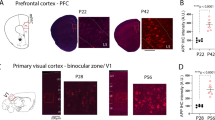
Abstract
Olfactory dysfunction has emerged as a promising target for the early diagnosis and treatment of Alzheimer’s disease (AD). However, the mechanisms underlying neural circuit disruption associated with olfactory dysfunction in AD remain poorly understood. We conducted single-cell RNA sequencing (RNA-seq) and ex vivo electrophysiological studies to determine the link between olfactory memory in AD and dynamic synaptic transmission disorders in PCx-IL engram cell circuits. Clinical functional magnetic resonance imaging (fMRI) data revealed that connectivity between the piriform cortex (PCx) and the infralimbic cortex (IL) was impaired during the early mild cognitive impairment (MCI) stage of AD. Optogenetic stimulation of IL-projecting PCx engram neurons successfully improved olfactory memory retrieval in 5xFAD mice. In addition, single-cell RNA sequencing was employed to investigate the mechanisms of damage in IL engram cells, which revealed increased glutamate expression and impaired synaptic function as key alterations. Guided by single-cell sequencing data, we analyzed glutamatergic synaptic transmission in the PCx-IL engram cell circuit in 5xFAD mice. These results indicated dynamic impairments in AMPA receptor-associated synaptic transmission within this circuit. Optical long-term potentiation (LTP) of synaptic transmission restored directional engram synaptic transmission and prevented olfactory memory decline. Therefore, dynamic impairment of synaptic transmission in the PCx-IL engram cell circuit underlies the early decline in olfactory memory in AD. Impairment of PCx-IL functional connectivity may represent a new target for the diagnosis and treatment of early-stage AD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Jia J, Ning Y, Chen M, Wang S, Yang H, Li F, et al. Biomarker changes during 20 years preceding Alzheimer’s disease. N Engl J Med. 2024;390:712–22.
Guo Y, You J, Zhang Y, Liu WS, Huang YY, Zhang YR, et al. Plasma proteomic profiles predict future dementia in healthy adults. Nat Aging. 2024;4:247–60.
Yan Y, Aierken A, Wang C, Song D, Ni J, Wang Z, et al. A potential biomarker of preclinical Alzheimer’s disease: the olfactory dysfunction and its pathogenesis-based neural circuitry impairments. Neurosci Biobehav Rev. 2022;132:857–69.
Murphy C. Olfactory and other sensory impairments in Alzheimer disease. Nat Rev Neurol. 2019;15:11–24.
Dan X, Wechter N, Gray S, Mohanty JG, Croteau DL, Bohr VA. Olfactory dysfunction in aging and neurodegenerative diseases. Ageing Res Rev. 2021;70:101416.
Dong Y, Li Y, Liu K, Han X, Liu R, Ren Y, et al. Anosmia, mild cognitive impairment, and biomarkers of brain aging in older adults. Alzheimer’s Dement. 2023;19:589–601.
Meyer C, Niedermeier T, Feyen PLC, Strübing FL, Rauchmann BS, Karali K, et al. Early locus coeruleus noradrenergic axon loss drives olfactory dysfunction in Alzheimer’s disease. Nat Commun. 2025;16:7338.
Chen B, Wang Q, Zhong X, Mai N, Zhang M, Zhou H, et al. Structural and functional abnormalities of olfactory-related regions in subjective cognitive decline, mild cognitive impairment, and Alzheimer’s disease. Int J Neuropsychopharmacol. 2022;25:361–74.
Tang Y, Yan Y, Mao J, Ni J, Qing H. The hippocampus associated GABAergic neural network impairment in early-stage of Alzheimer’s disease. Ageing Res Rev. 2023;86:101865.
Chen M, Wang C, Lin Y, Chen Y, Xie W, Huang X, et al. Dorsal raphe nucleus-hippocampus serotonergic circuit underlies the depressive and cognitive impairments in 5×FAD male mice. Transl Neurodegener. 2024;13:34.
Schoonover CE, Ohashi SN, Axel R, Fink AJP. Representational drift in primary olfactory cortex. Nature. 2021;594:541–6.
Terral G, Harrell E, Lepousez G, Wards Y, Huang D, Dolique T, et al. Endogenous cannabinoids in the piriform cortex tune olfactory perception. Nat Commun. 2024;15:1230.
Zhang L, Geng C, Li S, Tang Q, Liu P, Liu W, et al. Anterior piriform cortex dysfunction underlies autism spectrum disorders-related olfactory deficits in Fmr1 conditional deletion mice. Neuropsychopharmacology. 2024;50:798–807.
Takeuchi S, Shimizu K, Fukada Y, Emoto K. The circadian clock in the piriform cortex intrinsically tunes daily changes of odor-evoked neural activity. Commun Biol. 2023;6:332.
Calu DJ, Roesch MR, Stalnaker TA, Schoenbaum G. Associative encoding in posterior piriform cortex during odor discrimination and reversal learning. Cereb Cortex. 2007;17:1342–9.
Liu K, Aierken A, Liu M, Parhat N, Kong W, Yin X, et al. The decreased astrocyte-microglia interaction reflects the early characteristics of Alzheimer’s disease. iScience. 2024;27:109281.
Wang X, Hu M, Xie Q, Geng C, Jin C, Ren W, et al. Amyloid β oligomers disrupt piriform cortical output via a serotonergic pathway. Neurobiol Aging. 2023;121:64–77.
Martínez-García I, Hernández-Soto R, Villasana-Salazar B, Ordaz B, Peña-Ortega F. Alterations in piriform and bulbar activity/excitability/coupling upon amyloid-β administration in vivo related to olfactory dysfunction. J Alzheimer’s Dis. 2021;82:S19–s35.
Negrey JD, Dobbins DL, Howard TD, Borgmann-Winter KE, Hahn CG, Kalinin S, et al. Transcriptional profiles in olfactory pathway-associated brain regions of African green monkeys: associations with age and Alzheimer’s disease neuropathology. Alzheimer’s & Dementia. 2022;8:e12358.
Sun W, Liu Z, Jiang X, Chen MB, Dong H, Liu J, et al. Spatial transcriptomics reveal neuron-astrocyte synergy in long-term memory. Nature. 2024;627:374–81.
Poll S, Mittag M, Musacchio F, Justus LC, Giovannetti EA, Steffen J, et al. Memory trace interference impairs recall in a mouse model of Alzheimer’s disease. Nat Neurosci. 2020;23:952–8.
Lazarov O, Gupta M, Kumar P, Morrissey Z, Phan T. Memory circuits in dementia: the engram, hippocampal neurogenesis and Alzheimer’s disease. Prog Neurobiol. 2024;236:102601.
Gallo DA, Foster KT, Wong JT, Bennett DA. False recollection of emotional pictures in Alzheimer\“s disease. Neuropsychologia. 2010;48:3614–8.
Perusini JN, Cajigas SA, Cohensedgh O, Lim SC, Pavlova IP, Donaldson ZR, et al. Optogenetic stimulation of dentate gyrus engrams restores memory in Alzheimer’s disease mice. Hippocampus. 2017.
Roy DS, Arons A, Mitchell TI, Pignatelli M, Ryan TJ, Tonegawa S. Memory retrieval by activating engram cells in mouse models of early Alzheimer’s disease. Nature. 2016;531:508–12.
LaMontagne PJ, Benzinger TLS, Morris JC, Keefe S, Hornbeck R, Xiong C et al. OASIS-3: longitudinal neuroimaging, clinical, and cognitive dataset for normal aging and Alzheimer disease. medRxiv. 2019; 2019.2012.2013.19014902.
Dong Z, Han H, Li H, Bai Y, Wang W, Tu M, et al. Long-term potentiation decay and memory loss are mediated by AMPAR endocytosis. J Clin Invest. 2015;125:234–47.
Audronyte E, Pakulaite-Kazliene G, Sutnikiene V, Kaubrys G. Odor discrimination as a marker of early Alzheimer’s disease. J Alzheimer’s Dis. 2023;94:1169–78.
Igeta Y, Hemmi I, Yuyama K, Ouchi Y. Odor identification score as an alternative method for early identification of amyloidogenesis in Alzheimer’s disease. Sci Rep. 2024;14:4658.
Chatterjee P, Pedrini S, Doecke JD, Thota R, Villemagne VL, Doré V, et al. Plasma Aβ42/40 ratio, p-tau181, GFAP, and NfL across the Alzheimer’s disease continuum: a cross-sectional and longitudinal study in the AIBL cohort. Alzheimer’s Dement. 2023;19:1117–34.
O’Leary TP, Stover KR, Mantolino HM, Darvesh S, Brown RE. Intact olfactory memory in the 5xFAD mouse model of Alzheimer’s disease from 3 to 15 months of age. Behav Brain Res. 2020;393:112731.
Roberts ER, Dossat AM, Del Mar Cortijo M, Brundin P, Wesson DW. Alterations in odor hedonics in the 5XFAD Alzheimer’s disease mouse model and the influence of sex. Behav Neurosci. 2020;134:407–16.
Du Y, Du Y, Zhang Y, Huang Z, Fu M, Li J, et al. MKP-1 reduces Aβ generation and alleviates cognitive impairments in Alzheimer’s disease models. Signal Transduct Target Ther. 2019;4:58.
Du Y, Fu M, Huang Z, Tian X, Li J, Pang Y, et al. TRPV1 activation alleviates cognitive and synaptic plasticity impairments through inhibiting AMPAR endocytosis in APP23/PS45 mouse model of Alzheimer’s disease. Aging Cell. 2020;19:e13113.
Sasmita AO, Ong EC, Nazarenko T, Mao S, Komarek L, Thalmann M, et al. Parental origin of transgene modulates amyloid-β plaque burden in the 5xFAD mouse model of Alzheimer’s disease. Neuron. 2025;113:838–46.e834.
Son G, Yoo SJ, Kang S, Rasheed A, Jung DH, Park H, et al. Region-specific amyloid-β accumulation in the olfactory system influences olfactory sensory neuronal dysfunction in 5xFAD mice. Alzheimer’s Res Ther. 2021;13:4.
Ziegler-Waldkirch S, Friesen M, Loreth D, Sauer JF, Kemna S, Hilse A, et al. Seed-induced Aβ deposition alters neuronal function and impairs olfaction in a mouse model of Alzheimer’s disease. Mol Psychiatry. 2022;27:4274–84.
Williamson MR, Kwon W, Woo J, Ko Y, Maleki E, Yu K, et al. Learning-associated astrocyte ensembles regulate memory recall. Nature. 2024;637:478–86.
Song D, Wang D, Yang Q, Yan T, Wang Z, Yan Y, et al. The lateralization of left hippocampal CA3 during the retrieval of spatial working memory. Nat Commun. 2020;11:2901.
Chen BK, Murawski NJ, Cincotta C, McKissick O, Finkelstein A, Hamidi AB, et al. Artificially enhancing and suppressing hippocampus-mediated memories. Curr Biol. 2019;29:1885–94.e1884.
Choi JH, Sim SE, Kim JI, Choi DI, Oh J, Ye S, et al. Interregional synaptic maps among engram cells underlie memory formation. Science. 2018;360:430–5.
Redondo RL, Kim J, Arons AL, Ramirez S, Liu X, Tonegawa S. Bidirectional switch of the valence associated with a hippocampal contextual memory engram. Nature. 2014;513:426–30.
Nabavi S, Fox R, Proulx CD, Lin JY, Tsien RY, Malinow R. Engineering a memory with LTD and LTP. Nature. 2014;511:348–52.
Lacagnina AF, Brockway ET, Crovetti CR, Shue F, McCarty MJ, Sattler KP, et al. Distinct hippocampal engrams control extinction and relapse of fear memory. Nat Neurosci. 2019;22:753–61.
Li G, Chen Y, Chaudhary S, Li CS, Hao D, Yang L, et al. Sleep dysfunction mediates the relationship between hypothalamic-insula connectivity and anxiety-depression symptom severity bidirectionally in young adults. Neuroimage. 2023;279:120340.
Li G, Zhong D, Li B, Chen Y, Yang L, Li CR. Sleep deficits inter-link lower basal forebrain-posterior cingulate connectivity and perceived stress and anxiety bidirectionally in young men. Int J Neuropsychopharmacol. 2023;26:879–89.
Li G, Dong Y, Chen Y, Li B, Chaudhary S, Bi J, et al. Drinking severity mediates the relationship between hypothalamic connectivity and rule-breaking/intrusive behavior differently in young women and men: an exploratory study. Quant Imaging Med Surg. 2024;14:6669–83.
Zhang Z, Li G, Song Z, Han Y, Tang X. Relationship among number of close friends, subclinical geriatric depression, and subjective cognitive decline based on regional homogeneity of functional magnetic resonance imaging data. Front Aging Neurosci. 2022;14:978611.
Zhou G, Lane G, Cooper SL, Kahnt T, Zelano C. Characterizing functional pathways of the human olfactory system. eLife. 2019;8:e47177.
Nett KE, LaLumiere RT. Infralimbic cortex functioning across motivated behaviors: can the differences be reconciled? Neurosci Biobehav Rev. 2021;131:704–21.
Poldrack RA, Fletcher PC, Henson RN, Worsley KJ, Brett M, Nichols TE. Guidelines for reporting an fMRI study. NeuroImage. 2008;40:409–14.
Wen Y, Dong Z, Liu J, Axerio-Cilies P, Du Y, Li J, et al. Glutamate and GABA(A) receptor crosstalk mediates homeostatic regulation of neuronal excitation in the mammalian brain. Signal Transduct Target Ther. 2022;7:340.
Acknowledgements
We thank the Biological and Medical Engineering Core Facilities of Beijing Institute of Technology for supplying our experimental equipment. MRI data were provided by OASIS-3: Principal Investigators: T. Benzinger, D. Marcus, J. Morris; NIH P50 AG00561, P30 NS09857781, P01 AG026276, P01 AG003991, R01 AG043434, UL1 TR000448, R01 EB009352.
Funding
This work was supported by Shenzhen Municipal Key Project for Basic Research (Grant No. JCYJ20241202130511015), Shenzhen Natural Science Foundation General Project (Grant No. JCYJ20230807142704008), Open Project of the State Key Laboratory of Brain Cognition and Brain-inspired Intelligence Technology (Grant No. SKLBI-K2025002), Guangdong Province General Higher Education Institution Innovative Research Team Project (Natural Sciences) (Grant No. 2024KCXTD016) and.National Natural Science Foundation of China (Grant No. 82371446, 32371030, 32571170, 82501439), Natural Science Foundation of Hebei Province, H2024206232, Technology Research Program of Hebei Provincial Health Commission Grant No. ZF2024142. the Ministry of Science and Technology of China (STI2030-Major Projects 2022ZD0206800), the Scientific Project of Beijing Life Science Academy (2023000CB0010), the scientific research program of innovation platform in State Tobacco Monopoly Administration.
Author information
Authors and Affiliations
Contributions
Y Yan, DS, GFL, ZFD and HQ conceived and designed the studies and wrote the papers. Y Yan, YHT, YJ and YJD. performed optogenetics and behavioral tests. DYL, YJQ, carried out fibre photometry recordings and immunohistochemistry and cell counting. Y Yan, JJie L, YYan P, performed electrophysiology. DS, GL, XY L analyzed the data and revised papers. JM, ZW, HL and YC performed imaging and revised papers. All authors contributed to the data analysis and presentation in the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
Committee on animal Research. The research consent to institutional ethical regulations: Biological and Medical Ethics Committee, Minzu University of China. Ethical approval for the OASIS-3 dataset was obtained from the Institutional Review Board of Washington University, and all participants provided written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, Y., Song, D., Li, G. et al. The dynamic impairment of synaptic transmission in the PCx-IL engram circuit contributes to early olfactory memory decline in Alzheimer’s disease. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03550-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03550-2