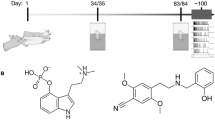
Abstract
Fragile X Syndrome (FXS) is the most common inherited intellectual disability and a leading monogenic cause of autism spectrum disorder (ASD). As a synaptic disorder, FXS involves the loss of Fragile X messenger ribonucleoprotein 1 (FMRP), leading to abnormal dendrite development and immature dendritic spines. Serotonergic signaling, essential for neuronal development and circuit remodeling, has been implicated in ASD and related conditions, including FXS, raising the possibility that serotonergic modulation could ameliorate neurodevelopmental impairments. This study investigated the therapeutic potential of psilocybin, a serotonergic compound, in the validated Fmr1-Δexon 8 rat model of FXS. Psilocybin microdosing rescued deficits in NOR. Importantly, its benefits on recognition memory persisted despite pretreatment with the 5HT2AR antagonist, volinanserin, or the 5HT1AR antagonist, WAY-100635, indicating that classical serotonergic receptor activation is not required. In contrast, pretreatment with the TrkB receptor antagonist, ANA-12, abolished psilocybin’s effects, implicating BDNF/TrkB signaling as essential. At the molecular level, psilocybin normalized mature BDNF (mBDNF), increased TrkB, and restored downstream AKT signaling in the prefrontal cortex of Fmr1-Δexon 8 rats, pathways strongly linked to synaptic plasticity and cognitive function. These findings demonstrate that psilocybin rescues object recognition memory deficits in this rat model of FXS via BDNF/TrkB-AKT signaling rather than serotonergic receptor mechanisms. By dissociating therapeutic effects from hallucinogenic pathways, our results highlight psilocybin microdosing as a promising therapeutic strategy for neurodevelopmental disorders such as FXS and ASD.
Similar content being viewed by others
Introduction
Fragile X Syndrome (FXS) is the most common inherited form of intellectual disability and the leading monogenic cause of Autism Spectrum Disorder (ASD), affecting ~1 in 7000 males and 1 in 11,000 females [1]. It results from a CGG repeat expansion in the FMR1 gene that silences expression of Fragile X Messenger Ribonucleoprotein 1 (FMRP) [2], a protein crucial for synaptic protein synthesis, mRNA transport, and activity-dependent synaptic plasticity. Its absence disrupts dendritic development and synaptic refinement, producing cognitive deficits, behavioral rigidity, and social dysfunction [2,3,4]. Nearly half of males and a significant portion of females with FXS meet criteria for ASD [5, 6].
Serotonergic dysregulation is a consistent feature of both ASD and FXS [7,8,9]. Serotonin regulates neurodevelopment and synaptic maturation, and aberrant serotonergic signaling has been consistently observed in individuals with ASD and FXS [8, 10]. Serotonergic drugs have been tested for their potential to improve social interaction and mood [11,12,13,14]. Psilocybin, a psychedelic tryptamine, is of particular interest: although its psychoactive effects are attributed to serotonin 2 A receptor (5HT2AR) agonism, its broader pharmacology suggests additional roles in neuroplasticity and cognition.
Psychedelics, including psilocybin, have recently been shown to promote neural plasticity by engaging brain-derived neurotrophic factor (BDNF) and its receptor TrkB [15, 16]. The BDNF/TrkB cascade is essential for synaptic development, learning, and memory [17, 18] and is disrupted in FXS and related neurodevelopmental disorders [19]. Given the functional interplay between serotonergic and neurotrophic systems, targeting BDNF/TrkB signaling in conjunction with serotonergic pathways may represent a new therapeutic strategy for FXS.
We previously reported that repeated low-dose psilocybin (“microdosing”) rescues object recognition deficits in the Fmr1-Δexon 8 rat [20], a validated model of FXS and ASD [21,22,23]. However, the mechanisms were unknown. While it is clear that psilocybin’s hallucinogenic effects depend on 5HT2AR activation [24, 25], it remains uncertain whether serotonergic receptors mediate its benefits on recognition memory. An alternative hypothesis is that psilocybin acts by restoring BDNF/TrkB-dependent plasticity.
In the present study, we investigated this hypothesis using pharmacological antagonism experiments and molecular analyses in Fmr1-Δexon 8 rats. We asked whether psilocybin’s benefits on short-term recognition memory require 5HT2A or 5HT1A receptor activation, and whether they depend on intact BDNF/TrkB signaling. Recognition memory was assessed with the novel object recognition (NOR) test, a widely used paradigm with prefrontal involvement. Molecular analyses examined Bdnf isoforms, mature BDNF (mBDNF), TrkB expression, and downstream AKT signaling in the prefrontal cortex (PFC).
By integrating behavioral and molecular approaches, we sought to determine whether psilocybin’s therapeutic effects in FXS derive from direct 5HT2A and 5HT1A receptor activation or involve neurotrophic mechanisms. Establishing this distinction has clinical importance: if efficacy can be achieved independently of serotonergic receptor activation, psilocybin or related compounds could provide benefits without hallucinogenic side effects, supporting microdosing as a safe and tolerable strategy for neurodevelopmental disorders.
Materials and methods
Experimental design and drugs
Wild-type (WT) and Fmr1-Δexon 8 rats were tested following the experimental design shown in Supplementary Fig. S1. Psilocybin was administered via oral gavage (0.1 mg/kg per os, p.o.) every other day for two weeks, from postnatal day (PND) 34 to PND 44. Animals were tested in the NOR test five days after the final administration to exclude any residual hallucinogenic effects and to capture enduring, post-acute outcomes [20]. Our dose regimen is consistent with a microdosing paradigm. Indeed, to provide quantitative support for our dosing choice, we applied standard FDA Km-based allometric scaling (rat Km = 6; human Km = 37) [26], yielding a human-equivalent dose (HED) of 0.016 mg/kg for our 0.1 mg/kg oral rat regimen — approximately 1.1 mg for a 70 kg adult — well within the empirically defined human microdose range of ~1–2.5 mg [27,28,29].
Experiment 1 investigated whether the recognition memory effects of psilocybin in Fmr1-Δexon 8 rats were dependent on 5HT2AR activation. The 5HT2AR antagonist volinanserin (M100907, Selleck Chemicals, USA) was dissolved in 5% Tween 80, 5% polyethylene glycol, and 90% saline, and administered intraperitoneally (i.p.) at the dose of 0.25 mg/kg 15 min before each psilocybin dose [30, 31]. This M100907 dose and pretreatment interval have been shown to suppress DOI- and LSD-induced head-twitch responses (HTRs) [31].
Experiment 2 examined the involvement of 5HT1AR in mediating the positive effects of psilocybin in the NOR test. The 5HT1AR antagonist WAY-100635 (MedChemExpress, USA) was dissolved in the same vehicle (VEH) and administered i.p. at the dose of 0.3 mg/kg 10 min prior to each psilocybin treatment [32].
Building on these results, Experiment 3 explored psilocybin-induced changes in the expression of 5HT1AR and 5HT2AR in the PFC by Western blot.
Experiments 4 and 5 tested the hypothesis that the beneficial effects of psilocybin in Fmr1-Δexon 8 rats might involve TrkB/BDNF signaling. The selective TrkB antagonist ANA-12 (Selleckchem, USA) was administered i.p. at the doses of 0.5 mg/kg (Experiment 4) and 1.0 mg/kg (Experiment 5), 3.5 h before each psilocybin administration. This pretreatment interval was selected based on ANA-12 pharmacokinetic profile, as the compound penetrates the brain within approximately 30 min but achieves maximal TrkB inhibition after 3–4 h, thereby ensuring effective receptor blockade during psilocybin exposure [33].
The timing of antagonist administration was further informed by pharmacokinetic data showing that oral psilocybin produces peak systemic psilocin levels and maximal HTRs within 15–30 min post-administration [34]. Thus, this schedule ensured overlap between the period of maximal TrkB inhibition and the peak of psilocybin’s central effects.
In Experiment 6, quantitative PCR (qPCR) was used to assess mRNA expression levels of total BDNF, as well as its exons I, IV, VI, and long isoforms, in PFC.
Experiment 7 further elucidated the molecular mechanisms underlying psilocybin’s effects by analyzing the expression of key neuroplasticity-related proteins (mBDNF, TrkB, pAKTS473, pAKT S473/AKT) via Western blot.
Animals
Male WT (Charles River Laboratories, Italy) and Fmr1-Δexon 8 rats (Horizon Discovery, formerly SAGE Labs, USA) on a Sprague-Dawley background were housed in groups of three under controlled environmental conditions: temperature 20–21 °C, humidity 55–65%, 12:12 h light/dark cycle (lights on at 07:00 h). Behavioral experiments and key experimental procedures were conducted between 9:00 a.m. and 2:30 p.m. Sample sizes (n) are indicated in the figure legends and were based on previous studies and power analyses conducted using G*Power software. A single cohort of animals was used to perform biochemical analyses in Experiments 3, 6, and 7, to reduce variability and limit animal use. Conversely, five independent cohorts were employed for the behavioral assessments, with one cohort dedicated to each behavioral experiment to ensure proper counterbalancing and avoid potential confounding effects of repeated testing. Outliers were identified using the Grubbs’ test in GraphPad Prism 8 (GraphPad Software, USA). The behavioral experiments were scored in blind conditions using the Observer 3.0 software (Noldus Information Technology, The Netherlands).
Procedures were approved by the Italian Ministry of Health (authorization n. 988/2020-PR) and conducted in accordance with the ARRIVE guidelines, the Italian Legislative Decree No. 26/2014, and the European Community Directive 2010/63/EU.
Novel object recognition (NOR) test
The test was conducted as previously described [35]. During the training phase, each rat was placed in an open field arena containing two identical objects (A1 and A2) and allowed to explore for 5 min. After a 30-min inter-trial interval, one familiar object (A3) and one novel object (B) were positioned in the exact location as during training. Each rat was then reintroduced to the arena for a 5-min test session.
Object exploration was scored when the animal was sniffing or touching the object with the nose and/or forepaws. The time spent exploring each object was recorded, and the discrimination index (DI) was calculated as the difference in time spent by each animal exploring the novel compared with the familiar object, divided by the total time spent exploring both objects in percentage.
To exclude potential confounding effects of locomotor or anxiety-like behavior, an independent cohort of Fmr1-Δexon 8 and WT rats was tested under identical housing, treatment, and experimental conditions in the open field and elevated plus-maze tasks (see Supplementary Information and Supplementary Fig. S2).
Biochemical analysis
Brain samples collection
Rats were decapitated, and their brains were rapidly removed from the skull on a cold plate. The PFC was manually dissected from fresh brains immediately following decapitation as a tissue block comprising the prelimbic, infralimbic and anterior cingulate cortex [36, 37]. Dissections were performed under microscopic guidance within 2 min and stored at −80 °C until analysis. Western blot and real-time PCR experiments were performed using PFC samples collected at PND 50.
Western blot analysis
PFC tissues were homogenized in ice-cold buffer (0.32 M sucrose, 0.1 mM PMSF, 1 mM HEPES, 0.1 mM EGTA, pH 7.4) supplemented with protease (Roche-Merk Life Science, Italy) and phosphatase (Sigma-Aldrich-Merk Life Science, Italy) inhibitors. Homogenates were sonicated, and protein concentrations were determined using the Bradford Protein Assay (Bio-Rad, Italy), with bovine serum albumin as standard. Samples were stored at −20 °C until analysis.
Eight micrograms of proteins per sample were separated by SDS-PAGE (14% gels) and transferred to nitrocellulose membranes (Bio-Rad, Italy). Membranes were blocked (I-Block, Life Technologies, Italy) in TBS + 0.1% Tween-20 buffer, incubated with phospho-specific antibodies, then stripped and reprobed for corresponding total proteins. Conditions of the primary antibodies are reported in the Supplementary Materials. Detection was performed using Clarity Western ECL substrate (Bio-Rad, USA) and chemiluminescence captured with a ChemiDoc XRS+ system (Bio-Rad, USA). Band intensities were quantified using ImageLab software, normalized to β-actin (43 kDa), and expressed as fold change relative to WT controls. Representative immunoblots are presented in Figs. 2C and 5F; examples of uncropped blots are shown in Supplementary Fig. S3. Each sample was analyzed in duplicate or triplicate gels, and results were averaged using a correction factor to adjust for inter-gel variability, calculated as: correction factor gel B =average of (OD protein of interest/OD β-actin for each sample loaded in gel A)/(OD protein of interest/OD β-actin for the same sample loaded in gel B). Correction factor gel C = average of (OD protein of interest/OD β-actin for each sample loaded in gel A)/(OD protein of interest/OD β-actin for the same sample loaded in gel C) [38].
Real-time PCR
Total RNA was isolated using PureZol reagent (Bio-Rad, Italy), quantified by Nanodrop spectrophotometry, and stored at −20 °C [39].
RNA samples were treated with DNase I (Thermo Scientific, RNase-free) for 30 min at 37 °C to remove genomic DNA, followed by EDTA inactivation (10 min, 65 °C). Gene expression was assessed by one-step RT-qPCR (CFX384 Real-Time System, Bio-Rad) using the iScriptTM kit (Bio-Rad, Italy). Each sample was run in triplicate in a 384-well plate. Data were analyzed with the comparative threshold cycle (ΔΔCt) using 36B4 as an internal standard. RT-qPCR analyses were performed to evaluate the gene expression of total Bdnf, Bdnf exon I, IV, VI, and Bdnf long. Sequences of primers and probes are reported in the Supplementary Materials.
Statistical analysis
Data are presented as mean ± standard error of the mean (SEM). Biochemical data were analyzed using two-way analysis of variance (ANOVA) to assess the effects of genotype and psilocybin. Behavioral data were analyzed using three-way ANOVA to evaluate the impact of genotype, psilocybin, and treatment with M100907, WAY-100635, or ANA-12. Tukey’s post hoc test was applied for multiple comparisons. Analyses were performed using GraphPad Prism version 8 (GraphPad Software, USA). Behavioral, biochemical (Western blot), and molecular (real-time PCR) data were analyzed by experimenters blinded to genotype and treatments.
Results
5HT1AR and 5HT2AR do not mediate the effects of psilocybin on object recognition in Fmr1- Δ exon 8 rats
As expected [20], psilocybin improved performance in the NOR task, and this effect was not secondary to changes in locomotor activity or emotionality (see the results of the open field and elevated plus-maze tests in Supplementary information). Pretreatment with the 5HT2AR antagonist M100907 (M100, 0.25 mg/kg) failed to block psilocybin’s rescue of object recognition in Fmr1-Δexon 8 rats (main effects of genotype: F1,66 = 12.57, p < 0.001; psilocybin: F1,66 = 11.95, p < 0.001; genotype × psilocybin: F1,66 = 20.40, p < 0.001; all other effects n.s.; Fig. 1A). Similarly, the 5HT1AR antagonist WAY-100635 (WAY, 0.3 mg/kg) failed to alter psilocybin’s effect (genotype: F1,59 = 11.43, p < 0.01; psilocybin: F1,59 = 4.95, p < 0.05; genotype × psilocybin: F1,59 = 23.31, p < 0.001; all other effects n.s.; Fig. 1C). Total objects exploration time was unchanged in both conditions (Figs. 1B, D and Supplementary Table 1), indicating that psilocybin’s enhancement of object recognition was not due to altered exploratory behavior. These results show that psilocybin’s pro-cognitive effects in the NOR task are independent of direct 5HT2AR and 5HT1AR activation.
Systemic administration of the 5HT2AR selective antagonist M100 (0.25 mg/kg, i.p.) did not counteract the positive effects of psilocybin (PSY) on object recognition in Fmr1-Δexon 8 rats (A). Total object exploration time during testing was unaffected by treatment across genotypes (B) (WT-VEH/VEH = 8, WT-VEH/PSY = 9, WT-M100/VEH = 9, WT-M100/PSY = 10, Fmr1-Δexon 8-VEH/VEH = 10, Fmr1-Δexon 8- VEH/PSY = 9, Fmr1-Δexon 8- M100/VEH = 10, Fmr1-Δexon 8-M100/PSY = 9). Similarly, administration of the 5HT1AR selective antagonist WAY (0.3 mg/kg, i.p.) failed to prevent the recognition memory-enhancing effects of psilocybin in Fmr1- Δexon 8 rats (C), and had no impact on total object exploration time (D) (WT-VEH/VEH = 9, WT-VEH/PSY = 10, WT-WAY/VEH = 6, WT-WAY/PSY = 9, Fmr1-Δexon 8-VEH/VEH = 8, Fmr1-Δexon 8- VEH/PSY = 8, Fmr1-Δexon 8- WAY/VEH = 7, Fmr1-Δexon 8-WAY/PSY = 10). Data represent mean ± SEM, **p < 0.01, ***p < 0.001 vs WT-VEH/VEH group, #p < 0.05, ##p < 0.01, vs Fmr1-Δexon 8-VEH/VEH group; (three-way ANOVA followed by Tukey’s post hoc test).
For all statistical details, see Supplementary Table 1.
Psilocybin selectively modulates 5HT1AR but not 5HT2AR protein levels in the PFC
No significant main effects of genotype, treatment, or genotype × treatment interaction were found on 5HT2AR expression (Fig. 2A). In contrast, 5HT1AR levels showed a significant main effect of treatment (psilocybin: F1,12 = 6.819, p < 0.05; all other effects n.s.; Fig. 2B), although post hoc comparisons did not reveal significant differences between individual groups.
Protein levels of 5HT2AR (A) and 5HT1AR (B). C shows representative immunoblots related to the expression levels of 5HT2AR (55 kDa) and 5HT1AR (60 kDa) in the PFC lysates. Each sample was analyzed in triplicate gels, and results were averaged after correction for inter-gel variability. Densitometric values were normalized to β-actin and expressed as fold change relative to WT vehicle-treated controls (WT-VEH). (WT-VEH = 4, Fmr1-Δexon 8-VEH = 4, WT-PSY = 4, Fmr1-Δexon 8-PSY = 4). Data represent means ± SEM (two-way ANOVA followed by Tukey’s post hoc test).
These results show that repeated psilocybin administration produces a modest treatment effect on 5HT1AR expression independent of genotype, while 5HT2AR levels remain unchanged in the PFC.
For all statistical details, see Supplementary Table 2.
TrkB receptor mediates the effects of psilocybin on object recognition in Fmr1- Δ exon 8 rats
Given prior evidence that psilocybin also acts via TrkB receptor activation [16], we investigated whether TrkB signaling contributes to the recognition memory-enhancing effects of psilocybin in Fmr1-Δexon 8 rats. At the lower dose (0.5 mg/kg), the TrkB antagonist ANA-12 did not modify psilocybin’s positive effect on object recognition (genotype: F1,64 = 24.23, p < 0.001; psilocybin: F1,64 = 5.004, p < 0.05; genotype × psilocybin: F1,64 = 16.77, p < 0.001; all other effects n.s.; Fig. 3A). Total object exploration time during test remained unaffected (Fig. 3B). Conversely, the higher dose of ANA-12 (1.0 mg/kg) abolished psilocybin’s beneficial effect on object recognition (genotype: F1,60 = 71.62, p < 0.001; psilocybin: F1,60 = 9.696, p < 0.01; genotype × psilocybin: F1,60 = 14.79, p < 0.001; ANA-12 × psilocybin: F1,60 = 5.156, p < 0.05; all other effects n.s.; Fig. 3C), leaving total exploration time unchanged (Fig. 3D). These results support a mechanistic link between psilocybin’s pro-cognitive actions in the NOR task and BDNF/TrkB pathway engagement.
Systemic administration of the selective TrkB antagonist ANA-12 at the dose of 0.5 mg/kg (i.p., ANA 0.5) did not prevent the recognition memory-enhancing effects of psilocybin (0.1 mg/kg, p.o.) in Fmr1-Δexon 8 rats, as measured by the novel object recognition (NOR) test (A). Total object exploration time during testing was unaffected across all groups (B). (WT-VEH/VEH = 7, WT-VEH/PSY = 8, WT-ANA0.5/VEH = 9, WT-ANA0.5/PSY = 9, Fmr1-Δexon 8-VEH/VEH = 10, Fmr1-Δexon 8- VEH/PSY = 10, Fmr1-Δexon 8- ANA0.5/VEH = 9, Fmr1-Δexon 8- ANA0.5/PSY = 10). In contrast, a higher dose of ANA-12 (1.0 mg/kg, i.p., ANA 1) effectively blocked psilocybin’s pro-cognitive effects in Fmr1-Δexon 8 rats (C), while still not altering total object exploration time (D). (WT-VEH/VEH = 9, WT-VEH/PSY = 8, WT-ANA1/VEH = 7, WT-ANA1/PSY = 9, Fmr1-Δexon 8-VEH/VEH = 8, Fmr1-Δexon 8- VEH/PSY = 9, Fmr1-Δexon 8- ANA1/VEH = 8, Fmr1-Δexon 8- ANA1/PSY = 10). Data represent mean ± SEM, ***p < 0.001 vs WT-VEH/VEH group, ###p < 0.001 vs Fmr1-Δexon 8-VEH/VEH group; $p < 0.05, vs Fmr1- Δexon 8-VEH/PSY (three-way ANOVA followed by Tukey’s post hoc test).
For all statistical details, see Supplementary Table 3.
Psilocybin enhances cortical Bdnf transcription in WT, but not in Fmr1- Δ exon 8 rats
Psilocybin significantly increased total Bdnf mRNA levels in WT rats, whereas no effect was observed in Fmr1-Δexon 8 animals, indicating a genotype-dependent impairment in transcriptional responsiveness (genotype: F1,16 = 35.65, p < 0.0001; genotype × psilocybin: F1,16 = 24.91, p = 0.0001; all other effects n.s.; Fig. 4A). In contrast, gene expression of Bdnf long, the pool of transcript with the long polyadenylation site at 3’UTR [40], was significantly affected by genotype only (genotype: F1,16 = 38.69; p < 0.0001; all other effects n.s.; Fig. 4B), suggesting that this transcript variant is not responsive to psilocybin. Conversely, a robust treatment effect was observed for Bdnf exon I mRNA, an activity-dependent transcript located in the soma [41]. Indeed, psilocybin markedly upregulated exon I expression in WT rats, while Fmr1-Δexon 8 rats exhibited significantly reduced baseline levels and failed to respond to treatment (genotype: F1,16 = 174.6, p < 0.0001; psilocybin: F1,16 = 26.14, p = 0.0001; genotype × psilocybin: F1,16 = 6.471, p < 0.05; Fig. 4C), reinforcing the notion of disrupted activity-dependent transcriptional regulation in the mutant genotype. Bdnf exon IV mRNA levels, an activity-dependent transcript targeted in the proximal dendrites [42], were altered only depending on the genotype (genotype: F1,16 = 22.31, p < 0.001; all other effects n.s.; Fig. 4D). Interestingly, Bdnf exon VI expression, an activity-dependent transcript targeted to the distal dendrites [43], was significantly reduced by psilocybin in WT rats compared to VEH controls, with Fmr1-Δexon 8 rats exhibiting overall lower Bdnf exon VI expression levels compared to WT-VEH (genotype: F1,16 = 8.062, p < 0.05; psilocybin: F1,16 = 8.707, p = 0.01; all other effects n.s.; Fig. 4E).
Data represent the mRNA levels for total Bdnf (A), Bdnf long (B), Bdnf exon I (C), Bdnf exon IV (D) and Bdnf exon VI (E) in the PFC expressed as a percentage of VEH-treated rats. (WT-VEH = 5, Fmr1-Δexon 8-VEH = 5, WT-PSY = 5, Fmr1-Δexon 8-PSY = 5). Data represent means ± SEM, *p < 0.05, **p < 0.01 and ***p < 0.001 vs WT-VEH group (two-way ANOVA followed by Tukey’s post hoc test).
Collectively, these findings demonstrate that psilocybin elicits a selective upregulation of total Bdnf and exon I transcripts in the PFC of WT rats, an effect that is absent in Fmr1-Δexon 8 animals. This suggests that FMRP is required for psilocybin-induced activation of specific Bdnf isoforms, pointing to impaired neuroplasticity-related transcriptional mechanisms in this rat model of FXS.
For all statistical details, see Supplementary Table 4.
Psilocybin enhances mBDNF-TrkB and AKT signaling in the PFC of Fmr1- Δ exon 8 rats
To investigate whether psilocybin modulates the BDNF/TrkB intracellular pathway, we assessed the expression of mBDNF, its high-affinity receptor TrkB, and one of its downstream signaling effector AKT by assessing both its levels of phosphorylation at Ser473 (pAKTS473) and expression in the PFC of WT and Fmr1-Δexon 8 rats.
Psilocybin did not alter mBDNF levels in WT rats but normalized its levels in Fmr1-Δexon 8 rats, which were significantly lower than those observed in WT controls (genotype: F1,16 = 5.103, p < 0.05; genotype × psilocybin: F1,16 = 6.946, p < 0.05; all other effects n.s.; Fig. 5A).
A mBDNF; B TrkB; C phosphorylated AKT (pAKTS473); D total AKT; E pAKT S473/AKT ratio measured in the whole homogenate of the PFC. Each sample was analyzed in duplicate gels, and results were averaged after correction for inter-gel variability. Densitometric values were normalized to β-actin and expressed as a percentage relative to vehicle-treated controls (WT-VEH). F shows representative immunoblots for TrkB (140 kDa), pAkt (ser473) and Akt (60 kDa), β-actin (43 kDa) and mBDNF (14 kDa). (WT-VEH = 5, Fmr1-Δexon 8-VEH = 5, WT-PSY = 5, Fmr1-Δexon 8-PSY = 5). Data represent means ± SEM, *p < 0.05, **p < 0.01 and ***p < 0.001 vs WT-VEH group; #p < 0.05, ##p < 0.01 vs Fmr1-Δexon 8-VEH group (two-way ANOVA followed by Tukey’s post hoc test).
In line with mBDNF levels, psilocybin did not change TrkB expression in WT animals while it increased TrkB protein levels in Fmr1-Δexon 8 rats (interaction genotype × psilocybin: F1,16 = 10.36, p < 0.01; all other effects n.s.; Fig. 5B).
Psilocybin reduced pAKTS473 levels in WT animals while it normalized the reduced phosphorylation of AKT observed in VEH-treated Fmr1-Δexon 8 rats (interaction genotype × psilocybin: F1,16 = 27.73, p < 0.001; all other effects n.s.; Fig. 5C).
Although AKT expression was altered by the genotype and treatment, no significant changes in the interaction between the two variables were observed (main effects of genotype: F1,16 = 103.5, p < 0.001; psilocybin: F1,16 = 25.88, p < 0.001; all other effects n.s.; Fig. 5D). As an indirect index of activation of TrkB downstream signaling, we measured the pAKT/AKT ratio. Psilocybin significantly reduced this ratio in WT animals and reversed the reduction observed in VEH-treated Fmr1-Δexon 8 rats, restoring levels comparable to WT controls (genotype: F1,16 = 5.286, p < 0.05; genotype × psilocybin: F1,16 = 37.39, p < 0.001; all other effects n.s.; Fig. 5E).
Collectively, these findings indicate that psilocybin selectively enhances mBDNF-mediated signaling and its downstream intracellular pathways in Fmr1-Δexon 8 rats.
For all statistical details, see Supplementary Table 5.
Discussion
Psychedelics, particularly psilocybin, are gaining increasing attention as treatments for psychiatric and neurodevelopmental disorders [20, 44,45,46,47,48,49]. Beyond their efficacy in anxiety and depression [50,51,52], recent work suggests that psychedelics may also enhance social behavior and cognitive performance, domains disrupted in ASD and FXS [14, 53]. Anecdotal reports further indicate that ASD individuals may experience lasting benefits, such as improved self-understanding and management of autistic traits [54]. Together, these findings support systematic investigation of psilocybin’s mechanisms in preclinical models of neurodevelopmental disorders.
Although psilocybin exerts its primary effects via activation of serotonergic 5HT2AR, the underlying mechanisms by which it may benefit ASD-related conditions remain poorly understood. Here, we confirmed that psilocybin rescues object recognition deficits in Fmr1-Δexon 8 rats, consistent with our prior findings [20]. Importantly, this benefit persisted despite pretreatment with selective antagonists of 5HT2AR or 5HT1AR, indicating that direct activation of these receptors is not required for psilocybin’s effects on recognition memory. Conversely, blockade of TrkB signaling abolished the effect, underscoring the necessity of BDNF-TrkB signaling in mediating psilocybin’s impact on object recognition.
This dissociation adds to a growing body of work showing that the therapeutic and hallucinogenic effects of psychedelics are not inseparable. While antagonism of 5HT2AR reliably prevents acute psychotomimetic responses, such as head-twitch behavior in rodents, it does not consistently block psilocybin-induced plasticity or sustained behavioral benefits [16, 25, 55]. For instance, Shao et al. [50] recently demonstrated that 5HT2ARs in pyramidal tract neurons are required for psilocybin’s anxiolytic and stress-alleviating effects in mice, underscoring a cell–type–specific contribution of cortical 5HT2AR signaling to its long-term impact on emotional reactivity. Conversely, Moliner et al. [16] reported that the effects of psychedelics on neurotrophic signaling, structural plasticity, and antidepressant-like behaviors in mice depend on TrkB binding and the enhancement of endogenous BDNF signaling, but are independent of 5HT2AR activation. Although our data suggest that classical membrane-bound 5HT2AR activation is unnecessary to mediate psilocybin’s benefits on object recognition, they do not exclude a contribution from intracellular 5HT2AR signaling, as recently suggested [56]. Indeed, it has been proposed that intracellular 5HT2AR activation contributes to neuroplastic effects of psychedelics [56], although the intracellular 5HT2AR hypothesis remains an evolving area of research. Future studies are therefore needed to disentangle the respective contributions of intracellular versus membrane-associated 5HT2AR receptor pools to psilocybin’s long-term effects.
Finally, although some authors argue that subjective psychedelic experiences may be necessary for therapeutic outcomes [57], growing evidence of 5HT2AR-independent antidepressant responses in the absence of hallucinogenic effects, together with our findings, support the possibility that psilocybin can engage distinct mechanisms to enhance cognition. This distinction is clinically important, as it opens the door to strategies that capture the therapeutic benefits of psychedelics while avoiding hallucinogenic liabilities.
The safety profile of the dosing regimen used in this study further supports this potential. Repeated administration of low-dose psilocybin was well tolerated in Fmr1-Δexon 8 rats, with no overt signs of adverse effects, including the absence of the “wet dog shake” response, a commonly used rodent behavioural correlate of hallucinogenic activity. Nonetheless, the lack of this behavioural marker does not exclude the possibility of more subtle central effects that may not be detectable using this measure alone. These findings align with reports that sub-perceptual doses in rodents produce beneficial effects without overt behavioral toxicity, supporting the feasibility of microdosing strategies for sensitive populations such as children and adolescents with neurodevelopmental disorders [58].
Within this context, elucidating the molecular mechanisms involved in psilocybin’s beneficial effects is crucial. Emerging evidence suggests that the therapeutic effects of psilocybin may depend, at least partially, on the modulation of the BDNF/TrkB pathway. Although psilocybin is not a direct TrkB agonist, it may act allosterically, enhancing the effects of BDNF released at active synapses [16]. Consistent with this model, previous work demonstrated that TrkB inhibition with ANA-12 abolishes psychedelic-induced dendritic growth [15]. Our findings align with an involvement of BDNF in psilocybin action, since the positive effects of psilocybin on object recognition in Fmr1-Δexon 8 rats were blocked by ANA-12, particularly at the higher dose (1.0 mg/kg). However, the upstream mechanisms linking psilocybin exposure to TrkB engagement were not directly addressed in the present study.
Importantly, psilocybin’s mechanism is unlikely to operate through an “either/or” process. Rather, it likely engages a cascade of interconnected signaling events. Activation of 5HT2AR may initiate upstream processes, including cortical burst firing, glutamate release, and immediate early gene induction, that converge on neurotrophin release and TrkB activation [15, 58, 59]. Recent evidence further demonstrates that serotonergic signaling via 5HT2AR can directly modulate TrkB receptor function through heteroreceptor complex formation [60], providing a mechanistic link between serotonergic and neurotrophic signaling pathways.
Taken together, our findings suggest that by the time psilocybin’s behavioral benefits emerge in this FXS model, engagement of the BDNF-TrkB pathway becomes indispensable, whereas isolated activation of 5HT2A/1A receptors is not sufficient to sustain the observed beneficial effects in the absence of TrkB signaling.
At the molecular level, our results reveal that psilocybin restores impaired neurotrophic signaling in Fmr1-Δexon 8 rats. Indeed, these animals exhibit reduced transcription of two of the major Bdnf exons, exon I and exon VI. The various isoforms of the complex Bdnf gene are under the control of distinct promoters and serve specialized functions in regulating BDNF expression and subcellular localization, thereby influencing whether BDNF primarily acts in the soma or dendrites [61]. Exon I is highly regulated by neuronal activity and calcium signaling within the soma [62], whereas exon VI, which is also activity-dependent, regulates structural and functional plasticity at distal dendrites [61]. Since exon I–containing mRNAs are important in activity-induced transcription in response to synaptic inputs, and can contribute to synaptic strengthening [41], their reduced transcription in Fmr1-Δexon 8 rats might contribute, at least in part, to dysfunction in the cortical-dependent behaviors, such as executive functions and object recognition [63,64,65,66,67]. Similarly, reduced exon VI transcription in these animals suggests a broader downregulation of PFC neuronal activation. The reduced expression of both transcripts, segregated in specific cellular compartments [61], may reflect a generalized impairment of cortical plasticity in Fmr1-Δexon 8 rats. Interestingly, in WT animals, psilocybin differently modulated the expression of the different exons, suggesting specific sub-mechanisms triggering BDNF response. Specifically, psilocybin enhanced total Bdnf gene expression, likely through activity-dependent upregulation of exon I. Since increased exon I expression is associated with adaptive coping responses to stress [68] as well as with enhanced mushroom spine density and size [62], these findings might suggest that psilocybin facilitates functional and structural plasticity under WT conditions. Conversely, the reduced expression of exon VI in WT rats may indicate reduced transcription of the specific neurotrophin pool at distal dendrites [43]. This reduction of Bdnf exon VI transcription in the PFC following psilocybin treatment in WT animals may represent a transient suppression of dendritically localized, activity-dependent BDNF signaling, facilitating a temporary weakening or pruning of existing synaptic connections that enables large-scale remodeling and reorganization of cortical microcircuits, an adaptive “reset” phase preceding the subsequent stabilization of newly configured synapses and network states [69]. More importantly, psilocybin rescued downstream deficits in Fmr1-Δexon 8 rats by normalizing mBDNF protein levels, increasing TrkB receptor expression, and restoring phosphorylation of AKT. This pattern suggests that psilocybin enhances the effectiveness of existing BDNF-TrkB signaling rather than driving new transcription, reactivating neuroplastic pathways that are otherwise blunted in FXS.
AKT signaling is a critical downstream effector of BDNF-TrkB and has been consistently implicated in the pathophysiology of FXS. Reduced activation of AKT has been documented in FXS patients, but not in patients with intellectual disability associated with obstetric complications, suggesting a role for this signaling pathway in the pathophysiology of FXS [70]. In the Fmr1-KO mouse model, both hyperactivation [71, 72] and hypoactivation [73] of the PI3K/AKT/mTOR pathway in the hippocampus and cortex have been reported. These seemingly divergent findings highlight the need for further investigation but consistently support the hypothesis that dysregulation of this pathway plays a critical role in FXS.
By reinstating AKT activation in Fmr1-Δexon 8 rats, psilocybin corrected a key molecular deficit linked to impaired synaptic plasticity and cognition. Interestingly, psilocybin modestly decreased the pAKT/AKT ratio in WT animals, perhaps reflecting a homeostatic mechanism to prevent excessive activation. Together, these data suggest that psilocybin selectively restores signaling imbalances in the FXS brain while maintaining stability in normal circuits.
Our results also align with earlier findings demonstrating altered BDNF and TrkB expression in Fmr1-KO mice and in neuronal cultures derived from these models [74, 75]. Moreover, exogenous enhancement of BDNF levels improved cognitive performance in Fmr1-KO mice, even in the absence of detectable baseline deficits in BDNF expression [76], underscoring the central role of this neurotrophin in supporting synaptic plasticity. The convergence of psilocybin-induced increases in BDNF and normalization of AKT activity provides a mechanistic explanation for its behavioral effects. Given the complex regulation of Bdnf isoforms [77], the selective changes we observed in exon-specific transcripts further suggest that psilocybin may fine-tune BDNF signaling at distinct subcellular compartments. However, since we have not provided a causal demonstration of their localization and direct function, this requires further study.
The parallels with ketamine provide additional insight. Despite acting on different primary targets, both psilocybin and ketamine rapidly engage BDNF-TrkB signaling [78] and produce long-lasting behavioral improvements at low, sub-perceptual doses. Prior work from our team has shown that ketamine’s antidepressant effects are separable from its reinforcing properties and are tightly linked to dynamic modulation of BDNF signaling [39, 79]. Our current findings suggest that psilocybin shares this capacity, pointing to BDNF-TrkB signaling as a convergent pathway across distinct classes of rapid-acting neuroplasticity-promoting agents. This convergence highlights the potential of targeting downstream neurotrophic signaling rather than receptor-specific pathways for therapeutic innovation.
Some limitations warrant consideration. First, our experiments were performed exclusively in male rats. Given the sex-specific differences documented in preclinical models of ASD [80] and the fact that FMRP loss can produce divergent effects in males and females [81], this represents an important limitation. However, a parallel cohort of female Fmr1-Δexon 8 rats is currently being tested to directly assess potential sex-dependent responses to psilocybin, ensuring that future work addresses this translationally relevant dimension.
Second, we focused on recognition memory using the NOR task, which involves prefrontal function, but it will be important to assess whether psilocybin also benefits other ASD-relevant domains such as social interaction, communication, and repetitive behaviors. Finally, while our data clearly implicate BDNF-TrkB-AKT signaling in mediating psilocybin’s effects, the specific cellular populations and circuit-level mechanisms remain to be elucidated. In particular, our molecular analyses were performed on PFC tissue, precluding cell-type–specific or circuit-resolved conclusions and not addressing potential contributions from other brain regions. Future studies employing regionally expanded, cell-type–specific, and circuit-level approaches will therefore be required to further refine the mechanistic interpretation of our findings.
In conclusion, we demonstrate that psilocybin rescues the object recognition deficits in Fmr1-Δexon 8 rats through reactivation of BDNF-TrkB-AKT signaling, independently of direct activation of 5HT2A and 5HT1A receptors (see Supplementary Fig. S4). By separating therapeutic from hallucinogenic effects, these findings support the hypothesis that low-dose psilocybin engages BDNF-TrkB-AKT signaling pathways implicated in neurodevelopmental disorders. At the same time, they underscore the need for careful preclinical and clinical evaluation before any conclusions regarding therapeutic applicability or safety can be drawn. More broadly, our results highlight BDNF-TrkB signaling as a shared pathway for therapeutic plasticity across diverse compounds, strengthening the rationale for interventions that modulate neurotrophic signaling to restore cognition in conditions characterized by impaired synaptic function.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Hunter J, Rivero-Arias O, Angelov A, Kim E, Fotheringham I, Leal J. Epidemiology of Fragile X syndrome: a systematic review and meta-analysis. Am J Med Genet Part A. 2014;164A:1648–58.
Maurin T, Zongaro S, Bardoni B. Fragile X syndrome: from molecular pathology to therapy. Neurosci Biobehav Rev. 2014;46:242–55.
Dictenberg JB, Swanger SA, Antar LN, Singer RH, Bassell GJ. A direct role for FMRP in activity-dependent dendritic mRNA transport links filopodial-spine morphogenesis to Fragile X syndrome. Dev cell. 2008;14:926–39.
Hagerman RJ, Berry-Kravis E, Hazlett HC, Bailey DB Jr., Moine H, Kooy RF, et al. Fragile X syndrome. Nat Rev Dis Primers. 2017;3:17065.
Kaufmann WE, Kidd SA, Andrews HF, Budimirovic DB, Esler A, Haas-Givler B, et al. Autism spectrum disorder in Fragile X syndrome: co-occurring conditions and current treatment. Pediatrics. 2017;139:S194–S206.
Hernandez RN, Feinberg RL, Vaurio R, Passanante NM, Thompson RE, Kaufmann WE. Autism spectrum disorder in Fragile X syndrome: a longitudinal evaluation. Am J Med Genet Part A. 2009;149A:1125–37.
Jiang CC, Lin LS, Long S, Ke XY, Fukunaga K, Lu YM, et al. Signalling pathways in autism spectrum disorder: mechanisms and therapeutic implications. Signal Transduct Target Ther. 2022;7:229.
Hanson AC, Hagerman RJ. Serotonin dysregulation in Fragile X Syndrome: implications for treatment. Intractable Rare Dis Res. 2014;3:110–7.
Muller CL, Anacker AMJ, Veenstra-VanderWeele J. The serotonin system in autism spectrum disorder: from biomarker to animal models. Neuroscience. 2016;321:24–41.
Chugani DC. Role of altered brain serotonin mechanisms in autism. Mol Psychiatry. 2002;7:S16–7.
Costa L, Sardone LM, Lacivita E, Leopoldo M, Ciranna L. Novel agonists for serotonin 5-HT7 receptors reverse metabotropic glutamate receptor-mediated long-term depression in the hippocampus of wild-type and Fmr1 KO mice, a model of Fragile X Syndrome. Front Behav Neurosci. 2015;9:65.
Davis AK, Barrett FS, May DG, Cosimano MP, Sepeda ND, Johnson MW, et al. Effects of psilocybin-assisted therapy on major depressive disorder: a randomized clinical trial. JAMA Psychiatry. 2021;78:481–89.
Nutt D, Erritzoe D, Carhart-Harris R. Psychedelic psychiatry’s brave new world. Cell. 2020;181:24–28.
Bhatt KV, Weissman CR. The effect of psilocybin on empathy and prosocial behavior: a proposed mechanism for enduring antidepressant effects. Npj Ment Health Res. 2024;3:7.
Ly C, Greb AC, Cameron LP, Wong JM, Barragan EV, Wilson PC, et al. Psychedelics promote structural and functional neural plasticity. Cell Rep. 2018;23:3170–82.
Moliner R, Girych M, Brunello CA, Kovaleva V, Biojone C, Enkavi G, et al. Psychedelics promote plasticity by directly binding to BDNF receptor TrkB. Nat Neurosci. 2023;26:1032–41.
Yoshii A, Constantine-Paton M. Postsynaptic BDNF-TrkB signaling in synapse maturation, plasticity, and disease. Dev Neurobiol. 2010;70:304–22.
Cohen-Cory S, Kidane AH, Shirkey NJ, Marshak S. Brain-derived neurotrophic factor and the development of structural neuronal connectivity. Dev Neurobiol. 2010;70:271–88.
Castren ML, Castren E. BDNF in Fragile X syndrome. Neuropharmacology. 2014;76:729–36.
Buzzelli V, Carbone E, Manduca A, Schiavi S, Feo A, Perederiy JV, et al. Psilocybin mitigates the cognitive deficits observed in a rat model of Fragile X syndrome. Psychopharmacology. 2023;240:137–47.
Golden CEM, Breen MS, Koro L, Sonar S, Niblo K, Browne A, et al. Deletion of the KH1 domain of Fmr1 Leads to transcriptional alterations and attentional deficits in rats. Cereb Cortex. 2019;29:2228–44.
Hamilton SM, Green JR, Veeraragavan S, Yuva L, McCoy A, Wu Y, et al. Fmr1 and Nlgn3 knockout rats: novel tools for investigating autism spectrum disorders. Behav Neurosci. 2014;128:103–9.
Schiavi S, Manduca A, Carbone E, Buzzelli V, Rava A, Feo A, et al. Anandamide and 2-arachidonoylglycerol differentially modulate autistic-like traits in a genetic model of autism based on FMR1 deletion in rats. Neuropsychopharmacology. 2023;48:897–907.
Vollenweider FX, Vollenweider-Scherpenhuyzen MF, Babler A, Vogel H, Hell D. Psilocybin induces schizophrenia-like psychosis in humans via a serotonin-2 agonist action. Neuroreport. 1998;9:3897–902.
Shao LX, Liao C, Gregg I, Davoudian PA, Savalia NK, Delagarza K, et al. Psilocybin induces rapid and persistent growth of dendritic spines in frontal cortex in vivo. Neuron. 2021;109:2535–44.e4.
Nair AB, Jacob S. A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm. 2016;7:27–31.
Syed OA, Petranker R, Tsang B. The effect of psychedelic microdosing on animal behavior: A review with recommendations for the field. Neurosci Biobehav Rev. 2025;174:106204.
MacCallum CA, Lo LA, Pistawka CA, Deol JK. Therapeutic use of psilocybin: practical considerations for dosing and administration. Front Psychiatry. 2022;13:1040217.
Cavanna F, Muller S, de la Fuente LA, Zamberlan F, Palmucci M, Janeckova L, et al. Microdosing with psilocybin mushrooms: a double-blind placebo-controlled study. Transl Psychiatry. 2022;12:307.
Madden JT, Reyna NC, Pentkowski NS. Antagonizing serotonin 2A (5-HT(2A)) receptors attenuates methamphetamine-induced reward and blocks methamphetamine-induced anxiety-like behaviors in adult male rats. Drug alcohol Depend. 2020;215:108178.
Jaster AM, Elder H, Marsh SA, de la Fuente Revenga M, Negus SS, Gonzalez-Maeso J. Effects of the 5-HT(2A) receptor antagonist volinanserin on head-twitch response and intracranial self-stimulation depression induced by different structural classes of psychedelics in rodents. Psychopharmacology. 2022;239:1665–77.
Misane I, Ogren SO. Selective 5-HT1A antagonists WAY 100635 and NAD-299 attenuate the impairment of passive avoidance caused by scopolamine in the rat. Neuropsychopharmacology. 2003;28:253–64.
Cazorla M, Premont J, Mann A, Girard N, Kellendonk C, Rognan D. Identification of a low-molecular-weight TrkB antagonist with anxiolytic and antidepressant activity in mice. J Clin Investig. 2011;121:1846–57.
Raithatha SA, Hagel JM, Matinkhoo K, Yu L, Press D, Cook SG, et al. Novel psilocin prodrugs with altered pharmacological properties as candidate therapies for treatment-resistant anxiety disorders. J Med Chem. 2024;67:1024–43.
Rava A, Buzzelli V, Feo A, Ascone F, Di Trapano M, Schiavi S, et al. Role of peroxisome proliferator-activated receptors alpha and gamma in mediating the beneficial effects of beta-caryophyllene in a rat model of Fragile X syndrome. Prog Neuropsychopharmacol Biol Psychiatry. 2025;136:111234.
Schiavi S, Iezzi D, Manduca A, Leone S, Melancia F, Carbone C, et al. Reward-related behavioral, neurochemical and electrophysiological changes in a rat model of autism based on prenatal exposure to valproic acid. Front Cell Neurosci. 2019;13:479.
Gray JM, Vecchiarelli HA, Morena M, Lee TT, Hermanson DJ, Kim AB, et al. Corticotropin-releasing hormone drives anandamide hydrolysis in the amygdala to promote anxiety. J Neurosci. 2015;35:3879–92.
Caffino L, Verheij MMM, Roversi K, Targa G, Mottarlini F, Popik P, et al. Hypersensitivity to amphetamine’s psychomotor and reinforcing effects in serotonin transporter knockout rats: glutamate in the nucleus accumbens. Br J Pharmacol. 2020;177:4532–47.
Caffino L, Mottarlini F, Piva A, Rizzi B, Fumagalli F, Chiamulera C. Temporal dynamics of BDNF signaling recruitment in the rat prefrontal cortex and hippocampus following a single infusion of a translational dose of ketamine. Neuropharmacology. 2024;242:109767.
Aid T, Kazantseva A, Piirsoo M, Palm K, Timmusk T. Mouse and rat BDNF gene structure and expression revisited. J Neurosci Res. 2007;85:525–35.
Ramnauth AD, Maynard KR, Kardian AS, Phan BN, Tippani M, Rajpurohit S, et al. Induction of Bdnf from promoter I following electroconvulsive seizures contributes to structural plasticity in neurons of the piriform cortex. Brain Stimul. 2022;15:427–33.
Chiaruttini C, Sonego M, Baj G, Simonato M, Tongiorgi E. BDNF mRNA splice variants display activity-dependent targeting to distinct hippocampal laminae. Mol Cell Neurosci. 2008;37:11–9.
Pattabiraman PP, Tropea D, Chiaruttini C, Tongiorgi E, Cattaneo A, Domenici L. Neuronal activity regulates the developmental expression and subcellular localization of cortical BDNF mRNA isoforms in vivo. Mol Cell Neurosci. 2005;28:556–70.
Lowe H, Toyang N, Steele B, Valentine H, Grant J, Ali A, et al. The Therapeutic Potential of Psilocybin. Molecules. 2021;26:2948.
Goodwin GM, Croal M, Feifel D, Kelly JR, Marwood L, Mistry S, et al. Psilocybin for treatment-resistant depression in patients taking a concomitant SSRI medication. Neuropsychopharmacology. 2023;48:1492–99.
Gukasyan N, Davis AK, Barrett FS, Cosimano MP, Sepeda ND, Johnson MW, et al. Efficacy and safety of psilocybin-assisted treatment for major depressive disorder: prospective 12-month follow-up. J Psychopharmacol. 2022;36:151–58.
Griffiths RR, Johnson MW, Carducci MA, Umbricht A, Richards WA, Richards BD, et al. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: a randomized double-blind trial. J Psychopharmacol. 2016;30:1181–97.
Markopoulos A, Inserra A, De Gregorio D, Gobbi G. Evaluating the potential use of serotonergic psychedelics in autism spectrum disorder. Front Pharmacol. 2021;12:749068.
Wang X, Lin C. Psychedelics and pro-social behaviors: a perspective on autism spectrum disorders. ACS Pharmacol Transl Sci. 2025;8:903–06.
Hibicke M, Landry AN, Kramer HM, Talman ZK, Nichols CD. Psychedelics, but not Ketamine, produce persistent antidepressant-like effects in a rodent experimental system for the study of depression. ACS Chem Neurosci. 2020;11:864–71.
Hernández-León A, Escamilla-Orozco RI, Tabal-Robles AR, Martínez-Vargas D, Romero-Bautista L, Escamilla-Soto G, et al. Antidepressant- and anxiolytic-like activities and acute toxicity evaluation of the Psilocybe cubensis mushroom in experimental models in mice. J Ethnopharmacol. 2024;320:117415.
Wang Z, Robbins B, Zhuang R, van Bruggen R, Sandini T, Li XM, et al. Psilocybin mitigates behavioral despair and cognitive impairment in treatment-resistant depression model using Wistar Kyoto rats. Sci Rep. 2025;15:18432.
Nardou R, Sawyer E, Song YJ, Wilkinson M, Padovan-Hernandez Y, de Deus JL, et al. Psychedelics reopen the social reward learning critical period. Nature. 2023;618:790–98.
Orsini AP. Autism on Acid: How LSD Helped Me Understand, Navigate, Alter & Appreciate My Autistic Perceptions. https://books.google.co.in/books?id=CCq5zgEACAAJ.
Hesselgrave N, Troppoli TA, Wulff AB, Cole AB, Thompson SM Harnessing psilocybin: antidepressant-like behavioral and synaptic actions of psilocybin are independent of 5-HT2R activation in mice. Proc Natl Acad Sci USA. 2021;118:e2022489118.
Vargas MV, Dunlap LE, Dong C, Carter SJ, Tombari RJ, Jami SA, et al. Psychedelics promote neuroplasticity through the activation of intracellular 5-HT2A receptors. Science. 2023;379:700–06.
Yaden DB, Griffiths RR. The subjective effects of psychedelics are necessary for their enduring therapeutic effects. ACS Pharmacol Transl Sci. 2021;4:568–72.
Bysiek A, Wojtas A, Szpregiel I, Wawrzczak-Bargiela A, Mackowiak M, Golembiowska K. The effect of low-dose psilocybin on brain neurotransmission and rat behavior. Prog Neuro Psychopharmacol Biol Psychiatry. 2025;138:111347.
Calder AE, Hasler G. Towards an understanding of psychedelic-induced neuroplasticity. Neuropsychopharmacology. 2023;48:104–12.
Ilchibaeva T, Tsybko A, Zeug A, Muller FE, Guseva D, Bischoff S, et al. Serotonin receptor 5-HT(2A) regulates trkB receptor function in heteroreceptor complexes. Cells. 2022;11:2384.
Baj G, Leone E, Chao MV, Tongiorgi E. Spatial segregation of BDNF transcripts enables BDNF to differentially shape distinct dendritic compartments. Proc Natl Acad Sci USA. 2011;108:16813–8.
Bach SV, Bauman AJ, Hosein D, Tuscher JJ, Ianov L, Greathouse KM, et al. Distinct roles of Bdnf I and Bdnf IV transcript variant expression in hippocampal neurons. Hippocampus. 2024;34:218–29.
Friedman NP, Robbins TW. The role of the prefrontal cortex in cognitive control and executive function. Neuropsychopharmacology. 2022;47:72–89.
Eichenbaum H. Prefrontal-hippocampal interactions in episodic memory. Nat Rev Neurosci. 2017;18:547–58.
Orsini CA, Kim JH, Knapska E, Maren S. Hippocampal and prefrontal projections to the basal amygdala mediate contextual regulation of fear after extinction. J Neurosci. 2011;31:17269–77.
Mumby DG. Perspectives on object-recognition memory following hippocampal damage: lessons from studies in rats. Behav Brain Res. 2001;127:159–81.
Morici JF, Bekinschtein P, Weisstaub NV. Medial prefrontal cortex role in recognition memory in rodents. Behav Brain Res. 2015;292:241–51.
Fumagalli F, Calabrese F, Luoni A, Bolis F, Racagni G, Riva MA. Modulation of BDNF expression by repeated treatment with the novel antipsychotic lurasidone under basal conditions and in response to acute stress. Int J Neuropsychopharmacol. 2012;15:235–46.
Kim J, He MJ, Widmann AK, Lee FS. The role of neurotrophic factors in novel, rapid psychiatric treatments. Neuropsychopharmacology. 2024;49:227–45.
Kovacs T, Bansagi B, Kelemen O, Keri S. Neuregulin 1-induced AKT and ERK phosphorylation in patients with Fragile X syndrome (FXS) and intellectual disability associated with obstetric complications. J Mol Neurosci. 2014;54:119–24.
Zhang B, Zhang J, Chen H, Qiao D, Guo F, Hu X, et al. Role of FMRP in AKT/mTOR pathway-mediated hippocampal autophagy in Fragile X syndrome. Prog Neuro Psychopharmacol Biol Psychiatry. 2024;134:111036.
Sharma A, Hoeffer CA, Takayasu Y, Miyawaki T, McBride SM, Klann E, et al. Dysregulation of mTOR signaling in Fragile X syndrome. J Neurosci. 2010;30:694–702.
Hu H, Qin Y, Bochorishvili G, Zhu Y, van Aelst L, Zhu JJ. Ras signaling mechanisms underlying impaired GluR1-dependent plasticity associated with Fragile X syndrome. J Neurosci. 2008;28:7847–62.
Louhivuori V, Vicario A, Uutela M, Rantamaki T, Louhivuori LM, Castren E, et al. BDNF and TrkB in neuronal differentiation of Fmr1-knockout mouse. Neurobiol Dis. 2011;41:469–80.
Chen YS, Zhang SM, Tan W, Zhu Q, Yue CX, Xiang P, et al. Early 7,8-Dihydroxyflavone administration ameliorates synaptic and behavioral deficits in the young fxs animal model by acting on BDNF-trkB pathway. Mol Neurobiol. 2023;60:2539–52.
Yuskaitis CJ, Mines MA, King MK, Sweatt JD, Miller CA, Jope RS. Lithium ameliorates altered glycogen synthase kinase-3 and behavior in a mouse model of Fragile X syndrome. Biochem Pharmacol. 2010;79:632–46.
You H, Lu B. Diverse Functions of Multiple Bdnf Transcripts Driven by Distinct Bdnf Promoters. Biomolecules. 2023;13:655.
Park D, Lee G, Lee WG, Kim B, Lee Y, Kim JW. The therapeutic potential of psilocybin beyond psychedelia through shared mechanisms with ketamine. Mol Psychiatry. 2025;30:4910–27.
Caffino L, Di Chio M, Giannotti G, Venniro M, Mutti A, Padovani L, et al. The modulation of BDNF expression and signalling dissects the antidepressant from the reinforcing properties of ketamine: Effects of single infusion vs. chronic self-administration in rats. Pharmacol Res. 2016;104:22–30.
Napolitano A, Schiavi S, La Rosa P, Rossi-Espagnet MC, Petrillo S, Bottino F, et al. Sex differences in autism spectrum disorder: diagnostic, neurobiological, and behavioral features. Front psychiatry. 2022;13:889636.
Nolan SO, Reynolds CD, Smith GD, Holley AJ, Escobar B, Chandler MA, et al. Deletion of Fmr1 results in sex-specific changes in behavior. Brain Behav. 2017;7:e00800.
Funding
The study was funded by the Italian Ministry of University and Research (MUR) with PRIN 2022 grants (2022CZTZ87 to Viviana Trezza and Lucia Caffino, 20227HRFPJ to Fabio Fumagalli). The study was partially funded by NOVA MENTIS Life Science Corp. At the time of study design, Dr. Marvin Hausman served as Scientific Director at NOVA MENTIS Life Science Corp. and contributed to the experimental design based on his clinical and translational expertise in drug development. The company had no role in data collection, data analysis, interpretation of the results, or manuscript preparation. NOVA MENTIS Life Science Corp. has since undergone corporate restructuring and has ceased its research operations. Dr. Francesca Mottarlini is a recipient of a fellowship from the Zardi-Gori Foundation. Open access funding provided by Università degli Studi Roma Tre within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Fabrizio Ascone, Valeria Buzzelli, Melania Di Trapano and Flavio Spano performed, analyzed, and contributed to the design of the behavioral experiments. Francesca Mottarlini, Paolo Miglioranza, Alessandro Rava, and Alessandro Feo performed, analyzed and contributed to the design of the biochemical experiments. Lucia Caffino and Fabio Fumagalli supervised and designed the biochemical experiments. Marvin Hausman and Kiminobu Sugaya contributed to the design of the behavioral and biochemical experiments. Fabrizio Ascone, Valeria Buzzelli and Viviana Trezza wrote the first draft of the paper. Viviana Trezza supervised the whole project. All authors contributed to the paper and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
Dr. Marvin Hausman previously served as Scientific Director at NOVA MENTIS Life Science Corp., which partially funded this study and provided the psilocybin used. All authors, including Dr. Hausman, declare that they have no current or prior financial, commercial, or advisory relationships with NOVA MENTIS Life Science Corp. beyond the contributions described above. NOVA MENTIS Life Science Corp. has since undergone corporate restructuring and has ceased its research operations. The authors declare no other competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ascone, F., Buzzelli, V., Mottarlini, F. et al. Psilocybin improves novel object recognition in a rat model of Fragile X Syndrome through the modulation of the BDNF/TrkB signaling pathway. Neuropsychopharmacol. (2026). https://doi.org/10.1038/s41386-026-02361-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41386-026-02361-x