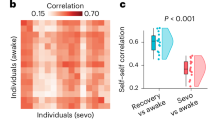
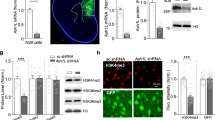
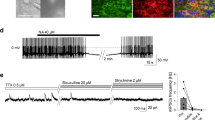
Abstract
General anesthetics (GAs) are conventionally thought to induce loss of consciousness (LOC) by acting on pre- and post-synaptic targets. However, the mechanism underlying the involvement of astrocytes in LOC remains unclear. Here we report that inhaled GAs cause reversible impairments in the fine processes of astrocytes within the somatosensory cortex, mediated by regulating the phosphorylation level of Ezrin, a protein critical for the fine morphology of astrocytes. Genetically deleting Ezrin or disrupting its phosphorylation was sufficient to decrease astrocyte-synapse interaction and enhance sensitivity to sevoflurane (Sevo) in vivo. Moreover, we show that disrupting astrocytic Ezrin phosphorylation boosted the inhibitory effect of Sevo on pyramidal neurons by enhancing tonic GABA and lowering excitability under anesthesia. Our work reveals previously unappreciated phosphorylation-dependent morphological dynamics, which enable astrocytes to regulate neuronal activity during the transition between two brain consciousness states.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials.
References
Bharioke A, Munz M, Brignall A, Kosche G, Eizinger MF, Ledergerber N, et al. General anesthesia globally synchronizes activity selectively in layer 5 cortical pyramidal neurons. Neuron. 2022;110:2024–40 e2010.
Suzuki M, Larkum ME. General anesthesia decouples cortical pyramidal neurons. Cell. 2020;180:666–76.e613.
Kelly EW, Solt K, Raines DE. Volatile aromatic anesthetics variably impact human gamma-aminobutyric acid type A receptor function. Anesth Analg. 2007;105:1287–92.
Wang D, Guo Y, Li H, Li J, Ran M, Guo J, et al. Selective optogenetic activation of orexinergic terminals in the basal forebrain and locus coeruleus promotes emergence from isoflurane anaesthesia in rats. Br J Anaesth. 2021;126:279–92.
Song Y, Chu R, Cao F, Wang Y, Liu Y, Cao J, et al. Dopaminergic neurons in the ventral tegmental-prelimbic pathway promote the emergence of rats from sevoflurane anesthesia. Neurosci Bull. 2022;38:417–28.
Speigel IA, Hemmings HC Jr. Selective inhibition of gamma aminobutyric acid release from mouse hippocampal interneurone subtypes by the volatile anaesthetic isoflurane. Br J Anaesth. 2021;127:587–99.
Denomme N, Hull JM, Mashour GA. Role of voltage-gated sodium channels in the mechanism of ether-induced unconsciousness. Pharmacol Rev. 2019;71:450–66.
Steinberg EA, Wafford KA, Brickley SG, Franks NP, Wisden W. The role of K2P channels in anaesthesia and sleep. Pflug Arch. 2015;467:907–16.
Covarrubias M, Barber AF, Carnevale V, Treptow W, Eckenhoff RG. Mechanistic insights into the modulation of voltage-gated ion channels by inhalational anesthetics. Biophys J. 2015;109:2003–11.
Woll KA, Zhou X, Bhanu NV, Garcia BA, Covarrubias M, Miller KW, et al. Identification of binding sites contributing to volatile anesthetic effects on GABA type A receptors. FASEB J. 2018;32:4172–89.
Baumgart JP, Zhou Z-Y, Hara M, Cook DC, Hoppa MB, Ryan TA, et al. Isoflurane inhibits synaptic vesicle exocytosis through reduced Ca2+ influx, not Ca2+ -exocytosis coupling. Proc Natl Acad Sci. 2015;112:11959–64.
Hemmings HC Jr, Riegelhaupt PM, Kelz MB, Solt K, Eckenhoff RG, Orser BA, et al. Towards a comprehensive understanding of anesthetic mechanisms of action: a decade of discovery. Trends Pharmacol Sci. 2019;40:464–81.
Yu X, Nagai J, Khakh BS. Improved tools to study astrocytes. Nat Rev Neurosci. 2020;21:121–38.
Verkhratsky A, Nedergaard M. Physiology of astroglia. Physiol Rev. 2018;98:239–389.
Parkhurst Christopher N, Yang G, Ninan I, Savas Jeffrey N, Yates John R, Lafaille Juan J, et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell. 2013;155:1596–609.
Henneberger C, Bard L, Panatier A, Reynolds JP, Kopach O, Medvedev NI, et al. LTP induction boosts glutamate spillover by driving withdrawal of perisynaptic astroglia. Neuron. 2020;108:919–36.e911.
Vainchtein ID, Chin G, Cho FS, Kelley KW, Miller JG, Chien EC, et al. Astrocyte-derived interleukin-33 promotes microglial synapse engulfment and neural circuit development. Science. 2018;359:1269–73.
Madry C, Kyrargyri V, Arancibia-Cárcamo IL, Jolivet R, Kohsaka S, Bryan RM, et al. Microglial ramification, surveillance, and interleukin-1β release are regulated by the two-pore domain K+ channel THIK-1. Neuron. 2018;97:299–12.e296.
Liu YU, Ying Y, Li Y, Eyo UB, Chen T, Zheng J, et al. Neuronal network activity controls microglial process surveillance in awake mice via norepinephrine signaling. Nat Neurosci. 2019;22:1771–81.
Sherpa AD, Xiao F, Joseph N, Aoki C, Hrabetova S. Activation of β-adrenergic receptors in rat visual cortex expands astrocytic processes and reduces extracellular space volume. Synapse. 2016;70:307–16.
McOmish CE, Demireva EY, Gingrich JA. Developmental expression of mGlu2 and mGlu3 in the mouse brain. Gene Expr Patterns. 2016;22:46–53.
He Y, Liu T, He Q, Ke W, Li X, Du J, et al. Microglia facilitate and stabilize the response to general anesthesia via modulating the neuronal network in a brain region-specific manner. eLife. 2023;12:RP92252.
Stowell RD, Sipe GO, Dawes RP, Batchelor HN, Lordy KA, Whitelaw BS, et al. Noradrenergic signaling in the wakeful state inhibits microglial surveillance and synaptic plasticity in the mouse visual cortex. Nat Neurosci. 2019;22:1782–92.
VanderZwaag J, Tremblay M. A neuronal activity-boosting microglial function in post-anesthetic emergence: how microglial-neuronal crosstalk may alter states of consciousness. Neurosci Bull. 2024;40:1590–2.
Haruwaka K, Ying Y, Liang Y, Umpierre AD, Yi M-H, Kremen V, et al. Microglia enhance post-anesthesia neuronal activity by shielding inhibitory synapses. Nat Neurosci. 2024;27:449–61.
Cao K, Qiu L, Lu X, Wu W, Hu Y, Cui Z, et al. Microglia modulate general anesthesia through P2Y12 receptor. Curr Biol. 2023;33:2187–2200.e2186.
Thrane AS, Thrane VR, Zeppenfeld D, Lou N, Xu Q, Nagelhus EA, et al. General anesthesia selectively disrupts astrocyte calcium signaling in the awake mouse cortex. Proc Natl Acad Sci USA. 2012;109:18974–9.
Neudecker V, Perez-Zoghbi JF, Martin LD, Dissen GA, Grafe MR, Brambrink AM. Astrogliosis in juvenile non-human primates 2 years after infant anaesthesia exposure. Br J Anaesth. 2021;127:447–57.
Zhou B, Chen L, Liao P, Huang L, Chen Z, Liao D, et al. Astroglial dysfunctions drive aberrant synaptogenesis and social behavioral deficits in mice with neonatal exposure to lengthy general anesthesia. PLoS Biol. 2019;17:e3000086.
Wang W, Lu R, Feng DY, Zhang H. Sevoflurane inhibits glutamate-aspartate transporter and glial fibrillary acidic protein expression in hippocampal astrocytes of neonatal rats through the janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway. Anesth Analg. 2016;123:93–102.
Stary CM, Sun X, Giffard RG. Astrocytes protect against isoflurane neurotoxicity by buffering pro-brain–derived neurotrophic factor. Anesthesiology. 2015;123:810–9.
Song S, Zhao W, Ji Y, Huang Q, Li Y, Chen S, et al. SHANK2 protein contributes to sevoflurane-induced developmental neurotoxicity and cognitive dysfunction in C57BL/6 male mice. Anesthesiol Perioper Sci. 2023;1:2.
Lin J, Cheng X, Wang H, Du L, Li X, Zhao G, et al. Activation of astrocytes in the basal forebrain in mice facilitates isoflurane-induced loss of consciousness and prolongs recovery. BMC Anesthesiol. 2023;23:213.
Liu P-C, Yao W, Chen X-Y, Su W-K, Zheng Z-H, Yan X-B, et al. Parabrachial nucleus astrocytes regulate wakefulness and isoflurane anesthesia in mice. Front Pharmacol. 2023;13:991238.
Ramadasan-Nair R, Hui J, Itsara LS, Morgan PG, Sedensky MM. Mitochondrial function in astrocytes is essential for normal emergence from anesthesia in mice. Anesthesiology. 2019;130:423–34.
Baldwin KT, Murai KK, Khakh BS. Astrocyte morphology. Trends Cell Biol. 2023;34:547–65.
Herrera Moro Chao D, Kirchner MK, Pham C, Foppen E, Denis RGP, Castel J, et al. Hypothalamic astrocytes control systemic glucose metabolism and energy balance. Cell Metab. 2022;34:1532–47.e1536.
Hösli L, Binini N, Ferrari KD, Thieren L, Looser ZJ, Zuend M, et al. Decoupling astrocytes in adult mice impairs synaptic plasticity and spatial learning. Cell Rep. 2022;38:110484.
Zhang Y, Reichel JM, Han C, Zuniga-Hertz JP, Cai D. Astrocytic process plasticity and IKKβ/NF-κB in central control of blood glucose, blood pressure, and body weight. Cell Metab. 2017;25:1091–1102.e1094.
Kruyer A, Angelis A, Garcia-Keller C, Li H, Kalivas PW. Plasticity in astrocyte subpopulations regulate heroin relapse. Sci Adv. 2022;8:eabo7044.
Octeau JC, Chai H, Jiang R, Bonanno SL, Martin KC, Khakh BS. An optical neuron-astrocyte proximity assay at synaptic distance scales. Neuron. 2018;98:49–66 e49.
Chung W, Ryu MJ, Heo JY, Lee S, Yoon S, Park H, et al. Sevoflurane exposure during the critical period affects synaptic transmission and mitochondrial respiration but not long-term behavior in mice. Anesthesiology. 2017;126:288–99.
Liang X, Jiang M, Xu H, Tang T, Shi X, Dong Y, et al. Maternal sevoflurane exposure increases the epilepsy susceptibility of adolescent offspring by interrupting interneuron development. BMC Med. 2023;21:510.
Zhou B, Zuo YX, Jiang RT. Astrocyte morphology: diversity, plasticity, and role in neurological diseases. CNS Neurosci Ther. 2019;25:665–73.
Santuy A, Rodríguez JR, DeFelipe J, Merchán-Pérez A. Study of the size and shape of synapses in the juvenile rat somatosensory cortex with 3D electron microscopy. eNeuro. 2018;5:ENEURO.0377–17.2017.
Kohtala S, Theilmann W, Suomi T, Wigren HK, Porkka-Heiskanen T, Elo LL, et al. Brief isoflurane anesthesia produces prominent phosphoproteomic changes in the adult mouse hippocampus. ACS Chem Neurosci. 2016;7:749–56.
Badia-Soteras A, Heistek TS, Kater MSJ, Mak A, Negrean A, van den Oever MC, et al. Retraction of astrocyte leaflets from the synapse enhances fear memory. Biol Psychiatry. 2023;94:226–38.
Endo F, Kasai A, Soto JS, Yu X, Qu Z, Hashimoto H, et al. Molecular basis of astrocyte diversity and morphology across the CNS in health and disease. Science. 2022;378:eadc9020.
Bernardinelli Y, Randall J, Janett E, Nikonenko I, Konig S, Jones EV, et al. Activity-dependent structural plasticity of perisynaptic astrocytic domains promotes excitatory synapse stability. Curr Biol. 2014;24:1679–88.
Vaidyanathan TV, Collard M, Yokoyama S, Reitman ME, Poskanzer KE. Cortical astrocytes independently regulate sleep depth and duration via separate GPCR pathways. eLife. 2021;10:e63329.
Soto JS, Neupane C, Kaur M, Pandey V, Wohlschlegel JA, Khakh BS. Astrocyte Gi-GPCR signaling corrects compulsive-like grooming and anxiety-related behaviors in Sapap3 knockout mice. Neuron. 2024;112:3412–23.e3416.
Chen L, Jiao J, Lei F, Zhou B, Li H, Liao P, et al. Ezrin-mediated astrocyte-synapse signaling regulates cognitive function via astrocyte morphological changes in fine processes in male mice. Brain Behav Immun. 2025;124:177–91.
Zhang X, Flores LR, Keeling MC, Sliogeryte K, Gavara N. Ezrin phosphorylation at T567 modulates cell migration, mechanical properties, and cytoskeletal organization. Int J Mol Sci. 2020;21:435.
Lavialle M, Aumann G, Anlauf E, Prols F, Arpin M, Derouiche A. Structural plasticity of perisynaptic astrocyte processes involves ezrin and metabotropic glutamate receptors. Proc Natl Acad Sci USA. 2011;108:12915–9.
Wu XS, Sun JY, Evers AS, Crowder M, Wu LG. Isoflurane inhibits transmitter release and the presynaptic action potential. Anesthesiology. 2004;100:663–70.
Pannasch U, Freche D, Dallerac G, Ghezali G, Escartin C, Ezan P, et al. Connexin 30 sets synaptic strength by controlling astroglial synapse invasion. Nat Neurosci. 2014;17:549–58.
Wang Z, Ma J, Miyoshi C, Li Y, Sato M, Ogawa Y, et al. Quantitative phosphoproteomic analysis of the molecular substrates of sleep need. Nature. 2018;558:435–9.
Brüning F, Noya SB, Bange T, Koutsouli S, Rudolph JD, Tyagarajan SK, et al. Sleep-wake cycles drive daily dynamics of synaptic phosphorylation. Science. 2019;366:eaav3617.
Hu J-J, Liu Y, Yao H, Cao B, Liao H, Yang R, et al. Emergence of consciousness from anesthesia through ubiquitin degradation of KCC2 in the ventral posteromedial nucleus of the thalamus. Nat Neurosci. 2023;26:751–64.
Derouiche A, Geiger KD. Perspectives for Ezrin and Radixin in Astrocytes: Kinases, Functions and Pathology. Int J Mol Sci. 2019;20:3776.
Pelaseyed T, Viswanatha R, Sauvanet C, Filter JJ, Goldberg ML, Bretscher A. Ezrin activation by LOK phosphorylation involves a PIP2-dependent wedge mechanism. eLife. 2017;6:e22759.
Bellesi M, de Vivo L, Tononi G, Cirelli C. Effects of sleep and wake on astrocytes: clues from molecular and ultrastructural studies. BMC Biol. 2015;13:66.
Khakh BS, Sofroniew MV. Diversity of astrocyte functions and phenotypes in neural circuits. Nat Neurosci. 2015;18:942–52.
Nelson LE, Lu J, Guo T, Saper CB, Franks NP, Maze M. The alpha2-adrenoceptor agonist dexmedetomidine converges on an endogenous sleep-promoting pathway to exert its sedative effects. Anesthesiology. 2003;98:428–36.
Korkmazhan E, Dunn AR. The membrane-actin linker ezrin acts as a sliding anchor. Sci Adv. 2022;8:eabo2779.
Kotani N, Akaike N. The effects of volatile anesthetics on synaptic and extrasynaptic GABA-induced neurotransmission. Brain Res Bull. 2013;93:69–79.
Ogawa SK, Tanaka E, Shin MC, Kotani N, Akaike N. Volatile anesthetic effects on isolated GABA synapses and extrasynaptic receptors. Neuropharmacology. 2011;60:701–10.
Oose Y, Miura M, Inoue R, Andou N, Aosaki T, Nishimura K. Imbalanced suppression of excitatory and inhibitory synaptic transmission onto mouse striatal projection neurons during induction of anesthesia with sevoflurane in vitro. Eur J Neurosci. 2012;35:1396–405.
Koh W, Kwak H, Cheong E, Lee CJ. GABA tone regulation and its cognitive functions in the brain. Nat Rev Neurosci. 2023;24:523–39.
Tong K, Song YT, Jing SQ, You Y, Wang SJ, Wu T, et al. Reactive astrocytes mediate postoperative surgery-induced anxiety through modulation of GABAergic signalling in the zona incerta of mice. Br J Anaesth. 2025;134:111–23.
Koh W, Park M, Chun YE, Lee J, Shim HS, Park MG, et al. Astrocytes render memory flexible by releasing D-serine and regulating NMDA receptor tone in the hippocampus. Biol Psychiatry. 2022;91:740–52.
Woo DH, Han KS, Shim JW, Yoon BE, Kim E, Bae JY, et al. TREK-1 and Best1 channels mediate fast and slow glutamate release in astrocytes upon GPCR activation. Cell. 2012;151:25–40.
Lee S, Yoon BE, Berglund K, Oh SJ, Park H, Shin HS, et al. Channel-mediated tonic GABA release from glia. Science. 2010;330:790–6.
Bieda MC, MacIver B. Major role for tonic GABAA conductances in anesthetic suppression of intrinsic neuronal excitability. J Neurophysiol. 2004;92:1658–67.
Rau V, Iyer SV, Oh I, Chandra D, Harrison N, Eger EI, et al. Gamma-aminobutyric acid type A receptor alpha 4 subunit knockout mice are resistant to the amnestic effect of isoflurane. Anesth Analg. 2009;109:1816–22.
Brickley Stephen G, Mody I. Extrasynaptic GABAA receptors: their function in the CNS and implications for disease. Neuron. 2012;73:23–34.
Lee V, Maguire J. The impact of tonic GABAA receptor-mediated inhibition on neuronal excitability varies across brain region and cell type. Front Neural Circuits. 2014;8:3.
Popov A, Brazhe N, Morozova K, Yashin K, Bychkov M, Nosova O, et al. Mitochondrial malfunction and atrophy of astrocytes in the aged human cerebral cortex. Nat Commun. 2023;14:8380.
Popov A, Brazhe A, Denisov P, Sutyagina O, Li L, Lazareva N, et al. Astrocyte dystrophy in ageing brain parallels impaired synaptic plasticity. Aging Cell. 2021;20:e13334.
Verkhratsky A, Augusto-Oliveira M, Pivoriūnas A, Popov A, Brazhe A, Semyanov A. Astroglial asthenia and loss of function, rather than reactivity, contribute to the ageing of the brain. Pflug Arch. 2021;473:753–74.
Verkhratsky A, Rodrigues JJ, Pivoriunas A, Zorec R, Semyanov A. Astroglial atrophy in Alzheimer’s disease. Pflug Arch. 2019;471:1247–61.
Lin SS, Zhou B, Chen BJ, Jiang RT, Li B, Illes P, et al. Electroacupuncture prevents astrocyte atrophy to alleviate depression. Cell Death Dis. 2023;14:343.
Li B, Zhang D, Verkhratsky A. Astrocytes in post-traumatic stress disorder. Neurosci Bull. 2022;38:953–65.
Verkhratsky A, Butt A, Li B, Illes P, Zorec R, Semyanov A, et al. Astrocytes in human central nervous system diseases: a frontier for new therapies. Signal Transduct Target Ther. 2023;8:396.
Pandin P, Estruc I, Van Hecke D, Truong HN, Marullo L, Hublet S, et al. Brain Aging and Anesthesia. J Cardiothorac Vasc Anesth. 2019;33(Suppl 1):S58–s66.
Chemali JJ, Kenny JD, Olutola O, Taylor NE, Kimchi EY, Purdon PL, et al. Ageing delays emergence from general anaesthesia in rats by increasing anaesthetic sensitivity in the brain. Br J Anaesth. 2015;115(Suppl 1(Suppl 1)):i58–i65.
Du J, Plas M, Absalom AR, van Leeuwen BL, de Bock GH. The association of preoperative anxiety and depression with neurocognitive disorder following oncological surgery. J Surg Oncol. 2020;121:676–87.
Subramaniyan S, Terrando N. Neuroinflammation and perioperative neurocognitive disorders. Anesth Analg. 2019;128:781–8.
Bushong EA, Martone ME, Ellisman MH. Maturation of astrocyte morphology and the establishment of astrocyte domains during postnatal hippocampal development. Int J Dev Neurosci. 2004;22:73–86.
Holt MG. Astrocyte heterogeneity and interactions with local neural circuits. Essays Biochem. 2023;67:93–106.
Winchenbach J, Düking T, Berghoff SA, Stumpf SK, Hülsmann S, Nave K-A, et al. Inducible targeting of CNS astrocytes in Aldh1l1-CreERT2 BAC transgenic mice. F1000Research. 2016;5:2934.
Tian T, Lindell SL, Lam M, Mangino MJ. Ezrin functionality and hypothermic preservation injury in LLC-PK1 cells. Cryobiology. 2012;65:60–67.
Perez-Alvarez A, Navarrete M, Covelo A, Martin ED, Araque A. Structural and functional plasticity of astrocyte processes and dendritic spine interactions. J Neurosci. 2014;34:12738–44.
Huang AY, Woo J, Sardar D, Lozzi B, Bosquez Huerta NA, Lin CJ, et al. Region-specific transcriptional control of astrocyte function oversees local circuit activities. Neuron. 2020;106:992–1008.e1009.
Tao CL, Liu YT, Sun R, Zhang B, Qi L, Shivakoti S, et al. Differentiation and Characterization of Excitatory and Inhibitory Synapses by Cryo-electron Tomography and Correlative Microscopy. J Neurosci. 2018;38:1493–510.
Tai SK, Ma J, Leung LS. Medial septal cholinergic neurons modulate isoflurane anesthesia. Anesthesiology. 2014;120:392–402.
Welch P. The use of fast Fourier transform for the estimation of power spectra: a method based on time averaging over short, modified periodograms. IEEE Trans Audio Electroacoustics. 2003;15:70–73.
Särkelä M, Mustola S, Seppänen T, Koskinen M, Lepola P, Suominen K, et al. Automatic analysis and monitoring of burst suppression in anesthesia. J Clin Monit Comput. 2002;17:125–34.
Mariotti L, Losi G, Lia A, Melone M, Chiavegato A, Gómez-Gonzalo M, et al. Interneuron-specific signaling evokes distinctive somatostatin-mediated responses in adult cortical astrocytes. Nat Commun. 2018;9:82.
Perea G, Yang A, Boyden ES, Sur M. Optogenetic astrocyte activation modulates response selectivity of visual cortex neurons in vivo. Nat Commun. 2014;5:3262.
González-Arias C, Sánchez-Ruiz A, Esparza J, Sánchez-Puelles C, Arancibia L, Ramírez-Franco J, et al. Dysfunctional serotonergic neuron-astrocyte signaling in depressive-like states. Mol Psychiatry. 2023;28:3856–73.
Acknowledgements
We are grateful to Xueke Tan and Xixia Li for helping with electron microscopy sample preparation and taking scanning/transmission electron microscopy images at the Center for Biological Imaging (CBI), Institute of Biophysics, Chinese Academy of Science. We are grateful to Dr. Zhian Hu, Dr. Liping Wang, Dr. Min Xu for helpful discussions and valuable comments.
Funding
This work was supported by the Natural Science Foundation of China (Grant no. 82001130 to JR, 81873808 to ZY, 82371281 to ZT, 82401506 to ZB), and partially funded by the Department of Science and Technology of Sichuan Province (22NSFSC2418 to ZB, 2025ZNSFSC1645 to SM) and the 1-3-5 Project for Disciplines of Excellence of West China Hospital of Sichuan University (ZYJC21034 to JR).
Author information
Authors and Affiliations
Contributions
ZB: study investigation, data analysis, article writing, and funding acquisition; LQ: study investigation, data analysis and article writing; SM: study investigation, data analysis, article writing and funding acquisition. LP, LR, YY, LY, LM, LF, LX, JJ, YL, WJ, YL, LD: study investigation and data analysis. ZC, ZX, XH: study design. ZY, ZT, LJ: study design, supervision, funding acquisition, lab resource. JR: study design, supervision, article writing and editing, lab resource and funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All methods were performed in accordance with the relevant guidelines and regulations. All animal experiments and procedures were approved by the Animal Research Committee at the West China Hospital of Sichuan University (20220223103, 20230220023).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, B., Li, Q., Su, M. et al. Astrocyte morphological remodeling regulates consciousness state transitions induced by inhaled general anesthesia. Mol Psychiatry 30, 4006–4022 (2025). https://doi.org/10.1038/s41380-025-02978-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41380-025-02978-2
This article is cited by
-
Research progress and future perspective in anaesthesiology
Anesthesiology and Perioperative Science (2025)
-
Astrocyte morphology: complex or trivial?
Biophysical Reviews (2025)