Abstract
A novel DEK-AFF2 fusion has been recently identified in four cases of basaloid to nonkeratinizing squamous cell carcinoma (SCC) in the sinonasal tract and middle ear with high-grade morphology. The exceptional response to immune checkpoint inhibitor in the first reported case highlights the potential clinical importance of identifying tumors with DEK-AFF2 fusions. We herein reported the first series of seven cases of DEK-AFF2 fusion-associated sinonasal SCC with deceptively bland morphology, including four cases of low-grade papillary Schneiderian carcinoma, which is a recently described tumor type with unknown molecular underpinnings. The DEK gene rearrangement was confirmed by DEK break-apart fluorescence in situ hybridization and DEK-AFF2 fusion transcripts were detected by reverse transcription polymerase chain reaction. In contrast to the previously reported DEK-AFF2 fusion-positive high-grade carcinomas, these tumors had a monotonous and bland morphology and were all initially diagnosed as sinonasal papilloma (SP) of various types, with or without dysplasia or carcinoma in situ. The tumor was characterized by mixed exophytic and inverted patterns, broad papillary fronds, acantholytic change, cellular monotony, dense neutrophilic infiltrates, and peripheral palisading. All tumors were diffusely positive for p40 or p63 and negative for NUT and p16. Molecular drivers associated with SP, including EGFR and KRAS mutations and both high and low-risk human papillomavirus infection, were negative in all cases. Although there was no overt stromal invasion or desmoplastic reaction in the initial specimens, these tumors tended to progress locoregionally through a prolonged clinical course and occasionally develop lymph node metastases, high-grade transformation, or extensively local destruction eventually leading to death. These justify more aggressive clinical management. Therefore, we propose the new terminology “DEK-AFF2 fusion-associated papillary SCC of the sinonasal tract” to better describe this clinicopathologically and molecularly distinct entity.
Similar content being viewed by others
Introduction
A novel DEK-AFF2 fusion was recently identified in a squamous cell carcinoma (SCC) from the skull base showing exceptional response to immune checkpoint inhibitor therapy [1]. Three additional SCCs with the same gene fusion, two from the middle ear and temporal bone and one involving the nasal cavity and orbit, were described in subsequent reports [2, 3]. Histologically, these tumors were high-grade and showed an appearance from basaloid to immature transitional epithelial-like nonkeratinizing SCC with relatively monotonous tumor cells, vesicular chromatin, and occasional to distinct nucleoli. They were characterized by syncytial nests and papillary growth with inverted ribbons and vague peripheral palisading. Increased mitotic activity and apoptotic bodies were present and focal tumor necrosis was noted.
We recently encountered a referral case initially diagnosed as an inverted sinonasal (Schneiderian) papilloma (SP) with dysplasia in the nasal cavity and nasopharynx. The tumor showed mixed inverted and exophytic papillary growth patterns with monotonous cytology, a morphology similar to previously described so-called low-grade papillary Schneiderian carcinomas (LGPSC) [4,5,6,7,8,9]. The tumor was negative for p16, NUT, and NKX2.2 immunostains, Epstein-Barr virus (EBV) in situ hybridization, and both high and low-risk human papillomavirus (HPV) DNA testing. Molecular drivers associated with SP, including EGFR and KRAS mutations [10,11,12], were also negative. With certain morphologically similar features to the previously described nonkeratinizing SCC with DEK-AFF2 fusion, the tumor was tested for DEK rearrangement by a break-apart fluorescence in situ hybridization (FISH), which turned out to be positive. Subsequent reverse transcription polymerase chain reaction (RT-PCR) confirmed the DEK-AFF2 fusion. In contrast to the previously reported fusion-positive tumors showing frank malignant and high-grade features, the current case was relatively low-grade in appearance without overt stromal invasion. This finding led us to investigate DEK-AFF2 fusion in a series of sinonasal/nasopharyngeal papillary tumors with monotonous and bland morphology, including cases previously described as LGPSC [4,5,6,7,8,9]. The aim of this study was to characterize the clinicopathologic features in this molecularly distinct tumor.
Material and methods
Case selection
The study was approved by the Institutional Review Board (IRB) of Taipei Veterans General Hospital (IRB no.: 2020-12-012CC). In the first step, a retrospective search of sinonasal and nasopharyngeal tumors diagnosed as SP with dysplasia, carcinoma ex-SP, Schneiderian carcinoma, papillary SCC, and nonkeratinizing SCC between 2000 and 2020 from the surgical pathology archives of Taipei Veterans General Hospital and the authors’ consultation files was performed. All pathology slides were retrieved and reviewed by two pathologists specialized in head and neck pathology (YJK and JFH) to exclude other possible differential diagnoses and to identify specific morphologic features. In the second step, the central authors contacted authors of the prior reports or series of LGPSC to recruit additional cases. All available slides of the initial and recurrent/metastatic specimens were reviewed and compared.
Immunohistochemistry (IHC) and EBV-encoded RNA (EBER) in situ hybridization
A panel of IHC stains, including antibodies against p40 (clone BC28; Biocare Medical, Pacheco, CA, USA), p16 (clone E6H4; Ventana Medical Systems, Tucson, AZ, USA), NUT (clone 52B1; Cell Signaling Technology, Danvers, MA, USA), and Ki-67 (clone MIB‐1; Dako, Glostrup, Denmark) were performed on 4-μm paraffin sections on a Leica Bond-Max autostainer (Leica Biosystems, Buffalo Grove, IL, USA). EBER in situ hybridization was also performed using Bond EBER probe (Leica Biosystems) on the autostainer based on the manufacturer’s protocol. Cases diffusely positive for p16 or NUT were excluded in this study. Cases with primary tumor involving the nasopharynx were tested for EBER in situ hybridization, and the positive cases were also excluded.
FISH and RT-PCR for DEK-AFF2 fusion
All selected cases were tested for DEK rearrangement using a break-apart FISH probe (CytoTest, Rockville, MD, USA). The results were analyzed under a fluorescence microscope (Zeiss Axio Imager.D2; Carl Zeiss Microscopy, White Plains, NY, USA) using Axio-Vision 4.5 software. Cases showing >20% of nuclei positive for break-apart signals in 50 non-overlapping nuclei of the tumor cells were regarded as positive for DEK rearrangement. Cases positive for DEK rearrangement by FISH were subjected to RT-PCR to identify DEK-AFF2 fusion. In brief, total RNA was extracted from the representative FFPE tumor tissue using the RecoverAll total nucleic acid isolation kit (Thermo Fisher Scientific, Waltham, MA, USA) and was converted to cDNA using SuperScript double-stranded cDNA synthesis kit (Thermo Fisher Scientific). PCR and direct sequencing were performed using the designed primers flanking the fusion sites of the reported DEK-AFF2 fusion variants (Table 1). The sequence was analyzed manually using the Chromas software version 2.6.6 (Technelysium Pty Ltd, South Brisbane, QLD, Australia).
EGFR and KRAS mutation analysis and HPV testing
Cases positive for DEK rearrangement were subjected to tumor genomic DNA extraction using PicoPure DNA extraction kit (Thermo Fisher Scientific). EGFR exons 19 and 20 mutation analysis was performed using PCR and Sanger sequencing as previously described [13]. KRAS codon 12, 13, and 61 mutation analysis was performed using a validated mass spectrometry assay as previously described [14]. HPV testing was performed using general primers GP5 + /6+ to amplify the L1 region of at least 27 common low-risk and high-risk HPV genotypes [15]. To exclude the possibility of L1 deletion during viral genome integration, additional high-risk HPV genotyping for type 16, 18, 31, 33, 45, 52, and 58 was performed using a validated multiplex quantitative PCR assay with type-specific primers to amplify the LCR/E6/E7 regions [16].
Statistical analysis
Categorical variables between two groups were compared using Fisher’s exact test. Two-sided P < 0.05 were considered to be statistically significant. Statistical analyses were performed using R statistical software version 3.5.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Clinical features
In the first step, we identified DEK-AFF2 fusions in three out of 14 initial cases, which included 11 in-house cases and three cases from the consultation files. In the second step, additional four LGPSCs were recruited and all tested positive for the DEK-AFF2 fusion. Three of the four cases were previously reported, including one case by Lewis et al. [4] and two cases by Zhai et al. (patients 4 and 5 in the original series) [9]. The clinical information of these seven cases is summarized in Table 2. There were five females and two males with a median age of 53 years (range: 28–79 years). The sites of initial presentation included the nasopharynx, nasal cavity, and paranasal sinuses (Fig. 1). One case (case 6) showed a very extensive regional involvement to the skull base and middle ear. All cases were initially diagnosed as SPs with or without dysplasia or carcinoma in situ (CIS) and were treated with local excision with or without adjuvant therapy. During the median follow-up of 26 months (range: 7 months to 18 years), all cases had at least one local recurrence. Two cases (case 4 and 6) developed nodal metastases. One case (case 4) had widely invasive tumor through multiple recurrences, and the patient eventually died of the disease 18 years after the initial diagnosis.
A Case 1 showed a tumor at right nasopharynx (asterisk) and superior turbinate. B Case 2 showed a tumor occupying right nasal cavity (asterisk). C Case 3 showed a tumor involving left frontal and anterior ethmoid sinuses (asterisk).
Histopathology and immunohistochemistry
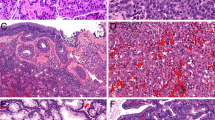
The pathologic and immunophenotypic features are summarized in Table 3. All primary tumors showed a similar histomorphology, which was characterized by basaloid to nonkeratinizing tumor cells arranged in a mixture of exophytic and inverted papillary growth patterns without overt invasion or desmoplastic stromal reaction (Fig. 2A, B). The exophytic tumor papillae were either thin with a delicate fibrovascular core or broad with occasional histiocytic aggregates (Fig. 2C, D). Some of the inverted trabeculae constituted a complex anastomosing labyrinth-like appearance and showed vague to prominent peripheral palisading (Fig. 2E). Focal peculiar acantholytic change, manifested by loss of cellular cohesion, was seen in all cases and gave rise to pseudopapillary structures (Fig. 2F) or a stellate reticulum-like appearance in the center of tumor trabeculae (Fig. 2G). The tumor cells generally showed very monotonous round nuclei with no or inconspicuous nucleoli (Fig. 2H). The amount of cytoplasm ranged from scant (basaloid) to abundant and pink (Fig. 3A). Dense neutrophilic infiltrates were focally present in both tumor nests and stroma of all cases (Fig. 3A). The other minor pathologic findings included cellular whorl formation (Fig. 3B), squamoid morules (Fig. 3C), clear cell change (Fig. 3D), microcysts (Fig. 3E), and focal ciliated epithelium (possibly from residual respiratory mucosa) superficially covering the tumor nests (Fig. 3F). The presence of broad papillary fronds, focal acantholytic change, and dense neutrophilic infiltrates were significantly more frequent in the DEK-AFF2 fusion-associated papillary SCC compared to the 11 DEK FISH negative papillary tumors of the sinonasal tract (7/7 versus 4/11, P = 0.0128), while the other major features, i.e., mixed exophytic and inverted pattern (7/7 versus 9/11, P = 0.4967), peripheral palisading (7/7 versus 9/11, P = 0.4967), and cellular monotony (7/7 versus 8/11, P = 0.2451) showed no differences.
A Basaloid tumor with a mixture of exophytic papillary architecture and inverted growth pattern. B Inverted growth with nonkeratinizing tumor cells without overt stromal invasion and desmoplastic reaction. C Broad tumor papillae with edematous vascular stroma. D Aggregates of foamy histiocytes in the papillary fronds. E Complex labyrinth-like structure composed of inverted and interconnecting tumor trabeculae. F Acantholytic change characterized by loss of cell cohesion giving rise to a pseudopapillary pattern. G Stellate reticulum-like features with peripheral palisading. H Monotonous tumor cells with bland nuclei and inconspicuous nucleoli.
A Dense neutrophilic infiltrates present in both tumor nests and stroma. B Cellular whorl-like formations. C Squamoid morules. D Focal clear cell change. E Microcystic pattern. F Ciliated epithelial caps.
Available recurrent tumor specimens were also examined and morphologic progression was observed in the two cases with aggressive clinical behavior. In case 4, the tumor invaded deeply to the orbital soft tissue and skin dermis (Fig. 4A). The tumor was composed of monotonous tumor cells with increased amounts of pink cytoplasm arranging in thickened papillae and solid nests reminiscent of a low-grade urothelial carcinoma (Fig. 4B, C) in contrast to the initial specimen showing tumor cells with scant cytoplasm and delicate thin papillae [4]. In case 6, the metastatic neck lymph node showed solid and cystic change (Fig. 4D). The tumor still preserved a mixed exophytic and inverted growth with thin papillae, vague peripheral palisading, and focal acantholytic change (Fig. 4E). However, the tumor cells were high-grade with vesicular nuclei, prominent eosinophilic nucleoli and an increased mitotic activity (up to 10/10HPF), while retaining the same monotonous appearance (Fig. 4F).
Case 4: A The tumor invaded deeply to the facial skin dermis. Note the epidermis on the left lower corner. B Thickened papillae and solid nests reminiscent of a low-grade urothelial carcinoma were noted. C The tumor was composed of monotonous tumor cells with increased amount of pink cytoplasm while remaining low-grade nuclear feature. Case 6: D The metastatic neck lymph node showed solid and cystic change. E The tumor still preserved a mixed exophytic and inverted growth with thin papillae, vague peripheral palisading, and focal acantholytic change. F Enlarged, crowded, and vesicular nuclei with accompanied prominent nucleoli were observed while retaining the monotonous appearance.
Low mitotic activity up to 1 per 10 high-power fields (HPFs) and low Ki-67 labeling index were noted in all primary tumors (Fig. 5A, C), except one case showing an increased mitotic activity and high Ki-67 labeling index up to 65% in focal areas (case 1) (Fig. 5B, D). Through disease recurrences, an increase of mitotic activity and Ki-67 labeling index was also observed in most of the cases. All tumors harbored a squamous immunophenotype and were diffusely positive for p40 or p63 (Fig. 5E) and negative for NUT. P16 IHC showed a patchy and mosaic staining pattern in <70% of the tumor cells (Fig. 5F).
A Low mitotic activity up to 1 per 10 high-power fields (HPFs). B An increase of mitotic activity up to 24 per 10 HPFs. C Corresponding area with low Ki-67 labeling index around 5%. D Corresponding area with high Ki-67 labeling index around 40%. E Diffuse and strong p40 immunoreactivity. F Mosaic p16 staining pattern in <70% of the tumor cells.
Molecular testing
The results of molecular testing are summarized in Table 3. In the first step, the DEK break-apart FISH was successfully performed in 14 of the 16 cases and three cases showed positive break-apart signals (Fig. 6A). In the second step, all four recruited LGPSCs were positive for DEK FISH. The subsequent RT-PCR confirmed the presence of in-frame DEK-AFF2 fusion transcript in all seven cases (Fig. 6B–E). Interestingly, a single strong DNA band was present in the reactions for both D7A4 and D7A6 primer sets in cases 6 and 7, and the band size of D7A4 reactions was shorter than the targeted product size of DEKexon7-AFF2exon4 transcript. Further Sanger sequencing demonstrated a novel DEKexon7-AFF2exon5 fusion variant and a known DEKexon7-AFF2exon6 fusion in both cases. Additional PCR using D7A4 forward primers and newly designed D7A5 reverse primers confirmed this finding. Given that the exon 5 of AFF2 was more proximal to the 5’-end than the exon 6, the novel DEKexon7-AFF2exon5 fusion was concluded to be the favored fusion variant and the presence of DEKexon7-AFF2exon6 transcript was most likely due to alternative splicing. All seven cases were negative for EGFR and KRAS hotspot mutations as well as high and low-risk HPV DNA.
A The DEK fluorescence in situ hybridization showed positive break-apart signals. B–E Different DEK-AFF2 fusion variants detected by reverse transcription polymerase chain reaction. F The DEK-AFF2 fusion is presumably resulted from a translocation between chromosomes 6p22 and Xq28 with multiple breakpoints. G The chimeric proteins are predicted to contain the major functional domains of both partners.
Discussion
With the advance of next-generation sequencing (NGS), more and more molecularly distinct novel entities have been identified, including the recently discovered DEK-AFF2 fusion-associated SCC of the head and neck region [1,2,3]. In the descriptions of the four reported cases to date, the DEK-AFF2 fusion-positive SCCs were clinically aggressive tumors that were usually unresectable and prone to metastasize. The exceptional response to immune checkpoint inhibitor therapy despite a low mutation burden and a lack of PD-L1 expression in the first reported case highlighted a potential clinical importance to the identification of tumors with DEK-AFF2 fusions [1]. Morphologically, these four DEK-AFF2 fusion-positive SCCs were composed of basaloid to nonkeratinizing tumor cells arranged in papillary architecture and inverted ribbons. High-grade features such as distinct nucleoli, comedo-type necrosis, apoptotic bodies, and an increased mitotic activity were noted. Although high-grade, the tumor cells in these reported cases had a monotonous appearance, typical of fusion-associated tumors in general, and in contrast to the pleomorphic morphology in usual SCCs.
In this study, we reported a series of seven DEK-AFF2 fusion-associated papillary SCCs of the sinonasal tract with unique clinical features and deceptively bland morphology. These tumors tended to progress locoregionally through a prolonged clinical course and occasionally develop lymph node metastases and local recurrences that lead to death. Extensive involvement of the paranasal sinuses, middle ear, and skull base can also occur. The morphologic features of these tumors are monotonous and bland. In fact, all seven of our cases were initially diagnosed as various types of SP with or without dysplasia or CIS due to the bland morphology. While these tumors shared some architectural similarities with the recently described high-grade DEK-AFF2 fusion-positive carcinomas, such as papillary and inverted growth, basaloid appearance, vague peripheral palisading, cellular monotony, and dense neutrophilic infiltrates, we also identified several peculiar features in our cases, including broad papillary fronds, acantholytic change, whorl-like formations, stellate reticulum-like features, occasional squamous morules, clear cell change, ciliated epithelial caps, and rare microcysts. The aforementioned clinicopathologic findings are similar to what has been reported as LGPSC in the prior literature.
LGPSC was first described by Lewis et al. in 2015 and was characterized by its deceptively bland morphology simulating an SP, locally destructive nature, prolonged period for disease progression, multiple recurrences, and potential for nodal metastasis [4]. So far, there have been 10 cases reported in the literature [4,5,6,7,8,9]. Among them, 6 were tested for EGFR and KRAS hotspot mutations [8, 9], which are known as the driver mutations in inverted and oncocytic SPs respectively, and their associated carcinomas [10,11,12, 17, 18], and all tested cases were negative. Although 2 of 9 cases were positive for high-risk HPV type 16 DNA, both were negative for diffuse and strong p16 expression [4, 5, 7,8,9]. Low-risk HPV DNA was only tested in two cases, and both were negative [4, 8]. Accordingly, none of the reported 10 cases had molecular changes associated with typical SPs. In our seven cases, including three LGPSCs that we collected from the previous reports [4, 9], all were positive for DEK-AFF2 fusion but negative for EGFR mutations, KRAS mutations, and high and low-risk HPV DNA. The above findings suggest that the so-called LGPSC is molecularly distinct from SP, despite the morphologic resemblance. Although all seven cases in this series lacked obvious stromal invasion in the initial specimens, the universal tendency for tumor recurrence and the occasional high-grade transformation and adverse behavior seem to justify a more aggressive management as would be undertaken for a frank carcinoma. Therefore, we propose the new terminology “DEK-AFF2 fusion-associated papillary SCC of the sinonasal tract” to better describe this clinicopathologically and molecularly distinct tumor. In this series, two cases developed morphological progression through multiple recurrences. In case 4, an increase in the amount of cytoplasm and cell proliferation with retention of low-grade nuclear features was noted in the tumor specimen that was deeply invasive into the orbit and facial skin dermis. In case 6, enlarged, crowded, and vesicular nuclei with prominent nucleoli were observed in the lymph node metastasis. The natural history and the relationship between the current DEK-AFF2 fusion-associated papillary SCC with deceptively bland morphological features and previously reported high-grade SCC with DEK-AFF2 fusion remain to be clarified.
The human DEK gene is located on chromosome 6p22.3 and has been demonstrated to be a proto-oncogene [19]. It was first discovered in a subset of acute myeloid leukemia (AML) characterized by a translocation (6;9) (p23;q34) resulting in the fusion of exon 2 of DEK and exon 6 of NUP214 (formerly known as CAN) [20]. The DEK-NUP214 fusion is sufficient to transform hematopoietic progenitor cells to induce AML [21]. The encoded DEK is a nuclear protein that binds to DNA with the SAP domain. Overexpression of DEK is required for cell proliferation in head and neck cancer [22]. The AFF2 gene is located on the long arm of the X chromosome (Xq28) and belongs to the AFF family of genes [23]. The encoded AFF2 is an RNA-binding protein through the C-terminal domain and functions as a transcriptional activator. While AFF2 was mainly linked to fragile X E (FRAXE) syndrome, the other three AFF family genes were reported to form fusion genes with MLL in acute lymphoblastic leukemia [24]. The DEK-AFF2 fusion presumably resulted from a translocation between chromosomes 6 and X (Fig. 6F). Multiple breakpoints in AFF2 have been identified, including a novel breakpoint at intron 4 forming the DEKexon7-AFF2exon5 fusion variant that we found in 2 cases in this study. The chimeric proteins are predicted to contain the major functional domains of both partners (Fig. 6G) and supposedly retains binding ability to both DNA and RNA. The oncogenic mechanism of DEK-AFF2 fusion requires further investigation. It is worth mentioning that most commercially available NGS panels to identify DEK fusion in AML are not applicable for the detection of DEK-AFF2 fusion-positive SCC due to the different breakpoints in these tumors.
SP is the most important entity in the differential diagnosis that needs to be distinguished from the DEK-AFF2 fusion-associated papillary SCC with deceptively bland morphological features. Papillary and inverted growth patterns and prominent neutrophilic infiltrates are also present in SP. However, it usually does not form the complex labyrinth-like structures and the downward nests typically have lumina rather than being completely solid. In addition, the tumor cells of SP are not monotonous and do not show acantholytic change. SPs with dysplasia or CIS, however, are much more challenging to distinguish from the DEK-AFF2 fusion-positive SCC. Nonkeratinizing SCC (with or without HPV infection), NUT carcinoma, and adamantinoma-like Ewing sarcoma are the other morphologic mimickers. In difficult cases, ancillary testing for EGFR/KRAS mutations, p16/HPV, NUT, NKX2.2, and DEK rearrangement would be helpful in confirming the diagnosis, since all of these markers are mutually exclusive based on current evidence.
In summary, we describe the first series of “DEK-AFF2 fusion-associated papillary SCC of the sinonasal tract” with deceptively bland morphological features, which includes a subset of previously reported LGPSCs. In contrast to the reported high-grade DEK-AFF2 fusion-positive carcinomas, these tumors have a deceptively bland morphology without overt stromal invasion that could easily be misdiagnosed as an SP at initial presentation. We show several diagnostic features for the distinction, most importantly the presence of broad papillary fronds, focal acantholytic change, and dense neutrophilic infiltrates. All cases tested negative for known drivers of SPs, such as EGFR/KRAS mutations and high and low-risk HPV, suggesting a completely different oncogenic driver mechanism. The tumors tend to progress locoregionally through a prolonged clinical course and may occasionally develop lymph node metastases or high-grade transformation that seems to justify more aggressive clinical management and classification as a carcinoma.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Yang W, Lee KW, Srivastava RM, Kuo F, Krishna C, Chowell D, et al. Immunogenic neoantigens derived from gene fusions stimulate T cell responses. Nat Med. 2019;25:767–75.
Todorovic E, Truong T, Eskander A, Lin V, Swanson D, Dickson BC, et al. Middle ear and temporal bone nonkeratinizing squamous cell carcinomas with DEK-AFF2 fusion: an emerging entity. Am J Surg Pathol. 2020;44:1244–50.
Bishop JA, Gagan J, Paterson C, McLellan D, Sandison A. Nonkeratinizing squamous cell carcinoma of the sinonasal tract with DEK-AFF2: further solidifying an emerging entity. Am J Surg Pathol. 2021;45:718–20.
Lewis JS Jr., Chernock RD, Haynes W, El-Mofty SK. Low-grade papillary Schneiderian carcinoma, a unique and deceptively bland malignant neoplasm: report of a case. Am J Surg Pathol. 2015;39:714–21.
Brown CS, Abi Hachem R, Pendse A, Madden JF, Francis HW. Low-grade papillary Schneiderian carcinoma of the sinonasal cavity and temporal bone. Ann Otol Rhinol Laryngol. 2018;127:974–7.
Jeong HJ, Roh J, Lee BJ, Cho KJ. Low-grade papillary Schneiderian carcinoma: a case report. Head Neck Pathol. 2018;12:131–5.
Williamson A, Sharma R, Cooper L, McGarry G. Low-grade papillary Schneiderian carcinoma; rare or under-recognised? Otolaryngol Case Rep. 2019;11:100107.
Carnevale S, Ferrario G, Sovardi F, Benazzo M, Morbini P. Low-grade papillary schneiderian carcinoma: report of a case with molecular characterization. Head Neck Pathol. 2020;14:799–802.
Zhai C, Wang H, Li S, Wang D. Clinicopathological analysis of low-grade papillary Schneiderian carcinoma: report of five new cases and review of the literature. Histopathology. 2021. https://doi.org/10.1111/his.14347.
Udager AM, McHugh JB, Goudsmit CM, Weigelin HC, Lim MS, Elenitoba-Johnson KSJ, et al. Human papillomavirus (HPV) and somatic EGFR mutations are essential, mutually exclusive oncogenic mechanisms for inverted sinonasal papillomas and associated sinonasal squamous cell carcinomas. Ann Oncol. 2018;29:466–71.
Mehrad M, Stelow EB, Bishop JA, Wang X, Haynes W, Oliver D, et al. Transcriptionally active HPV and targetable EGFR mutations in sinonasal inverted papilloma: an association between low-risk HPV, condylomatous morphology, and cancer risk? Am J Surg Pathol. 2020;44:340–6.
Udager AM, McHugh JB, Betz BL, Montone KT, Livolsi VA, Seethala RR, et al. Activating KRAS mutations are characteristic of oncocytic sinonasal papilloma and associated sinonasal squamous cell carcinoma. J Pathol. 2016;239:394–8.
Ho HL, Chang FP, Ma HH, Liao LR, Chuang YT, Chang-Chien YC, et al. Molecular diagnostic algorithm for epidermal growth factor receptor mutation detection in Asian lung adenocarcinomas: comprehensive analyses of 445 Taiwanese patients with immunohistochemistry, PCR-direct sequencing and Scorpion/ARMS methods. Respirology. 2013;18:1261–70.
Chang HY, Hang JF, Wu CY, Lin TP, Chung HJ, Chang YH, et al. Clinicopathological and molecular characterization of papillary renal neoplasm with reverse polarity and its renal papillary adenoma analogue. Histopathology. 2021;78:1019–31.
de Roda Husman AM, Walboomers JM, van den Brule AJ, Meijer CJ, Snijders PJ. The use of general primers GP5 and GP6 elongated at their 3’ ends with adjacent highly conserved sequences improves human papillomavirus detection by PCR. J Gen Virol. 1995;76:1057–62.
Hang JF, Hsieh MS, Li WY, Chen JY, Lin SY, Liu SH, et al. Human papillomavirus-related carcinoma with adenoid cystic-like features: a series of five cases expanding the pathological spectrum. Histopathology. 2017;71:887–96.
Udager AM, Rolland DCM, McHugh JB, Betz BL, Murga-Zamalloa C, Carey TE, et al. High-frequency targetable EGFR mutations in sinonasal squamous cell carcinomas arising from inverted sinonasal papilloma. Cancer Res. 2015;75:2600–6.
Wang H, Li H, Hu L, Zhou J, Zhai C, Wang D, et al. EGFR and KRAS mutations in Chinese patients with sinonasal inverted papilloma and oncocytic papilloma. Histopathology. 2019;75:274–81.
Waldmann T, Scholten I, Kappes F, Hu HG, Knippers R. The DEK protein-an abundant and ubiquitous constituent of mammalian chromatin. Gene. 2004;343:1–9.
von Lindern M, Fornerod M, van Baal S, Jaegle M, de Wit T, Buijs A, et al. The translocation (6;9), associated with a specific subtype of acute myeloid leukemia, results in the fusion of two genes, dek and can, and the expression of a chimeric, leukemia-specific DEK-CAN mRNA. Mol Cell Biol. 1992;12:1687–97.
Qin H, Malek S, Cowell JK, Ren M. Transformation of human CD34+ hematopoietic progenitor cells with DEK-NUP214 induces AML in an immunocompromised mouse model. Oncogene. 2016;35:5686–91.
Adams AK, Hallenbeck GE, Casper KA, Patil YJ, Wilson KM, Kimple RJ, et al. DEK promotes HPV-positive and -negative head and neck cancer cell proliferation. Oncogene. 2015;34:868–77.
Bensaid M, Melko M, Bechara EG, Davidovic L, Berretta A, Catania MV, et al. FRAXE-associated mental retardation protein (FMR2) is an RNA-binding protein with high affinity for G-quartet RNA forming structure. Nucleic Acids Res. 2009;37:1269–79.
Melko M, Douguet D, Bensaid M, Zongaro S, Verheggen C, Gecz J, et al. Functional characterization of the AFF (AF4/FMR2) family of RNA-binding proteins: insights into the molecular pathology of FRAXE intellectual disability. Hum Mol Genet. 2011;20:1873–85.
Acknowledgements
The authors would like to thank Jen-Chieh Lee, MD, PhD for the technical advice and the Biobank, Taipei Veterans General Hospital for assistance with sample preparation in this study.
Funding
The study was supported by the research grants from Taipei Veterans General Hospital (Grant No.: V109B-029 and V110B-019).
Author information
Authors and Affiliations
Contributions
YJK and JFH performed study concept and design, analyzed the data, and wrote the paper; YJK, JSL, CZ, YAC, RDC, MSH, MYL, CKL, and JFH provided specimen acquisition and clinical data; JFH provided molecular methodology; IW provided consultation support; JSL, RDC, MSH, and IW performed critical review and editing. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
Rebecca Chernock is a member of Caris Life Science Precision Oncology Alliance and there is no financial remuneration for this position. The other authors have no personal, financial, or institutional interest in any of the drugs, materials, or devices described in this article.
Ethics approval/consent to participate
The study was approved by the Institutional Review Board (IRB) of Taipei Veterans General Hospital (IRB no.: 2020-12-012CC).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kuo, YJ., Lewis, J.S., Zhai, C. et al. DEK-AFF2 fusion-associated papillary squamous cell carcinoma of the sinonasal tract: clinicopathologic characterization of seven cases with deceptively bland morphology. Mod Pathol 34, 1820–1830 (2021). https://doi.org/10.1038/s41379-021-00846-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41379-021-00846-2