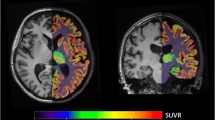
Abstract
Motor signs are common in patients with a major depressive disorder (MDD) and associated with functional disability and falls. They appear to be more pronounced in patients with late life depression, but it is unclear whether this is caused by global brain aging or by a specific pathology associated with depression. We therefore sought to investigate associations between motor signs in late life depression and aging related changes in synapses, gray and white matter. From the monocentric Leuven Late Life depression study, we included 75 participants (41 healthy controls, 34 currently depressed MDD patients) aged ≥ 60 years. Motor assessment included the MDS-Unified Parkinson’s Disease Rating Scale part III (MDS-UPDRSIII), Scale for Assessment and Rating of Ataxia (SARA), gait analysis and digitized drawing. Brain synaptic vesicle glycoprotein 2A binding as a proxy for synaptic density was determined in predefined cortico-subcortical volumes of interest (VOI) using 11C-UCB-J PET in 62 participants (25 patients). Brain T1 and FLAIR MR images were used to quantify gray matter volume and white matter hyperintensity volume in 69 participants (32 patients). Multiple linear regression analyses were performed with motor outcome as the dependent variable, diagnosis and VOI 11C-UCB-J SUVR and their interaction, age, whole brain white matter hyperintensity volume and gray matter VOI as independent variables. The study demonstrated that patients had significant impairments on all motor assessments, compared to healthy controls. Specifically in patients, right globus pallidus synaptic density was associated with MDS-UPDRSIII score and drawing speed; thalamic gray matter volume predicted SARA score, gait and drawing speed; and white matter hyperintensity volume predicted MDS-UPDRSIII score. We conclude that motor signs in late life depression are associated with specific synaptic density and gray matter volume differences in basal ganglia-thalamic structures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Grouped data supporting the study’s findings are provided in the supplementary materials. Due to ethical regulations concerning the specific participant profiles of older persons with depression, individual-level data are not made publicly available. However, anonymized data relevant to the study may be obtained from the corresponding author upon reasonable request.
References
Sobin C, Sackeim HA. Psychomotor symptoms of depression. Am J Psychiatry. 1997;154:4–17.
Rogers MA, Bradshaw JL, Phillips JG, Chiu E, Vaddadi K, Presnel I, et al. Parkinsonian motor characteristics in unipolar major depression. J Clin Exp Neuropsychol. 2000;22:232–44.
American Psychiatric Association D-TF Diagnostic and statistical manual of mental disorders: DSM-5™, 5th ed.: American Psychiatric Publishing; 2013.
Buyukdura JS, McClintock SM, Croarkin PE. Psychomotor retardation in depression: Biological underpinnings, measurement, and treatment. Prog neuro-Psychopharmacol Biol Psychiatry. 2011;35:395–409.
Belvederi Murri M, Triolo F, Coni A, Tacconi C, Nerozzi E, Escelsior A, et al. Instrumental assessment of balance and gait in depression: A systematic review. Psychiatry Res. 2020;284:112687.
Stubbs B, Stubbs J, Gnanaraj SD, Soundy A. Falls in older adults with major depressive disorder (MDD): a systematic review and exploratory meta-analysis of prospective studies. Int Psychogeriatr. 2016;28:23–9.
Fried EI, Nesse RM. The impact of individual depressive symptoms on impairment of psychosocial functioning. PLoS One. 2014;9:e90311.
Novick JS, Stewart JW, Wisniewski SR, Cook IA, Manev R, Nierenberg AA, et al. Clinical and demographic features of atypical depression in outpatients with major depressive disorder: preliminary findings from STAR*D. J Clin Psychiatry. 2005;66:1002–11.
Pier MPBI, Hulstijn W, Sabbe BGC. Psychomotor retardation in elderly depressed patients. J Affect Disord. 2004;81:73–7.
Shahpesandy H. Different manifestation of depressive disorder in the elderly. Neuro Endocrinol Lett. 2005;26:691–5.
Brodaty H, Luscombe G, Parker G, Wilhelm K, Hickie I, Austin MP, et al. Increased rate of psychosis and psychomotor change in depression with age. Psychol Med. 1997;27:1205–13.
Schrijvers D, Hulstijn W, Sabbe BGCC. Psychomotor symptoms in depression: A diagnostic, pathophysiological and therapeutic tool. J Affect Disord. 2008;109:1–20.
Shah PJ, Ogilvie AD, Goodwin GM, Ebmeier KP. Clinical and psychometric correlates of dopamine D2 binding in depression. Psychol Med. 1997;27:1247–56.
Martinot MLPP, Bragulat V, Artiges E, Dollé F, Hinnen F, Jouvent R, et al. Decreased presynaptic dopamine function in the left caudate of depressed patients with affective flattening and psychomotor retardation. Am J Psychiatry. 2001;158:314–6.
Bragulat V, Paillère-Martinot ML, Artiges E, Frouin V, Poline JB, Martinot JL. Dopaminergic function in depressed patients with affective flattening or with impulsivity: [18F]Fluoro-l-dopa positron emission tomography study with voxel-based analysis. Psychiatry Res - Neuroimaging. 2007;154:115–24.
Meyer JH, McNeely HE, Sagrati S, Boovariwala A, Martin K, Verhoeff NP, et al. Elevated putamen D(2) receptor binding potential in major depression with motor retardation: an [11C]raclopride positron emission tomography study. Am J Psychiatry. 2006;163:1594–602.
Wüthrich F, Lefebvre S, Mittal VA, Shankman SA, Alexander N, Brosch K, et al. The neural signature of psychomotor disturbance in depression. Mol Psychiatry. 2024;29:317–26.
Liberg B, Klauser P, Harding IH, Adler M, Rahm C, Lundberg J, et al. Functional and structural alterations in the cingulate motor area relate to decreased fronto-striatal coupling in major depressive disorder with psychomotor disturbances. Front Psychiatry. 2014;5:176.
Mayberg HS, Lewis PJ, Regenold W, Wagner HN Jr. Paralimbic hypoperfusion in unipolar depression. J Nucl Med. 1994;35:929–34.
Videbech P, Ravnkilde B, Pedersen TH, Hartvig H, Egander A, Clemmensen K, et al. The Danish PET/depression project: Clinical symptoms and cerebral blood flow. A regions-of-interest analysis. Acta Psychiatr Scandinavica. 2002;106:35–44.
Yin Y, Wang M, Wang Z, Xie C, Zhang H, Zhang H, et al. Decreased cerebral blood flow in the primary motor cortex in major depressive disorder with psychomotor retardation. Prog Neuropsychopharmacol Biol Psychiatry. 2018;81:438–44.
Walther S, Hugli S, Hofle O, Federspiel A, Horn H, Bracht T, et al. Frontal white matter integrity is related to psychomotor retardation in major depression. Neurobiol Dis. 2012;47:13–9.
Cantisani A, Stegmayer K, Bracht T, Federspiel A, Wiest R, Horn H, et al. Distinct resting-state perfusion patterns underlie psychomotor retardation in unipolar vs. bipolar depression. Acta Psychiatr Scandinavica. 2016;134:329–38.
Pan F, Xu Y, Zhou W, Chen J, Wei N, Lu S, et al. Disrupted intrinsic functional connectivity of the cognitive control network underlies disease severity and executive dysfunction in first-episode, treatment-naive adolescent depression. J Affect Disord. 2020;264:455–63.
Zitser J, Casaletto KB, Staffaroni AM, Sexton C, Weiner-Light S, Wolf A, et al. Mild motor signs matter in typical brain aging: the value of the UPDRS score within a functionally intact cohort of older adults. Front Aging Neurosci. 2021;13:594637.
Louis ED, Bennett DA. Mild Parkinsonian signs: An overview of an emerging concept. Mov Disord. 2007;22:1681–8.
Buchanan SM, Richards M, Schott JM, Schrag A. Mild Parkinsonian signs: a systematic review of clinical, imaging, and pathological associations. Mov Disord. 2021;36:2481–93.
Van Cauwenberge MGA, Delva A, Vande Casteele T, Laroy M, Radwan A, Vansteelandt K, et al. Mild Motor Signs in Healthy Aging Are Associated with Lower Synaptic Density in the Brain. Mov Disord. 2023;38:1786–94.
Emsell L, Laroy M, Van Cauwenberge M, Vande Casteele T, Vansteelandt K, Van Laere K, et al. The Leuven late life depression (L3D) study: PET-MRI biomarkers of pathological brain ageing in late-life depression: study protocol. BMC Psychiatry. 2021;21:64.
Van Cauwenberge MGA, Vande Casteele T, Laroy M, Vansteelandt K, Van den Stock J, Bouckaert F, et al. Motor dysfunction in late life depression: A mood or movement disorder? J Affect Disord. 2025;381:680–91.
Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;23:2129–70.
Schmitz-Hübsch T, du Montcel ST, Baliko L, Berciano J, Boesch S, Depondt C, et al. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology. 2006;66:1717–20.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin psychiatry. 1998;59:22–33. quiz 4-57
Henschel L, Conjeti S, Estrada S, Diers K, Fischl B, Reuter M. FastSurfer - A fast and accurate deep learning based neuroimaging pipeline. Neuroimage. 2020;219:117012.
Safai A, Prasad S, Chougule T, Saini J, Pal PK, Ingalhalikar M. Microstructural abnormalities of substantia nigra in Parkinson’s disease: A neuromelanin sensitive MRI atlas based study. Hum Brain Mapp. 2020;41:1323–33.
Ma D, Popuri K, Bhalla M, Sangha O, Lu D, Cao J, et al. Quantitative assessment of field strength, total intracranial volume, sex, and age effects on the goodness of harmonization for volumetric analysis on the ADNI database. Hum Brain Mapp. 2019;40:1507–27.
Mertens N, Maguire RP, Serdons K, Lacroix B, Mercier J, Sciberras D, et al. Validation of Parametric Methods for [(11)C]UCB-J PET Imaging Using Subcortical White Matter as Reference Tissue. Mol Imaging Biol. 2020;22:444–52.
Thomas BA, Erlandsson K, Modat M, Thurfjell L, Vandenberghe R, Ourselin S, et al. The importance of appropriate partial volume correction for PET quantification in Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2011;38:1104–19.
Tabachnick BG, Fidell LS Using Multivariate Statistics. Pearson; 2014, p. 1056.
Leucht S, Samara M, Heres S, Davis JM. Dose equivalents for antipsychotic drugs: the DDD method. Schizophrenia Bull. 2016;42:S90–S4.
Li X, Xing Y, Martin-Bastida A, Piccini P, Auer DP. Patterns of grey matter loss associated with motor subscores in early Parkinson’s disease. Neuroimage Clin. 2018;17:498–504.
Levy-Tzedek S, Arbelle D, Forman D, Zlotnik Y. Improvement in upper-limb UPDRS motor scores following fast-paced arm exercise: A pilot study. Restor Neurol Neurosci. 2018;36:535–45.
Holmes SE, Honhar P, Tinaz S, Naganawa M, Hilmer AT, Gallezot J-D, et al. Synaptic loss and its association with symptom severity in Parkinson’s disease. npj Parkinson’s Dis. 2024;10:42.
Matuskey D, Tinaz S, Wilcox KC, Naganawa M, Toyonaga T, Dias M, et al. Synaptic changes in Parkinson disease assessed with in vivo imaging. Ann Neurol. 2020;87:329–38.
Delva A, Van Weehaeghe D, Koole M, Van Laere K, Vandenberghe W. Loss of presynaptic terminal integrity in the substantia nigra in early Parkinson’s Disease. Mov Disord. 2020;35:1977–86.
Lauterbach EC Mood Disorders and the Globus Pallidus. In: Bédard M-A, Agid Y, Chouinard S, Fahn S, Korczyn AD, Lespérance P, editors. Mental and behavioral dysfunction in movement disorders. Totowa, NJ: Humana Press; 2003, pp. 305–20.
Alexander GE, DeLong MR, Strick PL. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci. 1986;9:357–81.
Mizuno Y, Ashok AH, Bhat BB, Jauhar S, Howes OD. Dopamine in major depressive disorder: A systematic review and meta-analysis of in vivo imaging studies. J Psychopharmacol. 2023;37:1058–69.
Meyer JH, Krüger S, Wilson AA, Christensen BK, Goulding VS, Schaffer A, et al. Lower dopamine transporter binding potential in striatum during depression. Neuroreport. 2001;12:4121–5.
Kazmi H, Walker Z, Booij J, Khan F, Shah S, Sudre CH, et al. Late onset depression: dopaminergic deficit and clinical features of prodromal Parkinson’s disease: a cross-sectional study. J Neurol Neurosurg Psychiatry. 2021;92:158–64.
Van Cauwenberge MGA, Bouckaert F, Vansteelandt K, Adamson C, De Winter FL, Sienaert P, et al. A longitudinal study of the association between basal ganglia volumes and psychomotor symptoms in subjects with late life depression undergoing ECT. Transl Psychiatry. 2021;11:199.
Belge JB, Van Diermen L, Schrijvers D, Sabbe B, Constant E, de Timary P, et al. The basal ganglia: A central hub for the psychomotor effects of electroconvulsive therapy. J Affect Disord. 2020;265:239–46.
Dong J, Hawes S, Wu J, Le W, Cai H. Connectivity and functionality of the globus pallidus externa under normal conditions and Parkinson’s disease. Front Neural Circuits. 2021;15:645287.
Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–75.
Williams D. Why so slow? Models of parkinsonian bradykinesia. Nat Rev Neurosci. 2024;25:573–86.
Marsden CD, Obeso JA. The functions of the basal ganglia and the paradox of stereotaxic surgery in Parkinson’s disease. Brain. 1994;117:877–97.
Poston KL, Eidelberg D. FDG PET in the evaluation of Parkinson’s Disease. PET Clin. 2010;5:55–64.
Giossi C, Rubin JE, Gittis A, Verstynen T, Vich C. Rethinking the external globus pallidus and information flow in cortico-basal ganglia-thalamic circuits. Eur J Neurosci. 2024;60:6129–44.
Naismith S, Hickie I, Ward PB, Turner K, Scott E, Little C, et al. Caudate nucleus volumes and genetic determinants of homocysteine metabolism in the prediction of psychomotor speed in older persons with depression. Am J Psychiatry. 2002;159:2096–8.
Desmidt T, Andersson F, Brizard B, Cottier JP, Patat F, Gissot V, et al. Cerebral blood flow velocity positively correlates with brain volumes in long-term remitted depression. Prog Neuropsychopharmacol Biol Psychiatry. 2018;81:243–9.
Wilson J, Allcock L, Mc Ardle R, Taylor JP, Rochester L. The neural correlates of discrete gait characteristics in ageing: A structured review. Neurosci Biobehav Rev. 2019;100:344–69.
Dumurgier J, Crivello F, Mazoyer B, Ahmed I, Tavernier B, Grabli D, et al. MRI atrophy of the caudate nucleus and slower walking speed in the elderly. NeuroImage. 2012;60:871–8.
Callisaya ML, Beare R, Phan TG, Chen J, Srikanth VK. Global and regional associations of smaller cerebral gray and white matter volumes with gait in older people. PLOS ONE. 2014;9:e84909.
van Diermen L, van den Ameele S, Kamperman AM, Sabbe BCG, Vermeulen T, Schrijvers D, et al. Prediction of electroconvulsive therapy response and remission in major depression: meta-analysis. Br J Psychiatry. 2018;212:71–80.
van Diermen L, Schrijvers D, Cools O, Birkenhäger TK, Fransen E, Sabbe BGCC. Distinguishing subgroups based on psychomotor functioning among patients with major depressive disorder. Neuropsychobiology. 2018;76:199–208.
van Diermen L, Walther S, Cools O, Fransen E, Birkenhäger TK, Sabbe BCG, et al. Observer-rated retardation but not agitation corresponds to objective motor measures in depression. Acta Neuropsychiatr. 2018;30:359–64.
Beheydt LL, Schrijvers D, Docx L, Bouckaert F, Hulstijn W, Sabbe B. Psychomotor retardation in elderly untreated depressed patients. Front Psychiatry. 2015;5:196.
Zapparoli L, Mariano M, Paulesu E. How the motor system copes with aging: a quantitative meta-analysis of the effect of aging on motor function control. Commun Biol. 2022;5:79.
Li KZ, Lindenberger U. Relations between aging sensory/sensorimotor and cognitive functions. Neurosci Biobehav Rev. 2002;26:777–83.
Asan L, Falfán-Melgoza C, Beretta CA, Sack M, Zheng L, Weber-Fahr W, et al. Cellular correlates of gray matter volume changes in magnetic resonance morphometry identified by two-photon microscopy. Sci Rep. 2021;11:4234.
Korczyn AD. Vascular parkinsonism—characteristics, pathogenesis and treatment. Nat Rev Neurol. 2015;11:319–26.
Taylor WD, Aizenstein HJ, Alexopoulos GS. The vascular depression hypothesis: mechanisms linking vascular disease with depression. Mol Psychiatry. 2013;18:963–74.
Pimontel MA, Reinlieb ME, Johnert LC, Garcon E, Sneed JR, Roose SP. The external validity of MRI-defined vascular depression. Int J Geriatr Psychiatry. 2013;28:1189–96.
Dalby RB, Frandsen J, Chakravarty MM, Ahdidan J, Sorensen L, Rosenberg R, et al. Correlations between Stroop task performance and white matter lesion measures in late-onset major depression. Psychiatry Res. 2012;202:142–9.
Park JH, Lee SB, Lee JJ, Yoon JC, Han JW, Kim TH, et al. Depression Plays a Moderating Role in the Cognitive Decline Associated With Changes of Brain White Matter Hyperintensities. J Clin Psychiatry. 2018;79:17m11763.
Reinlieb ME, Persaud A, Singh D, Garcon E, Rutherford BR, Pelton GH, et al. Vascular depression: overrepresented among African Americans? Int J Geriatr Psychiatry. 2014;29:470–7.
Brown PJ, Roose SP, O’Boyle KR, Ciarleglio A, Maas B, Igwe KC, et al. Frailty and its correlates in adults with late life depression. Am J Geriatr Psychiatry. 2020;28:145–54.
Hickie I, Scott E, Mitchell P, Wilhelm K, Austin MP, Bennett B. Subcortical hyperintensities on magnetic resonance imaging: Clinical correlates and prognostic significance in patients with severe depression. Biol Psychiatry. 1995;37:151–60.
Respino M, Jaywant A, Kuceyeski A, Victoria LW, Hoptman MJ, Scult MA, et al. The impact of white matter hyperintensities on the structural connectome in late-life depression: Relationship to executive functions. Neuroimage Clin. 2019;23:101852.
Coloigner J, Batail JM, Commowick O, Corouge I, Robert G, Barillot C, et al. White matter abnormalities in depression: A categorical and phenotypic diffusion MRI study. Neuroimage Clin. 2019;22:101710.
Bracht T, Federspiel A, Schnell S, Horn H, Höfle O, Wiest R, et al. Cortico-cortical white matter motor pathway microstructure is related to psychomotor retardation in major depressive disorder. PLoS ONE. 2012;7:1–8.
Hyett MP, Perry A, Breakspear M, Wen W, Parker GB. White matter alterations in the internal capsule and psychomotor impairment in melancholic depression. PLoS One. 2018;13:e0195672.
van Agtmaal MJM, Houben A, Pouwer F, Stehouwer CDA, Schram MT. Association of microvascular dysfunction with late-life depression: a systematic review and meta-analysis. JAMA Psychiatry. 2017;74:729–39.
Carvalho de Abreu DC, Pieruccini-Faria F, Son S, Montero-Odasso M, Camicioli R. Is white matter hyperintensity burden associated with cognitive and motor impairment in patients with parkinson’s disease? A systematic review and meta-analysis. Neurosci Biobehav Rev. 2024;161:105677.
Vande Casteele T, Laroy M, Van Cauwenberge M, Vanderlinden G, Vansteelandt K, Koole M, et al. Late life depression is not associated with Alzheimer-type tau: preliminary evidence from a next-generation tau ligand PET-MR study. Am J Geriatr Psychiatry. 2025;33:47–62.
Vande Casteele T, Laroy M, Van Cauwenberge M, Koole M, Dupont P, Sunaert S, et al. Preliminary evidence for preserved synaptic density in late-life depression. Transl Psychiatry. 2024;14:145.
Wilson H, Pagano G, de Natale ER, Mansur A, Caminiti SP, Polychronis S, et al. Mitochondrial Complex 1, Sigma 1, and Synaptic Vesicle 2A in Early Drug-Naive Parkinson’s Disease. Mov Disord. 2020;35:1416–27.
Johansen A, Armand S, Plavén-Sigray P, Nasser A, Ozenne B, Petersen IN, et al. Effects of escitalopram on synaptic density in the healthy human brain: a randomized controlled trial. Mol Psychiatry. 2023;28:4272–9.
Rossi R, Arjmand S, Bærentzen SL, Gjedde A, Landau AM. Synaptic Vesicle Glycoprotein 2A: Features and Functions. Front Neurosci. 2022;16:864514.
Serrano ME, Kim E, Petrinovic MM, Turkheimer F, Cash D. Imaging synaptic density: the next holy grail of neuroscience? Front Neurosci. 2022;16:796129.
Shanaki Bavarsad M, Spina S, Oehler A, Allen IE, Suemoto CK, Leite REP, et al. Comprehensive mapping of synaptic vesicle protein 2A (SV2A) in health and neurodegenerative diseases: a comparative analysis with synaptophysin and ground truth for PET-imaging interpretation. Acta Neuropathol. 2024;148:58.
Acknowledgements
We thank all participants for their contribution. We thank Jef Van Loock, Kwinten Porters and Michel Koole for their support at the PET-MR facility, Kim Serdons and radiopharmacy UZ Leuven team for tracer productions, Ruben Houbrechts at Icometrix® for technical support with WMH segmentation, Hans-Leo Teulings for support with Movalyzer®, Jari Mees, Aki Takamiya and Jeroen Blommaert for imaging software assistance.
Funding
M.V.C received support from KU Leuven grant PDMT2/24/089. M.L. received support from Research Foundation Flanders (FWO) grant 1168821 N. M.V., F.B., J.V.d.S., and L.E. are supported by FWO, grant G0C0319N, KU Leuven Fund C24/18/095, and the Sequoia Fund for Research on Aging and Mental Health. There was no industrial sponsoring related to the study.
Author information
Authors and Affiliations
Contributions
Concept: M.V.C., F.B., L.E., M.V.; Funding/resources: M.V.C., M.L., F.B., J.V.d.S, L.E., M.V., Data Curation & project administration: M.V.C., T.V.C., M.L., J.V.d.S., F.B., L.E., M.V.; Methodology: T.V.C, M.L., M.V.C.; Data analysis & statistical design: M.V.C. Statistical review: K.V.; Manuscript draft writing: M.V.C. Validation, visualization: M.V.C., K.V., K.V.L., L.E., M.V.; Supervision: F.B., L.E., M.V., Manuscript review/editing: M.V.C., T.V.C., M.L., K.V., J.V.d.S, F.B., L.E., M.V.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Van Cauwenberge, M.G.A., Vande Casteele, T., Laroy, M. et al. Motor signs in late life depression relate to altered subcortical synaptic density and gray matter volume: an 11C-UCB-J PET-MR study. Neuropsychopharmacol. 51, 661–671 (2026). https://doi.org/10.1038/s41386-025-02229-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-025-02229-6