Abstract
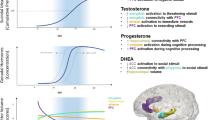
Puberty demarcates the start of adolescence, a critical period of marked changes in motivated behavior (e.g., approach, avoidance) and socio-affective processing supported by development of the mesocorticolimbic circuitry—prefrontal cortex (PFC), amygdala, and nucleus accumbens (NAcc). Puberty-related increases in psychiatric risk have been linked to alterations in mesocorticolimbic circuitry function and sensitivity to rewards and punishments. Yet, how puberty influences the development of mesocorticolimbic circuitry supporting motivational traits remains unclear. We examined resting-state functional connectivity (RSFC) in 126 adolescents, studied longitudinally (216 total scans over 2 years), assessed reward/punishment sensitivity via questionnaires, and collected multimodal measures of puberty. As hypothesized, fronto-striatal RSFC was associated with reward sensitivity, but both fronto-striatal and fronto-amygdala RSFC were linked to punishment sensitivity. Puberty moderated several associations in males but not females: weaker fronto-striatal RSFC related to higher reward sensitivity in males more advanced in pubertal maturation. Further, whereas in early puberty stages stronger fronto-striatal RSFC related to higher punishment sensitivity in males, by late puberty stages, stronger fronto-amygdala RSFC was related to higher punishment sensitivity. Testosterone levels moderated the association between anterior ventromedial PFC - NAcc RSFC and reward sensitivity such that weaker RSFC related to higher reward sensitivity in males with lower testosterone levels than expected for their age and pubertal status. These data support sex-specific puberty effects on the relationship between mesocorticolimbic circuitry connectivity and reward/punishment sensitivity. Future research is needed to determine how these findings represent markers of risk for or resilience against psychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Deidentified data from this study will be made available by the lead author upon reasonable request.
Notes
Throughout, we refer to youth as ‘males’ and ‘females’ based on the parent study demarcation (for literature reviews) or based on self-identification of binary sex stemming from sex assigned at birth based on visual inspection of genitalia; however, this approach denies how biological sex operates on a continuum (e.g., testosterone is biologically active across sexes), and that for many youth, sex assigned by chromosomes, endocrine, or genitalia are not dichotomous (e.g., ovarian streaks in girls with Turner syndrome). See [37] for a discussion.
References
Vijayakumar N, Macks de ZO, Shirtcliff EA, Pfeifer JH. Puberty and the human brain: insights into adolescent development. Neurosci Biobehav Rev. 2018;92:417–36.
Larsen B, Luna B. Adolescence as a neurobiological critical period for the development of higher-order cognition. Neurosci Biobehav Rev. 2018;94:179–95.
Spear LP. The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev. 2000;24:417–63.
Wahlstrom D, Collins P, White T, Luciana M. Developmental changes in dopamine neurotransmission in adolescence: behavioral implications and issues in assessment. Brain Cogn. 2010;72:146–59.
Gee DG, Bath KG, Johnson CM, Meyer HC, Murty VP, van den Bos W, et al. Neurocognitive development of motivated behavior: dynamic changes across childhood and adolescence. J Neurosci. 2018;38:9433–45.
Shulman EP, Harden KP, Chein JM, Steinberg L. The development of impulse control and sensation-seeking in adolescence: independent or interdependent processes? J Res Adolesc. 2016;26:37–44.
Luna B, Wright C. APA handbook of psychology and juvenile justice. 2016. 91–116. https://doi.org/10.1037/14643-005.
Crone EA, Dahl RE. Understanding adolescence as a period of social–affective engagement and goal flexibility. Nat Rev Neurosci. 2012;13:636–50.
Dahl RE, Allen NB, Wilbrecht L, Suleiman AB. Importance of investing in adolescence from a developmental science perspective. Nature. 2018;554:441–50.
Nelson EE, Jarcho JM, Guyer AE. Social re-orientation and brain development: an expanded and updated view. Dev Cogn Neurosci. 2016;17:118–27.
Paus T, Keshavan M, Giedd JN. Why do many psychiatric disorders emerge during adolescence?. Nat Rev Neurosci. 2008;9:947–57.
Dalsgaard S, Thorsteinsson E, Trabjerg BB, Schullehner J, Plana-Ripoll O, Brikell I, et al. Incidence rates and cumulative incidences of the full spectrum of diagnosed mental disorders in childhood and adolescence. JAMA Psychiatry. 2020;77:155–64.
Solmi M, Radua J, Olivola M, Croce E, Soardo L, Salazar de Pablo G, et al. Age at onset of mental disorders worldwide: large-scale meta-analysis of 192 epidemiological studies. Mol Psychiatry. 2022;27:281–95.
Somerville LH. The teenage brain. Curr Dir Psychol Sci. 2013;22:121–7.
Pfeifer JH, Allen NB. Puberty initiates cascading relationships between neurodevelopmental, social, and internalizing processes across adolescence. Biol Psychiatry. 2021;89:99–108.
Ladouceur CD, Peper JS, Crone EA, Dahl RE. White matter development in adolescence: the influence of puberty and implications for affective disorders. Dev Cogn Neurosci. 2012;2:36–54.
Eckert-Lind C, Busch AS, Petersen JH, Biro FM, Butler G, Bräuner EV, et al. Worldwide secular trends in age at pubertal onset assessed by breast development among girls. JAMA Pediatr. 2020;174:e195881.
Bräuner EV, Busch AS, Eckert-Lind C, Koch T, Hickey M, Juul A. Trends in the Incidence of Central Precocious Pubertyand Normal Variant Puberty Among Children in Denmark, 1998 to 2017. JAMA Netw Open 2020;3:e2015665.
Millner AJ, Zuromski KL, Joyce VW, Kelly F, Richards C, Buonopane RJ, et al. Increased severity of mental health symptoms among adolescent inpatients during COVID-19. Gen Hosp Psychiatry. 2022;77:77–79.
Keyes KM, Platt JM. Annual Research Review: Sex, gender, and internalizing conditions among adolescents in the 21st century – trends, causes, consequences. J Child Psychol Psychiatry. 2024;65:384–407.
Hyatt CJ, Wexler BE, Diefenbach GJ, Dichter GS, Mazefsky CA, Uscatescu LC, et al. Brain-wide connectivity changes due to social–emotional regulation during a naturalistic fMRI task. Cereb Cortex. 2025;35:bhaf118.
Palminteri S, Kilford EJ, Coricelli G, Blakemore S-J. The computational development of reinforcement learning during adolescence. PLoS Comput Biol. 2016;12:e1004953.
Ernst M, Pine DS, Hardin M. Triadic model of the neurobiology of motivated behavior in adolescence. Psychol Med. 2006;36:299–312.
van Duijvenvoorde ACK, Op de Macks ZA, Overgaauw S, Gunther Moor B, Dahl RW, Crone EA. et al. A cross-sectional and longitudinal analysis of reward-related brain activation: effects of age, pubertal stage, and reward sensitivity. Brain Cogn. 2014;89:3–14.
Gee DG, Humphreys KL, Flannery J, Goff B, Telzer EH, Shapiro M, et al. A developmental shift from positive to negative connectivity in human amygdala-prefrontal circuitry. J Neurosci J Soc Neurosci. 2013;33:4584–93.
Wu M, Kujawa A, Lu LH, Fitzgerald DA, Klumpp H, Fitzgerald KD, et al. Age-related changes in amygdala–frontal connectivity during emotional face processing from childhood into young adulthood. Hum Brain Mapp. 2016;37:1684–95.
Ernst M, Nelson EE, Jazbec S, McClure EB, Monk CS, Leibenluft E, et al. Amygdala and nucleus accumbens in responses to receipt and omission of gains in adults and adolescents. NeuroImage. 2005;25:1279–91.
Ferenczi EA, Zalocusky KA, Liston C, Grosenick L, Warden MR, Amatya D, et al. Prefrontal cortical regulation of brainwide circuit dynamics and reward-related behavior. Science. 2016;351:aac9698.
Baxter MG, Murray EA. The amygdala and reward. Nat Rev Neurosci. 2002;3:563–73.
Camara E. Functional connectivity of reward processing in the brain. Front Hum Neurosci. 2008;2:19.
Kim SH, Yoon H, Kim H, Hamann S. Individual differences in sensitivity to reward and punishment and neural activity during reward and avoidance learning. Soc Cogn Affect Neurosci. 2015;10:1219–27.
Yacubian J, Gläscher J, Schroeder K, Sommer T, Braus DF, Büchel C. Dissociable systems for gain- and loss-related value predictions and errors of prediction in the human brain. J Neurosci. 2006;26:9530–7.
Jonker NC, Ostafin BD, Glashouwer KA, Van Hemel-Ruiter ME, De Jong PJ. Reward and punishment sensitivity and alcohol use: the moderating role of executive control. Addict Behav. 2014;39:945–8.
Vergara-Lopez C, Lopez-Vergara HI, Colder CR. Executive functioning moderates the relationship between motivation and adolescent depressive symptoms. Personal Individ Differ. 2013;54:18–22.
Cardoso Melo RD, Groen RN, Hartman CA. Reward sensitivity at age 13 predicts the future course of psychopathology symptoms. Frontiers. https://doi.org/10.3389/fpsyt.2022.818047.
Forbes EE, Phillips ML, Silk JS, Ryan ND, Dahl RE. Neural systems of threat processing in adolescents: role of pubertal maturation and relation to measures of negative affect. Dev Neuropsychol. 2011;36:429–52.
Ruttle PL et al. Hormonal changes through a social justice lens. In: Encyclopedia of Adolescence, 191–204. Elsevier; 2024. https://doi.org/10.1016/B978-0-323-96023-6.00125-1.
Hankin BL, Abramson LY. Development of gender differences in depression: an elaborated cognitive vulnerability-transactional stress theory. Psychol Bull. 2001;127:773–96.
Kessler RC, McGonagle KA, Swartz M, Blazer DG, Nelson CB. Sex and depression in the National Comorbidity Survey I: Lifetime prevalence, chronicity and recurrence. J Affect Disord. 1993;29:85–96.
Nolen-Hoeksema S, Girgus JS. The emergence of gender differences in depression during adolescence. Psychol Bull. 1994;115:424–43.
Dunsmoor JE, Paz R. Fear generalization and anxiety: behavioral and neural mechanisms. Biol Psychiatry. 2015;78:336–43.
Guyer AE, Nelson EE, Perez-Edgar K, Hardin MG, Roberson-Nay R, Monk CS, et al. Striatal functional alteration in adolescents characterized by early childhood behavioral inhibition. J Neurosci. 2006;26:6399–405.
Hevey D, Thomas K, Laureano-Schelten S, Looney K, Booth R. Clinical depression and punishment sensitivity on the BART. Front Psychol. 2017;8:670.
Riley AL, Manke HN, Huang S. Impact of the aversive effects of drugs on their use and abuse. Behav Neurol. 2022;2022:8634176.
Simerly RB, Swanson LW, Chang C, Muramatsu M. Distribution of androgen and estrogen receptor mRNA-containing cells in the rat brain: an in situ hybridization study. J Comp Neurol. 1990;294:76–95.
Sato SM, Schulz KM, Sisk CL, Wood RI. Adolescents and androgens, receptors and rewards. Horm Behav. 2008;53:647–58.
Suleiman AB, Galván A, Harden KP, Dahl RE. Becoming a sexual being: the ‘elephant in the room’ of adolescent brain development. Dev Cogn Neurosci. 2017;25:209–20.
Epstein R, Blake J, Gonzzlez T. Girlhood interrupted: the erasure of Black girls childhood. SSRN Electron J. 2017. https://doi.org/10.2139/ssrn.3000695.
Delevich K, Klinger M, Okada NJ, Wilbrecht L. Coming of age in the frontal cortex: the role of puberty in cortical maturation. Semin Cell Dev Biol. 2021;118:64–72.
Goddings A-L, Mills KL, Clasen LS, Giedd JN, Viner RM, Blakemore S-J. The influence of puberty on subcortical brain development. NeuroImage. 2014;88:242–51.
Urošević S, Collins P, Muetzel R, Lim KO, Luciana M. Pubertal status associations with reward and threat sensitivities and subcortical brain volumes during adolescence. Brain Cogn. 2014;89:15–26.
Vijayakumar N, Youssef G, Allen NB, Anderson V, Efron D, Mundy L, et al. et al. The effects of puberty and its hormones on subcortical brain development. Compr Psychoneuroendocrinol. 2021;7:100074.
Chahal R, Delevich K, Kirshenbaum JS, Borchers LR, Ho TC, Gotlib IH. Sex differences in pubertal associations with fronto-accumbal white matter morphometry: Implications for understanding sensitivity to reward and punishment. NeuroImage. 2021;226:117598.
Duijvenvoorde ACK, Westhoff B, Vos F, Wierenga LM, Crone EA. A three-wave longitudinal study of subcortical–cortical resting-state connectivity in adolescence: Testing age- and puberty-related changes. Hum Brain Mapp. 2019;40:3769–83.
Spielberg JM, Forbes EE, Ladouceur CD, Worthman CM, Olino TM, Ryan ND, et al. Pubertal testosterone influences threat-related amygdala–orbitofrontal cortex coupling. Soc Cogn Affect Neurosci. 2015;10:408–15.
Ojha A, Parr AC, Foran W, Calabro FJ, Luna B. Puberty contributes to adolescent development of fronto-striatal functional connectivity supporting inhibitory control. Dev Cogn Neurosci. 2022;58:101183.
Ravindranath O, Calabro FJ, Foran W, Luna B. Pubertal development underlies optimization of inhibitory control through specialization of ventrolateral prefrontal cortex. Dev Cogn Neurosci. 2022;58:101162.
Fareri DS, Gabard-Durnam L, Goff B, Flannery J, Gee DG, Lumian DS, et al. Normative development of ventral striatal resting state connectivity in humans. NeuroImage. 2015;118:422–37.
Sanders AFP, Harms MP, Kandala S, Marek S, Somerville LH, Bookheimer SY, et al. Age-related differences in resting-state functional connectivity from childhood to adolescence. Cereb Cortex. 2023;33:6928–42.
Gabard-Durnam LJ, Flannery J, Goff B, Gee DG, Humphreys KL, Telzer E, et al. The development of human amygdala functional connectivity at rest from 4 to 23 years: a cross-sectional study. NeuroImage. 2014;95:193–207.
Ladouceur CD, Henry T, Ojha A, Shirtcliff EA, Silk JS. Fronto-amygdala resting state functional connectivity is associated with anxiety symptoms among adolescent girls more advanced in pubertal maturation. Dev Cogn Neurosci. 2023;60:101236.
Omary A, Curtis M, Cheng TW, Mair P, Shirtcliff EA, Barch DM, et al. Multimodal measurement of pubertal development: stage, timing, tempo, and hormones. Child Dev. 2025;96:980–99.
Ladouceur CD, Kerestes R, Schlund MW, Shirtcliff EA, Lee Y, Dahl R. Neural systems underlying reward cue processing in early adolescence: the role of puberty and pubertal hormones. Psychoneuroendocrinology. 2019;102:281–91.
Tobiansky DJ, Korol AM, Ma C, Hamden JE, Jalabert C, Tomm RJ, et al. Testosterone and corticosterone in the mesocorticolimbic system of male rats: effects of gonadectomy and caloric restriction. Endocrinology. 2018;159:450–64.
Loeber R, Hay D. Key issues in the development of aggression and violence from childhood to early adulthood. Annu Rev Psychol. 1997;48:371–410.
Forbes EE, Ryan ND, Phillips ML, Manuck SB, Worthman CM, Moyles DL, et al. Healthy adolescents’ neural response to reward: associations with puberty, positive affect, and depressive symptoms. J Am Acad Child Adolesc Psychiatry. 2010;49:162–172.e5.
Spielberg JM, Olino TM, Forbes EE, Dahl RE. Exciting fear in adolescence: does pubertal development alter threat processing?. Dev Cogn Neurosci. 2014;8:86–95.
White SF, Lee Y, Schlund MW, Shirtcliff EA, Ladouceur CD. Testosterone reactivity is associated with reduced neural response to reward in early adolescence. Behav Brain Res. 2020;387:112593.
Byrne ML, Whittle S, Vijayakumar N, Dennison M, Simmons JG, Allen NB. A systematic review of adrenarche as a sensitive period in neurobiological development and mental health. Dev Cogn Neurosci. 2017;25:12–28.
Dorn LD, Dahl RE, Woodward HR, Biro F. Defining the boundaries of early adolescence: a user’s guide to assessing pubertal status and pubertal timing in research with adolescents. Appl Dev Sci. 2006;10:30–56.
Marshall WA, Tanner JM. Variations in pattern of pubertal changes in girls. Arch Dis Child. 1969;44:291.
Marshall WA, Tanner JM. Variations in the pattern of pubertal changes in boys. Arch Dis Child. 1970;45:13.
Petersen AC, Crockett L, Richards M, Boxer A. A self-report measure of pubertal status: reliability, validity, and initial norms. J Youth Adolesc. 1988;17:117–33.
Shirtcliff EA, Granger DA, Schwartz EB, Curran MJ, Booth A, Overman WH. Assessing estradiol in biobehavioral studies using saliva and blood spots: simple radioimmunoassay protocols, reliability, and comparative validity. Horm Behav. 2000;38:137–47.
Torrubia R, Ávila C, Moltó J, Caseras X. The sensitivity to punishment and sensitivity to reward questionnaire (SPSRQ) as a measure of Gray’s anxiety and impulsivity dimensions. Personal Individ Differ. 2001;31:837–62.
Silvers JA, Insel C, Powers A, Franz P, Helion C, Martin RE, et al. vlPFC–vmPFC–amygdala interactions underlie age-related differences in cognitive regulation of emotion. Cereb Cortex. 2016;27:3502–14.
van Duijvenvoorde ACK, Achterberg M, Braams BR, Peters S, Crone EA. Testing a dual-systems model of adolescent brain development using resting-state connectivity analyses. NeuroImage. 2016;124:409–20.
Bates D, Mächler M, Bolker B, Walker S. Fitting linear mixed-effects models using lme4. J Stat Softw. 2015;67:1–48.
Kuznetsova A, Brockhoff PB, Christensen RHB. lmerTest package: tests in linear mixed effects models. J Stat Softw. 2017;82:1–26.
R Core Team. The R Project for Statistical Computing. 2023.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B Methodol. 1995;57:289–300.
Yankouskaya A, Humphreys G, Stolte M, Stokes M, Moradi Z, Sui J. An anterior–posterior axis within the ventromedial prefrontal cortex separates self and reward. Soc Cogn Affect Neurosci. 2017. https://doi.org/10.1093/scan/nsx112.
Smith DV, Clithero JA, Boltuck SE, Huettel SA. Functional connectivity with ventromedial prefrontal cortex reflects subjective value for social rewards. Soc Cogn Affect Neurosci. 2014. https://doi.org/10.1093/scan/nsu005.
O’Doherty JP. Contributions of the ventromedial prefrontal cortex to goal-directed action selection. Ann N Y Acad Sci. 2011;1239:118–29.
Peters J, Büchel C. Neural representations of subjective reward value. Behav Brain Res. 2010;213:135–41.
Stawicka ZM, Massoudi R, Horst NK, Koda K, Gaskin PLR, Alexander L, et al. Ventromedial prefrontal area 14 provides opposing regulation of threat and reward-elicited responses in the common marmoset. Proc Natl Acad Sci. 2020;117:25116–27.
Parr AC, Calabro F, Larsen B, Tervo-Clemmens B, Elliot S, Foran W, et al. Dopamine-related striatal neurophysiology is associated with specialization of frontostriatal reward circuitry through adolescence. Prog Neurobiol. 2021;201:101997.
Wen Z, Pace-Schott EF, Lazar SW, Rosén J, Åhs F, Phelps EA, et al. Distributed neural representations of conditioned threat in the human brain. Nat Commun. 2024;15:2231.
Silveri MM, Sneider JT, Crowley DJ, Covell MJ, Acharya D, Rosso IM, et al. Frontal lobe GABA levels during adolescence: associations with impulsivity and response inhibition. Biol Psychiatry. 2013;74:296.
Caballero A, Tseng KY. GABAergic function as a limiting factor for prefrontal maturation during adolescence. Trends Neurosci. 2016;39:441–8.
Reynolds LM, Flores C. Mesocorticolimbic dopamine pathways across adolescence: diversity in development. Front Neural Circuits. 2021;15:735625.
Tervo-Clemmens B, Karim ZA, Khan SZ, Ravindranath O, Somerville LH, Schuster RM, et al. The developmental timing but not magnitude of adolescent risk-taking propensity is consistent across social, environmental, and psychological factors. J Adolesc Health. 2024;74:613–6.
Greimel E, Bakos S, Landes I, Töllner T, Bartling J, Kohls G, et al. Sex differences in the neural underpinnings of social and monetary incentive processing during adolescence. Cogn Affect Behav Neurosci. 2018;18:296–312.
Chai Y, Chimelis-Santiago JR, Bixler KA, Aalsma M, Yu M, Hulvershorn LA. Sex-specific frontal-striatal connectivity differences among adolescents with externalizing disorders. NeuroImage Clin. 2021;32:102789.
de Jong JW, Afjei SA, Dorocic IP, Peck JR, Liu C, Kim CK, et al. A neural circuit mechanism for encoding aversive stimuli in the mesolimbic dopamine system. Neuron. 2019;101:133–151.e7.
Carlezon WA, Thomas MJ. Biological substrates of reward and aversion: a nucleus accumbens activity hypothesis. Neuropharmacology. 2009;56:122–32.
McClure SM, York MK, Montague PR. The neural substrates of reward processing in humans: the modern role of fMRI. Sage J. 2004. https://doi.org/10.1177/1073858404263526.
Jensen J, McIntosh AR, Crawley AP, Mikulis DJ, Remington G, Kapur S. Direct activation of the ventral striatum in anticipation of aversive stimuli. Neuron. 2003;40:1251–7.
Badrinarayan A, Wescott SA, Vander Weele CM, Saunders BT, Couturier BE, Maren S, et al. Aversive stimuli differentially modulate real-time dopamine transmission dynamics within the nucleus accumbens core and shell. J Neurosci. 2012;32:15779–90.
D’Argembeau A. On the role of the ventromedial prefrontal cortex in self-processing: the valuation hypothesis. Front Hum Neurosci. 2013;7:372.
Bartra O, McGuire JT, Kable JW. The valuation system: a coordinate-based meta-analysis of BOLD fMRI experiments examining neural correlates of subjective value. NeuroImage. 2013;76:412–27.
Floresco S. The nucleus accumbens: an interface between cognition, emotion, and action. Annu Rev Psychol. 2014;66:25–52.
Haber SN, Knutson B. The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology. 2010;35:4–26.
Phelps EA, LeDoux JE. Contributions of the amygdala to emotion processing: from animal models to human behavior. Neuron. 2005;48:175–87.
Sander D, Grafman J, Zalla T. The human amygdala: an evolved system for relevance detection. Rev Neurosci. 2003;14:303–16.
Phillips ML, Ladouceur CD, Drevets WC. A neural model of voluntary and automatic emotion regulation: implications for understanding the pathophysiology and neurodevelopment of bipolar disorder. Mol Psychiatry. 2008;13:833–57.
Silvers JA. Adolescence as a pivotal period for emotion regulation development. Curr Opin Psychol. 2022;44:258–63.
Zilverstand A, Parvaz MA, Goldstein RZ. Neuroimaging cognitive reappraisal in clinical populations to define neural targets for enhancing emotion regulation. A systematic review. NeuroImage. 2017;151:105–16.
Agustín-Pavón C, Braesicke K, Shiba Y, Santangelo AM, Mikheenko Y, et al. Lesions of ventrolateral prefrontal or anterior orbitofrontal cortex in primates heighten negative emotion. Biol Psychiatry. 2012;72:266–72.
Ghashghaei HT, Hilgetag CC, Barbas H. Sequence of information processing for emotions based on the anatomic dialogue between prefrontal cortex and amygdala. NeuroImage. 2007;34:905–23.
Ray RD, Zald DH. Anatomical insights into the interaction of emotion and cognition in the prefrontal cortex. Neurosci Biobehav Rev. 2012;36:479–501.
Quirk GJ, Beer JS. Prefrontal involvement in the regulation of emotion: convergence of rat and human studies. Curr Opin Neurobiol. 2006;16:723–7.
Kong M-S, Zweifel LS. Central amygdala circuits in valence and salience processing. Behav Brain Res. 2021;410:113355.
Silvers JA, Insel C, Powers A, Franz P, Helion C, Martin R, et al. The transition from childhood to adolescence is marked by a general decrease in amygdala reactivity and an affect-specific ventral-to-dorsal shift in medial prefrontal recruitment. Dev Cogn Neurosci. 2017;25:128–37.
Likhtik E, Pelletier JG, Paz R, Paré D. Prefrontal control of the amygdala. J Neurosci. 2005;25:7429–37.
Banks SJ, Eddy KT, Angstadt M, Nathan PJ, Phan KL. Amygdala–frontal connectivity during emotion regulation. Soc Cogn Affect Neurosci. 2007;2:303–12.
Davis M, Whalen PJ. The amygdala: vigilance and emotion. Mol Psychiatry. 2001;6:13–34.
Simmonds DJ, Hallquist MN, Asato M, Luna B. Developmental stages and sex differences of white matter and behavioral development through adolescence: a longitudinal diffusion tensor imaging (DTI) study. NeuroImage. 2014;92:356–68.
Tromp DPM, Williams LE, Fox AS, Oler JA, Roseboom PH, Rogers GM, et al. Altered uncinate fasciculus microstructure in childhood anxiety disorders in boys but not girls. Am J Psychiatry. 2019. https://doi.org/10.1176/appi.ajp.2018.18040425.
Olson J, Schrager SM, Belzer M, Simons LK, Clark LF. Baseline physiologic and psychosocial characteristics of transgender youth seeking care for gender dysphoria. J Adolesc Health. 2015;57:374–80.
Moyer DN, Connelly KJ, Holley AL. Using the PHQ-9 and GAD-7 to screen for acute distress in transgender youth: findings from a pediatric endocrinology clinic. J Pediatr Endocrinol Metab. 2019;32:71–74.
Strauss P, Cook A, Winter S, Watson V, Wright Toussaint D, Lin A. Associations between negative life experiences and the mental health of trans and gender diverse young people in Australia: findings from Trans Pathways. Psychol Med. 2020;50:808–17.
Fareri DS, Martin LN, Delgado MR. Reward-related processing in the human brain: developmental considerations. Dev Psychopathol. 2008;20:1191–211.
Palminteri S, Justo D, Jauffret C, Pavlicek B, Dauta A, Delmaire C, et al. Critical roles for anterior insula and dorsal striatum in punishment-based avoidance learning. Neuron. 2012;76:998–1009.
Gueguen MCM, Lopez-Persem A, Billeke P, Lachaux J, Rheims S, Kahane P, et al. Anatomical dissociation of intracerebral signals for reward and punishment prediction errors in humans. Nat Commun. 2021;12:3344.
Roy M, Shohamy D, Daw N, Jepma M, Wimmer GE, Wager TD. Representation of aversive prediction errors in the human periaqueductal gray. Nat Neurosci. 2014;17:1607–12.
Poulton A, Hester R. Transition to substance use disorders: impulsivity for reward and learning from reward. Soc Cogn Affect Neurosci. 2020;15:1182–91.
Miu AC, Bîlc MI, Bunea I, Szentágotai-Tătar A. Childhood trauma and sensitivity to reward and punishment: Implications for depressive and anxiety symptoms. Personal Individ Differ. 2017;119:134–40.
Katz BA, Matanky K, Aviram G, Yovel I. Reinforcement sensitivity, depression and anxiety: a meta-analysis and meta-analytic structural equation model. Clin Psychol Rev. 2020;77:101842.
Jernigan TL, Brown SA, Dowling GJ. The adolescent brain cognitive development study. J Res Adolesc J Soc Res Adolesc. 2018;28:154–6.
Cheng TW, Magis-Weinberg L, Guazzelli Williamson V, Ladouceur CD, Whittle SL, Herting MM, et al. A researcher’s guide to the measurement and modeling of puberty in the ABCD study® at baseline. Front Endocrinol. 2021;12:608575.
Acknowledgements
The study authors would like to thank the study participants, their families, and the research staff in the Cognitive-Affective Neuroscience & Development (CAN-D) Lab for making this work possible.
Funding
This research was supported by grants from the National Institute of Mental Health (R01MH099007, PI: Ladouceur).
Author information
Authors and Affiliations
Contributions
AO and CDL designed the study; AO led data processing and analysis, and writing; TRH, NPJ, and EAS assisted with data processing and analysis. All authors contributed to interpretation, writing, and approving the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ojha, A., Henry, T.R., Jones, N.P. et al. Mesocorticolimbic connectivity and motivational sensitivity: sex-specific effects of puberty in early adolescence. Neuropsychopharmacol. (2026). https://doi.org/10.1038/s41386-026-02378-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41386-026-02378-2