Abstract
Perovskite quantum dots (PQDs) show promise in light-emitting diodes (LEDs). However, near-infrared (NIR) LEDs employing PQDs exhibit inferior external quantum efficiency related to the PQD emitting in the visible range. One fundamental issue arises from the PQDs dynamic surface: the ligand loss and ions migration to the interfacial sites serve as quenching centers, resulting in trap-assisted recombination and carrier loss. In this work, we developed a chemical treatment strategy to eliminate the interface quenching sites and achieve high carrier utilization. We employ a bidentate and liquid agent (Formamidine thiocyanate, FASCN) with tight binding to suppress the ligand loss and the formation of interfacial quenching sites: the FASCN-treated films exhibit fourfold higher binding energy than the original oleate ligands. Furthermore, the short ligands (carbon chain <3) enable the treated films to show eightfold higher conductivity; and the liquid characteristics of FASCN avoid the use of high polar solvents and guarantee better passivation. The high conductivity ensures efficient charge transportation, enabling PQD-based NIR-LEDs to have a record-low voltage of 1.6 V at 776 nm. Furthermore, the champion EQE of the treated LEDs is ~23%: this is twofold higher than the control, and represents the highest among reported PQD-based NIR-LEDs.
Similar content being viewed by others
Introduction
Quantum dots (QDs) are an ideal class of emitters for light-emitting diodes (LEDs)1,2,3. Metal-halide perovskite quantum dots (PQDs) unite structural diversity, bandgap tunability, color purity, and low-cost solution processing4,5,6,7. LEDs employing PQDs have shown external quantum efficiency (EQE) of over 20% in the visible range from 400 to 700 nm8,9,10. However, in the near-infrared I region (NIR-I, >750 nm), until now, the efficiency is still relatively low11.
The presence of surface and interfacial trap sites is the primary issue accounting for the inferior performance of PQD-based NIR-LEDs. Surface traps are formed by uncoordinated lead (Pb2+) during the synthesis. This is caused by the dynamic ligand binding of the oleate ligand and the surface12,13,14. The oleate ligands have long organic chains and form steric-repulsing interactions when capped on the QD surface, preventing full surface coverage. Ligand exchange employing short organic ligands without steric hindrance is an ideal way to improve the surface coverage and passivate the surface traps (Fig. 1a)15,16,17. However, the introduction of some short ligands would cause the occurrence of interfacial trap sites due to the incompact binding to the QD surface. Since most ligands provide high surface coverage only when present in excess in solution and have liable features, the ligands tend to desorbed from the surface and to the interfacial sites during the spin-coating process (Fig. 1b); these desorbed ions at interface the act as interfacial quenching centers, which deteriorate the device performance18,19,20.
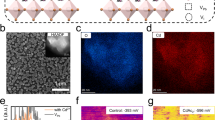
a Schematic illustrating oleate ligands and common halide ligands with incompact binding, and this work employed the bidentate and liquid reactive agent to enable full coverage and well-passivated QDs. b Schematic illustrating the dynamic ligand binding and c tight ligand binding on QDs surface during the film-preparation process. d Histogram of the binding energy to FAPbI3 QDs of various ligands. e PLQY and f TRPL spectra of FAPbI3 QDs after ligands exchange
We reasoned that this issue could be resolved only when the ligand has a short carbon chain to ensure high surface coverage, and at the same time, provide tight ligand-surface binding. We here employ a bidentate and liquid agent (Formamidine thiocyanate, FASCN) that satisfies these two requirements at the same time (Fig. 1c)21,22,23,24. The FASCN could form coordinate bonds simultaneously by using soft sulfur and nitrogen atoms and exhibits fourfold higher binding energy than the oleate ligands and threefold higher than FAI and MAI, preventing ligand desorption during the film-preparation process. The tight binding and short chain of FASCN ensures full coverage to the QD surface and improved conductivity of 3.95 × 10−7 S m−1, which is eightfold higher than the control, as evidenced by the two-terminal device. The FASCN-treated QD films exhibited an exciton binding energy of 76.3 meV, which is nearly twofold higher than the control films (39.1 meV). As a result, we fabricated PQD-based NIR-LEDs without carrier loss and achieved a recorded EQE of 23%, which is twofold higher than the control LEDs and represents the highest among reported PQD-based NIR-LEDs.
Results
The inferior efficiency of PQD-based NIR-LEDs can be ascribed to the high trap density of QDs and dynamic surface ligands, which results in carrier loss and low mobility. Surface ligand exchange is widely recognized as an effective way to replenish the trap sites and replace the long oleate ligands, but the incompact ligand-QD binding usually leads to undesired ligand desorption and new trap sites during the film-preparation process, so the appropriate ligand is a prerequisite for high-performance QDs. We chose organic-inorganic hybrid FAPbI3 QDs to verify the advantage of the FASCN treatment and synthesized the FAPbI3 QDs with oleate acid (OA−) and oleylammonium (OAm+) capping ligands (Figs. S1, and S2). We treated the QDs with various ligands during the posttreatment. Initially, we calculated the binding energy (Eb) of FASCN, oleate ligands (OA, OAm) and common halide ligands (FAI, MAI) on the host lattice FAPbI3 QDs through density-functional theory (DFT) (Figs. 1d, and S3). The Eb of FASCN (−0.91 eV) is fourfold larger than that of OAm (−0.18 eV) and OA (−0.22 eV), and is also visible higher than FAI (−0.31 eV) and MAI (−0.30 eV), indicating that the FASCN is thermodynamically more favorable for binding to the QDs surface. As shown in Fig. 1e, the photoluminescence quantum yield (PLQY) of QD got the most notable improvement after FASCN treatment, suggesting the effective passivation to surface trap sites, which was also demonstrated by the prolonged lifetime observed from time-resolved photoluminescence (TRPL) (Fig. 1f)25,26.
We then performed temperature-dependent photoluminescence (PL) spectra to investigate the intrinsic characteristics of photogenerated excitons (Figs. 2a, and S4). We observed a notable increase in integrated PL intensity as the temperature dropped from 300 to 80 K attributed to decreased exciton dissociation and nonradiative trapping processes at lower temperatures (Fig. 2b). We calculated the exciton binding energy for each film and applied the Arrhenius equation to establish the functional relationship between the integrated PL emission intensity and inverse temperature27.
Where I0 is the integrated PL intensity at 0 K, Eb is the exciton binding energy, kB is the Boltzmann constant, and A is the coefficient. The Eb of FASCN-treated QD film is 76.3 meV, nearly twofold higher than control film (39.1 meV). The higher binding energy of the FASCN-treated film indicates a reduced chance of exciton dissociation into free carriers in the absence of radiation. The possibility of exciton nonradiative complexation decreases as the exciton binding energy increases.
a Temperature-dependent PL spectra from 80 to 300 K for control and FASCN-treated FAPbI3 QD films. b, c Integrated PL emission intensity and the FWHM as a function of temperature for control and FASCN-treated FAPbI3 QD films. d Normalized decay kinetics of the photoinduced bleaching and e, f TA spectra at selected pump-probe delays for control and treated FAPbI3 QDs. g The emission peak dependence on aging time (Inset: initial PL spectra of the control and FASCN-treated QD films, and after 200 min). h Pb 4f and i I 3d core-level spectra of control and FASCN-treated QD films
The full width at half maximum (FWHM) of the PL spectra for the FASCN-treated film increased from 26.85 to 35.40 meV as the temperature rose from 80 to 300 K (Fig. 2c). Similarly, the FWHM of the control film increased from 23.56 to 36.23 meV. The reduction of structural vibration and deformation at lower temperatures helps to reduce electron-phonon coupling. To assess the strength of the electron-phonon coupling, we employed a fitting procedure to evaluate the temperature dependence of FWHM using the following equations:
Where ELO represents the longitudinal optical (LO) phonon energy, and ГLO signifies the coupling strength. The parameter C encompasses both the inhomogeneous broadening coefficient (Γinh) and the exciton-acoustic phonon coupling coefficient (σ). At low temperatures (<100 K), exciton-phonon interactions are dominated by acoustic phonons, and thermal diffusion has little effect on the system. Therefore, only factor C is considered in this regard. The fitting results demonstrate that the observed narrowing of the full width at half maximum (FWHM) can be explained by the decrease in LO phonon coupling28.
To further study the charge transfer and recombination dynamics of photons after generation, we employed femtosecond transient absorption (TA) spectroscopy (190 fs pulse width, 450 nm excited, 10 mW). The TA spectra of QDs are illustrated in Fig. S5, with probes ranging from 550 to 900 nm and scan delay times of 0.2 ps to 1 ns. We observed a strong ground bleaching signal centered at 750 nm, and the bleaching agent recovered within about 1 ps, indicating a rapid transfer of charge from the excited to the emissive state (Fig. 2d–f)29. The absorption feature decreased more rapidly after FASCN treatment, suggesting the introduction of additional energy carrier transfer and recombination pathways during the treatment30. In addition, we observed a consistent bleaching peak with an unsharp shift of FASCN-treated QDs.
We also assessed the thermal stability of the QD films under continuous heating at 100 °C by recording the PL intensity and the emission wavelength as a function of time, and found that the control films exhibited severe thermal quenching (Fig. 2g). We observed the FASCN-treated films exhibited good thermal stability with no observable emission shift with increasing heating time (Δλ = 1 nm), while the control films suffered from a significant shift with a Δλ of 12 nm, and the improved thermal stability was also clearly revealed by the PL intensity map (Fig. S6). In addition to thermal stability, we also investigated the humidity stability by exposing the QD films to humidity >99% (Fig. S7). After 30 min, the control film had corroded heavily while the target film was still intact, indicating the full ligand coverage and more compact QD structure ensure better water-oxygen stability. Then, we performed X-ray diffraction (XRD) of QD films after annealing over time to investigate the phase stability (Fig. S8), we observed a more obvious peak intensity attenuation of the control film than that of the target film during 2 h of annealing.
We also performed X-ray photoelectron spectroscopy (XPS) to further investigate the mechanism of FASCN treatment on QDs surface. In Fig. 2h, the Pb 4f peak position of the FASCN-treated sample is shifted to higher binding energy, suggesting the environmental composition changes and the electron density around Pb2+ increases with the introduction of FASCN. In addition, the I 3d peak position shifts 0.2 eV to the higher binding energy of the treated films, indicating the passivation of I vacancies (Fig. 2i)31. We also monitored the presence of the S 2p peak on QDs before and after ligand exchange (Fig. S9). The distinct S 2p peak was observed on FASCN-treated QDs, indicating that the FASCN ligand was successfully induced into the QDs surface23. The existence of the SCN− around QDs can also be verified by Fourier-transform infrared spectroscopy (FTIR), where the characteristic peak at 2248 cm−1 can be assigned to the stretching vibration of triple bonds from SCN− (Fig. S10)32. In addition, we observed the decreased intensity of FTIR peaks for FASCN-treated QD films, indicating the oleate ligands were further replaced. In addition, the ligand density decreases when increasing the FASCN concentration, the result can also be observed in 1H nuclear magnetic resonance spectra (Fig. S11). The atomic force microscopy (AFM) height images (Fig. S12) suggest that the FASCN-treated QDs films produce more homogeneous surface morphology. The root-mean-square roughness value of the FASCN-treated film is 3.77 nm, which is 1.9-fold smaller than that of the control one (7.36 nm).
In addition to the strong binding of the ligand to the QD surface, we also calculated the formation energy of the iodide vacancy (Iv). The formation energies for QDs treated with MAI, FAI, and FASCN are 1.99 eV, 2.00 eV, and 2.49 eV, respectively. These results indicate that FASCN exhibits superior defect passivation ability, which effectively reduces the density of traps and, consequently, enhances the conductivity of the QD films treated by FASCN (Fig. 3a). We then examine the conductivity of QD films through a two-terminal device (Fig. 3b). The resulting current-voltage (I-V) curve of the FASCN-treated films exhibited a notable increase in slope compared to the control films. Calculating from the I-V slope, with fixed channel length and width, the FASCN-treated films showed a conductivity (σ) of 3.95 × 10−7 S m−1, which is nearly eightfold higher than the control (4.94 × 10−8 S m−1), indicating the removal of surface insulting ligands (Fig. 3c)33. For the out-of-plane mobility, we built electron-only and hole-only devices to study the electrical transport mobility of QD films using the space charge limited current (SCLC) method. The current density-voltage (J-V) curves of electron-only and hole-only devices exhibit three modes: Ohmic contact, trap-filling limit, and child region (Fig. 3d, e). By fitting the child region and integrating the fit using the Mott–Gurney equation, the charge mobility (μ) of QD films can be obtained using the following equation (Tables S2, and 3)34,35:
Where J, d, and V are the dark current density measured in the SCLC region, the thickness of QDs films, and applied voltage, respectively. ε0 is the vacuum permittivity (ε0 = 8.854 × 10−14 F cm−1), εr is the relative permittivity of perovskite (εr = 46.9). We obtained electron mobility of 4.2 × 10−4 cm2 V−1 s−1 and hole mobility of 1.01 × 10−3 cm2 V−1 s−1 for the target device, which is 4.8-fold/2.3-fold higher than control device (8.67 × 10−5 cm2 V−1 s−1 and 4.29 × 10−4 cm2 V−1 s−1). Moreover, the smaller difference for FASCN-treated films between electron and hole mobility also suggests a more balanced carrier recombination, especially when these films were utilized in LEDs (Fig. 3f).
a Histogram of the defect formation energy of the iodide vacancy. b Schematic diagram of the two-terminal device. c The I–V curve of control and target two-terminal devices. d, e Space charge-limited current for electron-only and hole-only devices. f Schematic illustration of the carrier dynamic in NIR-LEDs
We also investigated the trap density of the QD films after FASCN treatment. The electron-only devices present trap-filling behavior at a lower voltage, a metric we refer to as trap-filled limit voltage (VTFL). We compared the trap density (ntrap) of the QD films using the following equation:
Where VTFL is the trap-filled limit voltage, e is the elemental charge, and d is film thickness36. The estimated trap density of FASCN-treated QD films is 2.95 × 10−17 cm−3, which is threefold lower than that of control films, indicating the significant passivation effect of FASCN treatment.
Encouraged by the combined high PLQY and high carrier mobility, we employed FASCN-treated QD films as emitters to fabricate the NIR-LEDs. We built a device configuration of indium tin oxide (ITO)/(PEDOT:PSS)/poly[N,N’bis(4-butylphenyl)-N,N’-bis(phenyl)-benzidine](poly-TPD)/QDs/(1,3,5-triazine-2,4,6-triyl)tris(benzene-3,1-diyl) tris (diphenylp-hosphine oxide)(PO-T2T)/lithium 8-quinolate (Liq)/Al, and the cross-sectional scanning electron microscopy (SEM) image shows each corresponding layer (Fig. 4a).
a Energy level diagram of each layer and cross-sectional SEM image of FASCN-treated NIR-LEDs. The energy levels of the FAPbI3 QDs were calculated from ultraviolet photoelectron spectroscopy (UPS in Fig. S13). b Current density-voltage curves, c EQE-current density curves and d radiance-voltage curves of control and FASCN-treated NIR-LEDs. e EL spectra from 1.6 V to 6 V and f peak EQE and radiance histogram of FASCN-treated NIR-LEDs. g Operational stability measurement of control and FASCN-treated NIR-LEDs at an initial radiance of 1 W sr−1 m−2 at ambient environment
The LED-based on FASCN-treated FAPbI3 QDs displayed a low turn-on voltage of 1.6 V. The NIR-LEDs showed a peak radiance of 24 W sr−1 m−2, representing a fourfold improvement compared to the control LEDs (Fig. 4d). The larger current density observed, compared to the control device, is attributed to higher conductivity, stemming from the decrease of trap density in the target LEDs. The electroluminescence (EL) spectra of these NIR-LEDs reveal a symmetrical EL peak at 776 nm with a full width at half maximum (FWHM) of 45 nm, importantly, the spectra remain constant within the voltage range of 1.6 to 6 V (Fig. 4e). The FASCN-treated LEDs achieved a champion EQE of 22.86%, which is nearly twofold higher than the control, and represents the highest among reported PQD-based NIR-LEDs (Fig. 4c). Furthermore, FASCN-treated LEDs exhibited consistent reproducibility, we fabricated twenty LEDs using identical procedures, and their peak EQE and peak radiance histograms are presented in Fig. 4f, revealing a mean EQE of 20.7% with a minimal standard deviation of 0.15% and a mean radiance of 22.75 W sr−1 m−2. We recorded the stability of LEDs at an initial radiance of 1 W sr−1 m−2 in the ambient environment (Fig. 4g), the FASCN-treated LEDs exhibited a half-life (T50) of 115 min, which is nearly fivefold higher than the control.
Discussion
In summary, we employed a bidentate and liquid agent (FASCN) that ensures high surface coverage and provides tight ligand-surface binding on the QDs surface, and the liquid characteristics of FASCN avoid the use of high polar solvents and guarantee better passivation. This strategy enabled us to fabricate trap-free perovskite QD films with higher conductivity, stability, and exciton binding energy. Implementing this strategy, we reported the PQD-based NIR-LEDs at 776 nm with an EQE of ≈23%, which is twofold higher than the control, and represents the highest among reported PQD-based NIR-LEDs. This chemical treatment strategy will contribute to much-needed further progress in resurfacing PQD films for NIR LEDs.
Materials and methods
Materials
Lead (II) iodide (PbI2, 99%, Sigma-Aldrich), Formamidine acetate (HN=CHNH2·CH3COOH, 99%, Alfa Aesar), Oleylamine (OAm, technical grade 70%, Sigma-Aldrich), Oleic acid (OA, technical grade 90%, Sigma-Aldrich), 1-Octadecene (ODE, technical grade 90%, Adamas), Methyl acetate (Reagent Plus, 99%, Sigma-Aldrich), Octane (anhydrous, ≥99%, Sigma-Aldrich). PEDOT: PSS solution (Clevios PVP AI 4083), poly-TPD, and PO-T2T were purchased from Xi’an Polymer Light Technology Corp. All the chemicals were used directly as received.
Synthesis of FAPbI3 QDs
In a typical synthesis, 650 mg FA(AC), and 6.0 ml OA were stirred in a 3-necked round bottom flask at room temperature for 5 min. The reaction mixture was degassed for 5 min at room temperature rapidly heated to 140 °C and continued to be degassed for 120 min. After the reaction form a clear FA-oleate precursor solution. Nitrogen (N2) was introduced, and the temperature was kept at 120 °C. Before hot injection, the system temperature of the FA-precursor was decreased to 100 °C.
In another flask, 350 mg PbI2 was mixed with 2 mL of OAm/OA mixture and 20 mL of ODE followed by vacuum drying at 100 °C for 100 min. Under the N2 atmosphere, the lead halide precursors were kept at a reaction temperature of 80 °C until all solids were dissolved. Then, 4.0 mL FA-precursor solution was rapidly injected into the flask containing the lead halide precursor. The solution was then swiftly cooled using an ice water bath. The solution was centrifuged at 7800 rpm to remove unreacted precursors. The supernatant was discarded, and the precipitation was collected. Then dissolve the precipitate with octane. During the two-step purification process, methyl acetate was added to the QDs solution. The mixture was then centrifuged to collect the precipitant and dissolve in the octane. The crude solution was centrifuged at 7800 rpm to remove unreacted precursors. The supernatant was discarded, and the precipitation was collected. Then dissolve the precipitate with octane to obtain FAPbI3 crude QDs solution.
Two-step purification strategy
Methyl acetate was added to the crude QDs solution. The mixture was then centrifuged to collect the precipitant and dissolve in the octane. Then add methyl acetate into the solution and centrifuge again. Collect the precipitant and dissolve it in the octane to obtain a purified FAPbI3 solution. The QDs solution was stored at 4 °C until further use.
FASCN ligand exchange strategy
During the second purification process, replace the methyl acetate with the 0.1 mg mL−1 FASCN solution (dissolved in methyl acetate) and centrifuge. Collect the precipitant and dissolve it in the octane to obtain FASCN-treated FAPbI3 QDs solution. The QDs solution was stored at 4 °C until further use.
FAPbI3-based NIR-LEDs fabrication
For fabricating FAPbI3 - based NIR-LEDs, ITO/glass substrates were cleaned using deionized water, acetone, and 2-propanol ultrasonically for 60, 60, and 60 min, respectively. After this, the substrate was dried with nitrogen flow and treated with UV-ozone for 15 min. Then, PEDOT: PSS was spin-coated at 4000 rpm for 30 s, followed by annealing on a hotplate at 150 °C for 15 min. After the substrate was cooled to room temperature, a solution of 8 mg mL−1 poly-TPD chlorobenzene was spin-coated onto the PEDOT: PSS layer at 4000 rpm for 40 s and bake for 15 min at 100 °C under a nitrogen atmosphere. FAPbI3 QDs were then spin-coated onto the Poly-TPD at 3000 rpm for 40 s and anneal for 60 to evaporate the solvent. Finally, ~60 nm of POT2T, ~2 nm of Liq and ~80 nm of Al were deposited using a thermal evaporation system (Suzhou Fang sheng FS380-S12) through shadow masks under a high vacuum (<10−4 Pa). Deposited using a thermal evaporation system (Suzhou Fang sheng FS380-S12) through shadow masks under a high vacuum (<10−4 Pa).
Characterization of QDs
UV-vis absorption spectra were measured by a Shimadzu UV-3600PC scanning spectrophotometer. The photoluminescence (PL) spectra were collected by a fluorescence spectrometer (NIR-VIS, FL3). PL decay spectra were obtained by using the PL-TCSPC fluorescence lifetime measurement system (C12132-38, Hamamatsu Photonics Co.). The FTIR spectra were measured by VERTEX 70 FT-IR Spectrometer under transmission mode. The samples were prepared by dripping the solution into KBr powder and pressed in powder compaction presses. Transient PL decays were measured by HORIB-FM-2015. 1H NMR spectra were measured by utilizing the JNM-ECZ400S/L1 with a frequency of 400 MHz. XRD patterns were measured using a Bruker AXS D8 diffractometer with Cu Kα radiation (λ = 1.54178 Å). TEM overview images were taken through a Talos F200X electron microscope operated at 200 kV and analyzed through the Velox software. The UPS and XPS measurements were conducted using KRATOS AXIS Ultra DLD with a base pressure of ~10−9 torr. Atomic force microscopy (AFM) measurements were operated by using a Cypher-S atomic force microscope (Asylum Research, Oxford Instruments, UK).
Characterization of PQD-based NIR-LEDs
Current density–voltage (J–V) characteristics were monitored by using a computer-controlled Keithley 2400 source meter. Electroluminescence (EL) spectra and external quantum efficiency (EQE) were tested by using a calibrated photonic multichannel PMA-12 analyzer system (Hamamatsu C10027-01 (360–950 nm) and C10028-01 (950–1600 nm)). The PMA-12 system was connected to an integrating sphere (3.3 in, collecting device forward light) and a power supply system (controlling current output) while a PR-745 instrument (Photo Research, 380–1060 nm) was used to calibrate the absolute radiance. All the measurements assumed Lambertian emission. All tests were conducted under ambient air conditions (T = 25–30 °C, H = 50–60%). During the lifetime test, the initial radiance was consistently set at 1 W sr−1 m−2, with the radiance value recorded every 5 min to track the performance over time.
Density functional theory calculations
Density functional theory (DFT) as implemented in the Vienna ab initio simulation package (VASP), was used to carry out the calculations presented here. The projector augmented wave (PAW) method was used to treat the effective interaction of the core electrons and nucleus with the valence electrons, while exchange and correlation were described using the Perdew–Burke–Ernzerhof (PBE) functional. The cut-off energy is set at 400 eV for the plane-wave basis restriction in all calculations. K-points are sampled under the Monkhorst–Pack scheme for the Brillouin-zone integration (K-points were sampled using the Gamma Point). In all calculations, the forces acting on all atoms are <0.02 eV Å−1 in fully relaxed structures, and self-consistency accuracy of 10−5 eV is reached for electronic loops. The binding energy was calculated as follows:
Where E is the total energy of the adsorbed system, Ea and Eb represent the total energy of tree species and bare surface, respectively.
Two-terminal device calculations
The conductivity (σ) of the quantum dot films has been calculated using the parameters from the Two-Terminal Device. Specifically, it is derived from the length (L), width (W), and thickness (T) of the light-emitting layer, along with the slope (S) obtained from the I-V curve. The calculation formula is as follows:
Encapsulation technique and storage condition of LEDs
We encapsulated LEDs in a glovebox using a glass encapsulation cover and UV-curable adhesive to protect the device structure. The LED devices were stored in the glovebox under N2 (H2O and O2 < 0.1 ppm) at a temperature between 25 °C and 30 °C, with humidity levels maintained between 15 and 20%.
Data availability
The data that supports the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Li, X. Y. et al. Bright colloidal quantum dot light-emitting diodes enabled by efficient chlorination. Nat. Photonics 12, 159–164 (2018).
Feng, H. W. et al. Highly efficient near-infrared light-emitting diodes based on chloride treated CdTe/CdSe type-II quantum dots. Front. Chem. 8, 266 (2020).
Gao, L. et al. Efficient near-infrared light-emitting diodes based on quantum dots in layered perovskite. Nat. Photonics 14, 227–233 (2020).
Hazarika, A. et al. Perovskite quantum dot photovoltaic materials beyond the reach of thin films: full-range tuning of a-site cation composition. ACS Nano 12, 10327–10337 (2018).
Li, M. X. et al. In situ surface reconstruction toward planar heterojunction for efficient and stable FAPbI3 quantum dot solar cells. Adv. Mater. 36, 2309890 (2024).
Yuan, S. et al. Efficient blue electroluminescence from reduced-dimensional perovskites. Nat. Photonics 18, 425–431 (2024).
Li, F. C. et al. Solution-mediated hybrid FAPbI3 perovskite quantum dots for over 15% efficient solar cell. Adv. Funct. Mater. 33, 2302542 (2023).
Duan, H. W. et al. Bi-ligand synergy enables threshold low voltage and bandgap stable pure-red mix-halide perovskite LEDs. Adv. Funct. Mater. 34, 2310697 (2024).
Wang, Y. K. et al. Self-assembled monolayer–based blue perovskite LEDs. Sci. Adv. 9, eadh2140 (2023).
Wei, Y., Cheng, Z. Y. & Lin, J. Correction: an overview on enhancing the stability of lead halide perovskite quantum dots and their applications in phosphor-converted LEDs. Chem. Soc. Rev. 48, 405–405 (2019).
Tseng, Z. L. et al. Aggregation control, surface passivation, and optimization of device structure toward near-infrared perovskite quantum-dot light-emitting diodes with an EQE up to 15.4%. Adv. Mater. 34, 2109785 (2022).
De Roo, J. et al. Highly dynamic ligand binding and light absorption coefficient of cesium lead bromide perovskite nanocrystals. ACS Nano 10, 2071–2081 (2016).
Shynkarenko, Y. et al. Direct synthesis of quaternary alkylammonium-capped perovskite nanocrystals for efficient blue and green light-emitting diodes. ACS Energy Lett. 4, 2703–2711 (2019).
Ten Brinck, S., Zaccaria, F. & Infante, I. Defects in lead halide perovskite nanocrystals: analogies and (many) differences with the bulk. ACS Energy Lett. 4, 2739–2747 (2019).
Bai, Y. et al. Surface chemistry engineering of perovskite quantum dots: strategies, applications, and perspectives. Adv. Mater. 34, 2105958 (2022).
Wang, Y. et al. All-inorganic colloidal perovskite quantum dots: a new class of lasing materials with favorable characteristics. Adv. Mater. 27, 7101–7108 (2015).
Yang, D. D. et al. CsPbBr3 quantum dots 2.0: benzenesulfonic acid equivalent ligand awakens complete purification. Adv. Mater. 31, 1900767 (2019).
Zhao, F. et al. Iodotrimethylsilane as a reactive ligand for surface etching and passivation of perovskite nanocrystals toward efficient pure-red to deep-red LEDs. Angew. Chem. Int. Ed. 62, e202311089 (2023).
Akkerman, Q. A. et al. Genesis, challenges and opportunities for colloidal lead halide perovskite nanocrystals. Nat. Mater. 17, 394–405 (2018).
Xue, J. J. et al. Surface ligand management for stable FAPbI3 perovskite quantum dot solar cells. Joule 2, 1866–1878 (2018).
Lu, H. Z. et al. Vapor-assisted deposition of highly efficient, stable black-phase FAPbI3 perovskite solar cells. Science 370, eabb8985 (2020).
Sun, B. et al. Pseudohalide-exchanged quantum dot solids achieve record quantum efficiency in infrared photovoltaics. Adv. Mater. 29, 1700749 (2017).
Zhang, X. L. et al. Ligand-assisted coupling manipulation for efficient and stable FAPbI3 colloidal quantum dot solar cells. Angew. Chem. Int. Ed. 62, e202214241 (2023).
Zhang, Z. Y. et al. Dual ligand capped quantum dots improving loading amount for high-efficiency quantum dot-sensitized solar cells. ACS Energy Lett. 8, 647–656 (2023).
Koscher, B. A. et al. Essentially trap-free CsPbBr3 colloidal nanocrystals by postsynthetic thiocyanate surface treatment. J. Am. Chem. Soc. 139, 6566–6569 (2017).
Zheng, X. P. et al. Defect passivation in hybrid perovskite solar cells using quaternary ammonium halide anions and cations. Nat. Energy 2, 17102 (2017).
Li, H. M. et al. Efficient and stable red perovskite light‐emitting diodes with operational stability >300 h. Adv. Mater. 33, 2008820 (2021).
Yuan, F. L. et al. Bright high-colour-purity deep-blue carbon dot light-emitting diodes via efficient edge amination. Nat. Photonics 14, 171–176 (2020).
Ding, C. et al. Photoexcited hot and cold electron and hole dynamics at FAPbI3 perovskite quantum dots/metal oxide heterojunctions used for stable perovskite quantum dot solar cells. Nano Energy 67, 104267 (2020).
Hu, L. et al. Flexible and efficient perovskite quantum dot solar cells via hybrid interfacial architecture. Nat. Commun. 12, 466 (2021).
Jin, H. E. et al. Eco-friendly Solvent-Processible and highly luminescent perovskite nanocrystals with polymer zwitterions for Air-Stable optoelectronics. Chem. Eng. J. 459, 141531 (2023).
Koh, W. et al. Thiocyanate-capped PbS nanocubes: ambipolar transport enables quantum dot based circuits on a flexible substrate. Nano Lett. 11, 4764–4767 (2011).
Li, H. J. et al. Thermal management towards ultra-bright and stable perovskite nanocrystal-based pure red light-emitting diodes. Nat. Commun. 15, 6561 (2024).
Duijnstee, E. A. et al. Toward understanding space-charge limited current measurements on metal halide perovskites. ACS Energy Lett. 5, 376–384 (2020).
Wang, Y. et al. Ligand‐solvent coordination enables comprehensive trap passivation for efficient near‐infrared quantum dot light-emitting diodes. Angew. Chem. Int. Ed. 63, e202407833 (2024).
Wang, H. L. et al. Extracting ammonium halides by solvent from the hybrid perovskites with various dimensions to promote the crystallization of CsPbI3 perovskite. Nano Energy 94, 106925 (2022).
Acknowledgements
Z.-S. L., W.Y. and F.Z. contributed equally to this work. This work was supported by the National Natural Science Foundation of China (NSFC) (62474119, 62175171, 61961160731 and 62205230), Suzhou Key Laboratory of Functional Nano & Soft Materials, Collaborative Innovation Center of Suzhou Nano Science & Technology, the 111 Project, Joint International Research Laboratory of Carbon-Based Functional Materials and Devices. This project is also funded by the Collaborative Innovation Center of Suzhou Nano Science and Technology (CIC-Nano).
Author information
Authors and Affiliations
Contributions
L.-S.L. and Y.-K.W. conceived and directed the project. Z.-S.L. designed and fabricated the NIR devices and performed the device characterizations under the guidance of Y.-K.W. Z.-S.L. synthesized the FAPbI3 QDs under the guidance of Y.-K.W. W.Y. and F.Z. characterized FAPbI3 QDs under the guidance of Y.-K.W. W.-Z.L. assisted with device tests. H.-H.L. contributed to the experimental works. Z.-S.L., Y.-K.W., and W.S.S. wrote the manuscript. All authors discussed the results and commented on this manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, ZS., Wang, Y., Zhao, F. et al. Liquid bidentate ligand for full ligand coverage towards efficient near-infrared perovskite quantum dot LEDs. Light Sci Appl 14, 35 (2025). https://doi.org/10.1038/s41377-024-01704-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41377-024-01704-x
This article is cited by
-
Green Synthesis Innovations and Robust Stabilization of Perovskite Quantum Dots for Sustainable High-Efficiency LED Technologies
Journal of Electronic Materials (2026)