Abstract
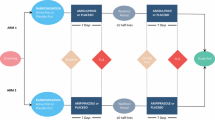
Determining the effects of antipsychotics on MRI brain structural metrics without the potential confounding effects related to the natural course of a psychotic illness is challenging. However, it is crucial to understand these effects to interpret the results of cross-sectional and longitudinal studies in medicated patients and, ultimately, to understand better the biological mechanisms driving antipsychotics’ effects. In this work, we aim to determine whether exposure to antipsychotics is associated with alterations in brain MRI structural metrics in the absence of disease effects. A randomized, double-blind, counter-balanced order, crossover, placebo-controlled study in healthy volunteers was performed. The study comprised two arms. Within arms, participants were randomized to receive daily doses of either the active compound (Arm 1= amisulpride 400 mg/day, N = 24; Arm 2= aripiprazole 10 mg/day, N = 24) for one week, followed by placebo or vice versa. We found increased MRI volume estimates in the left putamen and in the right caudate in the amisulpride condition as compared to placebo and increased right putamen volume estimates after aripiprazole compared to placebo. No other effects were found in cortical volume estimates, cortical thickness, cortical surface area, and T1-relaxation time. Striatal changes reversed within weeks of drug withdrawal. Short-term exposure to either one of two different antipsychotics results in a transient increase in striatal volume measured with T1-weighted MRI that normalizes rapidly on stopping treatment without cortical changes. Our findings suggest that striatal volumetric MRI differences detected in people with schizophrenia taking antipsychotics are, at least in part, attributable to pharmacological effects.
This is a preview of subscription content, access via your institution
Access options



Similar content being viewed by others
Data availability
The ethical approval conditions for this study do not permit unrestricted access to the raw data. De-identified individual participant data are available for research purposes from the corresponding authors from the publication date, subject to a data-sharing agreement, except data from a minority of subjects who did not consent to de-identified data being used to support future research. Requests will be responded to within 15 working days. The conditions of the ethical approval of the study stipulate that access to data that may allow the identification of volunteers will only be permitted for research that an ethics committee has independently reviewed.
References
McCutcheon RA, Reis Marques T, Howes OD. Schizophrenia-an overview. JAMA Psychiatry. 2020;77:201–10.
Huhn M, Nikolakopoulou A, Schneider-Thoma J, Krause M, Samara M, Peter N, et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394:939–51.
Kaar SJ, Natesan S, McCutcheon R, Howes OD. Antipsychotics: Mechanisms underlying clinical response and side-effects and novel treatment approaches based on pathophysiology. Neuropharmacology. 2020;172:107704.
Howes OD, Cummings C, Chapman GE, Shatalina E. Neuroimaging in schizophrenia: an overview of findings and their implications for synaptic changes. Neuropsychopharmacology. 2023;48:151–67.
Correll CU, Rubio JM, Kane JM. What is the risk-benefit ratio of long-term antipsychotic treatment in people with schizophrenia? World Psychiatry. 2018;17:149–60.
Fountoulakis KN, Stahl SM. The effect of first- and second-generation antipsychotics on brain morphology in schizophrenia: A systematic review of longitudinal magnetic resonance studies with a randomized allocation to treatment arms. J Psychopharmacol. 2022;36:428–38.
Lawrie SM. Do antipsychotic drugs shrink the brain? Probably not. J Psychopharmacol. 2022;36:425–27.
Amato D, Beasley CL, Hahn MK, Vernon AC. Neuroadaptations to antipsychotic drugs: Insights from pre-clinical and human post-mortem studies. Neurosci Biobehav Rev. 2017;76:317–35.
Turkheimer FE, Selvaggi P, Mehta MA, Veronese M, Zelaya F, Dazzan P, et al. Normalizing the abnormal: do antipsychotic drugs push the cortex into an unsustainable metabolic envelope? Schizophr Bull. 2020;46:484–95.
Hamer AnderssonC, Lawler RM, Mailman CP, Lieberman RB. JA. Striatal volume changes in the rat following long-term administration of typical and atypical antipsychotic drugs. Neuropsychopharmacology. 2002;27:143–51.
Guma E, Rocchetti J, Devenyi GA, Tanti A, Mathieu A, Lerch JP, et al. Regional brain volume changes following chronic antipsychotic administration are mediated by the dopamine D2 receptor. Neuroimage. 2018;176:226–38.
Vernon AC, Natesan S, Crum WR, Cooper JD, Modo M, Williams SC, et al. Contrasting effects of haloperidol and lithium on rodent brain structure: a magnetic resonance imaging study with postmortem confirmation. Biol Psychiatry. 2012;71:855–63.
Vernon AC, Natesan S, Modo M, Kapur S. Effect of chronic antipsychotic treatment on brain structure: a serial magnetic resonance imaging study with ex vivo and postmortem confirmation. Biol Psychiatry. 2011;69:936–44.
Drazanova E, Kratka L, Vaskovicova N, Skoupy R, Horska K, Babinska Z, et al. Olanzapine exposure diminishes perfusion and decreases volume of sensorimotor cortex in rats. Pharm Rep. 2019;71:839–47.
Vernon AC, Crum WR, Lerch JP, Chege W, Natesan S, Modo M, et al. Reduced cortical volume and elevated astrocyte density in rats chronically treated with antipsychotic drugs-linking magnetic resonance imaging findings to cellular pathology. Biol Psychiatry. 2014;75:982–90.
Dorph-Petersen KA, Pierri JN, Perel JM, Sun Z, Sampson AR, Lewis DA. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30:1649–61.
Andersen HG, Raghava JM, Svarer C, Wulff S, Johansen LB, Antonsen PK, et al. Striatal volume increase after six weeks of selective dopamine D(2/3) receptor blockade in first-episode, antipsychotic-naive schizophrenia patients. Front Neurosci. 2020;14:484.
Chakos MH, Lieberman JA, Bilder RM, Borenstein M, Lerner G, Bogerts B, et al. Increase in caudate nuclei volumes of first-episode schizophrenic patients taking antipsychotic drugs. Am J Psychiatry. 1994;151:1430–6.
Dazzan P, Morgan KD, Orr K, Hutchinson G, Chitnis X, Suckling J, et al. Different effects of typical and atypical antipsychotics on grey matter in first episode psychosis: the AESOP study. Neuropsychopharmacology. 2005;30:765–74.
Keshavan MS, Bagwell WW, Haas GL, Sweeney JA, Schooler NR, Pettegrew JW. Changes in caudate volume with neuroleptic treatment. Lancet. 1994;344:1434.
Lieberman JA, Tollefson GD, Charles C, Zipursky R, Sharma T, Kahn RS, et al. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62:361–70.
Fusar-Poli P, Smieskova R, Kempton MJ, Ho BC, Andreasen NC, Borgwardt S. Progressive brain changes in schizophrenia related to antipsychotic treatment? A meta-analysis of longitudinal MRI studies. Neurosci Biobehav Rev. 2013;37:1680–91.
Haijma SV, Van Haren N, Cahn W, Koolschijn PC, Hulshoff Pol HE, Kahn RS. Brain volumes in schizophrenia: a meta-analysis in over 18 000 subjects. Schizophr Bull. 2013;39:1129–38.
Huhtaniska S, Jaaskelainen E, Heikka T, Moilanen JS, Lehtiniemi H, Tohka J, et al. Long-term antipsychotic and benzodiazepine use and brain volume changes in schizophrenia: The Northern Finland Birth Cohort 1966 study. Psychiatry Res Neuroimaging. 2017;266:73–82.
van Erp TGM, Walton E, Hibar DP, Schmaal L, Jiang W, Glahn DC, et al. Cortical Brain Abnormalities in 4474 Individuals With Schizophrenia and 5098 Control Subjects via the Enhancing Neuro Imaging Genetics Through Meta Analysis (ENIGMA) Consortium. Biol Psychiatry. 2018;84:644–54.
Andreasen NC, Liu D, Ziebell S, Vora A, Ho BC. Relapse duration, treatment intensity, and brain tissue loss in schizophrenia: a prospective longitudinal MRI study. Am J Psychiatry. 2013;170:609–15.
Ho BC, Andreasen NC, Ziebell S, Pierson R, Magnotta V. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68:128–37.
Moncrieff J, Leo J. A systematic review of the effects of antipsychotic drugs on brain volume. Psychol Med. 2010;40:1409–22.
Navari S, Dazzan P. Do antipsychotic drugs affect brain structure? A systematic and critical review of MRI findings. Psychol Med. 2009;39:1763–77.
Smieskova R, Fusar-Poli P, Allen P, Bendfeldt K, Stieglitz RD, Drewe J, et al. The effects of antipsychotics on the brain: what have we learnt from structural imaging of schizophrenia?-a systematic review. Curr Pharm Des. 2009;15:2535–49.
Lawrie SM. Are structural brain changes in schizophrenia related to antipsychotic medication? A narrative review of the evidence from a clinical perspective. Ther Adv Psychopharmacol. 2018;8:319–26.
Tost H, Braus DF, Hakimi S, Ruf M, Vollmert C, Hohn F, et al. Acute D2 receptor blockade induces rapid, reversible remodeling in human cortical-striatal circuits. Nat Neurosci. 2010;13:920–2.
Hawkins PCT, Wood TC, Vernon AC, Bertolino A, Sambataro F, Dukart J, et al. An investigation of regional cerebral blood flow and tissue structure changes after acute administration of antipsychotics in healthy male volunteers. Hum Brain Mapp. 2018;39:319–31.
Lorio S, Kherif F, Ruef A, Melie-Garcia L, Frackowiak R, Ashburner J, et al. Neurobiological origin of spurious brain morphological changes: A quantitative MRI study. Hum Brain Mapp. 2016;37:1801–15.
Weiskopf N, Mohammadi S, Lutti A, Callaghan MF. Advances in MRI-based computational neuroanatomy: from morphometry to in-vivo histology. Curr Opin Neurol. 2015;28:313–22.
Marques JP, Kober T, Krueger G, van der Zwaag W, Van de Moortele PF, Gruetter R. MP2RAGE, a self bias-field corrected sequence for improved segmentation and T1-mapping at high field. Neuroimage. 2010;49:1271–81.
Taylor D, Barnes TRE, Young A The Maudsley prescribing guidelines in psychiatry. 14th edition / ed. Wiley Blackwell: Hoboken, NJ; 2021.
Lako IM, van den Heuvel ER, Knegtering H, Bruggeman R, Taxis K. Estimating dopamine D(2) receptor occupancy for doses of 8 antipsychotics: a meta-analysis. J Clin Psychopharmacol. 2013;33:675–81.
Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. 1999;9:179–94.
Desikan RS, Segonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31:968–80.
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33:341–55.
Osugo M, Wall MB, Selvaggi P, Zahid U, Finelli V, Chapman GE, et al. Striatal dopamine D2/D3 receptor regulation of human reward processing and behaviour. Nat Commun. 2025;16:1852.
Chopra S, Fornito A, Francey SM, O’Donoghue B, Cropley V, Nelson B, et al. Differentiating the effect of antipsychotic medication and illness on brain volume reductions in first-episode psychosis: A Longitudinal, Randomised, Triple-blind, Placebo-controlled MRI Study. Neuropsychopharmacology. 2021;46:1494–501.
Voineskos AN, Mulsant BH, Dickie EW, Neufeld NH, Rothschild AJ, Whyte EM, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77:674–83.
Camps M, Cortes R, Gueye B, Probst A, Palacios JM. Dopamine receptors in human brain: autoradiographic distribution of D2 sites. Neuroscience. 1989;28:275–90.
Lobo MC, Whitehurst TS, Kaar SJ, Howes OD. New and emerging treatments for schizophrenia: a narrative review of their pharmacology, efficacy and side effect profile relative to established antipsychotics. Neurosci Biobehav R. 2022;132:324–61.
Selvaggi P, Hawkins PCT, Dipasquale O, Rizzo G, Bertolino A, Dukart J, et al. Increased cerebral blood flow after single dose of antipsychotics in healthy volunteers depends on dopamine D2 receptor density profiles. Neuroimage. 2019;188:774–84.
Sander CY, Hooker JM, Catana C, Normandin MD, Alpert NM, Knudsen GM, et al. Neurovascular coupling to D2/D3 dopamine receptor occupancy using simultaneous PET/functional MRI. Proc Natl Acad Sci USA. 2013;110:11169–74.
Leucht S, Samara M, Heres S, Patel MX, Furukawa T, Cipriani A, et al. Dose equivalents for second-generation antipsychotic drugs: the classical mean dose method. Schizophr Bull. 2015;41:1397–402.
Hiemke C, Bergemann N, Clement HW, Conca A, Deckert J, Domschke K, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51:e1.
Stuber C, Morawski M, Schafer A, Labadie C, Wahnert M, Leuze C, et al. Myelin and iron concentration in the human brain: a quantitative study of MRI contrast. Neuroimage. 2014;93:95–106.
Cousins DA, Aribisala B, Nicol Ferrier I, Blamire AM. Lithium, gray matter, and magnetic resonance imaging signal. Biol Psychiatry. 2013;73:652–7.
Deoni SC. Magnetic resonance relaxation and quantitative measurement in the brain. Methods Mol Biol. 2011;711:65–108.
Franklin TR, Wang Z, Shin J, Jagannathan K, Suh JJ, Detre JA, et al. A VBM study demonstrating ‘apparent’ effects of a single dose of medication on T1-weighted MRIs. Brain Struct Funct. 2013;218:97–104.
Salgado-Pineda P, Delaveau P, Falcon C, Blin O. Brain T1 intensity changes after levodopa administration in healthy subjects: a voxel-based morphometry study. Br J Clin Pharm. 2006;62:546–51.
Weinberger DR, Radulescu E. Structural magnetic resonance imaging all over again. JAMA Psychiatry. 2021;78:11–12.
Abrantes A, Giusti-Rodriguez P, Ancalade N, Sekle S, Basiri ML, Stuber GD, et al. Gene expression changes following chronic antipsychotic exposure in single cells from mouse striatum. Mol Psychiatry. 2022;27:2803–12.
Andreassen OA, Jorgensen HA. Neurotoxicity associated with neuroleptic-induced oral dyskinesias in rats - Implications for tardive dyskinesia? Prog Neurobiol. 2000;61:525–41.
McCullumsmith RE, Stincic TL, Agrawal SM, Meador-Woodruff JH. Differential effects of antipsychotics on haloperidol-induced vacuous chewing movements and subcortical gene expression in the rat. Eur J Pharm. 2003;477:101–12.
Weinberger DR, Radulescu E. Finding the elusive psychiatric “lesion” with 21st-century neuroanatomy: a note of caution. Am J Psychiatry. 2016;173:27–33.
van Erp TG, Hibar DP, Rasmussen JM, Glahn DC, Pearlson GD, Andreassen OA, et al. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol Psychiatry. 2016;21:585.
Brugger SP, Howes OD. Heterogeneity and homogeneity of regional brain structure in schizophrenia: a meta-analysis. JAMA Psychiatry. 2017;74:1104–11.
Howes OD, Onwordi EC. The synaptic hypothesis of schizophrenia version III: a master mechanism. Mol Psychiatry. 2023;28:1843–56.
Acknowledgements
We thank all the volunteers who took part in this challenging study. We also thank the staff at the Robert Steiner MRI Unit Imperial College London and at the Maudsley Pharmacy South London and Maudsley NHS Foundation Trust for their crucial support in this study. The authors wish to thank the reviewers for their insightful comments and constructive suggestions, which greatly contributed to improving the quality and clarity of this manuscript. We also sincerely appreciate their positive evaluation of the study.
Funding
This study was funded by Medical Research Council-UK (no. MC_U120097115), Maudsley Charity (no. 666), and Wellcome Trust (no. 094849/Z/10/Z) grants to Prof. Howes. This paper represents an independent research part funded by the National Institute for Health Research Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London that supported Pierluigi Selvaggi, Ottavia Dipasquale, and Mitul A Mehta. Pierluigi Selvaggi has been supported by a Ph.D. studentship jointly funded by the NIHR-BRC at SLaM and the Department of Neuroimaging, King’s College London. Pierluigi Selvaggi is currently supported by the European Union’s Horizon 2020 Research and Innovation Program under grant agreement No. 964874 (REALMENT). George Chapman is currently a National Institute for Health and Care Research (NIHR) Academic Clinical Fellow. The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: PS, TRS, MAM, ODH, Methodology: PS, OD, BS, TCW, MBW, MAM, ODH; Investigation: PS, MO, UZ, TW, EO, GC, VF; Visualization: PS, MO, UZ, TCW, MBW, RM, MAM, TRM, ODH; Funding acquisition: ODH; Project administration: TRM, ODH; Supervision: RM, MAM, TRM, ODH; Writing – original draft: PS, ODH; Writing – review & editing: PS, MO, UZ, OD, TW, EO, GC, VF, BS, TCW, MBW, RM, MAM, TRM, ODH. All authors approved the manuscript and are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding authors
Ethics declarations
Competing interests
In the past three years, MAM has been an advisory board member for Lundbeck and Forum Pharmaceuticals. He also received research funding from Lundbeck, Takeda, and Johnson & Johnson. O.D.H. has received investigator-initiated research funding from and/or participated in advisory/speaker meetings organized by Angellini, Autifony, Biogen, Boehringer-Ingelheim, Eli Lilly, Elysium, Heptares, Global Medical Education, Invicro, Jansenn, Karuna, Lundbeck, Merck, Neurocrine, Ontrack/ Pangea, Otsuka, Sunovion, Recordati, Roche, Rovi and Viatris/Mylan. He was previously a part-time employee of Lundbeck A/v. Neither O.D.H. nor his family have holdings/a financial stake in any pharmaceutical company. ODH has a patent for the use of dopaminergic imaging. PS participated in advisory/speaker meetings organized by Angelini and Lundbeck. M.B.W. is an employee of Perceptive Inc., London. T.R.M. is an employee and founder of Pasithea Therapeutics. Other authors have reported no biomedical financial interests or potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Selvaggi, P., Osugo, M., Zahid, U. et al. Antipsychotics cause reversible structural brain changes within one week. Neuropsychopharmacol. 50, 1275–1283 (2025). https://doi.org/10.1038/s41386-025-02120-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-025-02120-4