Abstract
Alterations in cortical morphology have consistently been reported in obsessive-compulsive disorder (OCD). However, the microstructural properties of the cortex in OCD, including intracortical myelination, remain far less explored. The contrast between signal intensity in gray and subjacent white matter from T1-weighted magnetic resonance imaging (MRI), i.e. the gray/white matter contrast (GWC), is linked to intracortical myelination and may offer novel insights into the cortical microstructure of OCD. Here, we compared multivariate patterns of GWC defined from an independent component analysis between 454 adults with OCD and 394 healthy controls from eight international sites. To contextualize GWC results with the macrostructure of gray matter in OCD, we also investigated the association between GWC and each individual’s similarity with the pattern of gray matter morphology derived from ENIGMA-OCD using the Regional Vulnerability Index (RVI). Finally, we investigated the association of GWC with demographic and clinical characteristics of participants with OCD. Individuals with OCD showed significantly higher GWC in occipital and frontal regions relative to healthy controls. Moreover, OCD individuals had elevated OCD RVI, and individuals with a higher OCD RVI showed widespread higher GWC across the cortex. Finally, sexual/religious symptoms in OCD individuals were associated with higher GWC in frontal regions. In conclusion, we present new evidence of cortical microstructural alterations in OCD, with microstructural alterations relating to both the gray matter macrostructure and the clinical presentation of the disorder.
Similar content being viewed by others
Introduction
Obsessive-compulsive disorder (OCD) is a disabling mental disorder characterized by intrusive and anxiety-provoking obsessions as well as repetitive and time-consuming compulsions [1]. The disorder is clinically and etiologically heterogeneous [2]. Although effective psychological and pharmacological treatments for OCD exist, approximately 50% of patients either relapse or do not recover sufficiently after treatment [1]. Understanding the neurobiological underpinnings of the disorder may eventually lead to the development of new biologically informed treatments that may also benefit today’s non-responders [3].
Case-control studies in the Enhancing Neuroimaging and Genetics through Meta‐analysis working group for OCD (ENIGMA-OCD), a consortium spanning more than 50 sites and five continents, have found that individuals with OCD have subtle alterations in both cortical morphology and subcortical volumes [4,5,6]. This includes reduced thickness in parts of the frontoparietal cortex, smaller surface area in the temporal cortex, as well as smaller hippocampus and larger pallidum volumes [6], which are moderated by patient age, age at disease onset, disease duration, and medication use [4,5,6]. Interestingly, recent studies suggest that the neural correlate of OCD may be even more distributed, and include parts of the visual, sensorimotor and primary motor cortices [3]. Indeed, studies have found hyperactivation in the visual cortex during symptom provocation [7], larger volume of the postcentral gyrus [8], and hyperactivation in the postcentral and visual cortices during body-focused processing in individuals with OCD compared to healthy controls [9]. However, far less is known about the microstructural properties of the cortex in individuals with OCD. The neocortex is a layered anatomical structure composed of highly interconnected and specialized neurons in addition to a plethora of glial cells. It also contains myelin, a lipid-rich insulation of the axons ensuring efficient neural signaling, especially in its deeper layers [10]. The intracortical signal intensity in T1-weighted magnetic resonance imaging (MRI), which reflects the speed of tissue proton longitudinal relaxation, is highly sensitive to tissue myelination [11]. Lately, intracortical signal intensities in mental disorders have been studied using gray/white matter contrast (GWC) or the ratio between T1-weighted and T2- weighted signal intensities (T1w/T2w ratio), with findings of altered intracortical signal intensities in both schizophrenia and bipolar disorder [12,13,14].
Few studies have investigated cortical microstructure based on GWC or T1w/T2w ratio in OCD. Preliminary evidence from a small single-site study [15] suggested that GWC may be altered in the occipital, parietal, middle frontal and anterior cingulate cortices in individuals with OCD. However, the study did not have the statistical power to explore associations between GWC and clinical or demographical variables. Individuals with OCD show altered white matter microstructure in the posterior thalamic radiation and sagittal stratum as indicated by less fractional anisotropy and more mean and radial diffusivity, which could reflect aberrant myelination [16]. Further, aberrant developmental trajectories of GWC and magnetization transfer MRI have been linked to compulsivity and impulsivity symptoms in youths from the general population [17, 18]. Finally, genetic studies have found an association between OCD and oligodendrocyte lineage transcription factor 2 (OLIG2), which regulates the development of oligodendrocytes and the production of myelin [19]. Together, these findings suggest that cortical myelination may be affected in OCD, but a direct comparison of GWC between individuals with OCD and healthy controls (HC) in a well-powered study is missing. Moreover, the association between regional changes in cortical T1w signal intensity and clinical characteristics of individuals with OCD remain largely unexplored. Finally, if and how cortical T1w signal intensities relate to morphological changes of cortical and subcortical gray matter in OCD, including cortical thickness and subcortical volumes, has not been addressed.
An increasing number of studies have used machine learning methods on neuroimaging data to classify individuals with OCD versus healthy controls, with the aim of aiding diagnosis in the future [20,21,22]. However, the majority of these studies have not been successful in accurately classifying individuals with or without OCD, likely due to heterogeneity in neurobiology, clinical presentation, developmental stage, and disease stage [20,21,22]. A recently developed alternative is the regional vulnerability index (RVI, 23). RVI attempts to quantify the degree to which an individual’s brain structure or functional connectivity aligns with the established structural or functional pattern of a given disorder, through estimating an individuals’ degree of similarity with the meta-analytical patterns of for instance altered regional cortical thickness, subcortical volume, or white matter microstructure derived from large collaborative projects such as ENIGMA. This method has been used to investigate mental and neurodegenerative disorders, including schizophrenia and OCD [23,24,25]. Interestingly, youths with persistent and distressing psychotic like experiences showed higher schizophrenia RVI, suggesting that RVI may serve as an early brain-based risk score of the disorder [26]. Moreover, higher multimodal or subcortical gray matter RVI were associated with higher symptom load in individuals with schizophrenia [23] and OCD [27], respectively. However, studies investigating how RVI may be associated with cortical microstructure in OCD are lacking.
The present case-control study aimed to investigate cortical microstructure in adult individuals with OCD compared to HC in a multicenter mega-analysis from eight sites. We used independent component analysis (ICA) to identify regional patterns of GWC in the whole sample and compared individuals with OCD and HC. ICA and other dimensionality reduction techniques aid in decomposing the cortex into fewer, distinct anatomical components, and has been used in previous studies of GWC in mental disorders [18, 28, 29]. We also compared cortical GWC between OCD and HC using univariate vertex-wise analyses. Next, we investigated the relationship between GWC and RVI for OCD. Finally, we performed post-hoc within-group (OCD) analyses examining the association of regional GWC with demographic and clinical characteristics of the patients.
Materials and methods
Participants
T1w MRI and clinical data from adult individuals with OCD and HC acquired between 1997 and 2017 by seven international centers participating in the OCD Brain Imaging Consortium (OBIC), as well as Haukeland University Hospital in Norway, were used in the present study, see Supplemental Materials for the full list of consortium members. [30, 31]. Individuals with OCD were recruited through specialist OCD clinics while HC were recruited through advertisements. Exclusion criteria were age under 18 or over 65 years, a current psychotic disorder, recent substance use disorder, developmental disability, and severe somatic or neurological pathology except tics. HC were required to be free from any current axis I psychiatric disorders. After quality control (see Supplemental Materials) the final sample included 454 individuals with OCD (54.8% female, mean age = 32.27 [SD = 9.76]) and 394 HC (49% female, mean age = 30.34 [SD = 9.30]) from Brazil, Japan, the Republic of Korea, the Netherlands, Norway, Spain, and the United Kingdom. Standardized structured interviews were used to diagnose OCD and psychiatric comorbidities [32, 33]. The Yale-Brown Obsessive Compulsive Scale (Y-BOCS) was used to rate the severity of OCD symptoms [34]. For more information regarding demographic and clinical variables, please see Table S1. The study was performed in accordance with the relevant guidelines and regulations where all participants signed written informed consent and each site was granted local ethical approval. The multicenter mega-analysis was approved by the Norwegian Regional Ethical Committee West (#248045).
MRI acquisition and processing
T1w MRI were acquired on 1.5 T or 3 T MRI scanners (See Table S2 and S3 for details regarding MRI manufactures and sequence details). The images were processed using Freesurfer 7.1.1 [35], including non-uniformity correction, segmentation, and cortical reconstruction. We extracted the number of holes or topological deficits in the cortical reconstruction as this measure reflects both the quality of the T1w image and the cortical reconstruction, which are crucial for placing the gray-white matter border [36]. GWC was estimated based on signal intensities sampled from the nonuniform intensity normalized volume (nu.mgz) according to previous studies [12, 18]. Gray matter intensity was sampled at six points starting from 10–60% into the cortical gray matter while white matter intensity was sampled at 10 points from 0.15 mm to 1.5 mm into the subcortical white matter to avoid bias from cortical thickness [12]. The average intensity for each tissue type was then calculated and used to estimate GWC as 100×(white–gray)/[(white+gray)/2]. Higher GWC values reflect greater differences between the tissue signal intensities, while lower values indicate more similar intensities [12, 18]. Higher GWC values may reflect less intracortical myelination, but the exact neurobiological underpinning of GWC remains to be determined and may be multifactorial [10, 37, 38]. We next used ICA to reduce the number of dependent variables from the full cortex-wide map to fewer components using FastICA in MATLAB 2021a. This decomposes the cortex-wide GWC surface map into spatially independent components of cross-subject variation and associated subject weights. We used ICASSO 1.22 to rerun the ICA models 100 times for 5–20 components solutions. Component solutions were evaluated based on the overlap and separability between components, as well as their stability across runs [39], where the seven-component solution was chosen based on its superior stability, minimal overlap, and clean separability compared to other solutions (See Figure S1 for details).
Regional vulnerability index
We used the latest mega-analysis from ENIGMA-OCD as the reference for calculating RVI [6], as this represents the largest dataset of individuals with OCD and HC worldwide. To avoid overlap, we excluded all participants from the present samples from the ENIGMA-OCD cohort and reanalyzed the standardized difference in between the remaining 1389 adult individuals with OCD and 1259 HC from 25 sites within the ENIGMA-OCD. In the ENIGMA-OCD dataset, we quantified the standardized mean difference (Cohen’s d) for cortical thickness between adult individuals with OCD and HC for 34 regions in the Desikan-Killiany atlas (averaged across hemispheres) as well as the subcortical volume of the ventricles, thalamus, caudate nucleus, putamen, pallidum, hippocampus, amygdala and nucleus accumbens (averaged across hemispheres) using linear models adjusted for age, sex, and site [6] (See Table S4). For participants in the present manuscript, RVI was then calculated using the individual’s regional cortical thickness and subcortical volume after regressing out variance explained by age and sex (as well as intracranial volume for subcortical structures). The difference between the individual and the mean HC for each structure was then divided by the standard deviation in HC to produce a normalized difference score for every region. The similarity between the regional difference scores for each participant was then compared to the standardized difference between individuals with OCD, resulting in a Pearson’s correlation coefficient indicating to what extent the person resembled the gray matter morphological pattern of OCD in ENIGMA-OCD. The RVI was estimated separately for cortical thickness and subcortical volume, and the mean RVI across both the subcortex and cortex was used in subsequent analyses. RVI was estimated using the “RVIpkg” package (version 0.3.0) in R.
Statistical analyses
Statistical analyses were performed in R version 4.1.3. Group differences between individuals with OCD and HC and the relation between RVI and the seven GWC components were investigated using linear multilevel models which included age, sex and number of topological deficits as fixed effects covariates and site as a random effects factor. Statistical significance was set at false discovery rate (FDR) q < 0.05 to adjust for multiple comparisons. For components with a significant group difference, we used linear multilevel models with age, sex and number of topological deficits as fixed effects covariates and site as a random effects factor to test for associations with symptom severity (Y-BOCS), presence of specific symptoms (aggression/checking, contamination/cleaning, symmetry/ordering, sexual/religious and hoarding based on previously empirically identified dimensions [2, 40] and work in ENIGMA-OCD [5]), age of onset (dichotomized to under or over 18 years of age), medication status (unmedicated versus medicated at the time of scanning), and comorbid depression or anxiety disorders in individuals with OCD only. Finally, vertex-wise analyses comparing GWC in individuals with OCD versus HC, and the relation with RVI, was performed using Permutation Analysis of Linear Models (PALM, version 1.19) with 1000 permutations, thresholded to corrected p < 0.05 using threshold-free cluster enhancement (TFCE) adjusted for two hemispheres with tail approximation for faster estimation [41]. Here, age, sex, topological deficits and seven dummy variables for the different sites were added as covariates as PALM does not allow for modeling site as a random effects factor. Analyses linking RVI with GWC additionally included diagnosis (OCD vs HC) as a covariate to rule out a confounding effect of the group difference in RVI. All code for this manuscript is available at https://github.com/anderslthorsen/OBIC-GWC. Raw data is not openly available due to ethical and data protection constraints.
Results
ICA-based decomposition of GWC
The mean GWC across all participants was highly similar to previous studies of GWC and T1w/T2w ratio (Fig. 1) [18, 42]. Figure 2 shows the seven independent components estimated from each subject’s cortical GWC, which were largely bilateral and symmetric. The components were labeled as the global (explaining 87.25% of the variance), pre/postcentral-temporal (1.6% of the variance), fronto-occipital (0.97% of the variance), parietal-frontal (0.90% of the variance), insula-cingulate/fusiform (0.35% of the variance), temporal (0.26% of the variance), and fronto-temporal (0.19% of the variance) components according to the strongest positive and negative peaks of the component. We then investigated if individuals scanned at 3 T (one site, n = 74) showed a difference in component weights compared to individuals scanned at 1.5 T (seven sites, n = 774) using linear models adjusted for age, sex, and the number of topological deficits. The results revealed that individuals scanned at 3 T had significantly weaker weights for the global and pre/postcentral components, stronger weights for parietal-frontal, temporal, and fronto-temporal components, while the fronto-occipital and insula-cingulate/fusiform components showed no significant differences compared to 1.5 T sites (Table S5).
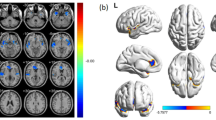
A illustrates the estimation of gray/white matter contrast (GWC). The pial surface is marked in red while the gray-white matter border is marked in white. Blue lines indicate points where gray and white matter signal intensities were extracted. Gray matter signal intensity was measured from six points from 10–60% into gray matter from the gray-white matter border to avoid partial volume effects and contamination from CSF. White matter signal intensity was extracted from 10 points from 0.15 mm to 1.5 mm into the subjacent white matter to avoid bias from cortical thickness. B shows the mean GWC across the cortex, where regions with more similar signal intensities in gray and white matter have lower GWC and are highlighted in red.
Component maps show the 10% strongest vertices except for the first (global) component which shows the 90% strongest vertices due to the component’s distribution across the cortex. In terms of explained variance, the global component explained 87.25%, pre/postcentral-temporal 1.6%, fronto-occipital 0.97%, parietal-frontal 0.90%, insula-cingulate/fusiform 0.35%, temporal 0.26%, and the fronto-temporal 0.19%.
Comparing GWC components in OCD versus HC
Individuals with OCD showed a more negative component weight for the fronto-occipital GWC component in comparison to HC (t = −2.74, FDR-p = 0.046, Cohen’s d = 0.19, Table 1). A leave-one-site-out analysis revealed that the effect size of the difference between individuals with OCD and HC was robust to iteratively leaving out each site from the analysis (t = −2.86 - −2.55, FDR-p = 0.01 - 0.05, d = 0.12 - 0.214), although the finding fell below statistical significance after leaving out the largest site with 81 individuals with OCD and 100 HC (t = −1.80, FDR-p = 0.10, d = 0.14). No significant group differences were found for the other components (Table 1).
Relating GWC components to RVI for OCD
Individuals with OCD showed a significantly higher RVI (t = 3.29, p = 0.001, d = 0.23, Figure S2) for OCD (mean = −0.004, median=0.007 SD = 0.22) than HC (mean = −0.05, median = −0.05, SD = 0.22). We then investigated the relation between RVI and GWC components (after adjusting for the presence of an OCD diagnosis to rule out a confounding effect of the group difference in RVI). These analyses showed that RVI was positively related to the global (t = 6.49, FDR-p < 0.001, β = 0.08), pre/post-motor components (t = 3.82, FDR-p < 0.001, β = 0.09), and negatively related to the temporal GWC component (t = −3.03, FDR-p = 0.01, β = −0.07, Table 1). The results were unchanged in sensitivity analyses not adjusting for the presence of an OCD diagnosis (Fig. 3).
A shows the regional standardized mean difference between individuals with OCD and HC for GWC. B shows the standardized beta for the relation between regional GWC and regional vulnerability index for OCD across all participants.
Vertex-Wise analyses of GWC
Post-hoc vertex-wise analyses comparing individuals with OCD versus HC did not show any significant findings at TFCE-p < 0.05. Inspection of uncorrected findings showed the largest difference between OCD and HC in the right lingual gyrus (Figure S4 and Table S6). RVI was positively associated with GWC across the cortex at TFCE-p < 0.05, with the strongest finding in the bilateral lateral occipital gyrus (Figure S5 and Table S7). The finding was attenuated in sensitivity analyses not adjusting for the presence of an OCD diagnosis, where RVI instead was positively associated with GWC in the bilateral entorhinal cortex, right insula, and right isthmus cingulate (Figure S6 and Table S8). The site with 3 T MRI showed significantly widespread higher GWC across the cortex compared to sites with 1.5 T MRI at TFCE-p < 0.05 (Figure S7 and Table S9).
Associations with demographic and clinical characteristics
Linear mixed models testing the association between demographic and clinical characteristics and the fronto-occipital GWC component did not result in significant group-by-age or group-by-sex interactions. When restricting the analyses of the fronto-occipital GWC component to individuals with OCD only, we found no significant association with total Y-BOCS severity, illness onset under or over 18 years of age, comorbid anxiety or depressive disorders, or medication status. However, a linear mixed model on the symptom dimensions showed a significant positive association between the presence of sexual/religious symptoms and fronto-occipital GWC (t = 2.66, p = 0.01, d = 0.28), but found no significant associations with aggressive/checking, contamination/cleaning, symmetry/ordering or hoarding symptoms. Follow-up vertex-wise analysis showed that sexual/religious symptoms were associated with widespread higher GWC across the cortex (Table S9 and Figure S10).
Discussion
The present study leverages the power of a large multicenter sample to show that adult individuals with OCD present an altered pattern of fronto-occipital cortical microstructure compared to HC, a finding that was driven by higher GWC in occipital and frontal regions in individuals with OCD. Moreover, cortex-wide higher GWC, reflecting a more widespread pattern of microstructural alterations, was found in individuals who also tended to have brain morphology resembling individuals with OCD from an independent cohort. The group difference in fronto-occipital GWC was not significantly related to individual differences in symptom severity, medication status, adult vs childhood onset of OCD or comorbidity. However, the fronto-occipital GWC component was positively associated with having sexual and religious symptoms. Together, the present findings suggest that cortical microstructure may be altered in frontal and occipital areas in OCD, particularly in individuals with sexual/religious obsessions. These findings were further supported when investigating the association between cortical microstructure and RVI, which revealed more global alterations in cortical microstructure in participants whose brain more strongly resembled the structural brain alterations found in ENIGMA-OCD.
Very few previous studies have investigated cortical microstructure in individuals with OCD. This stands in contrast to a high number of studies reporting alterations in cortical morphology in OCD, where the distribution of morphological alterations overlaps with several other mental disorders, including schizophrenia and bipolar disorder [43,44,45,46]. Notably, individuals suffering from schizophrenia and bipolar disorder also show altered GWC in visual and sensorimotor cortices [12, 15]. Interestingly, while medicated individuals with OCD tend to show substantially larger alterations in cortical thickness and surface area than unmedicated individuals [6, 47,48,49], medication status does not appear to influence case-control differences in GWC in OCD or schizophrenia [12, 14]. Accordingly, the group comparisons are likely to capture case-control differences in tissue properties beyond those that are related to the use of medications. However, medication effects should be interpreted with caution as most studies are cross-sectional and lack extensive information on medication history [50].
While the physiological underpinning of GWC remains an active area of investigation, there are reasons to believe that it is influenced by cortical myelination [10, 37, 38]. Cholesterol in myelin is a major determinant of white and gray signal intensities in T1-weighted magnetic resonance images [11], and the relation between myelin and T1w signal intensity in motor and somatosensory cortex is supported by histologically obtained myelin profiles [10, 37, 38]. Notably, intracortical myelination may also be approximated by T1w/T2w ratio, quantitative R1, and magnetization transfer [10]. However, there is evidence for strong spatial overlap between GWC, quantitative R1, and T1w/T2w ratio [51], supporting that GWC can be used to estimate intracortical myelination, reducing the need for more specialized MRI sequences. However, correlations between various MRI and histologically derived myelin profiles are imperfect, suggesting that other neurobiological processes are also at play [10, 52]. For example, some studies found a strong correlation between the T1w/T2w ratio and dendrite density, suggesting that neuronal structure may also influence T1w signal intensities [53]. The relation between cortical macro- and microstructure is complex, with cortical thickness being negatively associated with T1w/T2w ratio and positively associated with GWC [51]. Moreover, the association between micro- and macrostructure is likely heterogenous over the lifespan where increased myelination in childhood may lead to apparent cortical thinning [54], and demyelination in old age may obscure cortical thinning due to the cortex and subadjacent white matter becoming less separable [55]. Recently, multiscale cortical gradients have been found to bridge cortical macrostructure, microstructure, resting-state functional connectivity, cytoarchitecture, and distributions of neurotransmitters [56]. Notably, a cross-disorder pattern of altered cortical thickness in OCD, autism spectrum disorder, attention deficit/hyperactivity disorder, major depression disorder, bipolar disorder, and schizophrenia showed significant overlap with T1w/T2w ratio and functional connectivity gradients [46]. Still, the exact neurobiological underpinning of the observed GWC differences, and their relation to brain macrostructure, remains to be determined and may only be resolved through histological analyses.
The recently developed RVI is meant to translate case-control differences in brain structure or function from large studies into useful markers at the individual level by estimating the individual’s similarity to the disorder’s phenotype at the group level [23, 25]. We are the first to show that RVI for OCD is elevated in individuals with OCD compared to HC, extending previous validations of RVI in schizophrenia, Alzheimer’s disease, and major depressive disorder [23, 25]. We also found that RVI was positively related to GWC across the cortex, though this was attenuated in vertex-wise analyses not adjusting for diagnosis. This may suggest that having a macrostructural pattern more similar to the OCD phenotype is associated with more widespread alterations in cortical myelination.
We did not find an association between the fronto-occipital GWC component and symptom severity in individuals with OCD, but found the presence of sexual/religious symptoms to be associated with the fronto-occipital GWC component. Follow-up vertex-wise analyses showed that individuals with OCD with sexual or religious symptoms had higher GWC in both relatively highly myelinated regions (pre- and postcentral gyri) as well as more lightly myelinated regions such as the dorsal and subgenual anterior cingulate, temporal pole and orbitofrontal cortices. Sexual/religious OCD symptoms have previously been related to lower temporal lobe (including the amygdala), insular and orbitofrontal cortex [57] volumes, functional activation in fronto-temporal regions during symptom provocation, as well as resting-state connectivity in sensorimotor and fronto-limbic circuits [1, 57]. Together, this suggests an association between sexual/religious symptoms and alterations in fronto-limbic regions involved in affective processes across macrostructural, microstructural and functional MRI measures. Interestingly, research has shown that individuals with OCD with taboo obsessions, especially sexual and religious obsessions, may respond less well to standard psychological treatments that are primarily based on exposure with response prevention [58,59,60].
A strength of the present study is the application of a harmonized processing pipeline on multisite raw data, which allows the use of mega-analytic approaches and investigation of the link between individual differences in GWC, demographic and clinical characteristics. However, the present study also has several limitations. Several differences emerged for ICA-based versus vertex-wise analyses. Comparing GWC from the vertex-wise analyses between individuals with OCD and HC revealed no significant results which stands in contrast to the regional differences observed in the ICA-based analyses. Notably, a stronger association between GWC and psychopathology when using ICA-based versus vertex-wise approaches has also been observed in a previous study [18]. A possible explanation for this is the multivariate nature of ICA and other dimensionality reduction techniques, which integrate information across multiple related regions, thereby reducing the number of comparisons and possibly increasing the biological specificity of our analyses [61]. All multivariate techniques require considering the validity of the latent variables, or in this case, independent components. In the present analyses, the components correspond to previously observed patterns reflecting highly versus lightly myelinated regions [10, 42]. Still our findings should be replicated in future large datasets with more harmonized clinical measures and neuroimaging. Multisite studies are likely to be impacted by using different scanners, MRI sequences and heterogeneity between individuals with OCD. We aimed to control for this in the analyses using site-specific intercepts in the linear mixed models. The analyses revealed that sites with 3 T MRI had higher GWC across several components compared to sites with 1.5 T MRI, in line with the findings from a previous study where 25 participants where scanned at both field strengths [62].
Conclusion
GWC is higher in fronto-occipital regions in individuals with OCD compared to HC, which may be attributable to differences in cortical myelination in OCD. The group difference in fronto-occipital GWC was not related to individual differences in age of onset, comorbidity, or medication use, but was associated with sexual and religious symptoms. Finally, individuals more strongly resembling an independently derived macrostructural phenotype of OCD were more likely to have OCD and show a more widespread pattern of microstructural alterations. These results provide new evidence of microstructural alterations in OCD, with the alterations relating to both the cortical macrostructure and the clinical presentation of the disorder.
References
Stein DJ, Costa DLC, Lochner C, Miguel EC, Reddy YCJ, Shavitt RG, et al. Obsessive–compulsive disorder. Nat Rev Dis Primers. 2019;5:1–21.
Mataix-Cols D, Rosario-Campos MC, Leckman JF. A multidimensional model of obsessive-compulsive disorder. Am J Psychiatry. 2005;162:228–38.
Shephard E, Stern ER, van den Heuvel OA, Costa DLC, Batistuzzo MC, Godoy PBG, et al. Toward a neurocircuit-based taxonomy to guide treatment of obsessive-compulsive disorder. Mol Psychiatry. 2021;26:4583–604.
Boedhoe PSW, Schmaal L, Abe Y, Ameis SH, Arnold PD, Batistuzzo MC, et al. Distinct subcortical volume alterations in pediatric and adult ocd: a worldwide meta- and mega-analysis. Am J Psychiatry. 2017;174:60–69.
Boedhoe PSW, Schmaal L, Abe Y, Alonso P, Ameis SH, Anticevic A, et al. Cortical abnormalities associated with pediatric and adult obsessive-compulsive disorder: findings from the ENIGMA obsessive-compulsive disorder working group. Am J Psychiatry. 2018;175:453–62.
Boedhoe PSW, van Rooij D, Hoogman M, Twisk JWR, Schmaal L, Abe Y, et al. Subcortical brain volume, regional cortical thickness, and cortical surface area across disorders: findings from the ENIGMA ADHD, ASD, and OCD working groups. Am J Psychiatry. 2020;177:02019030331.
Thorsen AL, Hagland P, Radua J, Mataix-Cols D, Kvale G, Hansen B, et al. Emotional processing in obsessive-compulsive disorder: a systematic review and meta-analysis of 25 functional neuroimaging studies. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3:563–71.
Subirà M, Sato JR, Alonso P, do Rosário MC, Segalàs C, Batistuzzo MC, et al. Brain structural correlates of sensory phenomena in patients with obsessive-compulsive disorder. J Psychiatry Neurosci. 2015;40:232–40.
Brown C, Shahab R, Collins K, Fleysher L, Goodman WK, Burdick KE, et al. Functional neural mechanisms of sensory phenomena in obsessive-compulsive disorder. J Psychiatr Res. 2019;109:68–75.
Norbom LB, Ferschmann L, Parker N, Agartz I, Andreassen OA, Paus T, et al. New insights into the dynamic development of the cerebral cortex in childhood and adolescence: Integrating macro- and microstructural MRI findings. Prog Neurobiol. 2021;204:102109.
Koenig SH. Cholesterol of myelin is the determinant of gray-white contrast in MRI of brain. Magn Reson Med. 1991;20:285–91.
Jørgensen KN, Nerland S, Norbom LB, Doan NT, Nesvåg R, Mørch-Johnsen L, et al. Increased MRI-based cortical grey/white-matter contrast in sensory and motor regions in schizophrenia and bipolar disorder. Psychol Med. 2016;46:1971–85.
Iwatani J, Ishida T, Donishi T, Ukai S, Shinosaki K, Terada M, et al. Use of T1-weighted/T2-weighted magnetic resonance ratio images to elucidate changes in the schizophrenic brain. Brain Behav. 2015;5:e00399.
Kong L, Herold C, Stieltjes B, Essig M, Seidl U, Wolf RC, et al. Reduced gray to white matter tissue intensity contrast in schizophrenia. PLoS One. 2012;7:e37016.
Niu Q, Li J, Yang L, Huang Z, Niu M, Song X, et al. Association between gray/white matter contrast and white matter microstructural alterations in medication-naïve obsessive-compulsive disorder. Neuroimage Clin. 2022;35:103122.
Piras F, Piras F, Abe Y, Agarwal SM, Anticevic A, Ameis S, et al. White matter microstructure and its relation to clinical features of obsessive-compulsive disorder: findings from the ENIGMA OCD Working Group. Transl Psychiatry. 2021;11:173.
Ziegler G, Hauser TU, Moutoussis M, Bullmore ET, Goodyer IM, Fonagy P, et al. Compulsivity and impulsivity traits linked to attenuated developmental frontostriatal myelination trajectories. Nat Neurosci. 2019;22:992–9.
Norbom LB, Doan NT, Alnæs D, Kaufmann T, Moberget T, Rokicki J, et al. Probing brain developmental patterns of myelination and associations with psychopathology in youths using gray/white matter contrast. Biol Psychiatry. 2019;85:389–98.
Stewart SE, Platko J, Fagerness J, Birns J, Jenike E, Smoller JW, et al. A genetic family-based association study of OLIG2 in obsessive-compulsive disorder. Arch Gen Psychiatry. 2007;64:209–14.
Bruin WB, Abe Y, Alonso P, Anticevic A, Backhausen LL, Balachander S, et al. The functional connectome in obsessive-compulsive disorder: resting-state mega-analysis and machine learning classification for the ENIGMA-OCD consortium. Mol Psychiatry. 2023;28:4307–19.
Kim BG, Kim G, Abe Y, Alonso P, Ameis S, Anticevic A, et al. White matter diffusion estimates in obsessive-compulsive disorder across 1653 individuals: machine learning findings from the ENIGMA OCD Working Group. Mol Psychiatry. 2024;29:1063–74.
Bruin WB, Taylor L, Thomas RM, Shock JP, Zhutovsky P, Abe Y, et al. Structural neuroimaging biomarkers for obsessive-compulsive disorder in the ENIGMA-OCD consortium: medication matters. Transl Psychiatry. 2020;10:342.
Kochunov P, Fan F, Ryan MC, Hatch KS, Tan S, Jahanshad N, et al. Translating ENIGMA schizophrenia findings using the regional vulnerability index: association with cognition, symptoms, and disease trajectory. Hum Brain Mapp. 2022;43:566–75.
Ma Y, Kvarta MD, Adhikari BM, Chiappelli J, Du X, van der Vaart A, et al. Association between brain similarity to severe mental illnesses and comorbid cerebral, physical, and cognitive impairments. Neuroimage. 2023;265:119786.
Kochunov P, Ryan MC, Yang Q, Hatch KS, Zhu A, Thomopoulos SI, et al. Comparison of regional brain deficit patterns in common psychiatric and neurological disorders as revealed by big data. Neuroimage Clin. 2021;29:102574.
Karcher NR, Modi H, Kochunov P, Gao S, Barch DM. Regional vulnerability indices in youth with persistent and distressing psychoticlike experiences. JAMA Netw Open. 2023;6:e2343081.
Koch K, Manrique DR, Gigl S, Ruan H, Gürsel DA, Rus-Oswald G, et al. Decoding obsessive-compulsive disorder: the regional vulnerability index and its association with clinical symptoms. Biol Psychiatry Cogn Neurosci Neuroimaging. 2025;10:877–82.
Norbom LB, Rokicki J, Eilertsen EM, Wiker T, Hanson J, Dahl A, et al. Parental education and income are linked to offspring cortical brain structure and psychopathology at 9–11 years. JCPP Adv. 2024;4:e12220.
Makowski C, Lewis JD, Lepage C, Malla AK, Joober R, Lepage M, et al. Structural associations of cortical contrast and thickness in first episode psychosis. Cereb Cortex. 2019;29:5009–21.
de Wit SJ, Alonso P, Schweren L, Mataix-Cols D, Lochner C, Menchon JM, et al. Multicenter voxel-based morphometry mega-analysis of structural brain scans in obsessive-compulsive disorder. Am J Psychiatry. 2014;171:340–9.
Fouche JP, du Plessis S, Hattingh C, Roos A, Lochner C, Soriano-Mas C, et al. Cortical thickness in obsessive-compulsive disorder: multisite mega-analysis of 780 brain scans from six centres. Br J Psychiatry. 2017;210:67–74.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The mini-international neuropsychiatric interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59:22–33.
First M, Spitzer RL, Gibbon M, Williams JB. Structured Clinical Interview for DSM-IV-TR Axis I Disorders: New York State Psychiatric Institute, New York; 2002
Goodman WK, Price LH, Rasmussen SA, Mazure C, Fleischmann RL, Hill CL, et al. The yale-brown obsessive compulsive scale. I. development, use, and reliability. Arch Gen Psychiatry. 1989;46:1006–11.
Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I Segmentation Surf reconstruction Neuroimage. 1999;9:179–94.
Rosen AFG, Roalf DR, Ruparel K, Blake J, Seelaus K, Villa LP, et al. Quantitative assessment of structural image quality. Neuroimage. 2018;169:407–18.
Stüber C, Morawski M, Schäfer A, Labadie C, Wähnert M, Leuze C, et al. Myelin and iron concentration in the human brain: a quantitative study of MRI contrast. Neuroimage. 2014;93:95–106.
Eickhoff S, Walters NB, Schleicher A, Kril J, Egan GF, Zilles K, et al. High-resolution MRI reflects myeloarchitecture and cytoarchitecture of human cerebral cortex. Hum Brain Mapp. 2005;24:206–15.
Himberg J, Hyvarinen A, editors. Icasso: software for investigating the reliability of ICA estimates by clustering and visualization. 2003 IEEE XIII Workshop on Neural Networks for Signal Processing (IEEE Cat No03TH8718); 2003 17–19 Sept. 2003.
Bloch MH, Landeros-Weisenberger A, Rosario MC, Pittenger C, Leckman JF. Meta-analysis of the symptom structure of obsessive-compulsive disorder. Am J Psychiatry. 2008;165:1532–42.
Winkler AM, Ridgway GR, Webster MA, Smith SM, Nichols TE. Permutation inference for the general linear model. Neuroimage. 2014;92:381–97.
Glasser MF, Goyal MS, Preuss TM, Raichle ME, Van Essen DC. Trends and properties of human cerebral cortex: correlations with cortical myelin content. Neuroimage. 2014;93:165–75.
Opel N, Goltermann J, Hermesdorf M, Berger K, Baune BT, Dannlowski U. Cross-Disorder analysis of brain structural abnormalities in six major psychiatric disorders: a secondary analysis of mega- and meta-analytical findings from the ENIGMA consortium. Biol Psychiatry. 2020;88:678–86.
Radonjić NV, Hess JL, Rovira P, Andreassen O, Buitelaar JK, Ching CRK, et al. Structural brain imaging studies offer clues about the effects of the shared genetic etiology among neuropsychiatric disorders. Mol Psychiatry. 2021;26:2101–10.
Hettwer MD, Larivière S, Park BY, van den Heuvel OA, Schmaal L, Andreassen OA, et al. Coordinated cortical thickness alterations across six neurodevelopmental and psychiatric disorders. Nat Commun. 2022;13:6851.
Park BY, Kebets V, Larivière S, Hettwer MD, Paquola C, van Rooij D, et al. Multiscale neural gradients reflect transdiagnostic effects of major psychiatric conditions on cortical morphology. Commun Biol. 2022;5:1024.
van Erp TGM, Walton E, Hibar DP, Schmaal L, Jiang W, Glahn DC, et al. Cortical brain abnormalities in 4474 individuals with schizophrenia and 5098 control subjects via the enhancing neuro imaging genetics through meta analysis (ENIGMA) consortium. Biol Psychiatry. 2018;84:644–54.
Schmaal L, Hibar DP, Sämann PG, Hall GB, Baune BT, Jahanshad N, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA major depressive disorder working group. Mol Psychiatry. 2017;22:900–9.
Hibar DP, Westlye LT, Doan NT, Jahanshad N, Cheung JW, Ching CRK, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA bipolar disorder working group. Mol Psychiatry. 2018;23:932–42.
van den Heuvel OA, Boedhoe PSW, Bertolin S, Bruin WB, Francks C, Ivanov I, et al. An overview of the first 5 years of the ENIGMA obsessive-compulsive disorder working group: The power of worldwide collaboration. Hum Brain Mapp. 2020;43:23–36.
Parent O, Olafson E, Bussy A, Tullo S, Blostein N, Dai A, et al. High spatial overlap but diverging age-related trajectories of cortical magnetic resonance imaging markers aiming to represent intracortical myelin and microstructure. Hum Brain Mapp. 2023;44:3023–44.
van der Weijden CWJ, García DV, Borra RJH, Thurner P, Meilof JF, van Laar PJ, et al. Myelin quantification with MRI: a systematic review of accuracy and reproducibility. Neuroimage. 2021;226:117561.
Righart R, Biberacher V, Jonkman LE, Klaver R, Schmidt P, Buck D, et al. Cortical pathology in multiple sclerosis detected by the T1/T2-weighted ratio from routine magnetic resonance imaging. Ann Neurol. 2017;82:519–29.
Natu VS, Gomez J, Barnett M, Jeska B, Kirilina E, Jaeger C, et al. Apparent thinning of human visual cortex during childhood is associated with myelination. Proc Natl Acad Sci USA. 2019;116:20750–9.
Grydeland H, Vértes PE, Váša F, Romero-Garcia R, Whitaker K, Alexander-Bloch AF, et al. Waves of maturation and senescence in micro-structural mri markers of human cortical myelination over the lifespan. Cereb Cortex. 2019;29:1369–81.
Bazinet V, Hansen JY, Misic B. Towards a biologically annotated brain connectome. Nat Rev Neurosci. 2023;24:747–60.
Thorsen AL, Kvale G, Hansen B, van den Heuvel OA. Symptom dimensions in obsessive-compulsive disorder as predictors of neurobiology and treatment response. Curr Treat Options Psychiatry. 2018;5:182–94.
Alonso P, Menchon JM, Pifarre J, Mataix-Cols D, Torres L, Salgado P, et al. Long-term follow-up and predictors of clinical outcome in obsessive-compulsive patients treated with serotonin reuptake inhibitors and behavioral therapy. J Clin Psychiatry. 2001;62:535–40.
Mataix-Cols D, Marks IM, Greist JH, Kobak KA, Baer L. Obsessive-compulsive symptom dimensions as predictors of compliance with and response to behaviour therapy: results from a controlled trial. Psychother Psychosom. 2002;71:255–62.
Rufer M, Fricke S, Moritz S, Kloss M, Hand I. Symptom dimensions in obsessive-compulsive disorder: prediction of cognitive-behavior therapy outcome. Acta Psychiatr Scand. 2006;113:440–6.
Makowski C, Nichols TE, Dale AM. Quality over quantity: powering neuroimaging samples in psychiatry. Neuropsychopharmacology. 2024;50:58–66.
Phal PM, Usmanov A, Nesbit GM, Anderson JC, Spencer D, Wang P, et al. Qualitative comparison of 3-T and 1.5-T MRI in the evaluation of epilepsy. AJR Am J Roentgenol. 2008;191:890–5.
Acknowledgements
The present mega-analysis was supported by grants from the Trond Mohn Foundation, Helse Bergen, and University of Bergen. Data collection for studies in OBIC were supported by the Dutch Organization for Scientific Research (NWO) (grants 912-02-050, 907-00-012, 940-37- 018 and 916.86.038); the Carlos III Health Institute (PI09/01331, PI10/01753, PI10/01003, CP10/00604, PI13/01958 and CIBER-CB06/03/0034); the Agency for Administration of University and Research (AGAUR, Barcelona; 2009SGR1554); a ‘Miguel Servet’ contract from the Carlos III Health Institute (CP10/00604); Ministry of Education, Culture, Sports, Science, and Technology (Japan) Grants-in-Aid for Young Scientists (24791223); Wellcome Trust project grant 064846; a grant from the Foundation for the Support of Research in the State of São Paulo (FAPESP), a FAPESP scholarship (2005/ 04206-6); a National Research Foundation of Korea grant funded by the Korean government (Ministry of Education, Science, and Technology, 2012-0005150); Italian ministry of Health (Ricerca Corrente 23) and grants from Helse Vest Health Authority (911754 and 911880). Part of the data in the present paper have previously been presented at the Norwegian Research School in Neuroscience in September 2022 and at Amsterdam Neuroscience in October 2022.
Author information
Authors and Affiliations
Consortia
Contributions
Conceptualization: ALT, VB, OTO. Methodology: ALT, VB, DA, OTO. Software: ALT, OTO, DA. Formal analysis: ALT, OTO, VB. Investigation: ALT, VB, DA, DMC, JSK, JMM, YA, YS, MLP, BH, MH, JR, FB, BPB, YC, DD, YH, KK, TN, ELN, HBS, FP, DFT, ERS, ZW, JB, PM, JCB, CL, DJS, OAvdH, OTO. Data curation: ALT, VB, DA, DMC, JSK, JMM, YA, YS, MLP, BH, MH, JR, FB, BPB, YC, DD, YH, KK, TN, ELN, HBS, FP, DFT, ERS, ZW, JB, PM, JCB, CL, DJS, OAvdH, OTO. Writing – Original Draft: ALT, VB, OTO. Writing – Review & Editing: ALT, VB, DA, DMC, JSK, JMM, YA, YS, MLP, BH, MH, JR, FB, BPB, YC, DD, YH, KK, TN, ELN, HBS, FP, DFT, ERS, ZW, JB, PM, JCB, CL, DJS, OAvdH, OTO. Visualization: ALT. Supervision: ALT, OTO, OAvdH.
Corresponding author
Ethics declarations
Competing interests
Prof Mataix-Cols reports receiving royalties from UpToDate, Inc, outside the current work. Thorsen, Brecke, Alnæs, Kwon, Menchon, Abe, Sakai, Phillips, Hansen, Hoexter, Reddy, Benedetti, Brennan, Cheng, Denys, Hirano, Koch, Nakao, Nurmi, Simpson, Piras, Tolin, Stern, Wang, Buitelaar, Morgado, Beucke, Lochner, Stein, van den Heuvel, and Ousdal reported no biomedical financial interests or potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Thorsen, A.L., Brecke, V., Alnæs, D. et al. Altered frontal and occipital cortical microstructure in obsessive-compulsive disorder - a multisite mega-analysis. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03500-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03500-y