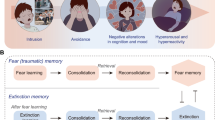
Abstract
Post-traumatic stress disorder (PTSD) remains one of psychiatry’s most challenging disorders, common, disabling, and biologically complex. Despite decades of research, only sertraline and paroxetine are FDA-approved, both with modest efficacy. Even first-line trauma-focused psychotherapies leave up to half of patients with persistent symptoms. However, advances in neurobiology are reframing PTSD as a disorder of maladaptive stress circuitry, neuroplasticity, and memory reconsolidation, opening new therapeutic possibilities. This comprehensive review examines current PTSD pharmacotherapy, emerging neurobiological targets, and investigational treatments, highlighting 45 actively enrolling clinical trials. Emerging approaches target the hypothalamic-pituitary-adrenal (HPA) axis, adrenergic signaling, glutamatergic/GABAergic systems, endocannabinoids, neuropeptides, and serotonergic-based psychedelics. Rapid-acting interventions represent a major advance in PTSD therapeutics. Ketamine produces symptom reductions within hours, MDMA-assisted psychotherapy has demonstrated Phase 3 efficacy, and neurosteroids offer novel approaches to targeting hyperarousal. However, questions remain regarding durability, optimal dosing, patient selection, precision approaches, and long-term safety. Several early-stage assets also show preliminary promise but require rigorous evaluation. At the same time, the development pipeline remains unforgiving: many mechanistically plausible candidates, from NK-1 antagonists to AMPA modulators, have failed in late-stage trials, often due to high placebo responses, patient heterogeneity despite subtype-specific medication targets, and translational gaps. Brexpiprazole’s recent FDA rejection, despite supportive early data, underscores persistent regulatory hurdles. While traditional approaches remain standard of care, breakthrough therapies represent paradigm shifts toward disease modification. Future progress depends on biomarker-guided precision medicine, novel trial designs to mitigate placebo effects, and integration of pharmacologic innovations with evidence-based psychotherapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Goldstein RB, Smith SM, Chou SP, Saha TD, Jung J, Zhang H, et al. The epidemiology of DSM-5 posttraumatic stress disorder in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions-III. Soc Psychiatry Psychiatr Epidemiol. 2016;51:1137–48. https://doi.org/10.1007/s00127-016-1208-5.
Schein J, Houle C, Urganus A, Cloutier M, Patterson-Lomba O, Wang Y, et al. Prevalence of post-traumatic stress disorder in the United States: a systematic literature review. Curr Med Res Opin. 2021;37:2151–61. https://doi.org/10.1080/03007995.2021.1978417.
Dewhurst E, Ettman CK, Hare Bork R, Thornburg B, Abdalla SM, Galea S, et al. Symptoms of posttraumatic stress during the COVID-19 pandemic in the governmental public health workforce and general population. J Public Health Manag Pr. 2024;30:E14–20. https://doi.org/10.1097/PHH.0000000000001837.
Yunitri N, Chu H, Kang XL, Jen HJ, Pien LC, Tsai HT, et al. Global prevalence and associated risk factors of posttraumatic stress disorder during COVID-19 pandemic: A meta-analysis. Int J Nurs Stud. 2022;126:104136. https://doi.org/10.1016/j.ijnurstu.2021.104136.
Gillespie CF, Bradley B, Mercer K, Smith AK, Conneely K, Gapen M, et al. Trauma exposure and stress-related disorders in inner city primary care patients. Gen Hosp Psychiatry. 2009;31:505–14. https://doi.org/10.1016/j.genhosppsych.2009.05.003.
Gluck RL, Hartzell GE, Dixon HD, Michopoulos V, Powers A, Stevens JS, et al. Trauma exposure and stress-related disorders in a large, urban, predominantly African-American, female sample. Arch Women’s Ment Health. 2021;24:893–901.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5), 2013.
Brewin CR, Cloitre M, Hyland P, Shevlin M, Maercker A, Bryant RA, et al. A review of current evidence regarding the ICD-11 proposals for diagnosing PTSD and complex PTSD. Clin Psychol Rev. 2017;58:1–15. https://doi.org/10.1016/j.cpr.2017.09.001.
Cohen E, Dr, Chazan S, Lerner M, Maimon E. Posttraumatic play in young children exposed to terrorism: An empirical study. Infant Ment Health J. 2010;31:159–81. https://doi.org/10.1002/imhj.20250.
Bryant RA, O’Donnell ML, Creamer M, McFarlane AC, Silove D. A multisite analysis of the fluctuating course of posttraumatic stress disorder. JAMA Psychiatry. 2013;70:839–46. https://doi.org/10.1001/jamapsychiatry.2013.1137.
Merz J, Schwarzer G, Gerger H. Comparative efficacy and acceptability of pharmacological, psychotherapeutic, and combination treatments in adults with posttraumatic stress disorder: a network meta-analysis. JAMA Psychiatry. 2019;76:904–13. https://doi.org/10.1001/jamapsychiatry.2019.0951.
Merians AN, Spiller T, Harpaz-Rotem I, Krystal JH, Pietrzak RH. Post-traumatic stress disorder. Med Clin North Am. 2023;107:85–99. https://doi.org/10.1016/j.mcna.2022.04.003.
Larsen SE, Fleming CJE, Resick PA. Residual symptoms following empirically supported treatment for PTSD. Psychol Trauma. 2019;11:207–15. https://doi.org/10.1037/tra0000384.
Kline AC, Otis NP, Crocker LD, Glassman LH, Michael Hunt W, Walter KH. Residual symptoms following trauma-focused treatment for comorbid posttraumatic stress disorder and major depressive disorder. J Psychiatr Res. 2025;182:42–9. https://doi.org/10.1016/j.jpsychires.2025.01.010.
Ressler KJ, Berretta S, Bolshakov VY, Rosso IM, Meloni EG, Rauch SL, et al. Post-traumatic stress disorder: clinical and translational neuroscience from cells to circuits. Nat Rev Neurol. 2022;18:273–88. https://doi.org/10.1038/s41582-022-00635-8.
Pfizer Labs. Zoloft [package insert]. U.S. Food and Drug Administration; 2016 Jul. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/019839s74s86s87_20990s35s44s45lbl.pdf.
GlaxoSmithKline. Paxil [package insert]. U.S. Food and Drug Administration; 2024 Jan. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/020031s083%2C020710s051lbl.pdf.
Williams T, Phillips NJ, Stein DJ, Ipser JC. Pharmacotherapy for post traumatic stress disorder (PTSD). Cochrane Database Syst Rev. 2022;3:CD002795. https://doi.org/10.1002/14651858.CD002795.pub3.
Martin A, Naunton M, Kosari S, Peterson G, Thomas J, Christenson JK. Treatment Guidelines for PTSD: A Systematic Review. J Clin Med. 2021;10:4175 https://doi.org/10.3390/jcm10184175.
Fisher PM, Meltzer CC, Price JC, Coleman RL, Ziolko SK, Becker C, et al. Medial prefrontal cortex 5-HT(2A) density is correlated with amygdala reactivity, response habituation, and functional coupling. Cereb Cortex. 2009;19:2499–507. https://doi.org/10.1093/cercor/bhp022.
Andolina D, Maran D, Valzania A, Conversi D, Puglisi-Allegra S. Prefrontal/amygdalar system determines stress coping behavior through 5-HT/GABA connection. Neuropsychopharmacology. 2013;38:2057–67. https://doi.org/10.1038/npp.2013.107.
Zoellner LA, Schulz PM, Campbell‑Law L, et al. Clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults [Internet]. Washington (DC): American Psychological Association; 2025 [cited 2025 Jul 12]. Available from: https://www.apa.org/ptsd-guideline/ptsd.pdf.
Bailey CR, Cordell E, Sobin SM, Neumeister A. Recent progress in understanding the pathophysiology of post-traumatic stress disorder: implications for targeted pharmacological treatment. CNS Drugs. 2013;27:221–32. https://doi.org/10.1007/s40263-013-0051-4.
Davidson J, Baldwin D, Stein DJ, Kuper E, Benattia I, Ahmed S, et al. Treatment of posttraumatic stress disorder with venlafaxine extended release: a 6-month randomized controlled trial. Arch Gen Psychiatry. 2006;63:1158–65. https://doi.org/10.1001/archpsyc.63.10.1158.
Dunlop BW, Rakofsky JJ, Newport DJ, Mletzko-Crowe T, Barone K, Nemeroff CB, et al. Efficacy of Vortioxetine Monotherapy for Posttraumatic Stress Disorder: A Randomized, Placebo-Controlled Trial. J Clin Psychopharmacol. 2021;41:172–9. https://doi.org/10.1097/JCP.0000000000001363.
Walderhaug E, Kasserman S, Aikins D, Vojvoda D, Nishimura C, Neumeister A. Effects of duloxetine in treatment-refractory men with posttraumatic stress disorder. Pharmacopsychiatry. 2010;43:45–9. https://doi.org/10.1055/s-0029-1237694.
Sowa-Kućma M, Pańczyszyn-Trzewik P, Misztak P, Jaeschke RR, Sendek K, Styczeń K, et al. Vortioxetine: A review of the pharmacology and clinical profile of the novel antidepressant. Pharm Rep. 2017;69:595–601. https://doi.org/10.1016/j.pharep.2017.01.030.
Sanchez C, Asin KE, Artigas F. Vortioxetine, a novel antidepressant with multimodal activity: review of preclinical and clinical data. Pharm Ther. 2015;145:43–57. https://doi.org/10.1016/j.pharmthera.2014.07.001.
de Moraes Costa G, Zanatta FB, Ziegelmann PK, Soares Barros AJ, Mello CF. Pharmacological treatments for adults with post-traumatic stress disorder: A network meta-analysis of comparative efficacy and acceptability. J Psychiatr Res. 2020;130:412–20. https://doi.org/10.1016/j.jpsychires.2020.07.046.
Zhao YF, Huang ZD, Gu HY, Guo GL, Yuan RX, Zhang C. Key Clinical interest outcomes of pharmaceutical administration for veterans with post-traumatic stress disorder based on pooled evidences of 36 randomised controlled trials with 2,331 adults. Front Pharm. 2020;11:602447. https://doi.org/10.3389/fphar.2020.602447.
Han C, Pae CU, Wang SM, Lee SJ, Patkar AA, Masand PS, et al. The potential role of atypical antipsychotics for the treatment of posttraumatic stress disorder. J Psychiatr Res. 2014;56:72–81. https://doi.org/10.1016/j.jpsychires.2014.05.003.
Liu XH, Xie XH, Wang KY, Cui H. Efficacy and acceptability of atypical antipsychotics for the treatment of post-traumatic stress disorder: a meta-analysis of randomized, double-blind, placebo-controlled clinical trials. Psychiatry Res. 2014;219:543–9. https://doi.org/10.1016/j.psychres.2014.05.027.
Ahearn EP, Juergens T, Cordes T, Becker T, Krahn D. A review of atypical antipsychotic medications for posttraumatic stress disorder. Int Clin Psychopharmacol. 2011;26:193–200. https://doi.org/10.1097/YIC.0b013e3283473738.
Pae CU, Lim HK, Peindl K, Ajwani N, Serretti A, Patkar AA, et al. The atypical antipsychotics olanzapine and risperidone in the treatment of posttraumatic stress disorder: a meta-analysis of randomized, double-blind, placebo-controlled clinical trials. Int Clin Psychopharmacol. 2008;23:1–8. https://doi.org/10.1097/YIC.0b013e32825ea324.
Villarreal G, Hamner MB, Cañive JM, Robert S, Calais LA, Durklaski V, et al. Efficacy of Quetiapine monotherapy in posttraumatic stress disorder: a randomized, placebo-controlled trial. Am J Psychiatry. 2016;173:1205–12. https://doi.org/10.1176/appi.ajp.2016.15070967.
Britnell SR, Jackson AD, Brown JN, Capehart BP. Aripiprazole for post-traumatic stress disorder: a systematic review. Clin Neuropharmacol. 2017;40:273–8. https://doi.org/10.1097/WNF.0000000000000251.
Naylor JC, Kilts JD, Bradford DW, Strauss JL, Capehart BP, Szabo ST, et al. A pilot randomized placebo-controlled trial of adjunctive aripiprazole for chronic PTSD in US military Veterans resistant to antidepressant treatment. Int Clin Psychopharmacol. 2015;30:167–74. https://doi.org/10.1097/YIC.0000000000000061.
Youssef NA, Marx CE, Bradford DW, Zinn S, Hertzberg MA, Kilts JD, et al. An open-label pilot study of aripiprazole for male and female veterans with chronic post-traumatic stress disorder who respond suboptimally to antidepressants. Int Clin Psychopharmacol. 2012;27:191–6. https://doi.org/10.1097/YIC.0b013e328352ef4e.
Robert S, Hamner MB, Durkalski VL, Brown MW, Ulmer HG. An open-label assessment of aripiprazole in the treatment of PTSD. Psychopharmacol Bull. 2009;42:69–80.
Morgenthaler TI, Auerbach S, Casey KR, Kristo D, Maganti R, Ramar K, et al. Position paper for the treatment of nightmare disorder in adults: An American Academy of Sleep Medicine Position Paper. J Clin Sleep Med. 2018;14:1041–55. https://doi.org/10.5664/jcsm.7178.
Boehnlein JK, Kinzie JD. Pharmacologic reduction of CNS noradrenergic activity in PTSD: the case for clonidine and prazosin. J Psychiatr Pr. 2007;13:72–8. https://doi.org/10.1097/01.pra.0000265763.79753.c1.
Raskind MA, Peskind ER, Hoff DJ, Hart KL, Holmes HA, Warren D, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61:928–34. https://doi.org/10.1016/j.biopsych.2006.06.032.
Raskind MA, Peskind ER, Kanter ED, Petrie EC, Radant A, Thompson CE, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160:371–3. https://doi.org/10.1176/appi.ajp.160.2.371.
Germain A, Richardson R, Moul DE, Mammen O, Haas G, Forman SD, et al. Placebo-controlled comparison of prazosin and cognitive-behavioral treatments for sleep disturbances in US Military Veterans. J Psychosom Res. 2012;72:89–96. https://doi.org/10.1016/j.jpsychores.2011.11.010.
Taylor FB, Martin P, Thompson C, Williams J, Mellman TA, Gross C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63:629–32. https://doi.org/10.1016/j.biopsych.2007.07.001.
Raskind MA, Peterson K, Williams T, Hoff DJ, Hart K, Holmes H, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170:1003–10. https://doi.org/10.1176/appi.ajp.2013.12081133.
Raskind MA, Peskind ER, Chow B, Harris C, Davis-Karim A, Holmes HA, et al. Trial of Prazosin for Post-Traumatic Stress Disorder in Military Veterans. N Engl J Med. 2018;378:507–17. https://doi.org/10.1056/NEJMoa1507598.
Trincavelli ML, Da Pozzo E, Daniele S, Martini C. The GABAA-BZR complex as target for the development of anxiolytic drugs. Curr Top Med Chem. 2012;12:254–69. https://doi.org/10.2174/1568026799078787.
Lund BC, Bernardy NC, Alexander B, Friedman MJ. Declining benzodiazepine use in veterans with posttraumatic stress disorder. J Clin Psychiatry. 2012;73:292–6. https://doi.org/10.4088/JCP.10m06775.
Delaney AJ, Sah P. GABA receptors inhibited by benzodiazepines mediate fast inhibitory transmission in the central amygdala. J Neurosci. 1999;19:9698–704. https://doi.org/10.1523/JNEUROSCI.19-22-09698.1999.
Campos B, Vinder V, Passos RBF, Coutinho ESF, Vieira NCP, Leal KB, et al. To BDZ or not to BDZ? That is the question! Is there reliable scientific evidence for or against using benzodiazepines in the aftermath of potentially traumatic events for the prevention of PTSD? A systematic review and meta-analysis. J Psychopharmacol. 2022;36:449–59. https://doi.org/10.1177/02698811221080464.
Schumacher S, Niemeyer H, Engel S, Cwik JC, Laufer S, Klusmann H, et al. HPA axis regulation in posttraumatic stress disorder: A meta-analysis focusing on potential moderators. Neurosci Biobehav Rev. 2019;100:35–57. https://doi.org/10.1016/j.neubiorev.2019.02.005.
Ross DA, Arbuckle MR, Travis MJ, Dwyer JB, van Schalkwyk GI, Ressler KJ. An integrated neuroscience perspective on formulation and treatment planning for posttraumatic stress disorder: an educational review. JAMA Psychiatry. 2017;74:407–15. https://doi.org/10.1001/jamapsychiatry.2016.3325.
Florido A, Velasco ER, Monari S, Cano M, Cardoner N, Sandi C, et al. Glucocorticoid-based pharmacotherapies preventing PTSD. Neuropharmacology. 2023;224:109344. https://doi.org/10.1016/j.neuropharm.2022.109344.
Kothgassner OD, Pellegrini M, Goreis A, Giordano V, Edobor J, Fischer S, et al. Hydrocortisone administration for reducing post-traumatic stress symptoms: A systematic review and meta-analysis. Psychoneuroendocrinology. 2021;126:105168. https://doi.org/10.1016/j.psyneuen.2021.105168.
Schelling G, Kilger E, Roozendaal B, de Quervain DJ, Briegel J, Dagge A, et al. Stress doses of hydrocortisone, traumatic memories, and symptoms of posttraumatic stress disorder in patients after cardiac surgery: a randomized study. Biol Psychiatry. 2004;55:627–33. https://doi.org/10.1016/j.biopsych.2003.09.014.
Weis F, Kilger E, Roozendaal B, de Quervain DJ, Lamm P, Schmidt M, et al. Stress doses of hydrocortisone reduce chronic stress symptoms and improve health-related quality of life in high-risk patients after cardiac surgery: a randomized study. J Thorac Cardiovasc Surg. 2006;131:277–82. https://doi.org/10.1016/j.jtcvs.2005.07.063.
Zohar J, Yahalom H, Kozlovsky N, Cwikel-Hamzany S, Matar MA, Kaplan Z, et al. High dose hydrocortisone immediately after trauma may alter the trajectory of PTSD: interplay between clinical and animal studies. Eur Neuropsychopharmacol. 2011;21:796–809. https://doi.org/10.1016/j.euroneuro.2011.06.001.
Delahanty DL, Gabert-Quillen C, Ostrowski SA, Nugent NR, Fischer B, Morris A, et al. The efficacy of initial hydrocortisone administration at preventing posttraumatic distress in adult trauma patients: a randomized trial. CNS Spectr. 2013;18:103–11. https://doi.org/10.1017/S1092852913000096.
Carmi L, Zohar J, Weissman T, Juven-Wetzler A, Bierer L, Yehuda R, et al. Hydrocortisone in the emergency department: a prospective, double-blind, randomized, controlled posttraumatic stress disorder study. Hydrocortisone during golden hours. CNS Spectr. 2023;28:457–63. https://doi.org/10.1017/S1092852922000852.
Shaked G, Shaked D, Sebbag G, Czeiger D. The effect of steroid treatment on whiplash associated syndrome: a controlled randomized prospective trial. Eur J Trauma Emerg Surg. 2021;47:1115–22. https://doi.org/10.1007/s00068-019-01282-3.
Kok L, Hillegers MH, Veldhuijzen DS, Cornelisse S, Nierich AP, van der Maaten JM, et al. Dexamethasone for Cardiac Surgery Study Group. The effect of Dexamethasone on symptoms of posttraumatic stress disorder and depression after cardiac surgery and intensive care admission: longitudinal follow-up of a randomized controlled trial. Crit Care Med. 2016;44:512–20. https://doi.org/10.1097/CCM.0000000000001419.
Graebener AH, Michael T, Holz E, Lass-Hennemann J. Repeated cortisol administration does not reduce intrusive memories - A double blind placebo controlled experimental study. Eur Neuropsychopharmacol. 2017;27:1132–43. https://doi.org/10.1016/j.euroneuro.2017.09.001.
Aerni A, Traber R, Hock C, Roozendaal B, Schelling G, Papassotiropoulos A, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiatry. 2004;161:1488–90. https://doi.org/10.1176/appi.ajp.161.8.1488. AugPMID: 15285979.
Yehuda R, Bierer LM, Pratchett LC, Lehrner A, Koch EC, Van Manen JA, et al. Cortisol augmentation of a psychological treatment for warfighters with posttraumatic stress disorder: Randomized trial showing improved treatment retention and outcome. Psychoneuroendocrinology. 2015;51:589–97. https://doi.org/10.1016/j.psyneuen.2014.08.004.
Lehrner A, Hildebrandt T, Bierer LM, Flory JD, Bader HN, Makotkine I, et al. A randomized, double-blind, placebo-controlled trial of hydrocortisone augmentation of Prolonged Exposure for PTSD in U.S. combat veterans. Behav Res Ther. 2021;144:103924. https://doi.org/10.1016/j.brat.2021.103924.
Surís A, North C, Adinoff B, Powell CM, Greene R. Effects of exogenous glucocorticoid on combat-related PTSD symptoms. Ann Clin Psychiatry. 2010;22:274–9.
Ludäscher P, Schmahl C, Feldmann RE Jr, Kleindienst N, Schneider M, Bohus M. No evidence for differential dose effects of hydrocortisone on intrusive memories in female patients with complex post-traumatic stress disorder-a randomized, double-blind, placebo-controlled, crossover study. J Psychopharmacol. 2015;29:1077–84. https://doi.org/10.1177/0269881115592339.
Mehta D, Gonik M, Klengel T, Rex-Haffner M, Menke A, Rubel J, et al. Using polymorphisms in FKBP5 to define biologically distinct subtypes of posttraumatic stress disorder: evidence from endocrine and gene expression studies. Arch Gen Psychiatry. 2011;68:901–10. https://doi.org/10.1001/archgenpsychiatry.2011.50.
Li H, Su P, Lai TK, Jiang A, Liu J, Zhai D, et al. The glucocorticoid receptor-FKBP51 complex contributes to fear conditioning and posttraumatic stress disorder. J Clin Invest. 2020;130:877–89. https://doi.org/10.1172/JCI130363.
Göver T, Slezak M. Targeting glucocorticoid receptor signaling pathway for treatment of stress-related brain disorders. Pharm Rep. 2024;76:1333–45. https://doi.org/10.1007/s43440-024-00654-w.
Hartmann J, Klengel C, Dillmann LJ, Hisey EE, Hafner K, Shukla R, et al. SKA2 enhances stress-related glucocorticoid receptor signaling through FKBP4-FKBP5 interactions in neurons. Proc Natl Acad Sci USA. 2024;121:e2417728121. https://doi.org/10.1073/pnas.2417728121.
Strawn JR, Geracioti TD Jr. Noradrenergic dysfunction and the psychopharmacology of posttraumatic stress disorder. Depress Anxiety. 2008;25:260–71. https://doi.org/10.1002/da.20292.
Brunet A, Saumier D, Liu A, Streiner DL, Tremblay J, Pitman RK. Reduction of PTSD Symptoms With Pre-Reactivation Propranolol Therapy: A Randomized Controlled Trial. Am J Psychiatry. 2018;175:427–33. https://doi.org/10.1176/appi.ajp.2017.17050481.
Raut SB, Canales JJ, Ravindran M, Eri R, Benedek DM, Ursano RJ, et al. Effects of propranolol on the modification of trauma memory reconsolidation in PTSD patients: A systematic review and meta-analysis. J Psychiatr Res. 2022;150:246–56. https://doi.org/10.1016/j.jpsychires.2022.03.045.
Kamboj SK, Gong AT, Sim Z, Rashid AA, Baba A, Iskandar G, et al. Reduction in the occurrence of distressing involuntary memories following propranolol or hydrocortisone in healthy women. Psychol Med. 2020;50:1148–55. https://doi.org/10.1017/S0033291719001028.
Li H, Zhang Z, Yang S, Zhu G. Systematic review and meta-analysis of propranolol in the prevention and treatment of post-traumatic stress disorder. Front Pharm. 2025;16:1545493. https://doi.org/10.3389/fphar.2025.1545493.
Leal Santos S, Stackmann M, Muñoz Zamora A, Mastrodonato A, De Landri AV, Vaughan N, et al. Propranolol Decreases Fear Expression by Modulating Fear Memory Traces. Biol Psychiatry. 2021;89:1150–61. https://doi.org/10.1016/j.biopsych.2021.01.005.
Zhou J, Luo Y, Zhang JT, Li MX, Wang CM, Guan XL, et al. Propranolol decreases retention of fear memory by modulating the stability of surface glutamate receptor GluA1 subunits in the lateral amygdala. Br J Pharm. 2015;172:5068–82. https://doi.org/10.1111/bph.13272.
Southwick SM, Morgan CA 3rd, Charney DS, High JR. Yohimbine use in a natural setting: effects on posttraumatic stress disorder. Biol Psychiatry. 1999;46:442–4. https://doi.org/10.1016/s0006-3223(99)00107-9.
Rasmusson AM, Hauger RL, Morgan CA, Bremner JD, Charney DS, Southwick SM. Low baseline and yohimbine-stimulated plasma neuropeptide Y (NPY) levels in combat-related PTSD. Biol Psychiatry. 2000;47:526–39. https://doi.org/10.1016/s0006-3223(99)00185-7.
Bremner JD, Krystal JH, Southwick SM, Charney DS. Noradrenergic mechanisms in stress and anxiety: II. Clinical studies. Synapse. 1996;23:39–51.
Southwick SM, Bremner JD, Rasmusson A, Morgan CA 3rd, Arnsten A, Charney DS. Role of norepinephrine in the pathophysiology and treatment of posttraumatic stress disorder. Biol Psychiatry. 1999;46:1192–204. https://doi.org/10.1016/s0006-3223(99)00219-x.
Mahan AL, Ressler KJ. Fear conditioning, synaptic plasticity and the amygdala: implications for posttraumatic stress disorder. Trends Neurosci. 2012;35:24–35. https://doi.org/10.1016/j.tins.2011.06.007.
Iqbal J, Huang GD, Xue YX, Yang M, Jia XJ. The neural circuits and molecular mechanisms underlying fear dysregulation in posttraumatic stress disorder. Front Neurosci. 2023;17:1281401. https://doi.org/10.3389/fnins.2023.1281401.
Averill LA, Purohit P, Averill CL, Boesl MA, Krystal JH, Abdallah CG. Glutamate dysregulation and glutamatergic therapeutics for PTSD: Evidence from human studies. Neurosci Lett. 2017;649:147–55. https://doi.org/10.1016/j.neulet.2016.11.064.
Huang J, Xu F, Yang L, Tuolihong L, Wang X, Du Z, et al. Involvement of the GABAergic system in PTSD and its therapeutic significance. Front Mol Neurosci. 2023;16:1052288. https://doi.org/10.3389/fnmol.2023.1052288.
Feder A, Costi S, Rutter SB, Collins AB, Govindarajulu U, Jha MK, et al. A randomized controlled trial of repeated ketamine administration for chronic posttraumatic stress disorder. Am J Psychiatry. 2021;178:193–202. https://doi.org/10.1176/appi.ajp.2020.20050596.
Feder A, Parides MK, Murrough JW, Perez AM, Morgan JE, Saxena S, et al. Efficacy of intravenous ketamine for treatment of chronic posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2014;71:681–8. https://doi.org/10.1001/jamapsychiatry.2014.62.
Sicignano DJ, Kurschner R, Weisman N, Sedensky A, Hernandez AV, White CM. The impact of ketamine for treatment of post-traumatic stress disorder: a systematic review with meta-analyses. Ann Pharmacother. 2024;58:669–77. https://doi.org/10.1177/10600280231199666.
Albuquerque TR, Macedo LFR, Delmondes GA, Rolim Neto ML, Almeida TM, Uchida RR, et al. Evidence for the beneficial effect of ketamine in the treatment of patients with post-traumatic stress disorder: A systematic review and meta-analysis. J Cereb Blood Flow Metab. 2022;42:2175–87. https://doi.org/10.1177/0271678X221116477.
Rasmusson AM, Pineles SL, Brown KD, Pinna G. A role for deficits in GABAergic neurosteroids and their metabolites with NMDA receptor antagonist activity in the pathophysiology of posttraumatic stress disorder. J Neuroendocrinol. 2022;34:e13062. https://doi.org/10.1111/jne.13062.
Chen S, Gao L, Li X, Ye Y. Allopregnanolone in mood disorders: Mechanism and therapeutic development. Pharm Res. 2021;169:105682. https://doi.org/10.1016/j.phrs.2021.105682.
Rasmusson AM, King MW, Valovski I, Gregor K, Scioli-Salter E, Pineles SL, et al. Relationships between cerebrospinal fluid GABAergic neurosteroid levels and symptom severity in men with PTSD. Psychoneuroendocrinology. 2019;102:95–104. https://doi.org/10.1016/j.psyneuen.2018.11.027.
Locci A, Pinna G. Neurosteroid biosynthesis down-regulation and changes in GABAA receptor subunit composition: a biomarker axis in stress-induced cognitive and emotional impairment. Br J Pharm. 2017;174:3226–41. https://doi.org/10.1111/bph.13843.
Almeida FB, Pinna G, Barros HMT. The Role of HPA Axis and Allopregnanolone on the Neurobiology of Major Depressive Disorders and PTSD. Int J Mol Sci. 2021;22:5495 https://doi.org/10.3390/ijms22115495.
Pinna G, Rasmusson AM. Ganaxolone improves behavioral deficits in a mouse model of post-traumatic stress disorder. Front Cell Neurosci. 2014;8:256 https://doi.org/10.3389/fncel.2014.00256.
Locci A, Pinna G. Stimulation of Peroxisome Proliferator-Activated Receptor-α by N-Palmitoylethanolamine Engages Allopregnanolone Biosynthesis to Modulate Emotional Behavior. Biol Psychiatry. 2019;85:1036–45. https://doi.org/10.1016/j.biopsych.2019.02.006.
Evans-Strong A, Walton N, Blandino K, Roper ATC, Donaldson ST, Lewis M, et al. Witnessed trauma exposure induces fear in mice through a reduction in endogenous neurosteroid synthesis. J Neuroendocrinol. 2024;36:e13378. https://doi.org/10.1111/jne.13378.
Marecki R, Kałuska J, Kolanek A, Hakało D, Waszkiewicz N. Zuranolone - synthetic neurosteroid in treatment of mental disorders: narrative review. Front Psychiatry. 2023;14:1298359. https://doi.org/10.3389/fpsyt.2023.1298359.
Henner RL, Keshavan MS, Hill KP. Review of potential psychedelic treatments for PTSD. J Neurol Sci. 2022;439:120302. https://doi.org/10.1016/j.jns.2022.120302.
Wolfgang AS, Fonzo GA, Gray JC, Krystal JH, Grzenda A, Widge AS, et al. MDMA and MDMA-Assisted Therapy. Am J Psychiatry. 2025;182:79–103. https://doi.org/10.1176/appi.ajp.20230681.
Bostoen T, Tap S, Breeksema J, Schoevers R. Post-traumatic stress disorder in psychedelic research. Int Rev Neurobiol. 2025;181:329–55. https://doi.org/10.1016/bs.irn.2025.02.004.
Mitchell JM, Bogenschutz M, Lilienstein A, Harrison C, Kleiman S, Parker-Guilbert K, et al. MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study. Nat Med. 2021;27:1025–33. https://doi.org/10.1038/s41591-021-01336-3.
Vizeli P, Straumann I, Duthaler U, Varghese N, Eckert A, Paulus MP, et al. Effects of 3,4-Methylenedioxymethamphetamine on conditioned fear extinction and retention in a crossover study in healthy subjects. Front Pharm. 2022;13:906639. https://doi.org/10.3389/fphar.2022.906639.
Young MB, Andero R, Ressler KJ, Howell LL. 3,4-Methylenedioxymethamphetamine facilitates fear extinction learning. Transl Psychiatry. 2015;5:e634. https://doi.org/10.1038/tp.2015.138.
van der Kolk BA, Wang JB, Yehuda R, Bedrosian L, Coker AR, Harrison C, et al. Effects of MDMA-assisted therapy for PTSD on self-experience. PLoS One. 2024;19:e0295926 https://doi.org/10.1371/journal.pone.0295926.
Smith KW, Sicignano DJ, Hernandez AV, White CM. MDMa-assisted psychotherapy for treatment of posttraumatic stress disorder: a systematic review with meta-analysis. J Clin Pharm. 2022;62:463–71. https://doi.org/10.1002/jcph.1995.
Hoyer D. Psychedelics, entactogens and psychoplastogens for depression and related disorders. Br J Pharm. 2025. https://doi.org/10.1111/bph.70088.
Stoliker D, Novelli L, Vollenweider FX, Egan GF, Preller KH, Razi A. Neural mechanisms of resting-state networks and the amygdala underlying the cognitive and emotional effects of Psilocybin. Biol Psychiatry. 2024;96:57–66. https://doi.org/10.1016/j.biopsych.2024.01.002.
Du Y, Li Y, Zhao X, Yao Y, Wang B, Zhang L, et al. Psilocybin facilitates fear extinction in mice by promoting hippocampal neuroplasticity. Chin Med J (Engl). 2023;136:2983–92. https://doi.org/10.1097/CM9.0000000000002647.
Rogers SA, Heller EA, Corder G. Psilocybin-enhanced fear extinction linked to bidirectional modulation of cortical ensembles. Nat Neurosci. 2025;28:1311–26. https://doi.org/10.1038/s41593-025-01964-9.
Anderson BT, Danforth A, Daroff PR, Stauffer C, Ekman E, Agin-Liebes G, et al. Psilocybin-assisted group therapy for demoralized older long-term AIDS survivor men: An open-label safety and feasibility pilot study. EClinicalMedicine. 2020;27:100538. https://doi.org/10.1016/j.eclinm.2020.100538.
Davis AK, Levin AW, Nagib PB, Armstrong SB, Lancelotta RL. Study protocol of an open-label proof-of-concept trial examining the safety and clinical efficacy of psilocybin-assisted therapy for veterans with PTSD. BMJ Open. 2023;13:e068884 https://doi.org/10.1136/bmjopen-2022-068884.
Meshkat S, Zeifman J, Stewart R, Janssen-Aguilar K, Lou R, Jetly W, et al. V. Psilocybin-assisted massed cognitive processing therapy for chronic posttraumatic stress disorder: Protocol for an open-label pilot feasibility trial. PLoS One. 2025;20:e0313741 https://doi.org/10.1371/journal.pone.0313741.
Chhatwal JP, Davis M, Maguschak KA, Ressler KJ. Enhancing cannabinoid neurotransmission augments the extinction of conditioned fear. Neuropsychopharmacology. 2005;30:516–24. https://doi.org/10.1038/sj.npp.1300655.
Mayo LM, Rabinak CA, Hill MN, Heilig M. Targeting the Endocannabinoid system in the treatment of posttraumatic stress disorder: a promising case of preclinical-clinical translation? Biol Psychiatry. 2022;91:262–72. https://doi.org/10.1016/j.biopsych.2021.07.019.
Morena M, Berardi A, Colucci P, Palmery M, Trezza V, Hill MN, et al. Enhancing Endocannabinoid neurotransmission augments the efficacy of extinction training and ameliorates traumatic stress-induced behavioral alterations in rats. Neuropsychopharmacology. 2018;43:1284–96. https://doi.org/10.1038/npp.2017.305.
Segev A, Korem N, Mizrachi Zer-Aviv T, Abush H, Lange R, Sauber G, et al. Role of endocannabinoids in the hippocampus and amygdala in emotional memory and plasticity. Neuropsychopharmacology. 2018;43:2017–27. https://doi.org/10.1038/s41386-018-0135-4.
Ney LJ, Crombie KM, Mayo LM, Felmingham KL, Bowser T, Matthews A. Translation of animal endocannabinoid models of PTSD mechanisms to humans: Where to next? Neurosci Biobehav Rev. 2022;132:76–91. https://doi.org/10.1016/j.neubiorev.2021.11.040.
Mizuno I, Matsuda S. The role of endocannabinoids in consolidation, retrieval, reconsolidation, and extinction of fear memory. Pharm Rep. 2021;73:984–1003. https://doi.org/10.1007/s43440-021-00246-y.
Lisboa SF, Vila-Verde C, Rosa J, Uliana DL, Stern CAJ, Bertoglio LJ, et al. Tempering aversive/traumatic memories with cannabinoids: a review of evidence from animal and human studies. Psychopharmacol (Berl). 2019;236:201–26. https://doi.org/10.1007/s00213-018-5127-x.
Sbarski B, Akirav I. Cannabinoids as therapeutics for PTSD. Pharm Ther. 2020;211:107551. https://doi.org/10.1016/j.pharmthera.2020.107551.
Neumeister A, Seidel J, Ragen BJ, Pietrzak RH. Translational evidence for a role of endocannabinoids in the etiology and treatment of posttraumatic stress disorder. Psychoneuroendocrinology. 2015;51:577–84. https://doi.org/10.1016/j.psyneuen.2014.10.012.
Xie G, Gao X, Guo Q, Liang H, Yao L, Li W, et al. Cannabidiol ameliorates PTSD-like symptoms by inhibiting neuroinflammation through its action on CB2 receptors in the brain of male mice. Brain Behav Immun. 2024;119:945–64. https://doi.org/10.1016/j.bbi.2024.05.016.
Han X, Song X, Song D, Xie G, Guo H, Wu N, et al. Comparison between cannabidiol and sertraline for the modulation of post-traumatic stress disorder-like behaviors and fear memory in mice. Psychopharmacology. 2022;239:1605–20. https://doi.org/10.1007/s00213-022-06132-6.
Hill MN, Campolongo P, Yehuda R, Patel S. Integrating endocannabinoid signaling and cannabinoids into the biology and treatment of posttraumatic stress disorder. Neuropsychopharmacology. 2018;43:80–102. https://doi.org/10.1038/npp.2017.162.
Elms L, Shannon S, Hughes S, Lewis N. Cannabidiol in the treatment of post-traumatic stress disorder: a case series. J Alter Complement Med. 2019;25:392–7. https://doi.org/10.1089/acm.2018.0437.
Bonn-Miller MO, Sisley S, Riggs P, Yazar-Klosinski B, Wang JB, Loflin MJE, et al. The short-term impact of 3 smoked cannabis preparations versus placebo on PTSD symptoms: A randomized cross-over clinical trial. PLoS One. 2021;16:e0246990 https://doi.org/10.1371/journal.pone.0246990.
Mayo LM, Gauffin E, Petrie GN, Tansey R, Mazurka R, Haggarty CJ, et al. The efficacy of elevating anandamide via inhibition of fatty acid amide hydrolase (FAAH) combined with internet-delivered cognitive behavioral therapy in the treatment of post-traumatic stress disorder: a randomized, placebo-controlled clinical trial. Neuropsychopharmacology. 2025;50:1564–72. https://doi.org/10.1038/s41386-025-02128-w.
Yang L, Meng M, Tian M, Zhang C, Zou J, Cai L, et al. Aberrations in peripheral blood interleukin-6 levels in post-traumatic stress disorder: A systematic review and meta-analysis. Behav Brain Res. 2026;497:115881. https://doi.org/10.1016/j.bbr.2025.115881.
Passos IC, Vasconcelos-Moreno MP, Costa LG, Kunz M, Brietzke E, Quevedo J, et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry. 2015;2:1002–12. https://doi.org/10.1016/S2215-0366(15)00309-0.
Zehra A, Burns J, Liu CK, Manza P, Wiers CE, Volkow ND, et al. Cannabis Addiction and the Brain: a Review. J Neuroimmune Pharm. 2018;13:438–52. https://doi.org/10.1007/s11481-018-9782-9.
Schmeltzer SN, Herman JP, Sah R. Neuropeptide Y (NPY) and posttraumatic stress disorder (PTSD): A translational update. Exp Neurol. 2016;284:196–210. https://doi.org/10.1016/j.expneurol.2016.06.020.
Sah R, Geracioti TD. Neuropeptide Y and posttraumatic stress disorder. Mol Psychiatry. 2013;18:646–55. https://doi.org/10.1038/mp.2012.101.
Sah R, Ekhator NN, Jefferson-Wilson L, Horn PS, Geracioti TD Jr. Cerebrospinal fluid neuropeptide Y in combat veterans with and without posttraumatic stress disorder. Psychoneuroendocrinology. 2014;40:277–83. https://doi.org/10.1016/j.psyneuen.2013.10.017.
Tural U, Iosifescu DV. Neuropeptide Y in PTSD, MDD, and chronic stress: A systematic review and meta-analysis. J Neurosci Res. 2020;98:950–63. https://doi.org/10.1002/jnr.24589.
Sabban EL, Alaluf LG, Serova LI. Potential of neuropeptide Y for preventing or treating post-traumatic stress disorder. Neuropeptides. 2016;56:19–24. https://doi.org/10.1016/j.npep.2015.11.004.
Serova LI, Tillinger A, Alaluf LG, Laukova M, Keegan K, Sabban EL. Single intranasal neuropeptide Y infusion attenuates development of PTSD-like symptoms to traumatic stress in rats. Neuroscience. 2013;236:298–312. https://doi.org/10.1016/j.neuroscience.2013.01.040.
Cohen H, Liu T, Kozlovsky N, Kaplan Z, Zohar J, Mathé AA. The neuropeptide Y (NPY)-ergic system is associated with behavioral resilience to stress exposure in an animal model of post-traumatic stress disorder. Neuropsychopharmacology. 2012;37:350–63. https://doi.org/10.1038/npp.2011.230.
Vollmer LL, Schmeltzer S, Schurdak J, Ahlbrand R, Rush J, Dolgas CM, et al. Neuropeptide Y impairs retrieval of extinguished fear and modulates excitability of neurons in the infralimbic prefrontal cortex. J Neurosci. 2016;36:1306–15. https://doi.org/10.1523/JNEUROSCI.4955-13.2016.
Wang SC, Lin CC, Chen CC, Tzeng NS, Liu YP. Effects of Oxytocin on fear memory and neuroinflammation in a rodent model of posttraumatic stress disorder. Int J Mol Sci. 2018;19:3848 https://doi.org/10.3390/ijms19123848.
Koch SB, van Zuiden M, Nawijn L, Frijling JL, Veltman DJ, Olff M. Intranasal Oxytocin Normalizes Amygdala Functional Connectivity in Posttraumatic Stress Disorder. Neuropsychopharmacology. 2016;41:2041–51. https://doi.org/10.1038/npp.2016.1.
Koch SB, van Zuiden M, Nawijn L, Frijling JL, Veltman DJ, Olff M. Intranasal oxytocin as strategy for medication-enhanced psychotherapy of PTSD: salience processing and fear inhibition processes. Psychoneuroendocrinology. 2014;40:242–56. https://doi.org/10.1016/j.psyneuen.2013.11.018.
Sippel LM, Khalifian CE, Knopp KC, Webster K, Maglione J, Holcomb J, et al. Pilot test of intranasal oxytocin as an enhancer of brief couples therapy for posttraumatic stress disorder. J Psychiatr Res. 2023;161:165–9. https://doi.org/10.1016/j.jpsychires.2023.03.001.
Sack M, Spieler D, Wizelman L, Epple G, Stich J, Zaba M, et al. Intranasal oxytocin reduces provoked symptoms in female patients with posttraumatic stress disorder despite exerting sympathomimetic and positive chronotropic effects in a randomized controlled trial. BMC Med. 2017;15:40. https://doi.org/10.1186/s12916-017-0801-0.
Flanagan JC, Sippel LM, Wahlquist A, Moran-Santa Maria MM, Back SE. Augmenting Prolonged Exposure therapy for PTSD with intranasal oxytocin: A randomized, placebo-controlled pilot trial. J Psychiatr Res. 2018;98:64–9. https://doi.org/10.1016/j.jpsychires.2017.12.014.
Giovanna G, Damiani S, Fusar-Poli L, Rocchetti M, Brondino N, de Cagna F, et al. Intranasal oxytocin as a potential therapeutic strategy in post-traumatic stress disorder: A systematic review. Psychoneuroendocrinology. 2020;115:104605. https://doi.org/10.1016/j.psyneuen.2020.104605.
Schultebraucks K, Maslahati T, Wingenfeld K, Hellmann-Regen J, Kraft J, Kownatzki M, et al. Intranasal oxytocin administration impacts the acquisition and consolidation of trauma-associated memories: a double-blind randomized placebo-controlled experimental study in healthy women. Neuropsychopharmacology. 2022;47:1046–54. https://doi.org/10.1038/s41386-021-01247-4.
Frijling JL, van Zuiden M, Koch SB, Nawijn L, Veltman DJ, Olff M. Intranasal Oxytocin Affects Amygdala Functional Connectivity after Trauma Script-Driven Imagery in Distressed Recently Trauma-Exposed Individuals. Neuropsychopharmacology. 2016;41:1286–96. https://doi.org/10.1038/npp.2015.278.
Lipov E, Ritchie EC. A review of the use of stellate ganglion block in the treatment of PTSD. Curr Psychiatry Rep. 2015;17:599. https://doi.org/10.1007/s11920-015-0599-4.
Hanling SR, Hickey A, Lesnik I, Hackworth RJ, Stedje-Larsen E, Drastal CA, et al. Stellate Ganglion block for the treatment of posttraumatic stress disorder: a randomized, double-blind, controlled trial. Reg Anesth Pain Med. 2016;41:494–500. https://doi.org/10.1097/AAP.0000000000000402.
Cohen H, Kaplan Z, Kotler M, Kouperman I, Moisa R, Grisaru N. Repetitive transcranial magnetic stimulation of the right dorsolateral prefrontal cortex in posttraumatic stress disorder: a double-blind, placebo-controlled study. Am J Psychiatry. 2004;161:515–24. https://doi.org/10.1176/appi.ajp.161.3.515.
Osuch EA, Benson BE, Luckenbaugh DA, Geraci M, Post RM, McCann U. Repetitive TMS combined with exposure therapy for PTSD: a preliminary study. J Anxiety Disord. 2009;23:54–9. https://doi.org/10.1016/j.janxdis.2008.03.015.
Ahmadizadeh MJ, Rezaei M, Fitzgerald PB. Transcranial direct current stimulation (tDCS) for post-traumatic stress disorder (PTSD): A randomized, double-blinded, controlled trial. Brain Res Bull. 2019;153:273–8. https://doi.org/10.1016/j.brainresbull.2019.09.011.
van ‘t Wout-Frank M, Shea MT, Larson VC, Greenberg BD, Philip NS. Combined transcranial direct current stimulation with virtual reality exposure for posttraumatic stress disorder: Feasibility and pilot results. Brain Stimul. 2019;12:41–3. https://doi.org/10.1016/j.brs.2018.09.011.
Campbell-Sills L, Sun X, Choi KW, He F, Ursano RJ, Kessler RC, et al. Dissecting the heterogeneity of posttraumatic stress disorder: differences in polygenic risk, stress exposures, and course of PTSD subtypes. Psychol Med. 2021;52:1–9. https://doi.org/10.1017/S0033291721000428.
Davis LL, Behl S, Lee D, Zeng H, Skubiak T, Weaver S, et al. Brexpiprazole and Sertraline Combination Treatment in Posttraumatic Stress Disorder: A Phase 3 Randomized Clinical Trial. JAMA Psychiatry. 2025;82:218–27. https://doi.org/10.1001/jamapsychiatry.2024.3996.
July 18, 2025: Meeting of the Psychopharmacologic Drugs Advisory Committee Meeting Announcement.
Liu H, Petukhova MV, Sampson NA, Aguilar-Gaxiola S, Alonso J, Andrade LH, et al. World Health Organization World Mental Health Survey Collaborators. Association of DSM-IV Posttraumatic Stress Disorder With Traumatic Experience Type and History in the World Health Organization World Mental Health Surveys. JAMA Psychiatry. 2017;74:270–81. https://doi.org/10.1001/jamapsychiatry.2016.3783.
Shi C, Ren Z, Zhao C, Zhang T, Chan SH. Shame, guilt, and posttraumatic stress symptoms: A three-level meta-analysis. J Anxiety Disord. 2021;82:102443. https://doi.org/10.1016/j.janxdis.2021.102443.
Békés V, Szabó D, Lévay EE, Salgó E, Unoka Z. Moral injury and shame mediate the relationship between childhood trauma and borderline personality disorder, PTSD, and Complex PTSd symptoms in psychiatric inpatients. J Pers Disord. 2023;37:406–23. https://doi.org/10.1521/pedi.2023.37.4.406.
Maercker A, Cloitre M, Bachem R, Schlumpf YR, Khoury B, Hitchcock C, et al. Complex post-traumatic stress disorder. Lancet. 2022;400:60–72. https://doi.org/10.1016/S0140-6736(22)00821-2.
Reiff CM, Richman EE, Nemeroff CB, Carpenter LL, Widge AS, Rodriguez CI, et al. the Work Group on Biomarkers and Novel Treatments, a Division of the American Psychiatric Association Council of Research. Psychedelics and Psychedelic-Assisted Psychotherapy. Am J Psychiatry. 2020;177:391–410. https://doi.org/10.1176/appi.ajp.2019.19010035.
Wang Y, Liu L, Wang J, Li J, Li H, Wang R, et al. Time-Dependent Therapeutic Effect of S-Ketamine on PTSD Mediated by VTA-OFC Dopaminergic Neurocircuit. Adv Sci (Weinh). 2025;12:e00805. https://doi.org/10.1002/advs.202500805.
Duek O, Korem N, Li Y, Kelmendi B, Amen S, Gordon C, et al. Long term structural and functional neural changes following a single infusion of Ketamine in PTSD. Neuropsychopharmacology. 2023;48:1648–58. https://doi.org/10.1038/s41386-023-01606-3.
Shalev A, Liberzon I, Marmar C. Post-traumatic stress disorder. N Engl J Med. 2017;376:2459–69. https://doi.org/10.1056/NEJMra1612499.
Funding
The authors received no funding for this work.
Author information
Authors and Affiliations
Contributions
TO Conceptualization, literature review, data extraction, manuscript drafting, and figure/table preparation. KJR: Conceptualization, critical revision of the manuscript, manuscript drafting, and figure/table preparation.
Corresponding author
Ethics declarations
Competing interests
TO no disclosures. KJR has performed scientific consultation for Bioxcel, Bionomics, Acer, Leal Therapeutics, and Jazz Pharma; serves on Scientific Advisory Boards for Sage, Boehringer Ingelheim, Senseye, and the Brain Research Foundation; and he has received sponsored research support from Alto Neuroscience. He receives research funding from the NIH, DARPA, DOD, and the Wellcome Leap Foundation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Obi, T., Ressler, K.J. Investigational drugs in PTSD. Neuropsychopharmacol. (2026). https://doi.org/10.1038/s41386-026-02371-9
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41386-026-02371-9