Abstract
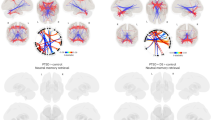
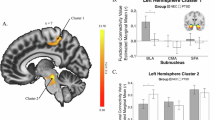
Amygdala hyperreactivity early-post trauma has been a demonstrable neurobiological correlate of future posttraumautic stress disorder (PTSD). The basolateral amygdala (BLA) particularly is vital for fear memory and threat processing, but BLA functional dynamics following a traumatic event are unexplored. BLA reactivity to threat may be a trait that can predict PTSD and persist over time. Alternatively, BLA responsivity to threat cues may change over time and be related to PTSD severity. As part of a larger, multisite study, AURORA, participants 18–75 years old were enrolled in an emergency department (ED) within 72 h of a traumatic event (N = 304, 199 female). At 2-weeks and 6-months post-trauma, PTSD symptoms, BLA responses to threat (fearful>neutral faces), and functional connectivity (FC) during fMRI were assessed. Generalizability of findings was assessed in an external replication sample of ED patients (n = 33). Two weeks post-trauma right BLA reactivity positively predicted later PTSD severity. However, left BLA reactivity to threat at 6 months post-trauma was negatively associated with PTSD severity at that timepoint (ΔPseudo-R2 = 0.04, IRR = 0.38, p < 0.001). In addition, a decrease in BLA reactivity from 2-weeks to 6-months predicted greater PTSD severity at 6 months (ΔPseudo-R2 = 0.03, IRR = 0.58, p < 0.001). This replicated in the external sample. A reduction in left BLA FC with the dorsal attention network predicted increased PTSD severity over time. These findings support a shift in BLA function within the first 6 months post-trauma that predicts PTSD pathology and stand in contrast to prior conceptualizations of amygdala hyperreactivity as a trait-like PTSD risk factor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Data and/or research tools used in the preparation of this manuscript were obtained from the National Institute of Mental Health (NIMH) Data Archive (NDA). NDA is a collaborative informatics system created by the National Institutes of Health to provide a national resource to support and accelerate research in mental health. Dataset identifier(s): NIMH Data Archive Digital Object Identifier (DOI) https://doi.org/10.15154/5t8n-fj46. Data and analysis code are available via https://github.com/aroeckner/BLA. Any additional information required to reanalyze the data reported in this paper is available from Jennifer Stevens, PhD (jennifer.stevens@emory.edu).
References
Diamond PR, Airdrie JN, Hiller R, Fraser A, Hiscox LV, Hamilton-Giachritsis C, et al. Change in prevalence of post-traumatic stress disorder in the two years following trauma: a meta-analytic study. Eur J Psychotraumatol. 2022;13:2066456.
Bonanno GA, Kaltman S. The varieties of grief experience. Clin Psychol Rev. 2001;21:705–34.
Galatzer-Levy IR, Huang SH, Bonanno GA. Trajectories of resilience and dysfunction following potential trauma: A review and statistical evaluation. Clin Psychol Rev. 2018;63:41–55.
Galatzer-Levy IR, Ankri Y, Freedman S, Israeli-Shalev Y, Roitman P, Gilad M, et al. Early PTSD symptom trajectories: persistence, recovery, and response to treatment: results from the Jerusalem Trauma Outreach and Prevention Study (J-TOPS). PLoS One. 2013;8:e70084.
Lanius RA, Rabellino D, Boyd JE, Harricharan S, Frewen PA, McKinnon MC. The innate alarm system in PTSD: conscious and subconscious processing of threat. Curr Opin Psychol. 2017;14:109–15.
Alexandra Kredlow M, Fenster RJ, Laurent ES, Ressler KJ, Phelps EA. Prefrontal cortex, amygdala, and threat processing: implications for PTSD. Neuropsychopharmacology. 2022;47:247–59.
Liberzon I, Sripada CS. The functional neuroanatomy of PTSD: a critical review. Progress Brain Res. 2007;167:151–69.
Rauch SL, Shin LM, Phelps EA. Neurocircuitry models of posttraumatic stress disorder and extinction: human neuroimaging research—past, present, and future. Biol Psychiatry. 2006;60:376–82.
Rauch SL, Whalen PJ, Shin LM, McInerney SC, Macklin ML, Lasko NB, et al. Exaggerated amygdala response to masked facial stimuli in posttraumatic stress disorder: a functional MRI study. Biol Psychiatry. 2000;47:769–76.
Shin LM, Rauch SL, Pitman RK. Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Ann N Y Acad Sci. 2006;1071:67–79.
Shin LM, Wright CI, Cannistraro PA, Wedig MM, McMullin K, Martis B, et al. A functional magnetic resonance imaging study of amygdala and medial prefrontal cortex responses to overtly presented fearful faces in posttraumatic stress disorder. Arch Gen Psychiatry. 2005;62:273–81.
Stevens JS, Jovanovic T, Fani N, Ely TD, Glover EM, Bradley B, et al. Disrupted amygdala-prefrontal functional connectivity in civilian women with posttraumatic stress disorder. J Psychiatr Res. 2013;47:1469–78.
Williams LM, Kemp AH, Felmingham K, Barton M, Olivieri G, Peduto A, et al. Trauma modulates amygdala and medial prefrontal responses to consciously attended fear. Neuroimage. 2006;29:347–57.
Patel R, Girard TA, Pukay-Martin N, Monson C. Preferential recruitment of the basolateral amygdala during memory encoding of negative scenes in posttraumatic stress disorder. Neurobiol Learn Mem. 2016;130:170–6.
Ousdal OT, Milde AM, Hafstad GS, Hodneland E, Dyb G, Craven AR, et al. The association of PTSD symptom severity with amygdala nuclei volumes in traumatized youths. Transl Psychiatry. 2020;10:288.
Roy AK, Shehzad Z, Margulies DS, Kelly AMC, Uddin LQ, Gotimer K, et al. Functional connectivity of the human amygdala using resting state fMRI. Neuroimage. 2009;45:614–26.
Terburg D, Morgan BE, Montoya ER, Hooge IT, Thornton HB, Hariri AR, et al. Hypervigilance for fear after basolateral amygdala damage in humans. Transl Psychiatry. 2012;2:e115–e115.
McLaughlin KA, Busso DS, Duys A, Green JG, Alves S, Way M, et al. Amygdala response to negative stimuli predicts PTSD symptom onset following a terrorist attack. Depress Anxiety. 2014;31:834–42.
Badura-Brack A, McDermott TJ, Heinrichs-Graham E, Ryan TJ, Khanna MM, Pine DS, et al. Veterans with PTSD demonstrate amygdala hyperactivity while viewing threatening faces: A MEG study. Biol Psychol. 2018;132:228–32.
Daniels JK, Coupland NJ, Hegadoren KM, Rowe BH, Densmore M, Neufeld RW, et al. Neural and behavioral correlates of peritraumatic dissociation in an acutely traumatized sample. J Clin Psychiatry. 2012;73:420–6.
Nicholson AA, Densmore M, Frewen PA, Théberge J, Neufeld RW, McKinnon MC, et al. The Dissociative Subtype of Posttraumatic Stress Disorder: Unique Resting-State Functional Connectivity of Basolateral and Centromedial Amygdala Complexes. Neuropsychopharmacology. 2015;40:2317–26.
Lanius RA, Vermetten E, Loewenstein RJ, Brand B, Schmahl C, Bremner JD, et al. Emotion modulation in PTSD: Clinical and neurobiological evidence for a dissociative subtype. Am J Psychiatry. 2010;167:640–7.
Sripada RK, King AP, Garfinkel SN, Wang X, Sripada CS, Welsh RC, et al. Altered resting-state amygdala functional connectivity in men with posttraumatic stress disorder. J Psychiatry Neurosci. 2012;37:241–9.
Belleau EL, Ehret LE, Hanson JL, Brasel KJ, Larson CL, deRoon-Cassini TA. Amygdala functional connectivity in the acute aftermath of trauma prospectively predicts severity of posttraumatic stress symptoms. Neurobiol Stress. 2020;12:100217.
Gilboa A, Shalev AY, Laor L, Lester H, Louzoun Y, Chisin R, et al. Functional connectivity of the prefrontal cortex and the amygdala in posttraumatic stress disorder. Biol Psychiatry. 2004;55:263–72.
Mcdonald AJ, Mascagni F, Guo L. Projections of the medial and lateral prefrontal cortices to the amygdala: a Phaseolus vulgaris leucoagglutinin study in the rat. Neuroscience. 1996;71:55–75.
Freese JL, Amaral DG. Synaptic organization of projections from the amygdala to visual cortical areas TE and V1 in the macaque monkey. J Comp Neurol. 2006;496:655–67.
Hart G, Leung BK, Balleine BW. Dorsal and ventral streams: the distinct role of striatal subregions in the acquisition and performance of goal-directed actions. Neurobiol Learn Mem. 2014;108:104–18.
Michely J, Rigoli F, Rutledge RB, Hauser TU, Dolan RJ. Distinct processing of aversive experience in amygdala subregions. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:291–300.
Brown VM, Workgroup M-AM, LaBar KS, Haswell CC, Gold AL, McCarthy G, et al. Altered resting-state functional connectivity of basolateral and centromedial amygdala complexes in posttraumatic stress disorder. Neuropsychopharmacology. 2014;39:351–9.
Liu T, Ke J, Qi R, Zhang L, Zhang Z, Xu Q, et al. Altered functional connectivity of the amygdala and its subregions in typhoon-related post-traumatic stress disorder. Brain Behav. 2021;11:e01952.
Shalev A, Liberzon I, Marmar C. Post-traumatic stress disorder. N. Engl J Med. 2017;376:2459–69.
Rothbaum BO, Schwartz AC. Exposure therapy for posttraumatic stress disorder. Am J Psychother. 2002;56:59–75.
Lissek S, van Meurs B. Learning models of PTSD: Theoretical accounts and psychobiological evidence. Int J Psychophysiol. 2015;98:594–605.
Admon R, Lubin G, Stern O, Rosenberg K, Sela L, Ben-Ami H, et al. Human vulnerability to stress depends on amygdala’s predisposition and hippocampal plasticity. Proc Natl Acad Sci. 2009;106:14120–5.
Stevens JS, Kim YJ, Galatzer-Levy IR, Reddy R, Ely TD, Nemeroff CB, et al. Amygdala reactivity and anterior cingulate habituation predict posttraumatic stress disorder symptom maintenance after acute civilian trauma. Biol Psychiatry. 2017;81:1023–9.
Van Wingen GA, Geuze E, Vermetten E, Fernández G. The neural consequences of combat stress: long-term follow-up. Mol Psychiatry. 2012;17:116–8.
Van Wingen GA, Geuze E, Vermetten E, Fernández G. Perceived threat predicts the neural sequelae of combat stress. Mol Psychiatry. 2011;16:664–71.
Shou H, Yang Z, Satterthwaite TD, Cook PA, Bruce SE, Shinohara RT, et al. Cognitive behavioral therapy increases amygdala connectivity with the cognitive control network in both MDD and PTSD. Neuroimage Clin. 2017;14:464–70.
Canli T, Zhao Z, Desmond JE, Kang E, Gross J, Gabrieli JD. An fMRI study of personality influences on brain reactivity to emotional stimuli. Behav Neurosci. 2001;115:33–42.
Castro JE, Diessler S, Varea E, Márquez C, Larsen MH, Cordero MI, et al. Personality traits in rats predict vulnerability and resilience to developing stress-induced depression-like behaviors, HPA axis hyper-reactivity and brain changes in pERK1/2 activity. Psychoneuroendocrinology. 2012;37:1209–23.
Hamann S, Canli T. Individual differences in emotion processing. Curr Opin Neurobiol. 2004;14:233–8.
Zhu X, Suarez-Jimenez B, Lazarov A, Helpman L, Papini S, Lowell A, et al. Exposure-based therapy changes amygdala and hippocampus resting-state functional connectivity in patients with posttraumatic stress disorder. Depress Anxiety. 2018;35:974–84.
McLean SA, Ressler K, Koenen KC, Neylan T, Germine L, Jovanovic T, et al. The AURORA Study: a longitudinal, multimodal library of brain biology and function after traumatic stress exposure. Mol Psychiatry. 2020;25:283–96.
Kim YJ, van Rooij SJH, Ely TD, Fani N, Ressler KJ, et al. Association between posttraumatic stress disorder severity and amygdala habituation to fearful stimuli. Depress Anxiety. 2019;36:647–58.
Blevins CA, Weathers FW, Davis MT, Witte TK, Domino JL. The Posttraumatic Stress Disorder Checklist for DSM-5 (PCL-5): Development and initial psychometric evaluation. J Trauma Stress. 2015;28:489–98.
Bernstein DP, Fink L, Handelsman L, Foote J. Childhood Trauma Questionnaire. Am J Psych Assess Family Violence. 1998. 1998. https://doi.org/10.1037/t02080-000.
Dalenberg C, Carlson E. Severity of Dissociative Symptoms - Adult (Brief Dissociative Experiences Scale (DES-B) – Modified). American Psychiatric Association: Online Assessment Measures. 2010. https://www.psychiatry.org/psychiatrists/practice/dsm/educational-resources/assessment-measures.
Carlson EB, Putnam FW. An update on the dissociative experiences scale. Dissociat Prog Dissociative Disord. 1993;6:16–27.
Esteban O, Birman D, Schaer M, Koyejo OO, Poldrack RA, Gorgolewski KJ. MRIQC: Advancing the automatic prediction of image quality in MRI from unseen sites. PLoS ONE. 2017;12:e0184661.
Esteban O, Markiewicz CJ, Blair RW, Moodie CA, Isik AI, Erramuzpe A, et al. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat Methods. 2019;16:111–6.
Gorgolewski K, Burns CD, Madison C, Clark D, Halchenko YO, Waskom ML, et al. Nipype: a flexible, lightweight and extensible neuroimaging data processing framework in python. Front Neuroinform. 2011;5:13.
Foa EB, Tolin DF. Comparison of the PTSD Symptom Scale-Interview Version and the Clinician-administered PTSD scale. J Trauma Stress. 2000;13:181–91.
Tyszka JM, Pauli WM. In vivo delineation of subdivisions of the human amygdaloid complex in a high-resolution group template. Hum Brain Mapp. 2016;37:3979–98.
Roeckner AR, Oliver KI, Lebois LAM, van Rooij SJH, Stevens JS. Neural contributors to trauma resilience: a review of longitudinal neuroimaging studies. Transl Psychiatry. 2021;11:508.
Admon R, Lubin G, Rosenblatt JD, Stern O, Kahn I, Assaf M, et al. Imbalanced neural responsivity to risk and reward indicates stress vulnerability in humans. Cereb Cortex. 2013;23:28–35.
Stevens JS, Harnett NG, Lebois LAM, Van Rooij SJH, Ely TD, Roeckner, A. et al. Brain-based biotypes of psychiatric vulnerability in the acute aftermath of trauma. Am J Psychiatry 2021;178:1037–49.
Phan KL, Britton JC, Taylor SF, Fig LM, Liberzon I. Corticolimbic blood flow during nontraumatic emotional processing in posttraumatic stress disorder. Arch Gen Psychiatry. 2006;63:184–92.
Britton JC, Phan KL, Taylor SF, Fig LM, Liberzon I. Corticolimbic blood flow in posttraumatic stress disorder during script-driven imagery. Biol Psychiatry. 2005;57:832–40.
Korem N, Duek O, Ben-Zion Z, Kaczkurkin AN, Lissek S, Orederu T, et al. Emotional numbing in PTSD is associated with lower amygdala reactivity to pain. Neuropsychopharmacology. 2022;47:1913–21.
Brashers-Krug T, Jorge R. Bi-Directional Tuning of Amygdala Sensitivity in Combat Veterans Investigated with fMRI. PLoS One. 2015;10:e0130246.
Kaldewaij R, Koch SBJ, Hashemi MM, Zhang W, Klumpers F, Roelofs K. Anterior prefrontal brain activity during emotion control predicts resilience to post-traumatic stress symptoms. Nat Hum Behav. 2021;5:1055–64.
Buffalari DM, Grace AA. Noradrenergic modulation of basolateral amygdala neuronal activity: opposing influences of α-2 and β receptor activation. J Neurosci. 2007;27:12358–66.
Goddard AW, Ball SG, Martinez J, Robinson MJ, Yang CR, Russell JM, et al. Current perspectives of the roles of the central norepinephrine system in anxiety and depression. Depress Anxiety. 2010;27:339–50.
Wang S, Liu X, Shi W, Qi Q, Zhang G, Li Y, et al. Mechanism of chronic stress-induced glutamatergic neuronal damage in the basolateral amygdaloid nucleus. Anal Cell Pathol. 2021;2021:8388527.
Shallcross J, Wu L, Wilkinson CS, Knackstedt LA, Schwendt M. Increased mGlu5 mRNA expression in BLA glutamate neurons facilitates resilience to the long-term effects of a single predator scent stress exposure. Brain Struct Funct. 2021;226:2279–93.
Braga MFM, Aroniadou-Anderjaska V, Manion ST, Hough CJ, Li H. Stress Impairs α1A Adrenoceptor-Mediated Noradrenergic Facilitation of GABAergic Transmission in the Basolateral Amygdala. Neuropsychopharmacology. 2003;29:45–58.
Poletti S, Locatelli C, Falini A, Colombo C, Benedetti F. Adverse childhood experiences associate to reduced glutamate levels in the hippocampus of patients affected by mood disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2016;71:117–22.
France J, Khatib D, Valbrun S, Basarkod S, Davie W, Diwadkar V, et al. 56. Trauma-exposed adolescents show reduced cortical glutamatergic neurotransmission during inhibitory control with negative emotional stimuli. Biol Psychiatry. 2024;95:S121–22.
Bremner JD. Long-term effects of childhood abuse on brain and neurobiology. Child Adolesc Psychiatr Clin N Am. 2003;12:271–92.
Felmingham KL, Bryant RA, Gordon E. Processing angry and neutral faces in post-traumatic stress disorder: an event-related potentials study. Neuroreport. 2003;14:777–80.
Brunetti M, Sepede G, Mingoia G, Catani C, Ferretti A, Merla A, et al. Elevated response of human amygdala to neutral stimuli in mild post traumatic stress disorder: neural correlates of generalized emotional response. Neuroscience. 2010;168:670–9.
Palomero-Gallagher N, Amunts K. A short review on emotion processing: a lateralized network of neuronal networks. Brain Struct Funct. 2022;227:673–84.
Ocklenburg S, Peterburs J, Mundorf A. Hemispheric asymmetries in the amygdala: A comparative primer. Prog Neurobiol. 2022;214:102283.
Gläscher J, Adolphs R. Processing of the arousal of subliminal and supraliminal emotional stimuli by the human amygdala. J Neurosci. 2003;23:10274–82.
Dyck M, Loughead J, Kellermann T, Boers F, Gur RC, Mathiak K. Cognitive versus automatic mechanisms of mood induction differentially activate left and right amygdala. Neuroimage. 2011;54:2503–13.
Vossel S, Geng JJ, Fink GR. Dorsal and ventral attention systems: distinct neural circuits but collaborative roles. Neuroscientist. 2014;20:150–9.
Evans TC, Alonso MR, Jagger-Rickels A, Rothlein D, Zuberer A, Bernstein J, et al. PTSD symptomatology is selectively associated with impaired sustained attention ability and dorsal attention network synchronization. NeuroImage Clin. 2022;36:103146.
White SF, Costanzo ME, Thornton LC, Mobley AM, Blair JR, Roy MJ. Increased cognitive control and reduced emotional interference is associated with reduced PTSD symptom severity in a trauma-exposed sample: A preliminary longitudinal study. Psychiatry Res Neuroimaging. 2018;278:7–12.
Acknowledgements
The investigators wish to thank the trauma survivors participating in the AURORA Study. Their time and effort during a challenging period of their lives make our efforts to improve recovery for future trauma survivors possible.
Funding
This research was supported by the National Institute of Mental Health K00 MH119603, K01 MH118467, U01 MH110925, and F31 MH126623, as well as the U.S. Department of Defense W81XWH-22-C-0122. This project was supported by NIMH under U01MH110925, the US Army MRMC, One Mind, and The Mayday Fund. The content is solely responsibility of the authors and does not necessarily represent the official views of any of the funders. This manuscript reflects the views of the authors and may not reflect the opinions or views of the NIH or of the Submitters submitting original data to NDA.
Author information
Authors and Affiliations
Contributions
SAM, RC, KJR, KCK, ARR, JSS, RH, SJHvR, TDE, NH, VPM, LAML, TJ, SLH and SEB contributed to the conceptualization, including formulation or evolution of overarching research goals and aims, of the study. FLB, XA, TCN, GDC, SDL, LTG, SLR, JJ, DAP, JFS, SEH, SAM, RCK, KJR, KCK, SJHvR, JSS, TDE, NH, VM, LL, TJ, SLH. and SB contributed to the methodology of the study, including development or design of methodology and creation of models. ARR, JSS, SJHvR, J TDE, NH, VM, LL, TJ, SLH, SB and KJR contributed to the neuroimaging data collection, formal analyses and validation of the study. SLH, FLB, XA, JSS, TCN, GDC, TJ, SDL, LTG, SLR, JPH, ABS, CL, PIMJr, PLH, SS, CWJ, BEP, RAS, JLP, MJS, CP, DAP, RCM, RMD, NKR, BJO, LDS, SAM, RCK, KJR and KCK conducted the research and investigation process, specifically, performing the experiments or data and evidence collection. SLH, FLB, XA, JSS, TCN, GDC, TJ, SDL, LTG, SLR, JPH, ABS, CL, PIMJr, PLH, SS, CWJ, BEP, RAS, JLP, MJS, CP, DAP, RCM, RMD, NKR, BJO, LDS, SAM, RCK., KJR. and KCK provided the resources for the study, including provision of study materials, patients, laboratory samples, instrumentation, computing resources or other analysis tools. SLH, FLB, XA, JSS, TCN, GDC, TJ, SDL, LTG, SLR, SAM, RCK, KJR and KCK were responsible for data curation, including management activities to annotate, scrub data and maintain research data for initial use and later reuse. ARR, ER-HL, and JSS were responsible for writing the original draft, including preparation, creation and presentation of the published work. All authors contributed to the paper by reviewing and editing the original draft. ARR and JSS were responsible for data visualization, including preparing, creating and presenting the published work, specifically, visualization and data presentation. SAM, RCK, KJR, KCK, and JSS were responsible for supervision, including oversight and leadership for the research activity planning and execution, including mentorship external to the core team. SLH, FLB, JSS, TJ, JPH, ABS, CL PIMJr, PLH, SS, CWJ, BEP, RAS, JLP, MJS, CP, RCM, RMD, NKR, NJO, LDS and SB were responsible for project administration, including management and coordination, responsibility for the research activity planning and execution. SAM, RCK, KJR and KCK were responsible for acquisition of the financial support for the project leading to this publication. ARR was responsible for funding supporting her effort on this publication.
Corresponding author
Ethics declarations
Competing interests
Dr. Harnett reports grant support from the National Institute of Mental Health, K00 MH119603. Dr. Lebois reports unpaid membership on the Scientific Committee for the International Society for the Study of Trauma and Dissociation (ISSTD) and grant support from the National Institute of Mental Health, K01 MH118467. ISSTD and NIMH were not involved in the analysis or preparation of the manuscript. Dr. van Rooij is supported by the NIMH (K01MH121653). Dr. Neylan has received research support from NIH, VA, and Rainwater Charitable Foundation, and consulting income from Jazz Pharmaceuticals. In the last three years Dr Clifford has received research funding from the NSF, NIH and LifeBell AI, and unrestricted donations from AliveCor Inc, Amazon Research, the Center for Discovery, the Gates Foundation, Google, the Gordon and Betty Moore Foundation, MathWorks, Microsoft Research, Nextsense Inc, One Mind Foundation, the Rett Research Foundation, and Samsung Research. Dr Clifford has financial interest in AliveCor Inc and Nextsense Inc. He also is the CTO of MindChild Medical and CSO of LifeBell AI and has ownership in both companies. These relationships are unconnected to the current work. Dr. Germine receives funding from the National Institute of Mental Health (R01 MH121617) and am on the board of the Many Brains Project. My family also has equity in Intelerad Medical Systems, Inc. Dr Rauch reported serving as secretary of the Society of Biological Psychiatry; serving as a board member of Community Psychiatry and Mindpath Health; serving as a board member of National Association of Behavioral Healthcare; serving as secretary and a board member for the Anxiety and Depression Association of America; serving as a board member of the National Network of Depression Centers; receiving royalties from Oxford University Press, American Psychiatric Publishing Inc, and Springer Publishing; and receiving personal fees from the Society of Biological Psychiatry, Community Psychiatry and Mindpath Health, and National Association of Behavioral Healthcare outside the submitted work. Dr. Sheikh has received funding from the Florida Medical Malpractice Joint Underwriter’s Association Dr. Alvin E. Smith Safety of Healthcare Services Grant; Allergan Foundation; the NIH/NIA-funded Jacksonville Aging Studies Center (JAX-ASCENT; R33AG05654); and the Substance Abuse and Mental Health Services Administration (1H79TI083101-01); and the Florida Blue Foundation. Dr. Jones has no competing interests related to this work, though he has been an investigator on studies funded by AstraZeneca, Vapotherm, Abbott, and Ophirex. Dr. Datner serves as Medical Advisor and on the Board of Directors for Cayaba Care. Dr. Joormann receives consulting payments from Janssen Pharmaceuticals. Dr. Harte has no competing interest related to this work, though in the last three years he has received research funding from Aptinyx and Arbor Medical Innovations, and consulting payments from Aptinyx. Dr. Koenen’s research has been supported by the Robert Wood Johnson Foundation, the Kaiser Family Foundation, the Harvard Center on the Developing Child, Stanley Center for Psychiatric Research at the Broad Institute of MIT and Harvard, the National Institutes of Health, One Mind, the Anonymous Foundation, and Cohen Veterans Bioscience. She has been a paid consultant for Baker Hostetler, Discovery Vitality, and the Department of Justice. She has been a paid external reviewer for the Chan Zuckerberg Foundation, the University of Cape Town, and Capita Ireland. She has had paid speaking engagements in the last three years with the American Psychological Association, European Central Bank. Sigmund Freud University – Milan, Cambridge Health Alliance, and Coverys. She receives royalties from Guilford Press and Oxford University Press. In the past 3 years, Dr. Kessler was a consultant for Cambridge Health Alliance, Canandaigua VA Medical Center, Holmusk, Partners Healthcare, Inc., RallyPoint Networks, Inc., and Sage Therapeutics. He has stock options in Cerebral Inc., Mirah, PYM, and Roga Sciences. Dr. McLean served as a consultant for Walter Reed Army Institute for Research and for Arbor Medical Innovations, and BioXcel Therapeutics, Inc. Dr. Ressler has performed scientific consultation for Bioxcel, Bionomics, Acer, and Jazz Pharma; serves on Scientific Advisory Boards for Sage, Boehringer Ingelheim, Senseye, and the Brain Research Foundation, and he has received sponsored research support from Alto Neuroscience. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Roeckner, A.R., Lin, E.RH., Hinrichs, R. et al. Sequential decreases in basolateral amygdala response to threat predict failure to recover from PTSD. Neuropsychopharmacol. 50, 1573–1582 (2025). https://doi.org/10.1038/s41386-025-02115-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-025-02115-1