dose real-time X-ray imaging with nontoxic double perovskite scintillators. Light Sci. Appl. 9, 112 (2020).
Wu, Y. T. et al. CsI: Tl+, Yb2+: ultra-high light yield scintillator with reduced afterglow. CrystEngComm 16, 3312–3317 (2014).
Lu, L. et al. All-inorganic perovskite nanocrystals: next-generation scintillation materials for high-resolution X-ray imaging. Nanoscale Adv. 4, 680–696 (2022).
Su, B. B. et al. Sb3+-doping in cesium zinc halides single crystals enabling high-efficiency near-infrared emission. Adv. Funct. Mater. 31, 2105316 (2021).
Li, B. Y. et al. Prominent reverse saturable absorption of lead-free two-dimensional organic-inorganic hybrid double perovskites promoted by defects. Mater. Today Phys. 38, 101219 (2023).
Seeman, V. et al. EPR of VHal centres in SrS. Phys. Status Solidi (B) 243, 1978–1982 (2006).
Sun, Y. N. et al. All-inorganic perovskite quantum dots based on InX3-trioctylphosphine oxide hybrid passivation strategies for high-performance and full-colored light-emitting diodes. ACS Appl. Electron. Mater. 3, 415–421 (2021).
Woo, J. Y. et al. Highly stable cesium lead halide perovskite nanocrystals through in situ lead halide inorganic passivation. Chem. Mater. 29, 7088–7092 (2017).
Qiao, J. W. et al. Near-infrared light-emitting diodes utilizing a europium-activated calcium oxide phosphor with external quantum efficiency of up to 54.7%. Adv. Mater. 34, 2201887 (2022).
Zhou, X. Q. et al. Interplay of defect levels and rare earth emission centers in multimode luminescent phosphors. Nat. Commun. 13, 7589 (2022).
Sebastian, M. et al. Excitonic emissions and above-band-gap luminescence in the single-crystal perovskite semiconductors CsPbBr3 and CsPbCl3. Phys. Rev. B 92, 235210 (2015).
Maddalena, F. et al. Effect of commensurate lithium doping on the scintillation of two-dimensional perovskite crystals. J. Mater. Chem. C 9, 2504–2512 (2021).
Samei, E., Flynn, M. J. & Reimann, D. A. A method for measuring the presampled MTF of digital radiographic systems using an edge test device. Med. Phys. 25, 102–113 (1998).
Wang, Z. H. et al. Needs, trends, and advances in scintillators for radiographic imaging and tomography. IEEE Trans. Nucl. Sci. 70, 1244–1280 (2023).
Acknowledgements
This research was supported by National Natural Science Foundations of China (22361132525) and Guangdong Provincial Science & Technology Project (2023A0505050084).
Author information
Authors and Affiliations
Contributions
X.Z.G. and H.K. conceived the initial concept. H.K. prepared the sample. H.K., J.J.C., and B.H.L. processed the experimental data. X.Z.G., H.K. and Z.X.Q. interpreted the theoretical and experimental results. H.K. wrote the paper, X.Z.G. supervised the work and revised the paper. All authors discussed and edited the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, K., Jin, J., Wang, Y. et al. Hybrid Eu(II)-bromide scintillators with efficient 5d-4f bandgap transition for X-ray imaging. Light Sci Appl 13, 222 (2024). https://doi.org/10.1038/s41377-024-01589-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41377-024-01589-w
This article is cited by
-
Hybrid cuprous halides enable high-sensitivity luminescence lifetime thermometry with exceptional water resistance
Light: Science & Applications (2025)
-
Suppressing electron–phonon coupling for narrow-band emitting Eu(II)-based perovskitoids
Nature Communications (2025)
-
Integrated copper-halide activated scintillator fiber array for remote high-resolution X-ray imaging
Nature Communications (2025)
-
Luminescence lifetime thermometers based on hybrid cuprous halides with exceptional water resistance and giant thermal expansion
Light: Science & Applications (2025)
-
Rational design and synthesis of narrow-band emitting Eu(II)-based hybrid halides via alkyl thermal cleavage
Nature Communications (2025)