Abstract
Cognitive-behavioral therapy (CBT) is a primary treatment for depression. Although previous research has underscored the significant roles of white matter (WM) alterations and maladaptive parenting in depression risk, their associations with CBT response remain largely unknown. This longitudinal study investigated the interplay of WM integrity changes over time, treatment response, and parenting style in patients with depression. Diffusion-tensor-imaging and clinical data were assessed in n = 65 (55% female) patients with depression before and after 20 CBT sessions and n = 65 (68% female) healthy controls (HC) in a naturalistic design. Linear-mixed-effect models compared changes in fractional anisotropy (FA) between groups and tested associations between FA changes and symptom changes. It was investigated whether parenting style predicts depressive symptoms at follow-up and whether FA changes mediate this association. Patients showed differential FA changes over time in the corpus callosum and corona radiata compared to HC (ptfce-FWE = 0.008). Increases in FA in the corpus callosum, corona radiata and superior longitudinal fasciculus were linked to symptom improvement after CBT in patients (ptfce-FWE = 0.023). High parental care (pFDR = 0.010) and low maternal overprotection (pFDR = 0.001) predicted fewer depressive symptoms at follow-up. The association between maternal overprotection and depressive symptoms at follow-up was mediated by FA changes (pFDR = 0.044). Robustness checks—controlling for outliers, non-linear age effects, clinical characteristics, and patient subgroups—supported these results. Overall, patients with depression show changes in WM integrity following CBT, which are linked to treatment response. The results highlight the significance of early life adversities and related microstructural changes in the effectiveness of CBT for treating depression.
Similar content being viewed by others
Introduction
Depression is one of the most prevalent mental diseases worldwide, with an estimated lifetime prevalence of 17% [1]. Depression is characterized by chronicity and recurrence, leading to severe impairment in social and occupational domains [2]. Cognitive-behavioral therapy (CBT) is a first-line psychotherapeutic treatment for depression that focuses on the identification and modification of dysfunctional cognitions and behaviors [3]. While previous studies have demonstrated the superiority of CBT compared to other antidepressant treatments in the long-term [4], there is still a substantial number of patients who do not benefit from CBT [5, 6]. Investigating the neural underpinnings of CBT and determining predictors of treatment outcome might help to improve our understanding of CBT and optimize antidepressant treatment options.
Neuroimaging research in depression has demonstrated microstructural alterations in white matter (WM) pathways responsible for connecting and transferring information between different regions of the human brain. Fractional anisotropy (FA) is a widely used measure in diffusion tensor imaging (DTI) that reflects the degree of directionality in water diffusion within white matter. Higher FA values generally indicate greater alignment of axonal fibers along the longitudinal axis and suggest intact microstructural organization, including myelin integrity. Cross-sectional DTI studies have identified lower FA in patients with depression compared to healthy controls (HC) in the corpus callosum, superior longitudinal fasciculus, and corona radiata [7, 8], with alterations most pronounced in individuals suffering from recurrent depression [9, 10]. To date, only few longitudinal DTI studies in patients with depression have been conducted [11,12,13], yielding a rather inconsistent pattern of results. However, recent evidence suggests that patients with depression show reductions in FA compared to HC over a 2-year follow-up interval [14]. Regarding antidepressant treatment, prospective DTI studies have revealed increases [15,16,17,18] and decreases [19, 20] of WM microstructural integrity after electroconvulsive or psychopharmacological therapy, with different DTI metrics associated with treatment response [16, 17, 20, 21]. Despite the high clinical relevance of CBT in treating depression, no DTI study has yet investigated the microstructural underpinnings of CBT and their associations with treatment response in depression.
Treatment response and microstructural correlates might be influenced by several environmental factors. For instance, childhood experiences of parental neglect and overprotection are important risk factors for the development and maintenance of depression [22,23,24]. Studies have reported that low parental care and high parental overprotection are associated with more depressive symptoms [25, 26] and severe disease courses [23, 27]. Moreover, these factors may also correlate with a lower likelihood of remission following antidepressant treatment [23, 28]. For example, Asano and colleagues demonstrated that high maternal overprotection is associated with a lower response to psychotherapy [28]. However, this finding was limited by small sample sizes and lacking correction for clinical measures (e.g., current psychiatric medication, comorbidities, previous disease course before treatment). At the biological level, poor parenting style has been linked to altered cortisol response [29, 30] and lower FA in healthy individuals [31]. This interplay between disease progression, parenting style, and WM microstructural integrity may potentially affect responsiveness to antidepressant treatments such as CBT and warrants investigation.
This prospective and naturalistic DTI study aimed to investigate the longitudinal associations between parenting style, changes in WM microstructural integrity, and treatment response following CBT in patients with depression. To this end, longitudinal changes in FA in patients with depression, receiving CBT during the interval, compared to HC were examined at the whole-brain level (analysis 1). Based on the heterogeneity of previous longitudinal findings, it was hypothesized that patients show differential changes in FA in the corpus callosum, superior longitudinal fasciculus, and corona radiata over time compared to HC. Furthermore, associations between FA changes and symptom changes after CBT in patients with depression were tested (analysis 2), with an increase in FA expected to be associated with symptom improvement after CBT. Lastly, the role of parenting style in the relationship between FA and CBT response in patients was investigated (analysis 3). Consistent with previous studies, it was assumed that better (i.e., more caring and less controlling) parenting style predicts fewer depressive symptoms after CBT and that this association is mediated by FA changes.
Materials and methods
Participants
This prospective and naturalistic study included n = 65 HC and n = 65 patients with depression from the ongoing study of the Prevention and Intervention Neuroimaging Cohort (PINC) (Table 1, Supplement 1). All patients received CBT during the study interval and were measured shortly before treatment initiation (i.e., waiting list or trial phase; baseline) and after approximately 20 sessions (M = 20.83, SD = 3.70) of naturalistic CBT (excluding the trial phase; follow-up) (Supplement 2). HC underwent assessments at equivalent time points (interscan interval in months: Mpatients = 8.09, SDpatients = 2.30; MHC = 8.19, SDHC = 1.72; p = 0.782). At both time points, participants completed a DTI measurement, a clinical interview and the Hamilton Depression Rating Scale (HDRS, [32]) to assess current depressive symptoms. At baseline, the Parental Bonding Instrument (PBI) was used, which evaluates participants’ perceptions of their caregivers’ parenting styles (four scores: maternal care, paternal care, maternal overprotection, paternal overprotection) before age 16, with higher scores reflecting higher parental care or higher overprotection [33]. Further, patients were included if they fulfilled criteria for an acute or partially remitted major depressive disorder, acute dysthymia or acute adjustment disorder with depressed mood. HC were included if they had no history of mental disorders. Psychiatric diagnoses were determined using the Structured Clinical Interview for the Diagnostic and Statistical Manual of Mental Disorders, Version 4, Text Revision (DSM-IV-TR) for Axis I disorders (SCID-I) by trained raters [34]. Patients were recruited from the psychotherapeutic outpatient unit of the University of Muenster between August 18, 2017, and September 26, 2022. HC were recruited through public notices and newspaper advertisements. The study received approval from the ethics committee of the medical faculty of the University of Münster (Amendment of 2016-173-f-S; 2020-205-f-S), and all experiments and procedures adhered to ethical guidelines and regulations. Written informed consent was obtained from all participants, who also received financial compensation.
DTI preprocessing
DTI data were measured using a 3 T magnetic resonance imaging (MRI) scanner (Prisma, Siemens, Erlangen, Germany). Preprocessing was performed in FSL6.0.1 (http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/, FMRIB, Oxford Center for Functional MRI of the Brain, University of Oxford, Department of Clinical Neurology, John Radcliffe Hospital, Oxford, United Kingdom) [35,36,37], using a longitudinal preprocessing stream (Supplement 3, Supplement 4) While this study focuses on FA, results on further diffusion metrics are reported in Supplement 5.
Statistical analyses
Demographic and clinical data were analyzed using R studio (version 4.0.2; R Core Team, 2020). To investigate the influence of CBT on the severity of depressive symptoms, a random intercepts fixed slopes linear mixed-effect (LME) model was performed in patients with HDRS as outcome variable and time (baseline vs. follow-up) as predictor variable, while correcting for age. Fixed slopes were used for all LME models, as the inclusion of random slopes did not result in a significant improvement in model fit (Supplement 6). Furthermore, for LME models, we did not control for variables that do not change between measurements, as these are already considered by the random intercepts of participants. Neuroimaging analyses were conducted voxel-wise at the whole-brain level using tract-based-spatial-statistics (TBSS, correction: TFCE, 5000 permutations for p < 0·05 FWE-correction, two-sided when based on non-directional hypotheses and one-sided when based on directional hypotheses) [38]. While tractography-based methods approaches have allowed for tract reconstruction at the subject level [39], we chose TBSS as it is a widely accepted tool [8] and offers a fully automated voxel-wise analysis of the whole brain. This approach minimizes the risk of overlooking patterns or relationships beyond predefined regions. In contrast, region of interest-based approaches are limited by potential biases in prior region selection [40, 41].
Analysis 1: differences in FA changes between patients with depression and HC
To test whether FA changes over time differ between patients versus HC, a random intercepts fixed slopes LME model was conducted in FSL with FA as outcome variable and time (baseline vs. follow-up) and diagnosis (HC vs. depression) as predictor variables, while controlling for total intracranial volume (TIV) and age. The main effect of time and the diagnosis×time interaction were analyzed. Since the LME model in FSL does not provide meaningful cross-sectional contrasts, cross-sectional group analyses for baseline and follow-up data were conducted with separate general linear models.
Analysis 2: FA changes and symptom changes after CBT
To analyze the association between FA changes and symptom changes after CBT, another random intercepts fixed slopes LME model was performed in patients with FA as outcome variable and time as well as HDRS as predictor variables, controlling for TIV and age. The HDRS×time interaction was analyzed. As an exploratory approach, it was further investigated whether responders (i.e., patients who achieved full or partial remission after CBT) and non-responders (i.e., patients who were still acutely depressed after CBT) differed in 1) longitudinal FA changes over the interval; and 2) cross-sectional FA at baseline or follow-up. Therefore, a random intercepts fixed slopes LME model was calculated with FA as outcome variable, time and response (non-responders vs. responders) as predictor variables, TIV, age, and remission status at baseline as covariates. The response×time interaction was investigated. For cross-sectional analyses, two separate general linear models were calculated (one for baseline and one for follow-up) with FA from each timepoint as outcome variable, response as predictor variable, and TIV, age, sex, and remission status at baseline as covariates. The main effect of response was investigated for each model. See Supplement 7 for a detailed description of the definition of remission status.
Analysis 3: parenting style, FA changes, and symptom severity after CBT
To test whether parenting style is associated with symptom changes after CBT and whether this association is mediated by FA changes, mediation analyses were performed in patients using a bootstrapping approach implemented in the macro PROCESS (http://www.processmacro.org). Parenting style was entered as predictor variable, mean ΔFA derived from significant clusters of analysis 2 as mediator, and HDRS at follow-up as outcome variable into the model. Separate mediation models were calculated for each of the four PBI scores, with false discovery rate (FDR) correction applied to correct for multiple testing. HDRSbaseline, TIVbaseline, ΔTIV, agebaseline, and interscan interval were included as covariates.
Results
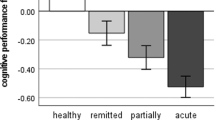
HDRS scores significantly decreased in patients from baseline to follow-up (t(1,63) = -5.62, p < .001, sr2 = 0.143), indicating symptom improvement after CBT. N = 51 (78.5%) patients achieved full or partial remission (responders) and n = 14 (21.5%) patients remained in an acute depressive state (non-responders).
Analysis 1: FA changes between patients with depression and HC
The analysis revealed a significant diagnosis×time interaction in the corpus callosum and corona radiata (ptfce-FWE = .006, sr2 = 0.010, k = 1445 voxel in 2 clusters, Supplement 8, Supplementary Tables 1–2). Post-hoc-t-contrasts showed that patients showed greater decreases in FA following CBT compared to HC (ptfce-FWE = .008, sr2 = 0.008, k = 2999 voxel in 1 cluster, Fig. 1, Supplement 8, Supplementary Tables 1–2). There was no significant main effect of time (ptfce-FWE = .532). Neither at baseline (ptfce-FWE = .576), nor at follow-up (ptfce-FWE = .314), could cross-sectional group differences be observed.
A This figure shows the distribution, means, individual measurements, and slopes of FA, derived from the significant t-contrast, for patients with depression and HC at baseline and follow-up. Each dot represents one participant. Patients with depression showed more decline in FA than HC over time. B Interaction effect of time and diagnosis. Mean FA values of the significant clusters were extracted using ‘fslstats’ in FSL and displayed onto the SPM152 template in the x = −-21, y = –35, z = 38 plane in MNI space using MRIcroGL. Red-yellow represents voxels, in which a significant interaction effect was found. FA fractional anisotropy.
Analysis 2: FA changes and symptom changes after CBT
The analysis of FA and symptom changes after CBT yielded a significant HDRS×time interaction in the corpus callosum, superior longitudinal fasciculus, and corona radiata: Symptom improvement after CBT was associated with increases in FA after CBT (ptfce-FWE = .023, sr2 = 0.034, k = 4741 voxel in 1 cluster, Fig. 2, Supplement 8, Supplementary Tables 1–2). The exploratory analysis revealed a significant response×time interaction in the corpus callosum and corona radiata: responders showed greater increase in FA over time compared to non-responders (ptfce-FWE = .016, sr2 = 0.012, k = 614 voxel in 1 cluster, Supplement 8, Supplementary Tables 1–2). Furthermore, responders compared to non-responders showed higher FA at baseline in the corpus callosum, corona radiata, and superior longitudinal fasciculus (ptfce-FWE = .020, sr2 = 0.255, k = 1165 voxel in 1 cluster, Supplement 8, Supplementary Tables 1–2) as well as follow-up in widespread fiber tracts, mainly affecting the corpus callosum, corona radiata, and superior longitudinal fasciculus (ptfce-FWE = 0.004, sr2 = 0.407, k = 24271 voxel in 1 cluster, Supplement 8, Supplementary Tables 1–2).
A Scatterplot depicting the longitudinal association between symptom improvement, measured with the Hamilton Depression Rating Scale (HDRS) and FA changes derived from the significant t-contrast in patients with depression. Symptom improvements over time were positively associated with FA changes. B Interaction effect of time and symptom improvement. Mean FA values of the significant clusters were extracted using ‘fslstats’ in FSL and displayed onto the SPM152 template in the x = 17, y = 20, z = 43 plane in MNI space using MRIcroGL. Red-yellow represents voxels, in which a significant interaction effect was found. CBT cognitive behavioral therapy, FA fractional anisotropy.
Analysis 3: parenting style, FA changes, and symptom severity after CBT
The four mediation analyses within patients showed a negative association between HDRS at follow-up and maternal care (total effect: β = –0.316, SE = 0.079, t = –2.77, pFDR = 0.010) as well as paternal care (total effect: β = -0.354, SE = 0.083, t = –2.77, pFDR = 0.010), and a positive association between HDRS at follow-up and maternal overprotection (total effect: β = 0.421, SE = 0.081, t = 3.74, pFDR = 0.001), but not paternal overprotection (total effect: pFDR = 0.989). This indicates that high maternal and paternal care as well as low maternal overprotection prospectively predict fewer depressive symptoms at follow-up. ΔFA was also negatively associated with HDRS at follow-up (maternal care: β = –0.531, SE = 65.881, t = –5.53, pFDR < 0.001; paternal care: β = –0.586, SE = 59.706, t = –6.63, pFDR < 0.001; maternal overprotection: β = –0.492, SE = 67.976, t = –4.97, pFDR < 0.001; paternal overprotection: β = –0.623, SE = 64.164, t = –6.55, pFDR < 0.001), indicating that FA increases over time are associated with fewer depressive symptoms at follow-up. Furthermore, a significant positive indirect (mediated) effect of maternal overprotection on HDRS at follow-up through ΔFA (β = 0.187, SE = 0.053, pFDR = 0.044, 95%-CI [0.041, 0.326], Fig. 3) was observed. The direct effect of maternal overprotection on HDRS at follow-up was not significant (β = 0.234, SE = 0.073, pFDR = 0.050, puncorrected = 0.025, 95%-CI [0.021, 0.318]), indicating that the association between maternal overprotection and depressive symptoms at follow-up tended to be fully mediated by FA changes. No indirect (mediated) effect of ΔFA was found for the associations between maternal care (95%-CI [-0.274, 0.027]), paternal care (95%-CI [-0.212, 0.112]), or paternal overprotection (95%-CI [–0.073, 0.287]) with HDRS at follow-up.
The figure depicts the mediator model with maternal overprotection as the predictor variable, fractional anisotropy changes (follow-up-baseline) as the mediator variable, and depressive symptom severity at follow-up as the outcome variable in patients with depression. standardized coefficients and standard errors for each path of the mediation model are presented. Note that c represents the total effect, c‘ the direct effect, and ab the indirect effect. *indicates significance at pFDR < 0.05.
Robustness checks
Several robustness checks (Supplement 9, Supplementary Tables 3–17) for main results were conducted, including the correction for outliers, non-linear age effects, additional covariates at baseline and clinical characteristics in patients (e.g., current medication adherence, comorbidities, previous disease course). Furthermore, analyses were repeated excluding patients suffering from acute dysthymia or acute adjustment disorder at baseline (n = 6). Overall, robustness checks revealed a comparable pattern of results.
Discussion
This longitudinal study investigated changes in WM microstructural integrity in patients with depression undergoing naturalistic CBT relative to HC, as well as the link of these changes with CBT response and parenting style. Patients and HCs experienced differential changes in FA over time, with patients showing reductions in FA in the corpus callosum and corona radiata, fiber tracts commonly affected in depression [8, 42, 43]. Notably, these FA reductions were mainly driven by patients not responding to CBT. In contrast, increases in FA were associated with symptom improvement after CBT. Moreover, responders had higher FA both before and after CBT compared to non-responders. Finally, high levels of parental care as well as low levels of maternal overprotection predicted fewer depressive symptoms after CBT. The association between low maternal overprotection and fewer depressive symptoms was mediated by FA changes. Robustness checks, including corrections for outliers, non-linear age effects, clinical characteristics, and the exclusion of specific patient subgroups, consistently revealed a comparable pattern of results, underscoring the reliability of the findings.
Aligning with prior research documenting declines in FA in patients with depression during the disease course compared to HC [11, 14], this study observed a significant decrease in FA in patients during CBT. While this finding may appear counterintuitive, similar inconsistencies in WM microstructural integrity changes have been noted in studies on antidepressant treatments [15,16,17,18,19,20]. One possible explanation is that the cumulative effects of prolonged depression and chronic stress, a key factor in the pathophysiology of depression [44, 45], prior to treatment, characterized by heightened hypothalamic-pituitary-adrenal axis activity and elevated glucocorticoid levels, may have precipitated neurodegeneration [44] and compromised WM fiber integrity [46, 47]. These detrimental effects could still be detectable as a delayed consequence during CBT, despite the intervention’s focus on reducing stress-inducing thought patterns.
At the same time, further analyses revealed that symptom improvement following CBT correlates with FA increases within the corpus callosum, superior longitudinal fasciculus, and corona radiata among patients with depression. This suggests that the overall FA decreases in patients compared to HC might be largely driven by the CBT non-responders, as demonstrated by our exploratory analysis comparing responders versus non-responders. Alternatively, the coexistence of responders and non-responders, each exhibiting distinct trajectories in WM integrity, may underlie the net FA decrease, thereby complicating the interpretation of these group-level results. Future research should investigate these subgroup effects more thoroughly to clarify the interplay between WM microstructure changes, chronic stress, and differential treatment responses. Observations of increased FA associated with CBT response suggest enhanced fiber coherence and organization [17]. The anterior corona radiata includes fibers that link limbic, thalamic and prefrontal regions [18, 48], crucial for emotion and cognitive processing [49,50,51]. Additionally, the superior longitudinal fasciculus plays a role in modulating the dorsolateral prefrontal cortex [52, 53]. Disruptions in WM connectivity may contribute to diminished top-down cognitive control, possibly leading to rumination [54] and emotion regulation deficits [49]. Reduced corpus callosum volume has been linked to diminished working memory capacity and processing speed [55], mirroring cognitive disruptions observed in depression [56]. Consequently, increases in FA of these fiber tracts may reflect improvements in top-down emotion and cognitive processing, thereby potentially highlighting their relevance for improvements in fronto-limbic dysfunctions [57,58,59] and the antidepressant mechanism. These results cautiously suggest that FA decreases may be attributed to the course of the disease of depression, while FA increases could indicate a positive effect of CBT in those responding to treatment. Additionally, responders showed higher baseline FA compared to non-responders, raising the speculation that pre-treatment FA could play a role in predicting treatment response. To determine whether these differential FA changes during CBT are ultimately attributable to the therapy itself and/or the progression of the disease over time, an untreated patient control group is essential in future research. Furthermore, while baseline FA shows potential as a predictive marker, individualized prediction requires the application of machine learning approaches in larger, multicenter studies.
In line with previous studies showing a link between poor parenting style and lower pharmacotherapy [23, 60] and CBT [28] responses, the findings demonstrate that high maternal and paternal care and low maternal overprotection are predictive of fewer depressive symptoms after CBT. This observation may be attributed to the development of secure attachments during childhood, potentially enabling individuals to develop effective emotion regulation strategies and robust coping mechanisms against adversities [61,62,63]. Such factors may likely enhance the efficacy of CBT, potentially augmenting responsiveness to techniques such as cognitive restructuring [61]. Additionally, a balance of high parental care and low overprotection could foster autonomy and self-efficacy [64], contributing to lower psychological distress [63] and serving as a protective factor against depressive symptoms [65, 66]. Asano and colleagues [28] also highlight the relevance of the therapeutic relationship in CBT’s success [67,68,69,70], suggesting that less controlling parenting styles may foster better therapeutic relationships, thereby improving treatment outcome.
Crucially, the relationship between low maternal overprotection and fewer depressive symptoms following CBT was mediated by changes in FA. These novel findings suggest that high maternal overprotection may result in decreases in WM microstructural integrity, subsequently impacting the depressive symptom severity after CBT and thereby CBT efficacy. Overprotective parenting may limit a child’s experiential learning and independent stress management [62]. This potentially affects the development and functioning of neural circuits essential for stress regulation and emotional control [71] and reduces brain plasticity. The effectiveness of CBT may depend on whether these neural pathways can be improved or compensated for during therapy despite the profound impact of poor parenting styles. Besides, individuals with a background of high parental overprotection might perceive therapy as a greater stressor, leading to heightened stress and thereby cortisol responses [29, 30]. Mothers, often being the primary caregivers, spend more time with their children than fathers [72]. This increased exposure means that a mother’s parenting style could have a more profound influence on the child’s developmental outcomes [72, 73].
This study has several strengths including its longitudinal design, the examination of a well-characterized clinical sample, inclusion of a HC group, and the integration of comprehensive clinical and imaging data. However, some limitations must be acknowledged: First, the PBI, a retrospective self-report questionnaire, is prone to negative recall biases, potentially distorting its impact on subsequent mental health outcomes. Nonetheless, empirical evidence highlights the temporal stability of the PBI [74] and this study demonstrated robustness against outliers and multiple sensitivity checks. Second, the PBI captures only a limited range of caregiving and environmental factors influencing white matter development, excluding aspects like parental mental health, socioeconomic status, and exposure to chronic stress, which should be considered when interpreting the findings. Third, the inclusion of patients with different depressive diagnoses and partial remission adds heterogeneity to the patient group, potentially explaining findings of lower FA in depression relative to HC cross-sectionally neither at baseline or at follow-up, which is in contrast to findings from prior cross-sectional studies [7, 8, 14]. This heterogeneity, however, offers a more realistic representation of individuals in our healthcare system seeking CBT for depression. Future research should consider including a waitlist or active control group and larger subgroups differentiated by remission status to evaluate the effects of CBT, remission status, and therapy-independent fluctuations in depressive symptoms on FA changes. Fourth, six patients with acute dysthymia or adjustment disorder at baseline were included, though there is evidence suggesting that individuals with dysthymia may show less responsiveness to therapy [75]. However, our robustness checks indicate that the results remain consistent even when accounting for varying levels of symptom severity at baseline, or when excluding patients with dysthymia and adjustment disorder. Fifth, while the naturalistic study design poses challenges in controlling factors during CBT, it is noteworthy that the supervised psychotherapists in training adhered to established manuals and national care guidelines for depression, ensuring a higher level of comparability in the administered treatment. Sixth, while TBSS offers several advantages, it is limited by its inability to reconstruct individual fiber tracts. Future studies could benefit from integrating tractography-based analyses to complement TBSS. Lastly, the mediation analysis was conducted on a relatively small sample, which may have resulted in insufficient statistical power to detect significant effects in the other parenting dimensions.
Overall, this study underscores the significance of WM microstructural integrity in depression and suggests that changes in WM microstructural integrity could serve as a potential neural mechanism linked to symptom improvement after CBT. Moreover, the findings indicate that positive parenting styles—characterized by high levels of care and low levels of overprotection—are predictive of fewer depressive symptoms following CBT, mediated by WM microstructural integrity changes. The results highlight the relevance of parenting styles and related microstructural changes in treating depression with CBT.
Data availability
The data of this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Vos T, Lim SS, Abbafati C, Abbas KM, Abbasi M, Abbasifard M, et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:1204–22.
Greenberg P, Chitnis A, Louie D, Suthoff E, Chen S-Y, Maitland J, et al. The economic burden of adults with major depressive disorder in the United States (2019). Adv Ther. 2023;40:4460–79.
Beck AT. Cognitive Therapy of Depression. Guilford Press; 1979.
Hofmann SG, Asnaani A, Vonk IJJ, Sawyer AT, Fang A. The efficacy of cognitive behavioral therapy: a review of meta-analyses. Cogn Ther Res. 2012;36:427–40.
Akil H, Gordon J, Hen R, Javitch J, Mayberg H, McEwen B, et al. Treatment resistant depression: a multi-scale, systems biology approach. Neurosci Biobehav Rev. 2018;84:272–88.
Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163:1905–17.
Flinkenflügel K, Meinert S, Thiel K, Winter A, Goltermann J, Strathausen L, et al. Negative stressful life events and social support are associated with white matter integrity in depressed patients and healthy controls: a diffusion tensor imaging study. Biol Psychiatry. 2023;94:650–60.
van Velzen, Kelly LS, Isaev S, Aleman D, Aftanas LI A, Bauer J, et al. White matter disturbances in major depressive disorder: a coordinated analysis across 20 international cohorts in the ENIGMA MDD working group. Mol Psychiatry. 2020;25:1511–25.
Chen G, Hu X, Li L, Huang X, Lui S, Kuang W, et al. Disorganization of white matter architecture in major depressive disorder: a meta-analysis of diffusion tensor imaging with tract-based spatial statistics. Sci Rep. 2016;6:21825.
Meinert S, Leehr EJ, Grotegerd D, Repple J, Förster K, Winter NR, et al. White matter fiber microstructure is associated with prior hospitalizations rather than acute symptomatology in major depressive disorder. Psychol Med. 2022;52:1166–74.
Doolin K, Andrews S, Carballedo A, McCarthy H, O’Hanlon E, Tozzi L, et al. Longitudinal diffusion weighted imaging of limbic regions in patients with major depressive disorder after 6 years and partial to full remission. Psychiatry Res: Neuroimaging. 2019;287:75–86.
Repple J, Zaremba D, Meinert S, Grotegerd D, Redlich R, Förster K, et al. Time heals all wounds? A 2-year longitudinal diffusion tensor imaging study in major depressive disorder. J Psychiatry Neurosci. 2019;44:407–13.
Shen X, Adams MJ, Ritakari TE, Cox SR, McIntosh AM, Whalley HC. White matter microstructure and its relation to longitudinal measures of depressive symptoms in mid- and late life. Biol Psychiatry. 2019;86:759–68.
Flinkenflügel K, Meinert S, Hirtsiefer C, Grotegerd D, Thiel K, Winter A, et al. Associations between white matter microstructure and cognitive decline in major depressive disorder versus controls in Germany: a prospective case-control cohort study. The Lancet Psychiatry. 2024;11:899–909.
Belge J-B, Mulders PCR, Van Diermen L, Schrijvers D, Sabbe B, Sienaert P, et al. White matter changes following electroconvulsive therapy for depression: a multicenter ComBat harmonization approach. Transl Psychiatry. 2022;12:517.
He X, Pueraro E, Kim Y, Garcia CM, Maas B, Choi J, et al. Association of white matter integrity with executive function and antidepressant treatment outcome in patients with late-life depression. Am J Geriatr Psychiatry. 2021;29:1188–98.
Lyden H, Espinoza RT, Pirnia T, Clark K, Joshi SH, Leaver AM, et al. Electroconvulsive therapy mediates neuroplasticity of white matter microstructure in major depression. Transl Psychiatry. 2014;4:e380–e380.
Seiger R, Gryglewski G, Klöbl M, Kautzky A, Godbersen GM, Rischka L, et al. The influence of acute SSRI administration on white matter microstructure in patients suffering from major depressive disorder and healthy controls. Int J Neuropsychopharmacol. 2021;24:542–50.
Dong Q, Liu J, Zeng L, Fan Y, Lu X, Sun J, et al. State-independent microstructural white matter abnormalities in major depressive disorder. Front Psychiatry. 2020;11:431.
Repple J, Meinert S, Bollettini I, Grotegerd D, Redlich R, Zaremba D, et al. Influence of electroconvulsive therapy on white matter structure in a diffusion tensor imaging study. Psychological Med. 2020;50:849–56.
Korgaonkar MS, Williams LM, Song YJ, Usherwood T, Grieve SM. Diffusion tensor imaging predictors of treatment outcomes in major depressive disorder. Br J Psychiatry. 2014;205:321–8.
Kidd KN, Prasad D, Cunningham JEA, de Azevedo Cardoso T, Frey BN. The relationship between parental bonding and mood, anxiety and related disorders in adulthood: A systematic review and meta-analysis. J Affect Disord. 2022;307:221–36.
Klein DN, Arnow BA, Barkin JL, Dowling F, Kocsis JH, Leon AC, et al. Early adversity in chronic depression: clinical correlates and response to pharmacotherapy. Depression Anxiety. 2009;26:701–10.
Yap MBH, Jorm AF. Parental factors associated with childhood anxiety, depression, and internalizing problems: a systematic review and meta-analysis. J Affect Disord. 2015;175:424–40.
Eun JD, Paksarian D, He J-P, Merikangas KR. Parenting style and mental disorders in a nationally representative sample of US adolescents. Soc Psychiatry Psychiatr Epidemiol. 2018;53:11–20.
Grotmol KS, Ekeberg Ø, Finset A, Gude T, Moum T, Vaglum P, et al. Parental bonding and self-esteem as predictors of severe depressive symptoms: a 10-year follow-up study of Norwegian physicians. J Nerv Ment Dis. 2010;198:22–27.
Saffer BY, Glenn CR, David Klonsky E. Clarifying the relationship of parental bonding to suicide ideation and attempts. Suicide Life-Threatening Behav. 2015;45:518–28.
Asano M, Esaki K, Wakamatsu A, Kitajima T, Narita T, Naitoh H, et al. Maternal overprotection score of the Parental Bonding Instrument predicts the outcome of cognitive behavior therapy by trainees for depression. Psychiatry Clin Neurosci. 2013;67:340–4.
Chen L, Du H, Zilioli S, Zhao S, Chi P. Effects of parental care and overprotection on adolescents’ diurnal cortisol profiles. Horm Behav. 2022;140:105121.
Shirtcliff EA, Skinner ML, Obasi EM, Haggerty KP. Positive parenting predicts cortisol functioning six years later in young adults. Dev Sci. 2017;20:e12461.
Farber MJ, Kim MJ, Knodt AR, Hariri AR. Maternal overprotection in childhood is associated with amygdala reactivity and structural connectivity in adulthood. Dev Cogn Neurosci. 2019;40:100711.
Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62.
Parker G, Tupling H, Brown LB. A parental bonding instrument. Br J Med Psychol. 1979;52:1–10.
Wittchen H-U, Wunderlich U, Gruschwitz S, Zaudig M. SKID I. Strukturiertes Klinisches Interview für DSM-IV. Achse I: Psychische Störungen. Interviewheft und Beurteilungsheft. Eine deutschsprachige, erweiterte Bearb. d. amerikanischen Originalversion des SKID I. 1997. 1997.
Jenkinson M, Beckmann CF, Behrens TEJ, Woolrich MW, Smith SM. FSL. Neuroimage. 2012;62:782–90.
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TEJ, Johansen-Berg H, et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage. 2004;23:S208–219.
Woolrich MW, Jbabdi S, Patenaude B, Chappell M, Makni S, Behrens T, et al. Bayesian analysis of neuroimaging data in FSL. Neuroimage. 2009;45:S173–186.
Smith SM, Nichols TE. Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. Neuroimage. 2009;44:83–98.
Farquharson S, Tournier J-D, Calamante F, Fabinyi G, Schneider-Kolsky M, Jackson GD, et al. White matter fiber tractography: why we need to move beyond DTI: Clinical article. J Neurosurg. 2013;118:1367–77.
Smith SM, Jenkinson M, Johansen-Berg H, Rueckert D, Nichols TE, Mackay CE, et al. Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. NeuroImage. 2006;31:1487–505.
Jeurissen B, Descoteaux M, Mori S, Leemans A. Diffusion MRI fiber tractography of the brain. NMR Biomed. 2019;32:e3785.
Murphy ML, Frodl T. Meta-analysis of diffusion tensor imaging studies shows altered fractional anisotropy occurring in distinct brain areas in association with depression. Biol Mood Anxiety Disord. 2011;1:3.
Wise T, Radua J, Nortje G, Cleare AJ, Young AH, Arnone D. Voxel-based meta-analytical evidence of structural disconnectivity in major depression and bipolar disorder. Biol Psychiatry. 2016;79:293–302.
Lupien SJ, McEwen BS, Gunnar MR, Heim C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci. 2009;10:434–45.
Willner P. The chronic mild stress (CMS) model of depression: history, evaluation and usage. Neurobiol Stress. 2017;6:78–93.
Jauregui-Huerta F, Ruvalcaba-Delgadillo Y, Gonzalez-Castañeda R, Garcia-Estrada J, Gonzalez-Perez O, Luquin S. Responses of glial cells to stress and glucocorticoids. Curr Immunol Rev. 2010;6:195–204.
Poletti S, Aggio V, Brioschi S, Bollettini I, Falini A, Colombo C, et al. Impact of early and recent stress on white matter microstructure in major depressive disorder. J Affect Disord. 2018;225:289–97.
Olivo G, Wiemerslage L, Swenne I, Zhukowsky C, Salonen-Ros H, Larsson E-M, et al. Limbic-thalamo-cortical projections and reward-related circuitry integrity affects eating behavior: A longitudinal DTI study in adolescents with restrictive eating disorders. PLOS ONE. 2017;12:e0172129.
Drevets WC, Price JL, Furey ML. Brain structural and functional abnormalities in mood disorders: implications for neurocircuitry models of depression. Brain Struct Funct. 2008;213:93–118.
Etkin A, Egner T, Kalisch R. Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci. 2011;15:85–93.
Ochsner KN, Ray RR, Hughes B, McRae K, Cooper JC, Weber J, et al. Bottom-up and top-down processes in emotion generation: common and distinct neural mechanisms. Psychol Sci. 2009;20:1322–31.
Schmahmann JD, Pandya DN, Wang R, Dai G, D’Arceuil HE, de Crespigny AJ, et al. Association fibre pathways of the brain: parallel observations from diffusion spectrum imaging and autoradiography. Brain. 2007;130:630–53.
Zuo N, Fang J, Lv X, Zhou Y, Hong Y, Li T, et al. White matter abnormalities in major depression: a tract-based spatial statistics and rumination study. PLOS ONE. 2012;7:e37561.
Pisner DA, Shumake J, Beevers CG, Schnyer DM. The superior longitudinal fasciculus and its functional triple-network mechanisms in brooding. NeuroImage: Clin. 2019;24:101935.
Fling BW, Chapekis M, Reuter-Lorenz PA, Anguera J, Bo J, Langan J, et al. Age differences in callosal contributions to cognitive processes. Neuropsychologia. 2011;49:2564–9.
Flinkenflügel K, Gruber M, Meinert S, Thiel K, Winter A, Goltermann J, et al. The interplay between polygenic score for tumor necrosis factor-α, brain structural connectivity, and processing speed in major depression. Mol Psychiatry. 2024;29:3151–9.
Borgers T, Zwiky E, Klug M, Enneking V, Fisch L, Klein L, et al. Reductions in limbic activity during negative emotion processing following cognitive-behavioural therapy in depressive disorders. PsyArXiv. 2024. https://doi.org/10.31234/osf.io/36prt.
Disner SG, Beevers CG, Haigh EAP, Beck AT. Neural mechanisms of the cognitive model of depression. Nat Rev Neurosci. 2011;12:467–77.
Roiser JP, Elliott R, Sahakian BJ. Cognitive mechanisms of treatment in depression. Neuropsychopharmacol. 2012;37:117–36.
Geerts E, van Os T, Gerlsma C. Nonverbal communication sets the conditions for the relationship between parental bonding and the short-term treatment response in depression. Psychiatry Res. 2009;165:120–7.
Jaffe M, Gullone E, Hughes EK. The roles of temperamental dispositions and perceived parenting behaviours in the use of two emotion regulation strategies in late childhood. J Appl Developmental Psychol. 2010;31:47–59.
Matheson K, Kelly O, Cole B, Tannenbaum B, Dodd C, Anisman H. Parental bonding and depressive affect: The mediating role of coping resources. Br J Soc Psychol. 2005;44:371–95.
Tani F, Pascuzzi D, Raffagnino R. The relationship between perceived parenting style and emotion regulation abilities in adulthood. J Adult Dev. 2018;25:1–12.
Givertz M, Segrin C. The association between overinvolved parenting and young adults’ self-efficacy, psychological entitlement, and family communication. Commun Res. 2014;41:1111–36.
Dentale F, Vecchione M, Alessandri G, Barbaranelli C. Investigating the protective role of global self-esteem on the relationship between stressful life events and depression: A longitudinal moderated regression model. Curr Psychol. 2020;39:2096–107.
Lakey CE, Hirsch JK, Nelson LA, Nsamenang SA. Effects of contingent self-esteem on depressive symptoms and suicidal behavior. Death Stud. 2014;38:563–70.
Zuroff DC, Blatt SJ. The therapeutic relationship in the brief treatment of depression: contributions to clinical improvement and enhanced adaptive capacities. J Consult Clin Psychol. 2006;74:130–40.
Carter JD, Crowe MT, Jordan J, McIntosh VVW, Frampton C, Joyce PR. Predictors of response to CBT and IPT for depression; the contribution of therapy process. Behav Res Ther. 2015;74:72–79.
Vîslă A, Constantino MJ, Newkirk K, Ogrodniczuk JS, Söchting I. The relation between outcome expectation, therapeutic alliance, and outcome among depressed patients in group cognitive-behavioral therapy. Psychother Res. 2018;28:446–56.
Arnow BA, Steidtmann D, Blasey C, Manber R, Constantino MJ, Klein DN, et al. The relationship between the therapeutic alliance and treatment outcome in two distinct psychotherapies for chronic depression. J Consult Clin Psychol. 2013;81:627–38.
Mayes LC, Swain JE, Leckman JF. Parental attachment systems: neural circuits, genes, and experiential contributions to parental engagement. Clin Neurosci Res. 2005;4:301–13.
Cabrera NJ, Fagan J, Wight V, Schadler C. Influence of mother, father, and child risk on parenting and children’s cognitive and social behaviors. Child Dev. 2011;82:1985–2005.
Laible DJ, Carlo G. The differential relations of maternal and paternal support and control to adolescent social competence, self-worth, and sympathy. J Adolesc Res. 2004;19:759–82.
Murphy E, Wickramaratne P, Weissman M. The stability of parental bonding reports: a 20-year follow-up. J Affect Disord. 2010;125:307–15.
Mathias L, Quagliato LA, Carta MG, Nardi AE, Cheniaux E. Challenges in the treatment of dysthymia: a narrative review. Expert Rev Neurother. 2024;24:633–42.
Acknowledgements
The study was supported by grants from the German Research Foundation (DFG grant RE4458/1-1 to RR, DA1151/5-1, DA1151/5-2, DA1151/9-1, DA1151/10-1, DA1151/11-1 to UD and SFB/TRR 393 project grant no 521379614 to UD, SM, EJL), the Interdisciplinary Center for Clinical Research (IZKF) of the medical faculty of Münster (grant Dan3/022/22 to UD), and by the Federal Ministry of Education and Research (BMBF) and the ministry of Saxony-Anhalt within the initial phase of the German Center for Mental Health (DZPG; BMBF grant 01EE2305C to RR).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
KF: Drafting the work, Conception and design of the work, Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. TB: Drafting the work, Conception and design of the work, Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. MK: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. MMM: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. EJL: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. SM: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. MG: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. JR: Critical revision of the paper for important intellectual content. TK: Critical revision of the paper for important intellectual content. NO: Critical revision of the paper for important intellectual content. JB: Critical revision of the paper for important intellectual content. EZ: Critical revision of the paper for important intellectual content. PK: Critical revision of the paper for important intellectual content. AK: Critical revision of the paper for important intellectual content. KS: Critical revision of the paper for important intellectual content. RK: Critical revision of the paper for important intellectual content. UD: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. VE: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. RR: Acquisition, analysis, or interpretation of data, Critical revision of the paper for important intellectual content. All authors have read and finally approved the submitted version of the paper, and agree to be accountable for all aspects of the work to ensure that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
JR received speaking honoraria from Janssen, Hexal and Novartis. Tilo Kircher received unrestricted educational grants from Servier, Janssen, Recordati, Aristo, Otsuka, neuraxpharm. All other authors state that they have no actual or potential conflict of interest to declare, including any financial, personal or other relationships with other people or organizations within three years of beginning the submitted work that could influence or bias their work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Flinkenflügel, K., Borgers, T., Klug, M. et al. Longitudinal associations between white matter integrity, early life adversities, and treatment response following cognitive-behavioral therapy in depression. Neuropsychopharmacol. 50, 1000–1007 (2025). https://doi.org/10.1038/s41386-025-02070-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-025-02070-x
This article is cited by
-
The SFB/TRR 393 Collaborative Research Centre: trajectories of affective disorders
Der Nervenarzt (2025)